Joseph D. Tobias, MD1-3
1Department of Anesthesiology & Pain Medicine, Nationwide Children’s Hospital, Columbus, Ohio
2Department of Anesthesiology & Pain Medicine, The Ohio State University College of Medicine, Columbus, Ohio
3Department of Pediatrics, The Ohio State University College of Medicine, Columbus, Ohio
Correspondence: Joseph D. Tobias, MD, Chairman, Department of Anesthesiology & Pain Medicine, Nationwide Children’s Hospital, 700 Children’s Drive, Columbus, Ohio 43205 (USA); Phone: (614) 722-4200; FAX: (614) 722-4203; E-mail: Joseph.Tobias@Nationwidechildrens.org
SUMMARY
In critically ill pediatric patients, central venous access may serve many objectives including the administration of life-saving medications, a secure source of vascular access, and a site for monitoring central venous pressure and obtaining intermittent blood samples. As with any invasive procedure, the risk-benefit ratio must be considered. Although a CVC may be used to provide life-saving therapies, complications and adverse effects may occur. These complications may occur during catheter placement or with its subsequent use. The two factors that have provided the most dramatic impact in decreasing complications include the use of ultrasound for CVC placement and the placement checklist recommended by Dr. Pronovost. Ultrasound has been shown to increase the success rate and decrease the complication rate by helping the clinical avoid inadvertent carotid puncture and excessive depth of needle insertion which may result in pneumothorax. With ongoing use in the ICU setting, a daily reassessment of the need for central venous access should be included into the rounding checklist so that consideration regarding removal of the line is discussed on a daily basis.
Key words: Catheterization, Cannulation; Central Venous; Catheterization, Peripheral; Ultrasonography; Diagnostic Ultrasound; Ultrasonography, Interventional; Checklist
Citation: Tobias JD. The safety of invasive monitoring in infants and children: Complications of central venous access. Anaesth Pain & Intensive Care 2016;20(1):3-7
INTRODUCTION
Although the first public display of the delivery of general anesthesia in the ether dome in Boston, Massachusetts was successful with no reported morbidity, this sentinel case was followed within 2 years by the first reported case of a death related to anesthesia in a child. Despite improvements in preoperative preparation, perioperative care, monitoring, and pharmacology, morbidity and mortality may still occur during anesthetic care.1-4 The incidence of such problems is increased in younger pediatric patients (neonates and infants), in the presence of co-morbid diseases, in higher American Society of Anesthesiologists (ASA) status, and by a lack of experience or training of the anesthesia provider.3-5 Data from the ‘Australian Incident Monitoring Study’ suggested that approximately half of intraoperative cardiac arrests were anesthesia-related. Of these events, more than half had a preventable factor that could be identified by the use of intraoperative monitoring technologies.6
One factor that may be instrumental in the early identification, prevention, and treatment of adverse events is the presence of central venous access. A central venous catheter (CVC) may be used during the perioperative period to provide access for the rapid delivery of resuscitation medications, for the administration of postoperative parenteral nutrition, for fluid administration to treat intravascular volume depletion, for monitoring of hemodynamic status or for long term venous access following complex surgical procedures. Despite its utility in various clinical scenarios, attaining central venous access is rarely if ever indicated in the emergency situation. When rapid vascular access is required for resuscitation, if peripheral venous access cannot be achieved within 60 seconds, intraosseous access should be obtained.7-9
Although anesthesiologists are frequently called upon to obtain central venous access during the perioperative period or in the ICU setting, familiarity with these techniques may be decreasing given the increased use of peripherally inserted central (PIC) catheters and the emerging trend of placement of invasive vascular devices by Interventional Radiologists. The following editorial reviews some of the more common complications associated with placement and subsequent use of a CVC. Preventive strategies are reviewed and treatment options for complications presented.
