Syed Muhammad Nadeem, FCPS1, Imran Khan Sandeelo, FCPS2
1Consultant Department of Anesthesiology & In-charge Surgical Intensive Care
Liaquat National Hospital & Medical College, Karachi (Pakistan)
2Senior Registrar, Department of Cardiology, Liaquat National Hospital & Medical College, Karachi (Pakistan)
Correspondence: Dr Syed Muhammad Nadeem, FCPS, Department of Anesthesiology & In-charge Surgical Intensive Care, Liaquat National Hospital National Stadium Road, Karachi 74800 (Pakistan); Phone: +92-21-34412536, : +92-21-34412539; E-mail: smnadeemlnh@gmail.com
ABSTRACT
Arrhythmias are common during the perioperative period. These abnormal rhythms can alter the hemodynamics by reducing cardiac output and may contribute to an increase in morbidity and mortality. Early diagnosis and identification of correctable causes is essential for appropriate management of arrhythmias. Some of these arrhythmias are benign requiring no intervention, while others may need urgent therapeutic intervention. This article focuses on the diagnosis and management of arrhythmias in the perioperative setting.
Key words: Rhythm disorders; Fibrillation; Heart block; Sinoatrial node; Bradycardia; Tachycardia.
Citation: Nadeem SM, Sandeelo IK. Peri-operative arrhythmias: diagnosis and management. Anaesth Pain & Intensive Care. 2016;20 Suppl 1:S64-S76
Received: 9 August 2016; Reviewed: 12 July, 16 August, 20 August 2016; Corrected: 4 August, 19 August, 30 August 2016; Accepted: 20 October 2016
INTRODUCTION
The reported incidence of perioperative arrhythmias vary according to the nature of surgery, underlying co-morbidities and type of monitoring (continuous vs non-continuous) employed. It can be as high as 90% in patients with underlying structural heart disease undergoing cardiothoracic surgery with continuous monitoring (pre, intra and postoperative).1,2 Majority of perioperative arrhythmias are supraventricular in origin with sinus tachycardia being the most common followed by atrial fibrillation as the most frequent in general surgical as well as cardio-thoracic surgery patients.3,4 After atrial fibrillation, ventricular premature complexes (VPC) is the next commonly observed arrhythmia.5 The arrhythmias can lead to an increase in perioperative morbidity and mortality. The tachyarrhythmias may cause hypotension by reducing cardiac output secondary to reduced diastolic filling time. The increase in heart rate increases myocardial oxygen demand. This increase in demand coupled with reduced supply due to hypotension may cause myocardial ischemia. Bradyarrhythmias may also cause a reduction in cardiac output in patients with a relatively fixed stroke volume. The loss of atrial contraction (atrial fibrillation) may reduce diastolic filling resulting in a marked increase in pulmonary venous pressures in patients with diastolic dysfunction.2 In adults, the most effective combination of heart rate and ventricular filling time is approximately 100-110 beats/min.6
Factors predisposing to perioperative arrhythmias
Perioperative arrhythmias occur in patients with underlying heart disease (ischemic or valvular heart disease, cardiomyopathy, heart failure), electrolyte or acid-base disorder (hypokalemia, hyperkalemia, hypomagnesemia, hypercalcemia, acidosis), endocrine disorder (thyrotoxicosis, pheochromocytoma), intracranial bleed (subarachnoid hemorrhage) or drug effects (digoxin, beta blockers, calcium channel blockers, theophylline, antidepressants).2 The intraoperative factors predisposing to arrhythmias include hypoxemia, hypercarbia, hypothermia, volatile anesthetics, inadequate depth of anesthesia, traction on extra-ocular muscles during squint surgery, traction on peritoneum, carotid sinus manipulation during neck dissection and mechanical stimulation of heart during chest tube, central venous or pulmonary artery catheter placement.7
THE CONDUCTION SYSTEM OF THE HEART
The primary pacemaker of the heart is the sinoatrial (SA) node. The SA node is located just beneath the epicardium in the upper part of the right atrium, at its junction with the superior vena cava. It is about 1-2 cm in length and extends along the crista terminalis.8 The pacemaker’s automaticity (ability to generate an impulse without being stimulated) is attributed to the spontaneous phase-4 diastolic depolarization of the cardiac action potential.8 This depolarization spreads from the right atrium through preferential pathways called the anterior, middle and posterior internodal tracts to the atrioventricular (AV) node located in the right atrium just above the septal leaflet of the tricuspid valve.9 The depolarization spreads between the two atria via another preferential pathway known as Bachmann’s bundle.10 The only route for impulse transmission from the atria to the ventricle is through the AV node or an abnormal accessory pathway (a strand of myocardium between the atria and the ventricles). The impulse is delayed in the AV node allowing both atria to empty out into ventricles. The impulse is then rapidly conducted down the bundle of His which divides at the upper part of the interventricular septum into right and left bundle branches.9 The right and the left bundle branches are located on the respective side of the interventricular septum. The left bundle subdivides into an anterior and a posterior fascicle. The left and right bundle branches carry the impulses down to the terminal Purkinje fibres at the apex of the ventricles: rapidly spreading the impulse to the entire ventricular myocardium from the apex of the heart towards its base. The intrinsic rate of impulse generation, and thus the ability to determine the heart rate, is fastest with the SA node followed by the AV node, bundle of His and slowest in the Purkinje fibres.
The electrocardiography diagnostic terms used in this article conform to the guidelines for the standardization and interpretation of the ECG by the American Heart Association, the American College of Cardiology and the Heart Rhythm Society.11
SINUS NODE RHYTHMS AND ARRHYTHMIAS
The rhythm is said to be sinus when it originates in the SA node. The sinus rhythm is characterized by presence of normal P waves (rounded in shape, 80-110 ms in duration) with a normal P-axis between +15º to +75º (positive P waves in leads I and II and negative P waves in aVR).10 The sinus P waves are biphasic in leads V1 and V2 but their initial deflection has to be positive.
Sinus tachycardia
The ECG shows sinus rhythm but with a heart rate greater than 100 beats/minute. Sinus tachycardia is the most common arrhythmia in the perioperative period4 and is commonly caused by increased sympathetic activity secondary to pain, anxiety, hypovolemia, fever, anemia, hypoxia, hypercarbia, low cardiac output states,6 sepsis and thyrotoxicosis. It may also be secondary to drugs like atropine, epinephrine, dopamine, dobutamine, ephedrine, aminophylline and salbutamol. Management involves correcting the underlying cause. Sinus tachycardia is generally benign but in patients with ischemic heart disease (IHD) myocardial ischemia may be precipitated due to increased myocardial oxygen demand induced by increased heart rate. In such patients, beta blockers like metoprolol (2.5-5 mg IV repeated every two minutes if required, to a total of 15 mg) can be used to slow the heart rate.4,6
Sinus bradycardia
The ECG shows characteristics of sinus rhythm but with a heart rate of less than 60 beats/min. The bradycardia may cause a drop in cardiac output and hypotension. Physiological sinus bradycardia is commonly seen by in young healthy individuals, especially trained athletes. Pathological causes of sinus bradycardia include increased vagal tone pain, nausea, vasovagal and carotid sinus syndromes,12 hypoxemia, hypothermia, hypothyroidism, raised intracranial pressure and drugs like beta blockers, calcium channel blockers, amiodarone, digoxin, opioids, neostigmine, lithium and amitriptyline.13 Bradycardia without hypotension does not require any treatment. When associated with hypotension or low cardiac output, the heart rate may be increased with antimuscarinic or beta agonist drugs:
- atropine 0.5 mg IV boluses to a total of 3 mg or glycopyrrolate 0.1 mg IV bolus repeated every 2-3 min if required.14
- adrenaline or isoprenaline infusion at 2-10 mcg/min titrated to response.15
- Dopamine at 2-10 mcg/kg/min titrated to response.15
Concurrently reversible causes of bradycardia are addressed. External trans-thoracic, esophageal or temporary transvenous pacing may be needed if there is no response to the above.