Complications associated with catheter placement:
Complications related to CVCs can broadlybe grouped into those which may occur during placement and those which occur during subsequent use (Table 1). The incidence of these complications will vary based on the experience of the practitioner, the size of the patient (adult vs. pediatric vs. neonate), the site of cannulation (internal jugular, subclavian, femoral), use of ultrasound, and the duration that the CVC is in place. Given the myriad of factors that impact the complication rate, it is generally not feasible to come up with a specific incidence regarding complications such as a pneumothorax or vascular injury, but rather make broad statements from the available evidence-based medicine.

In pediatric practice, especially neonates and infants, the size of the CVC that is placed is smaller than those used in adult practice (4F versus 7-8F). The 4F, double lumen kit generally has a 22G needle and a 0.018” wire. For the internal jugular (IJ) approach, in older patients and certainly in adults, it may be beneficial to use a 22G, finder needle to demonstrate the location of the vein, prior to insertion of the larger needle (16-18G) which is used to pass the larger wire necessary for vessel cannulation. The finder needle and syringe can be used to identify the vessel and then the larger needle and syringe inserted directly on top of and parallel with the other needle and syringe. This practice may help prevent inadvertent arterial puncture especially if ultrasound is not used. Others may prefer to use a standard peripheral intravenous cannula to identify the vein and then slide the cannula off the needle and into the vein. The wire is then passed through the cannula.Some of the complications associated with catheter placement are site specific. Obviously pneumothorax is only a concern with internal jugular or subclavian approaches and cannot occur with the femoral approach. Although vascular injury and perforation may occur with any site of placement given the proximity of the venous and arterial structures, direct pressure is generally more feasible with the femoral versus the internal jugular or subclavian approaches thereby limiting the morbidity of such issues if the femoral route is used.
After wire placement and prior to placement of the dilator and larger cannula, the correct location of the wire within the venous system should be confirmed. This can be done using ultrasound (see below) or by placement of a small, single lumen cannula (22 or 20G) over the wire. The small cannula can then be transduced or connected to a fluid column to demonstrate that it is within the venous system. If none of these modalities are available, a blood gas sample can be obtained from the catheter. Although the risks of serious sequelae are limited with a single inadvertent carotid puncture with a needle, these complications can be significantly more severe in patients with coagulation disturbances or thrombocytopenia. As such, these steps or the use of ultrasound is suggested to avoid complications, especially in high risk patients.
Ultrasound has rapidly gained acceptance and is now considered in many countries to be the standard of care for CVC placement. It may be particularly beneficial in smaller pediatric patients e.g. neonates and infants, because of the smaller caliber of the vessels and perhaps a greater likelihood of non-optimal anatomy (carotid artery and internal jugular vein overlapping). The problems posed by these anatomical variations are highlighted by the fact that the incidence of complications increases with the number of needle punctures and attempts.10-12 Without ultrasound, the site of CVC is identified using surface landmarks or the pulsation of the carotid artery.13,14 The use of surface landmarks is complicated by the variation in position of the deep vascular structures in relation to these surface landmarks.15,16 It has been suggested that there should be at least one systematic ultrasound screening before CVC access is attempted. Our current clinical practice is to use real time ultrasound to watch the displacement of the tissues as the needle is advanced from the skin into the lumen of the IJ. Evidence-based literature from the pediatric population has demonstrated a higher success rate, shorter procedure time, fewer attempts, and a decreased risk of carotid puncture when using ultrasound versus surface landmarks for CVC placement in the IJ.17-19 Similar results have been noted for CVC placement via the femoral or subclavian routes.20-23
Ultrasound allows for visualization of the anatomical relationship of the vessels, identification of the needle entry into the internal jugular vein, and when placed in the longitudinal view, can also be used to identify the wire within the vessel prior to cannula placement (Figure 1). The latter should be performed prior to dilator placement to ensure that the wire is within the vein thereby avoiding potential damage to the carotid artery.
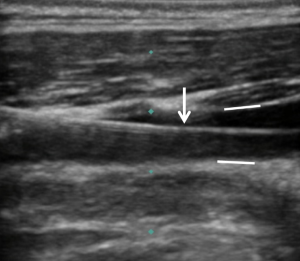
Figure 1: Longitudinal ultrasound image of the neck, showing the guide wire (arrow) within the lumen of the internal jugular vein. The anterior and posterior vessel walls are also clearly identified (straight white lines).