Sinus arrhythmia
The ECG shows characteristics of sinus rhythm but with variable P-P interval (the duration between two successive P waves), the variability exceeding 160 ms. This is attributable to the respiratory dependent changes in heart rate, the rate increases during inspiration and decreases during expiration.16 This rhythm is commonly seen in young healthy individuals and does not require any treatment. Intraoperatively, sinus arrhythmia has been suggested as a sign of light anesthesia.17
Sinoatrial (SA) block and sinus arrest
These rhythms may be seen with myocardial ischemia and infarction especially inferior wall myocardial infarction (MI), myocarditis, pericarditis, degenerative changes in SA node with aging and infiltration (amyloidosis, sarcoidosis, hemochromatosis) involving the SA node. The rhythms may also be drug induced secondary to digoxin, beta blockers, calcium channel blockers, amiodarone or lithium therapy. Intraoperatively, they may be due to increased vagal tone from surgical stimulation or pain.
The SA node comprises of two types of specialized cells, the pacemaker cells (P-cells) located in the centre which initiate depolarization and the transitional cells (T-cells) located peripherally that transmit the depolarization from the P-cells to the right atrial myocardium. 18 In SA blocks, depolarizations are initiated in the P-cells but intermittently they fail to be conducted to the right atrium resulting in a pause on ECG with no P-QRS complex. The sinus rhythm usually resumes after the pause. The SA block is of two types:
SA block type I: There is progressive delay in conduction of impulse from P-cells to the atrial myocardium ending with failure of conduction giving a pause on ECG. It appears as irregular rhythm with progressive shortening of P-P interval followed by a pause which is less than twice the preceding sinus P-P interval. The cycle then repeats itself giving a visual impression of ‘group beating’ (clusters of PQRS complexes separated by pauses).10
SA block type II: The P-P interval remains constant during sinus rhythm until a P-QRS complex drops abruptly producing a pause on ECG which is a near exact multiple (≥ 2) of preceding P-P interval10 (Figure 1).
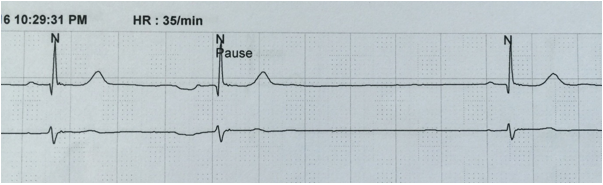
Figure 1: Sinoatrial block. After the first two complexes there is a pause which is exactly twice the preceding P-P interval. (Obtained from Holter monitoring)
The patients with asymptomatic sinoatrial blocks usually do not require any treatment.
Sinus arrest (sinus pause)
The P-cells fail to initiate depolarization producing a pause on ECG which can last from greater than 2 seconds to several minutes. The pause is not a multiple of preceding P-P interval10 (Figure 2). The pause is usually interrupted by an escape rhythm taking over as the pacemaker. If the pause is prolonged, syncope occurs. The initial management is the same as that described above for sinus bradycardia, but these patients may need temporary transvenous pacing and have to be evaluated by a cardiologist for permanent pacemaker insertion.
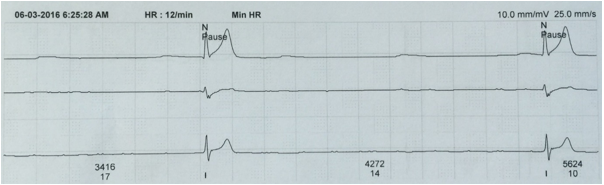
Figure 2: Sinus arrest. After the first complex a sinus pause lasting > 3 sec is seen. (Obtained from Holter monitoring)
SUPRAVENTRICULAR ARRHYTHMIAS
Atrial premature complexes (APCs)
Atrial premature complexes are commonly seen and are usually benign and asymptomatic. They may occur in normal subjects with anxiety, excessive caffeine or alcohol intake. The impulse can originate from an ectopic focus in either atrium. APCs are best seen in leads II and V1. As the name suggests, an APC is premature occurring earlier than the next expected sinus P wave (the distance between the APC and the preceding P is less than the sinus P-P interval). The morphology of the P-wave of APC differs from the sinus P-wave. Since they occur early, they may find the distal conducting tissue in a refractory state. Thus, they may be conducted after a delay resulting in a longer PR interval or may fail to get conducted (non-conducted APC) in which case no QRS complex follows. The P-wave of APC may be superimposed on and distort the shape of the preceding T-wave (Figure 3). The QRS complex after the APC resembles the sinus rhythm QRS. The most common cause of pauses in sinus rhythm are non-conducted APCs.19 Frequently occurring APCs have the potential to give rise to an atrial tachycardia. Generally, APCs do not require treatment, if needed, beta blockers may be employed.
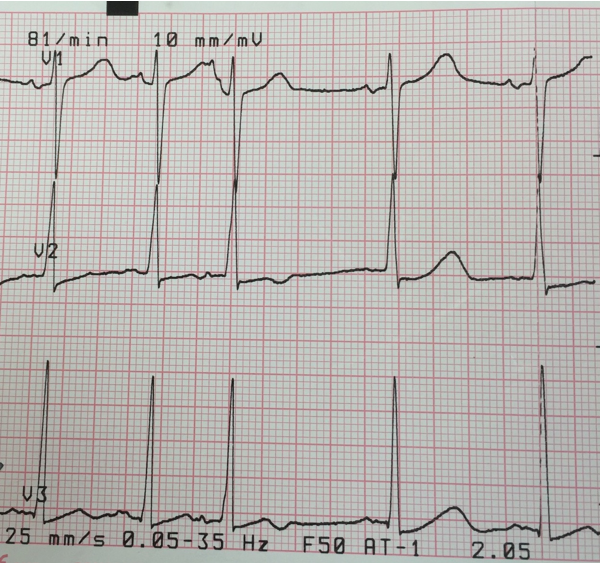 Figure 3: Atrial premature contraction. The fourth complex is an APC. The P-wave is seen distorting the shape of the preceding T-wave.
Figure 3: Atrial premature contraction. The fourth complex is an APC. The P-wave is seen distorting the shape of the preceding T-wave.