Many of the potential complications listed in Table 1 can be prevented or significantly decreased with ultrasound. However, others such as the occurrence of arrhythmias, wire embolization, and cardiac tamponade require additional vigilance to prevent. Once the vessel is cannulated and the wire placed, the dilator is gently advanced over the wire using a twisting motion while inserting. The wires used in pediatric patients are significantly smaller in diameter (0.018″ versus 0.035ʺ) than those used in adults and as such may more easily be kinked or a false passage made as the dilator is advanced. The dilator should not be advanced any deeper than the needle had been (generally 1-2 centimeters from the skin), to avoid the potential for damage to the great vessels or the heart. With longer dilators, perforation of the atrium is possible if the dilator is advanced too deeply. In general, the IJ is no more than 1-2 cm deep to the skin in a pediatric patient.
The operator should always have control of the wire above and below the dilator or CVC at all times. As the guide wires are generally 2.5-3 times the length of the catheter, excessive lengths should not be advanced through the needle as it may irritate the atrium or ventricle leading to arrhythmias. Constant observation of the electrocardiogram is suggested during wire advancement. Arrhythmias are generally easily treated by gentle withdrawal of the wire.
Free aspiration of air into the syringe may herald inadvertent pleural puncture and pneumothorax. However, it is more commonly seen when the syringe is not fixed firmly in the needle hub allowing for the entrainment of air. Suspected pleural puncture mandates close observation of the patient’s clinical condition as a progressive tension pneumothorax may result during positive pressure ventilation leading to hemodynamic compromise and the needle for prompt therapy. If hemodynamic compromise is noted and breath sounds are absent, immediate treatment by needle thoracostomy is indicated, even prior to obtaining a chest radiograph. The risk of pleural puncture can be limited by the use of ultrasound and assurance that the needle is not advanced too deeply into the neck. As lung volumes are high during positive pressure ventilation, it has also been suggested that the risk of pneumothorax can be lessened by needle advancement at the end exhalation. In all cases, a chest radiograph should be obtained following CVC placement.24
Complications associated with subsequent catheter use:
In addition to immediate complications from CVC placement, other adverse effects may be seen with their subsequent use. The most significant, related to both cost, duration of hospitalization, and patient morbidity, is the potential for infectious complications. Various factors have been shown, at least in the adult population, to increase the risk of infection including site of placement (femoral or internal jugular placement more than subclavian), use of the catheter for hemodialysis or parenteral nutrition (increased further when intralipids are administered), violations in sterile technique during placement, percutaneous versus tunneled, and non-use of antibiotic impregnated catheters.25-27 The most significant, modifiable factor remains strict attention to sterile technique during placement. A simple yet extremely effective checklist was developed and its efficacy documented by Dr. Peter Pronovost from the Johns Hopkins Hospital.28,29
Thrombotic complications may involve the superficial or deep venous systems. Superficial thrombotic complications due to peripheral catheters are generally self-limited once the catheter is removed without long-term implications. Thrombosis involving the deep veins can lead to pulmonary embolism. Risk factors for thrombotic complications related to a CVC include oncologic diseases, inherited pro-thrombotic states (Factor V Leiden), the presence of cyanotic congenital disease, administration of hypertonic solutions, and the use of a large catheter in relationship to the size of the vein. When clinical signs of venous obstruction such as limb swelling present, ultrasonography is useful to identify a clot and its extent. To date, there are limited data to help determine the optimal therapy for such patients, especially in infants and children. Current recommendations suggest removal of the CVC and instituting a course of anticoagulation for up to 6-8 weeks. In our practice, we generally remove the catheter as soon as it is feasible and initiate anticoagulation with a continuous infusion of heparin in the absence of contraindications to heparin therapy. If the CVC is still required, we may initiate therapy without CVC removal. Once the clinical situation is stable, anticoagulation therapy is switcher to intermittent therapy with subcutaneous low molecular weight heparin.