Junctional premature complexes (JPCs) and junctional rhythm
JPCs are premature in timing, occurring before the next expected sinus beat. They originate at the AV junction which includes AV node, bundle of His and atrial wall and septum adjacent to the annulus of AV valves.20 The P-waves are conducted retrogradely to the atria resulting in inverted P waves in inferior leads (II, III, aVF). The P-waves may be absent (buried in QRS), precede or follow the QRS complex.21 Since the impulse is transmitted to the ventricles normally, the QRS complex that follows is narrow resembling the sinus rhythm QRS. The inherent pacing rate of AV junction is around 40-60 beats/minute, a junctional rhythm with a rate faster than 60 but below 100 beats/minute is called accelerated junctional rhythm (Figure 4). The same rhythm occurring with a rate exceeding 100 beats/min is called junctional tachycardia. The junctional rhythm usually does not require any treatment but in symptomatic patients beta blockers can be used to suppress enhanced automaticity of the junctional pacemaker.1
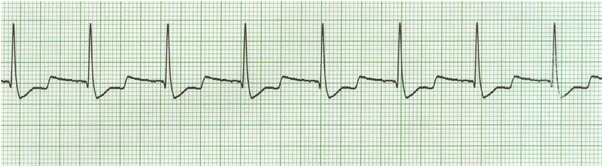
Figure 4: Accelerated junctional rhythm. Inverted P-waves following QRS complexes, distorting the T-wave. The rate is > 60 but < 100 beats/min
Ectopic atrial tachycardias
They may be unifocal or multifocal and can originate from either atrium. Unifocal ectopic atrial tachycardia is more commonly seen in children. Ectopic atrial tachycardia is frequently observed in critically ill patients.19 It is characterized by discrete P waves of similar morphology with isoelectric segment between P-waves. The isoelectric segment helps in differentiating it from atrial flutter.22 The P-wave precedes the QRS complex and the rate is 120-250 beats/min and the rhythm is regular.22 This arrhythmia also occurs in digoxin toxicity and in patients with past history of atrial surgery (congenital heart diseases).22
Multifocal atrial tachycardia is typically seen in patients with severe pulmonary disease, most commonly chronic obstructive pulmonary disease (COPD).22,23,24,25 It is recognized by the presence of three or more different P-wave morphologies with variable PR-intervals. The rate exceeds 100 beats/min and the rhythm is always irregular22 (Figure 5). It is frequently confused with atrial fibrillation. The presence of P-waves and isoelectric segment between P-waves differentiates it from atrial fibrillation where the baseline is undulating.22
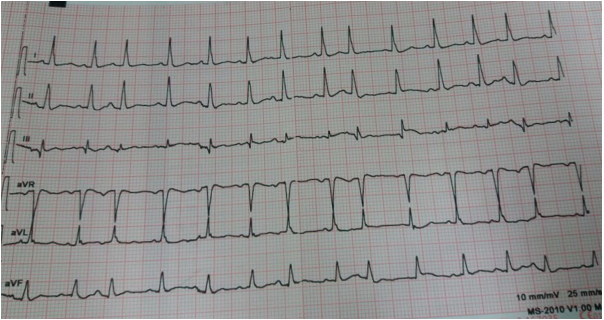
Figure 5: Multifocal ectopic atrial tachycardia. Narrow QRS complexes with an irregular rhythm. Three different P-wave morphologies can be seen.
Methods for unmasking underlying atrial rhythm:19
a) Vagal maneuvers (carotid sinus massage for 5 sec or Valsalva maneuver for 15 sec) may transiently reveal the underlying atrial rhythm by slowing AV conduction.
b) Intravenous administration of adenosine produces a transient AV block revealing the underlying atrial rhythm. It can also terminate the arrhythmia.
c) Using a modified ECG lead that accentuates P waves e.g. S5 lead (negative electrode over upper manubrium, positive electrode over middle of right sternal border).
During vagal maneuvers and adenosine injection, a continuous 12-lead ECG is recorded to look for the underlying rhythm after the AV conduction has been slowed by the above methods.23
Synchronized direct current (DC) cardioversion is used for hemodynamically unstable patients (hypotension, dyspnea, chest pain, altered mentation). In hemodynamically stable patients, AV nodal blocking agents (beta blockers, calcium channel blockers) can terminate the arrhythmia or control the ventricular rate. Commonly, calcium channel blockers (verapamil) are used since beta blockers may be contraindicated by the underlying severe pulmonary disease.22 Simultaneously underlying pulmonary disease and electrolyte abnormalities are optimized.22 Amiodarone, sotalol and flecainide can convert the arrhythmia to sinus rhythm.24
Supraventricular tachycardias (SVTs)
AV nodal reentrant tachycardia (AVNRT) and AV reentrant tachycardia (AVRT) are commonly referred to as paroxysmal SVTs because of their abrupt onset.23 These occur in young subjects with no underlying structural heart disease. AVNRT is the most common paroxysmal SVT and is twice as common in females than males.2,23 Atrial tachycardia and junctional tachycardia should also be included in the differential diagnosis.26 SVTs are characterized by a narrow-QRS, regular tachycardia at 140-250/min usually with no visible P-waves.
AVNRT: Two distinct pathways exist within the AV node: one with short refractory period and slow conduction and the other with long refractory period and fast conduction. AVNRT is usually initiated by an APC. In the typical more common type, slow-fast AVNRT the APC finds the fast pathway refractory and is conducted down the slow pathway to the ventricle and then up through the fast pathway thus creating a reentry mechanism and resultant tachycardia. Since the reentry circuit is short (within AV node), the P-waves are superimposed over the terminal QRS, giving pseudo ‘S‘ waves (inverted P-waves) in inferior leads and pseudo R-prime waves (positive P-waves) in V1 resembling RBBB.23 (Figure 6) Presence of a notch in aVL is also a sensitive and specific criteria for AVNRT.27 The R-P interval is < 70 ms.22 A regular, narrow-QRS tachycardia with no visible P-waves is most likely AVNRT.22
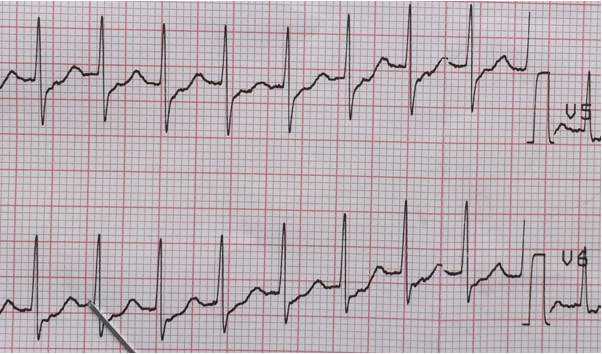
Figure 6: Supraventricular tachycardia. Positive P-waves are clearly seen preceding the T-waves and superimposed over ST-segments in lead V5
AVRT: here the reentry circuit is longer (a separate accessory pathway exists), the impulse takes longer time to reenter atria, the inverted P-waves are superimposed on T-waves giving a pointed shape to T-waves.28 It is usually initiated by a ventricular premature contraction (VPC). The QRS is usually narrow but may appear wide due to the presence of ‘delta’ wave (slurred upstroke of R-wave). The RP interval is usually > 70 ms.
Management of paroxysmal SVTs: the acute management of AVNRT and AVRT is the same.
Unstable rhythm requires immediate synchronized DC cardioversion.
In stable rhythms the following can be used: (Table 1)
- Vagal maneuvers may terminate the tachy-arrhythmia.