REFERENCES
- Morray JP. Anesthesia-related cardiac arrest in children. Anesth Clin North Amer. 2002 Mar;20(1):1-28. [PubMed]
- Morray JP, Geiduschek J, Ramamoorthy C, Haberkern CM, Hackel A, Caplan RA, et al. Anesthesia-related cardiac arrest in children: initial findings of the Pediatric Perioperative Cardiac Arrest (POCA) registry. Anesthesiology. 2000 Jul;93(1):6-14.[PubMed] [Free full text]
- Bhanaker SM, Ramamoorthy C, Geiduschek JM, Posner KL, Domino KB, Haberkern CM, et al. Anesthesia-related cardiac arrest in children: Update from the Pediatric Perioperative Cardiac Arrest Registry. Anesth Analg. 2007 Aug;105(2):344-50. [PubMed]
- Tiret L, Nivoche Y, Hatton F, Desmonts JM, Vourc’h G. Complications related to anesthesia in infants and children. A prospective survey of 40240 anesthetics. Br J Anaesth. 1988 Sep;61(3):263-9.[PubMed] [Free full text]
- Flick RP, Sprung J, Harrison TE, Gleich SJ, Schroeder RJ, Hanson AC, et al. Perioperative cardiac arrests in children between 1988 and 2005 at a tertiary referral center: a study of 92881 patients. Anesthesiology. 2007 Feb;106(2):226-237. [PubMed] [Free full text]
- Morgan CA, Webb RK, Cockings J, Williamson JA. The Australian Incident Monitoring Study. Cardiac arrest- an analysis of 2000 incident reports. Anaesth Intensive Care. 1993 Oct;21(5):626-637. [PubMed]
- Joseph G, Tobias JD. The use of intraosseous infusions in the operating room. J Clin Anesth. 2008 Sep;20(6):469-73. doi: 10.1016/j.jclinane. [PubMed]
- Tobias JD, Ross AK. Intraosseous infusions: A review for the anesthesiologist with a focus on pediatric use. Anesth Analg. 2010 Feb 1;110(2):391-401. [PubMed]
- Neuhaus D, Weiss M, Engelhardt T, et al. Semi-elective intraosseous infusion after failed intravenous access in pediatric anesthesia. Paediatr Anaesth 2010;20:168-71.
- Johnson EM, Saltzman DA, Suh G, Dahms RA, Leonard AS. Complications and risks of central venous catheter placement in children. Surgery. 1998 Nov;124(5):911-16. [PubMed]
- Karapinar B, Cura A. Complications of central venous catheterization in critically ill children. Pediatr Int. 2007 Oct;49(5):593-9. [PubMed]
- Casado-Flores J, Barja J, Martino R, Serrano A, Valdivelso A. Complications of central venous catheterization in critically ill children. Pediatr Crit Care Med. 2001 Jan;2(1):57-62. [PubMed]
- Chuan WX, Wei W, Yu L. A randomized-controlled study of ultrasound prelocation vs anatomical landmark-guided cannulation of the internal jugular vein in infants and children. Paediatr Anaesth. 2005;15:733-8.