- Adenosine: It can terminate the arrhythmia and has a quick onset and an extremely short half-life (10 sec). Adenosine can cause dyspnea, chest tightness, and facial-flushing and should be avoided in asthmatics22 and WPW syndrome.
- Non-dihydropyridine calcium channel blockers (verapamil, diltiazem)24
- Beta-1 blockers (metoprolol, esmolol)23
Atrial fibrillation (AF)
This is one of the most frequent arrhythmia during the postoperative period, especially after cardiothoracic surgery. The peak incidence of postoperative atrial fibrillation after open heart surgery is noted on day three.29 The risk of postoperative atrial fibrillation increases nearly 3-fold when the preoperative ECG shows P-wave duration > 110 ms.30 A new onset atrial fibrillation in patients with severe sepsis increases the risk of stroke and in-hospital mortality.31 Atrial fibrillation can occur in patients with heart failure, ischemic heart disease, mitral valve disease, cardiomyopathy, electrolyte abnormalities (potassium, magnesium, calcium) congenital heart diseases like atrial septal defect or past surgery involving the right atrium and in subjects with obstructive sleep apnea.
The ECG characteristics of atrial fibrillation are absent P-waves, narrow QRS (< 120 ms) with an irregular rhythm (varying R-R intervals) (Figure 7).
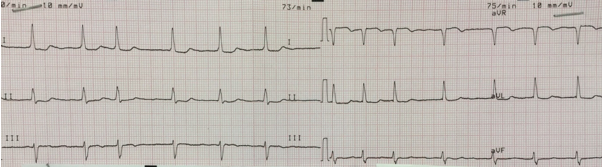
Figure 7: Atrial fibrillation. Narrow QRS complexes with an irregular rhythm. No visible P-waves.
Atrial flutter
Atrial flutter is less common than atrial fibrillation but share a common etiology and initial treatment.23 It is caused by a re-entry circuit in the right atrium. Classical atrial flutter is characterized by saw-tooth flutter waves preceding narrow QRS complexes and a regular rhythm. The flutter waves are best seen in inferior leads (II, III, aVF) or V1. Commonly, the atrial rate is 300 beats/min (250-350/min) with an AV conduction ratio of 2:1 giving a ventricular rate of 150 beats/min. In the common form, counter-clockwise atrial flutter, the re-entry wave travels up the atrial septum and down the lateral wall of the right atrium. This produces negative saw-tooth flutter waves in inferior leads and positive waves in V1 (Figure 8). In the uncommon clockwise atrial flutter, the impulse travels in a reverse direction giving positive saw-tooth waves in inferior leads and negative in V1. Atrial flutter should always be considered in a narrow QRS tachycardia with a rate of 150 beats/min.
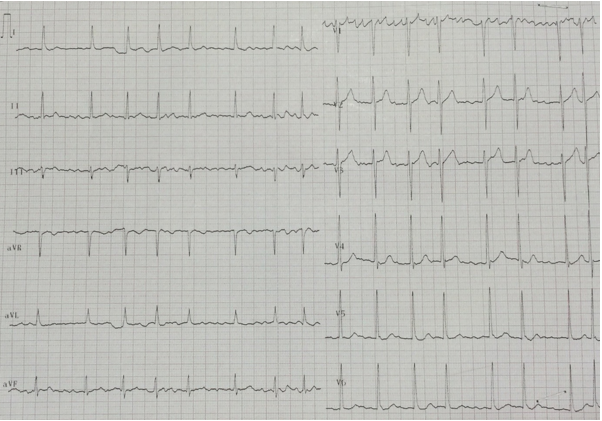
Figure 8: Atrial flutter. Narrow QRS complexes with preceding negative saw-tooth waves in inferior leads and positive in V1.
Differential diagnosis of a narrow QRS complex tachycardia:
- Atrial fibrillation
- Atrial flutter
- Unifocal ectopic atrial tachycardia
- Multifocal ectopic atrial tachycardia
- Paroxysmal SVTs (AVNRT, AVRT)32
- Sinus tachycardia33,34
In difficult but hemodynamically stable patients, use of vagal maneuvers or adenosine IV, as discussed above, may transiently slow AV conduction revealing the underlying rhythm.
Management of atrial fibrillation and flutter:
Correct underlying cause simultaneously with other treatment. Hemodynamically unstable patients with new-onset AF require immediate R-wave synchronized DC cardioversion starting at 100-200 J.23,35,36 If initial cardioversion fails or AF recurs, synchronized DC cardioversion can be repeated after starting rate and rhythm control therapy.35 In hemodynamically stable patients, the management involves ventricular rate control, conversion to sinus rhythm and long-term anticoagulation for prevention of embolic stroke.37 The preferred drugs for AF prophylaxis after cardiac surgery are Vaughan-Williams class II antiarrhythmics (carvedilol, atenolol, bisoprolol)38
Ventricular rate control: IV beta-1 blockers, non-dihydropyridine calcium channel blockers and digoxin39 are commonly employed but amiodarone may also be effective, particularly in patients with reduced LV function.23 (Table 1) Beta blockers and calcium channel blockers can cause hypotension, digoxin is less likely to cause hypotension but it’s rate controlling effect is weaker.39
- Metoprolol (beta blocker): Onset in 5 min.
- Esmolol (short acting beta blocker): Onset in 5 min. Hypotension or heart block can develop.
- Propranolol (non-selective beta blocker): Onset time 5 min.23
- Verapamil37: Onset occurs in 3-5 min.
- Diltiazem: Onset time is 2-7 min.
Beta blockers and calcium channel blockers can cause hypotension because of their negative inotropic effect. Avoid non-dihydropyridine calcium channel blockers in heart failure.37 Hypotension during beta blocker or calcium channel blocker therapy can be managed with IV phenylephrine infusion.40
- Digoxin: The loading dose is reduced to 50% in presence of renal impairment or age > 70 yr. The onset is slow taking more than 1 hr with a peak effect at 6 hr.37 Hypokalemia and hypercalcemia may enhance digoxin effect. Hypomagnesemia increases dose requirements of digoxin.41 These electrolyte abnormalities should be corrected before administering digoxin.