- Trojanos CA, Kuwik RJ, Pasqual JR, Lim AJ, Odasso DP. Internal jugular vein and carotid artery anatomic relation as determined by ultrasonography. Anesthesiology 1996 Jul;85(1):43-8. [PubMed] [Free full text]
- P Souza Neto E , Grousson S, Duflo F, Tahon F, Motoleese C, Dialler F. Ultrasonographic anatomic variations of the major veins in paediatric patients. Br J Anaesth. 2014 May;112(5):879-84. doi: 10.1093/bja/aet482. [PubMed] [Free full text]
- Alderson PJ, Burrows FA, Stemp LI, Holtby HM. Use of ultrasound to evaluate internal jugular vein anatomy and to facilitate central venous cannulation in paediatric patients. Br J Anaesth. 1993 Feb;70(2):145-8. [PubMed] [Free full text]
- Chuan WX, Wei W, Yu L. A randomized-controlled study of ultrasound prelocation vs anatomical landmark-guided cannulation of the internal jugular vein in infants and children. Paediatr Anaesth. 2005 Sep;15(9):733-8. [PubMed]
- Verghese ST, McGill WA, Patel RI, Sell JE, Midgley FM, Ruttiman UE. Ultrasound-guided internal jugular venous cannulation in infants: A prospective comparison with the traditional palpation method. Anesthesiology. 1999 Jul;91(1):71-7. [PubMed] [Free full text]
- Verghese ST, McGill WA, Patel RI, Sell JE, Midgley FM, Ruttimann UE. Comparison of three techniques for internal jugular vein cannulation in infants. Paediatr Anaesth. 2000;10(5):505-11. [PubMed]
- Pirotte T, Veyckemans F. Ultrasound-guided subclavian vein cannulation in infants and children: A novel approach. Br J Anaesth. 2007 Apr;98(4):509-14. [PubMed] [Free full text]
- Iwashima S, Ishikawa T, Ohzeki T. Ultrasound-guided versus landmark-guided femoral vein access in pediatric cardiac catheterization. Pediatr Cardiol. 2008 Mar;29(2):339-42. [PubMed]
- Alten JA, Borasino S, Gurley WQ, Law MA, Toms R, Dabal RJ. Ultrasound-guided femoral vein catheterization in neonates with cardiac disease. Pediatr Crit Care Med. 2012 Nov;13(6):654-9. doi: 10.1097/PCC.0b013e318250af0c. [PubMed]
- Guilbert AS, Xavier L, Ammouche C, Desprez P, Astruc D, Diemunsch P, et al. Supraclavicular ultrasound-guided catheterization of the subclavian vein in pediatric and neonatal ICUs: a feasibility study. Pediatr Crit Care Med. 2013 May;14(4):351-5. doi: 10.1097/PCC.0b013e3182745489. [PubMed]
- McGee DC, Gould MK. Preventing complications of central venous catheterization. N Engl J Med. 2003 Mar;348(20):1123-33. [PubMed] [Free full text]
- Richet H, Hubert B, Nitemberg G, Andremont A, Buu-Hoi A, Ourbak P, et al. Prospective multicenter study of vascular-catheter-related complications and risk factors for positive central-catheter cultures in intensive care unit patients. J Clin Microbiol. 1990 Nov;28(11):2520-6. [PubMed] [Free full text]
- Merrer J, De Jonghe B, Golliot F, Lefrant JY, Raffy B, Barre E, et al. Complications of femoral and subclavian venous catheterization in critically ill patients: a randomized controlled trial. JAMA. 2001 Aug 8;286(6):700-7. [PubMed] [Free full text]
- Raad II, Hohn DC, Gilbreath BJ, Sulaiman N, Hill LA, Bruso PA, et al. Prevention of central venous catheter-related infections by using maximal sterile barrier precautions during insertion. Infect Control Hosp Epidemiol. 1994 Apr;15(4 Pt 1):231-5. [PubMed]
- Goeschel CA, Holzmueller CG, Cosgrove SE, Ristaino P, Pronovost PJ. Infection preventionist checklist to improve culture and reduce central line-associated bloodstream infections. Jt Comm J Qual Patient Saf. 2010 Dec;36(12):571-5. [PubMed]
- Rodriguez-Paz JM, Pronovost P. Prevention of catheter-related bloodstream infections. Adv Surg. 2008;42:229-48. [PubMed]
- Ullman AJ, Marsh N, Mihala G, Cooke M, Rickard CM. Complications of central venous access devices: A Systematic Review. Pediatrics. 2015 Nov;136:e1331-44. doi: 10.1542/peds.2015-1507. [PubMed] [Free full text]




 Facebook
Facebook Twitter
Twitter GooglePlus
GooglePlus Youtube
Youtube