- Amiodarone: It’s rate controlling effect is rather slow (1-2 hr) but is useful when other therapies are ineffective or there is underlying LV dysfunction.23,37
The drugs which slow AV-conduction without increasing the refractory period of accessory pathway (non-dihydropyridine calcium channel blockers, digoxin and amiodarone) can accelerate the ventricular rate leading to ventricular fibrillation and should be avoided in AF with accessory pathway (WPW syndrome). Procainamide with continuous QRS monitoring for evidence of QT prolongation, ibutilide and flecainide are recommended in presence of an accessory pathway in hemodynamically stable patients.2,37 Sotalol and ibutilide can precipitate torsades de pointes if the QTc exceeds 500 ms.42 In patients with underlying heart failure with AF, beta blockers and digoxin are the first line drugs followed by amiodarone for ventricular rate control.43
Table 1: Common anti-arrhythmic drugs
| Drug | Dose | Repeat dose / infusion |
| Adenosine | 6 mg very rapid IV push | 12 mg after 2 min then another 12 mg if required |
| Amiodarone | 150 mg over 10 min, repeat 150 mg over 20 min | 900-1200 mg over 24 h |
| Digoxin | 10-15 mcg/kg lean body weight. Give 50% of the calculated dose over 20 min; 25% of the dose after 6 h, remaining 25% after another 6 h | 125-250 mcg/day |
| Diltiazem | 10-20 mg over 2 min. May be repeated after 15-30 min | 5-15 mg/h infusion |
| Esmolol | 500 mcg/kg over 1 min. May be repeated once | 50-200 mcg/kg/min infusion |
| Flecainide | 2 mg/kg with a max of 150 mg over 30 min. May repeat 1.5 mg/kg over 1 h | 100-250 mcg/kg/h infusion |
| Ibutilide | 1 mg over 10 min | Repeat once if required |
| Lignocaine | 1 mg/kg IV push. May repeat 0.5 mg/kg boluses to a total of 3 mg/kg | 1-4 mg/min infusion |
| Metoprolol | 2.5-5 mg over 2 min | 2.5-5 mg q 5 min to a total of 15 mg |
| Propranolol | 1 mg over 1 min | 1 mg q 2 min to a total of 10 mg |
| Procainamide | 15 mg/kg at 20 mg/min infusion | 1-4 mg/min infusion |
| Sotalol | 20-60 mg slow IV | |
| Verapamil | 5-10 mg over 2 min. May be repeated once after 15-30 min | 5-10 mg q 4-6 h or 5 mcg/kg/min infusion |
Over-drive pacing can be employed for atrial flutter, especially if pacing wire is in place (post-cardiac surgery).23
Patients with atrial flutter or atrial fibrillation need cardiology referral for conversion to sinus rhythm or assess the need for catheter ablation therapy.
VENTRICULAR ARRHYTHMIAS
The ventricular arrhythmias originate from sites distal to the bundle of His and are characterized by wide-QRS complexes greater than 110 ms.44
Ventricular premature complexes (VPC)
Since the impulse originates from an ectopic focus located in the ventricular myocardium no P-wave precedes the QRS of VPC. Moreover, the ectopic impulse is not conducted via the fast conducting normal pathway producing a wide (> 120 ms), abnormal shaped QRS complex. The complex is premature in timing, occurring before the next expected sinus beat. The T-wave is opposite in direction to the QRS (Figure 9). If the VPCs are similar in shape, they are arising from the same ventricular focus (unifocal) but VPCs of varying morphology, indicates a multifocal origin.
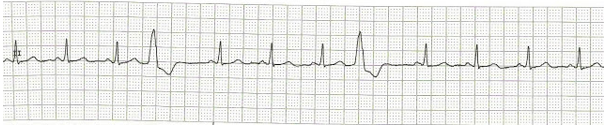
Figure 9: Ventricular premature complexes. The fourth and eighth complexes are VPCs. No preceding P-wave, wide abnormal shaped QRS occurring before the next expected sinus beat.
Two VPCs occurring in succession is referred to as a ‘couplet’.45 If the VPCs are frequent (> 30/hour), multifocal, occurring as couplets or falls on the preceding T wave (R-on-T phenomenon), the rhythm can degenerate into a sustained ventricular tachycardia.46 The management involves looking for and correcting any inciting cause like myocardial ischemia, hypoxemia, hypercarbia, electrolyte and acid-base abnormalities, pro-arrhythmic drugs and mechanical cardiac stimulation from central venous or pulmonary artery catheters.
Ventricular tachycardia (VT)
Ventricular tachycardia is defined as three or more ventricular premature complexes with the rate exceeding 100 beats/min.45 Classic VT is characterized by wide-QRS complexes
(> 120 ms) with a regular rhythm.
A VT lasting less than 30 sec is referred to as unsustained VT (Figure 10). This rhythm, generally, does not require any prophylactic anti-arrhythmic therapy.47 A ventricular tachycardia with single morphology QRS complexes at a rate of less than 120 beats/min is referred to as accelerated idioventricular rhythm. Like unsustained VT, this rhythm usually does not require treatment.48 This rhythm is almost always transient, alternating with sinus rhythm.45
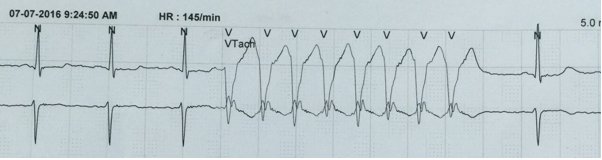
Figure 10: Unsustained VT. After three complexes a burst of VT is seen lasting < 30 sec.
Polymorphic ventricular tachycardia and Torsades de pointes
A specific type of ventricular tachycardia where the QRS complexes change their direction around the baseline, with runs of positive QRS complexes of varying morphology alternating with runs of negative QRS complexes of different morphology is referred to as torsades de pointes if the QT interval is prolonged between episodes of arrhythmia.2,49 The normal QT-interval corrected for heart rate , QTc is < 420 ms in males and less than 440 ms in females. The same rhythm with wide-QRS complexes of different morphology occurring in patients with normal QT-interval is referred to as polymorphic ventricular tachycardia.33
Differential diagnosis of a wide-QRS tachycardia:
A wide-QRS tachycardia resembling ventricular tachycardia can be of supraventricular origin as seen in SVTs with a bundle branch block, aberrant conduction (rate-related bundle branch block) or Wolff-Parkinson-White (WPW) syndrome.50 The ECG features suggesting a ventricular tachycardia are:
- wide-QRS complexes greater than 140 ms in duration.50
- marked QRS axis deviation45
- no RS complex in any precordial leads.46
- if RS complexes are visible, the RS duration (from beginning of QRS to the nadir of S wave) greater than 100 ms in any precordial lead45,46
- Concordance: all the QRS complexes in precordial leads are either positive (positive concordance) or negative (negative concordance).50
- Atrioventricular dissociation as evidenced by presence of dissociated P-waves. This may not be evident on rhythm strip and may require scrutiny of 12-lead ECG.48
- presence of a ‘capture beat’: a QRS complex of normal shape and duration produced by ventricular activation from an atrial focus. Presence of a single capture beat is sufficient to confirm ventricular tachycardia (Figure 11).
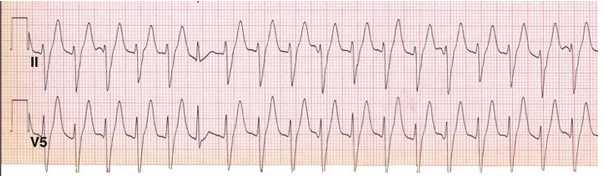
Figure 11: Ventricular tachycardia.
- presence of a ‘fusion beat’ (a QRS morphology that is intermediate between a normal QRS complex and a VPC) produced by simultaneous activation of ventricle from an atrial and a ventricular focus.48,51
- If the diagnosis remains uncertain, the broad complex tachycardia should be taken as VT.47
Common causes of VT include acute myocardial infarction or ischemia, cardiomyopathy (dilated, hypertrophic types), valvular heart diseases, mitral valve prolapse, myocarditis. Torsades de pointes may be secondary to hereditary (Romano-Ward syndrome) or acquired prolongation of QT-interval. The acquired QT-prolongation may be due to myocardial ischemia, electrolyte abnormalities (hypokalemia, hypomagnesemia, hypocalcemia), subarachnoid hemorrhage and several drugs like antiarrhythmics (quinidine, procainamide, disopyramide, sotalol, amiodarone, ibutilide), psychotropics (phenothiazines, tricyclic antidepressants, lithium, droperidol, haloperidol), antihistamines (terfenadine, astemizole), antimicrobials (clarithromycin, erythromycin, voriconazole, chloroquine) and domperidone.52
A ventricular tachycardia can occur with palpable major pulse or be pulseless (cardiac arrest). For pulseless ventricular tachycardia (pVT), the algorithm for pVT/VF in the recently updated guidelines of American Heart Association (AHA) for adult advanced cardiovascular life support (ACLS) should be followed.53
Ventricular tachycardia with pulse:
If VT is unstable, immediate DC cardioversion is required. Cardioversion is also indicated if anti-arrhythmic drugs fail to revert the VT or produce hypotension.
Stable VT:
If the left ventricular function is good, following drugs may be used: (Table 1)
- IV Amiodarone54
- Lignocaine: If a bolus is ineffective, the infusion is likely to fail as well.42
- Procainamide IV
- A refractory wide-QRS tachycardia may respond to sotalol.6,47
In patients with low ejection fraction (<40%), only amiodarone or lignocaine is recommended.47 Simultaneously, correction of underlying electrolyte abnormalities and ischemia is implemented. Target a serum potassium level of 4.5-5 mmol/L.47
For torsades de pointes, stop offending drugs and correct electrolyte abnormalities (hypokalemia, hypomagnesemia, hypocalcemia). Intravenous magnesium sulphate, 1-2 g over 1-2 minutes is recommended.47 Isoprenaline can be used as a temporary treatment in recurrent torsades de pointes who do not have a congenital prolongation of QT interval. Serum potassium should be maintained between 4.5-5 mmol/L.
Over-drive pacing is an option in torsades de pointes and may also be indicated in other VTs when drug therapy fails or VT keeps recurring after repeated DC cardioversion or if temporary pacing wire is already in place.39 Patients with recurrent VT need to be evaluated by a cardiologist for catheter ablation therapy or implantable cardioverter-defibrillator insertion.
Ventricular fibrillation (VF)
It is characterized by an abrupt onset of chaotic, irregular waveforms of different shape, duration and amplitude with no recognizable QRS-T waves due to the absence of organized ventricular depolarizations. Clinically, this is associated with an abrupt loss of effective forward blood flow resulting in absent major pulses with loss of consciousness within seconds.46,47 The recently updated ACLS protocol of AHA for pVT/VF should be initiated immediately.53
Asystole
There is complete absence of cardiac electrical activity producing an isoelectric, flat baseline on the ECG. This rhythm requires immediate initiation of ACLS protocol for asystole, as recommended by AHA.53 The survival outcome is better with VT/VF than for asystole.47
AV CONDUCTION DISORDERS
These may be secondary to varied causes like myocardial ischemia, drugs (digoxin, beta blockers, calcium channel blockers), infiltration, inflammation, infections or fibrosis involving the conduction system and post-valvular cardiac surgery.
Prolonged PR-interval: It is characterized by a PR-interval exceeding 200 ms. Each P-wave is followed by a QRS (P:QRS ratio 1:1). It is benign and asymptomatic and does not require any treatment or workup.55
Second-degree AV block: some atrial impulses fail to be conducted to the ventricles. It is of two types, Mobitz type I and type II.
Mobitz type I (Wenckebach): characterized by a progressive prolongation of PR-interval until a P-wave is not followed by a QRS producing a pause on the ECG. As the PR-interval lengthens, the R-R interval shortens. The cycle then repeats itself. This gives a characteristic visual impression of ‘group beating’, generally with clusters of 2, 3 or 4 QRS complexes separated by pauses (Figure 12). The conduction disturbance mostly resides in the AV node giving narrow QRS complexes. If the PR-interval after a pause is shorter than the PR-interval before the pause, the block is Mobitz type I second-degree block. This block is frequently seen on ambulatory ECG monitoring during nocturnal sleep and generally is harmless not requiring pacing.15
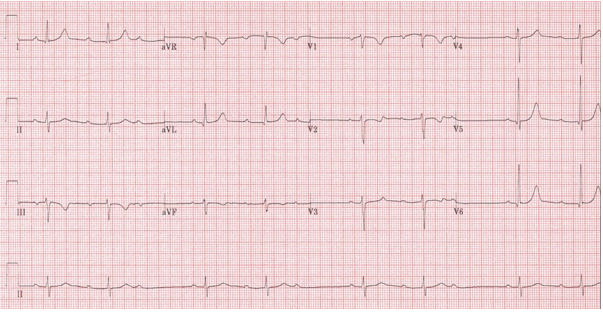
Figure 12: Second-degree AV block, Mobitz type-I (Wenckebach). Groups of two QRS complexes separated by pauses (‘group beating’) seen in long lead-II. The PR-interval following the pause is much shorter than the one preceding the pause.
Mobitz type II: the PR-intervals remain constant. There is an abrupt failure of a P-wave to get conducted resulting in a P-wave with no following QRS. The P:QRS ratio is > 1:1 (more P-waves than QRS complexes). When the P:QRS ratio is 3:1 or more, it is referred to as high-grade second-degree AV block56 (Figure 13). This block is almost always due to an infra-nodal conduction disturbance giving wide-QRS complexes.55 It is seen after anterior wall myocardial infarction. Mobitz type II block, particularly the high-grade block, is an inherently unstable rhythm and can progress to complete AV block.55
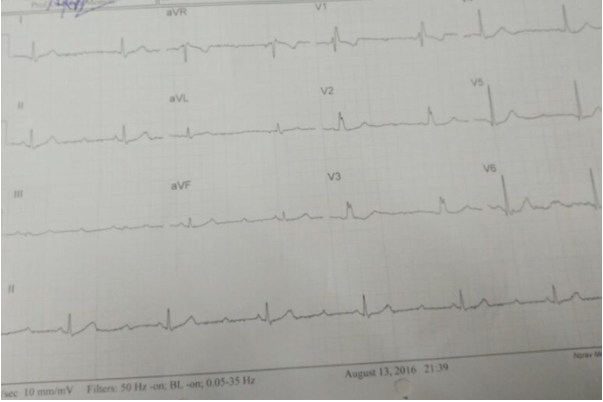
Figure 13: Second-degree AV block, Mobitz type-II
Complete AV block (third-degree): none of the P-wave is conducted to the ventricles so that the atria and ventricles function independently (AV dissociation). The P-P and R-R intervals are constant. The atrial rate is greater than the ventricular rate. On the ECG, the P-waves bear no relationship to QRS complexes and, typically, can be seen ‘marching through’ the QRS complexes because of the higher atrial rate (Figure 14). The PR-intervals are irregular. The escape rhythm may be junctional (narrow-QRS with a rate of 40-60 beat/min) or ventricular (wide-QRS with a rate of less than 40 beats/min), in which case, the patient is generally symptomatic with dyspnea on exertion, low cardiac output and syncope.55 Complete AV block may be congenital where the escape rhythm is junctional giving adequate ventricular rate. It is more common in females. An acquired complete heart block with broad QRS complexes nearly always requires pacing.15 Acquired complete AV block can occur after inferior wall MI, where it is usually temporary, or extensive anterior wall MI where it usually requires a permanent pacemaker.
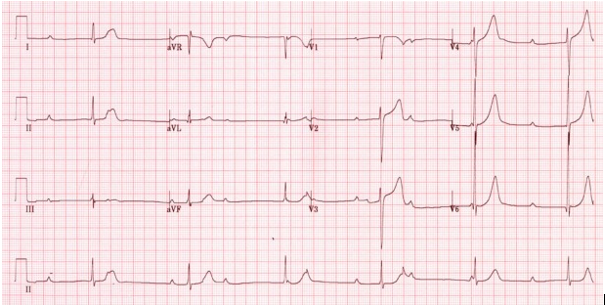
Figure 14. Complete heart block (third-degree AV block). P-waves can be seen ‘marching through’ QRS complexes. A P-wave is superimposed on the T-wave of first, third and fourth complexes.
Temporary transvenous pacing may be required in Mobitz type-II second-degree AV block, complete AV block, recurrent sinus pauses > 3 sec unresponsive to drugs, right bundle branch block with left anterior or left posterior fascicle block after MI, asystole, symptomatic bradycardia unresponsive to drugs and bradycardia with hyperkalemia.55 Permanent pacemaker is usually required for complete AV block with escape rhythm rate < 40 beats/min, high-grade second-degree AV block and sinus pauses lasting more than 3 seconds.57
Conflict of interest: None declared by the authors.
Authors’ Contribution: SMN: performed the literature search, conceived and drafted the manuscript. Both authors read and approved the final manuscript; IKS: reviewed and discussed the manuscript, provided most of the figures. Both authors read and approved the final manuscript
REFERENCES
- Atlee JL. Perioperative cardiac dysrhythmias: diagnosis and management. Anesthesiology. 1997;86:1397-1424. [PubMed] [Free full text]
- Hollenberg SM, Dellinger RP. Noncardiac surgery: postoperative arrhythmias. Crit Care Med 2000;28:N145-N150 . [PubMed]
- Walsh SR, Tang T, Wijewardena C, Yarham SI, Boyle JR, Gaunt ME. Postoperative arrhythmias in general surgical patients. Ann R Coll Surg Engl 2007;89:91-95. [PubMed] [Free full text]
- Bittner EA, Greco L, George E. Postoperative complications. In: Longnecker DE, Brown DL, Newman MF, Zapol WM. Editors. Anesthesiology. New York. McGraw-Hill Medical 2010. p 1284-1296 .
- Batra GS, Molyneux J, Scott NA. Colorectal patients and cardiac arrhythmias detected on the surgical high dependency unit. Ann R Coll Surg Engl 2001;83:174-176 . [PubMed] [Free full text]
- Postoperative complications involving the heart and lungs. In: Lemmer JH, Richenbacher WE, Vlahakes GJ. Editors. Handbook of patient care in cardiac surgery. Philadelphia. Lippincott Williams & Wilkins 2003. p 116-167 .
- Thompson A, Balser JR. Perioperative cardiac arrhythmias. Br J Anaesth 2004;93:86-94 . [PubMed] [Free full text]
- Park DS, Fishman GI. The cardiac conduction system. Circulation. 2011;123:904-915. [PubMed] [Free full text] doi: 10.1161/CIRCULATIONAHA.110.942284.
- Ramrakha P, Hill J. Arrhythmias. Oxford handbook of cardiology. Oxford University Press; 2012. 477-539 . [Free full text]
- Sinus rhythm and its discontents. In: Abedin Z, Conner R. Editors. ECG interpretation: the self-assessment approach. Massachusetts. Blackwell publishing 2008. p 59-78 .
- Mason JW, Hancock EW, Gettes LS. Recommendations for the standardization and interpretation of the electrocardiogram, part II: Electrocardiography diagnostic statement list: a scientific statement from the American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology; the American College of Cardiology Foundation; and the Heart Rhythm Society. Circulation. 2007;115:1325-1332. [PubMed] [Free full text]
doi: 10.1161/CIRCULATIONAHA.106.180201
- Arthur W, Kaye GC. The pathophysiology of common causes of syncope. Postrad Med J 2000;76:750-753 . [PubMed] [Free full text]
- Tomaselli GF. The bradyarrhythmias. In: Loscalzo J Editor. Harrison’s cardiovascular medicine. New York. McGraw-Hill Medical 2010. p 132-146 .
- Mets B, Redai I. Parasympathomimetic and parasympatholytic drugs. In: Evers AS, Maze M, Kharasch ED. Editors. Anesthetic pharmacology. Cambridge. Cambridge University Press. 2011. p 666-675.
- Gill HS, Gill JS. Causes, diagnosis and therapeutic strategy in Bradyarrhythmias. Oxford textbook of critical care. Oxford University Press.2016.730-736. [Free full text]
- Silbernagl S, Despopoulos A. Editors. Color atlas of physiology. Stuttgart. Thieme publishing group. 2009 .
- Pomfrett CJD, Barrie JR, Healy TJ. Respiratory sinus arrhythmia: an index of light anaesthesia. Br J Anaesth. 1993;71:212-217 . [PubMed] [Free full text]
- Boyett MR, Honjo H, Kodama I. The sinoatrial node, a heterogenous pacemaker structure. Cardiovasc Res. 2000;47:658-687 . [PubMed] [Free full text] doi:10.1016/S0008-6363(00)00135-8
- Abedin Z, Conner R. Atrial arrhythmias. ECG interpretation: the self-assessment approach. Massachusetts. Blackwell publishing.2008;91-95 . [Free full text]
- Abedin Z, Conner R. Junctional arrhythmia. ECG interpretation: the self-assessment approach. Massachusetts. Blackwell publishing.2008;161-164 . [Free full text]
- Ectopic beats. In: Bennett DH. Editor. Cardiac arrhythmias: practical notes on interpretation and treatment. London. Hodder Arnold. 2006. p 6-15 . [PubMed] [Free full text]
- Blomstrom-Lundqvist C, Scheinman MM, Aiot EM, Alpert JS, Calkins H, Camm AJ et al. ACC/AHA/ESC guidelines for the management of patients with supraventricular arrhythmias. Circulation. 2003;108:1871-1909 . [PubMed] [Free full text]
- Camm J, Savelieva I. Supraventricular arrhythmias. Textbook of critical care. Elsevier Saunders. 2011; 565-574. [Free full text]
- Marchlinski F. The tachyarrhythmias. In: Loscalzo J Editor. Harrison’s cardiovascular medicine. New York. McGraw-Hill Medical 2010. p 147-175 .
- McCord J, Borzak S. Multifocal atrial tachycardia. Chest 1998;113:203-209 . [PubMed]
- Katristsis DG, Camm AJ. Atrioventricular nodal reentrant tachycardia. Circulation. 2010;122:831-840 . [PubMed] [Free full text]
- Di Toro D, Hadid C, Lopez C, Fuselli J, Luis V, Labadet C. Utility of the aVL lead in the electrocardiographic diagnosis of atrioventricular node re-entrant tachycardia. Europace 2009;11:944-948 . [Free full text]
- Bennett, D. H. Atrioventricular Junctional Re‐entrant Tachycardias. Bennett’s Cardiac Arrhythmias: Practical Notes on Interpretation and Treatment, Eighth Edition, 56-66.
- Tsikouris JP, Kluger J, Song J, White CM. Changes in P wave dispersion and P wave duration after open heart surgery are associated with the peak incidence of atrial fibrillation. Heart Lung. 2001;30:466-471 . [PubMed]
- Chang CM, Lee SH, Lu MJ et al. The role of P wave in prediction of atrial fibrillation after coronary artery surgery. Intl J Cardiol. 1999;68:303-308 . [PubMed] [Free full text]
- Walkey AJ, Wiener RS, Ghobrial JM, Curtis LH, Benjamin EJ. Incident stroke and mortality associated with new onset atrial fibrillation in patients hospitalized with severe sepsis. JAMA 2011;306:2248-2255 . [PubMed] [Free full text]
doi: 10.1001/jama.2011.1615
- Walkey AJ, McManus DD. Causes and diagnosis of tachyarrhythmias. Oxford textbook of critical care. Oxford University Press.2016;722-725. [Free full text]
- Wang K. Editor. Atlas of electrocardiography. New Delhi. Jaypee Medical Pulishers (P) Ltd. 2013 .
- The supraventricular tachycardias. In: Bennett DH. Editor. Cardiac arrhythmias: practical notes on interpretation and treatment. London. Hodder Arnold. 2006. p 27-32 .
- Frendl G, Sodickson AC, Chung MK, Waldo AL, Gersh BJ, Tisdale JE et al. 2014 AATS guidelines for the prevention and management of perioperative atrial fibrillation and flutter for thoracic surgical procedures. J Thorac Cardiovasc Surg. 2014;148:e153-193 . [PubMed] [Free full text] doi: 10.1016/j.jtcvs.2014.06.036
- January CT, Wann LS, Alpert JS, Calkins H, Cigarroa JE, Cleveland JC et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation. J Am Coll Cardiol. 2014;64:2246-2280 . [PubMed] [Free full text]
doi: 10.1016/j.jacc.2014.03.022
- Olshansky B, Rosenfeld LE, Warner AL, Solomon AJ, O’Neill G, Sharma A et al. The atrial fibrillation follow-up investigation of rhythm management (AFFIRM) study; approaches to control rate in atrial fibrillation. J Am Coll Cardiol. 2004;43:1201-1208. . [PubMed] [Free full text] doi: 10.1016/j.jacc.2003.11.032
- Broadley D, Creswell LL, Hogue CW, Epstein AE, Prystowsky EN et al. American college of chest physicians guidelines for the prevention and management of postoperative atrial fibrillation after cardiac surgery. Chest.2005;128:39s-47s. [PubMed]
- Walkey AJ, Magnani J. Therapeutic strategy in tachyarrhythmias. Oxford textbook of critical care. Oxford University Press. 2016;726-729. . [Free full text]
- Tai CT, Chiou CW, Wen ZC, Hsieh MH, Tsai CF, Lin WS et al. Effect of phenylephrine on focal atrial fibrillation originating in the pulmonary veins and superior vena cava. J Am Coll Cardiol. 2000;36:788-793. [PubMed] [Free full text]
- Munger MA, Radwanski PB, Van Tassell BW. Digitalis. Textbook of critical care. New Delhi. Elsevier Saunders. 2011; 1317-1321. [Free full text]
- LeMaitre J, Kornder J. Anti-arrhythmics in critical illness. Oxford textbook of critical care. Oxford University Press. 2016; 165-169. [Free full text]
- Neuberger HR, Mewis C, Van Veldhuisen DJ, Schotten U, Van Gelder IC et al. Management of atrial fibrillation in patients with heart failure. Eur Heart J. 2007;28:2568-2577. [PubMed] [Free full text] doi: 10.1093/eurheartj/ehm341
- Surawicz B, Childers R, Deal BJ, Gettes LS. Recommendations for the standardization and interpretation of the electrocardiogram, part III: Intraventricular conduction disturbances: a scientific statement from the American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology; the American College of Cardiology Foundation; and the Heart Rhythm Society. Circulation. 2009;119:235-240 . [PubMed] [Free full text]
doi: 10.1161/CIRCULATIONAHA.108.191095
- Abedin Z, Conner R. Ventricular arrhythmias. ECG interpretation: the self-assessment approach. Massachusetts. Blackwell publishing.2008;165-182. [Free full text]
- Gazmuri RS, Santonocito C. Ventricular arrhythmias. Textbook of critical care. Elsevier Saunders. 2011; 575-586. [Free full text]
- Zipes DP, Camm J, Borggrefe M, Buxton AE, Chaitman B, Fromer M et al. ACC/AHA/ESC 2006 guidelines for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Circulation. 2006;114:e385-e484 . [PubMed] [Free full text] doi: 10.1161/CIRCULATIONAHA.106.178233
- Bennett, DH. Monomorphic ventricular tachycardia. Cardiac arrhythmias: practical notes on interpretation and treatment.2006; 91-114. [Free full text]
- Bennett, DH. Polymorphic ventricular tachycardia and ventricular fibrillation. Cardiac arrhythmias: practical notes on interpretation and treatment.2006;115-129 . [Free full text]
- Bennett, DH. Tachycardias with broad ventricular complexes. Cardiac arrhythmias: practical notes on interpretation and treatment.2006;130-140. [Free full text]
- Vecht R, Gatzoulis MA, Peters N. Rhythm disturbances. ECG diagnosis in clinical practice. London. Springer-Verlag.2009;113-168 . [Free full text]
- Vecht R, Gatzoulis MA, Peters N. Conduction impairment. ECG diagnosis in clinical practice. London. Springer-Verlag. 2009; 67-112 . [Free full text]
- Link MS, Berkow LC, Kudenchuk PJ, Halperin HR, Hess EP, Moitra VK et al. Adult advanced cardiovascular life support: 2015 American Heart Association guidelines update for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation. 2015;132:S444-S464. [PubMed] [Free full text]
doi: 10.1161/CIR.0000000000000261.
- Bronheim D, Nicoara A, Abel M. Cardiovascular drugs. In: Longnecker DE, Brown DL, Newman MF, Zapol WM. Editors. Anesthesiology. New York. McGraw-Hill Medical 2010. p 742-766 .
- Knight J, Sarko J. Conduction disturbances and cardiac pacemakers. Textbook of critical care. Elsevier Saunders. 2011;587-593 . [Free full text]
- Adamec J, Adamec R. Cardiac conduction troubles. ECG holter: guide to electrocardiographic interpretation. New York. Springer. 2008; 40-48 . [Free full text]
- Epstein AE, DiMarco JP, Ellenbogen KA, Estes NAM, Freedman RA, Gettes LS et al. ACC/AHA/HRS 2008 guidelines for device-based therapy of cardiac rhythm abnormalities. Heart rhythm 2008;5:1-22 . [PubMed] [Free full text]
doi: 10.1016/j.hrthm.2008.04.015




 Facebook
Facebook Twitter
Twitter GooglePlus
GooglePlus Youtube
Youtube