Varun Dua1, Nitin Ahuja2, Hemant Bhagat3, Tanvir Samra4, Venkata Ganesh5,
Saravdeep Dhatt6, Puneet Khanna4, Pravin Salunke7
1Assistant Professor, Maharishi Markandeshwar Institute of Medical Sciences and Research (MMIMSR), Mullana, Ambala, Haryana 133207 (India); E-mail: vrndua@gmail.com
2Senior Resident; 3Additional Professor; 4Assistant Professor; 5Junior Resident
Department of Anaesthesia and Intensive Care, PGIMER, Chandigarh (India)
6Associate Professor, Department of Orthopedics, PGIMER, Chandigarh (India)
7Associate Professor, Department of Neurosurgery, PGIMER, Chandigarh (India)
Correspondence: Tanvir Samra, MBBS, MD, House No. 262, Sector 33-A,
Chandigarh-160034 (India); Phone: 9910703609; E-mail: drtanvirsamra@yahoo.co.in
ABSTRACT
Introduction: Patient age and severity of brain injury are validated prognostic indicators in patients with polytrauma. This prospective observational study was conducted to study the influence of extracranial injuries on neurological outcome of patients with traumatic brain injury (TBI).
Methodology: Patients with TBI aged 20-60 years were enrolled and categorized into two groups: Group 1- with extracranial injuries; Group 2- without extracranial injuries. Patients with fixed and dilated pupils, brainstem injuries, delay of more than 24 hours for hospitalization, and patients who developed secondary insults were excluded.
Results: Complete follow up and data collection was feasible in 33 patients of Group 1 and in 47 patients of Group 2. Severity of head injury, SOFA score, co-morbid conditions, duration of hospital stay, mortality, GCS scores on admission and Δ GCS (GCS on admission – GCS at time of discharge) were comparable between the two groups. The duration of mechanical ventilation and the ISS scores were significantly higher in patients with extracranial injuries. Extracranial injuries did not influence the survival rate. Severity of head injury was the prime determinant of survival. Extracranial injuries were shown to have a synergistic effect on morbidity.
Conclusion: Presence of extracranial injuries does not influence the outcome of patients with head injuries in which secondary insults like hypoxia, hypercapnia / hypocapnia, hypotension, hyperpyrexia, hypoglycemia / hyperglycemia and intracranial hypertension are avoided.
Key words: Head injury; Multiple injuries; Polytrauma; Outcome; Secondary insults
Citation: Dua V, Ahuja N, Bhagat H, Samra T, Ganesh V, Dhatt S, Khanna P, Salunke P. Outcome in patients with head injury: Do extra-cranial injuries worsen prognosis? Anaesth Pain & Intensive Care 2016;20(4):411-416
Received: 10 Jun 2016; Reviewed: 9 Jul, 10 Oct 2016; Corrected: 9 Jun, 24 Aug 2016; Accepted: 4 Nov 2016
INTRODUCTION
Traumatic brain injury (TBI) is the major cause of morbidity and mortality worldwide.1Patient age and severity of brain injury (depth and duration of coma, other neurological abnormalities, intracranial pressure) are some of the validated prognostic indicators in patients with polytrauma.2,3 Injuries to the torso and/or extremities (TEI) have shown to worsen the outcome of patients with head injury with an Abbreviated Injury Scale (AIS) of 4 and more.4
The number of trauma victims in India in a year are estimated to be 1.5 to 2 million, whereas the number of deaths/year due to it are estimated to be 1 million.5 Pre-hospital care of trauma victims, dedicated trauma centers and facilities for rehabilitation of the injured are underdeveloped in our country. This prospective observational study was conducted to study the influence of other associated systemic injuries on neurological outcome of adult patients admitted with TBI in our trauma center.
METHODOLOGY
Patients with head injury aged between 20-60 years and admitted to our advanced trauma center (level III) were enrolled and categorized into two groups: Group 1- with extracranial injuries, and Group 2, with no extracranial injuries. Approval from institutional ethics committee was taken and data were collected after informed patient consent.
Patients with fixed and dilated pupils, brainstem injuries and with a delay of more than 24 hours from time of injury to presentation to the hospital were excluded. To minimize the effect of other confounding factors on clinical outcome, any patient developing a secondary insult like hypoxia, hypercapnia / hypocapnia, hypotension, hyperpyrexia, hypoglycemia / hyperglycemia and intracranial hypertension, was also excluded.
Standard resuscitative protocols in accordance to guidelines of Advanced Trauma Life Support6 and Brain Trauma Foundation were followed.7 Abbreviated Injury Scale (AIS)8, Injury severity scale (ISS)8 and SOFA9 were calculated on admission. Head injury was classified as mild, moderate and severe, based on Glasgow coma scale (GCS) value of > 12, 9-12 and < 8, respectively. An ISS value of more than 15 was labelled as severe trauma. Injury Severity Score (ISS) in patients with polytrauma (Group 1) was derived from AIS: the sum of the squared AIS in each of the three most severely injured of seven defined regions of the anatomy. Injuries were assigned AIS score from 1-6 based on severity; minor (1), moderate (2), serious (3), severe (4), critical (5), unsurvivable (6). Appendix 1 summarizes the AIS scores for various types of injuries.
Appendix: AIS Scores (examples)
| Area | Injury description | AIS |
| Head and Neck | Cerebral contusion
Subarachnoid hemorrhage subdural hematoma |
3
3 4/5 |
| Blunt Trauma Chest | Flail chest:
Unilateral (≥3 ribs) Bilateral Contusion Laceration (skin, subcutaneous) |
3-4 5 3 1 |
| Blunt Trauma Abdomen | Liver/spleen injury scale:
Depending on amount of subcapsular hematoma, capsular tear and depth of laceration Vascular injuries |
2-3
5 |
| Long bone Fracture | Femur | 3 |
The neurological status of the patient (GCS scores) was evaluated on admission, at 24 hours and then every 48 hours till time of discharge, or a total duration of 28 days, which ever was earlier. Δ GCS i.e. difference between GCS on admission and GCS at time of discharge was compared between the two groups. Co-morbid conditions, duration of hospital stay, days of mechanical ventilation and mortality were also noted.
The primary outcome was to evaluate the effect of extracranial injuries on neurological recovery in patients with head injury. Δ Glasgow coma scores (ΔGCS) (difference between GCS on admission and GCS at time of discharge) was compared in patients with TBI and additional extracranial injuries (Group 1) and in patients with isolated head injury (Group 2). To identify independent risk factors for neurological deterioration, linear regression analysis of age, co-morbidities, ISS and SOFA scores with ΔGCS were performed. Kaplan-Meier survival analysis was done to study the effect of extracranial injuries on mortality. The difference between the curves were analyzed using the log-rank test.
This was a pilot study so we did not do a sample size estimation. Statistical analyses were performed using the statistical software package (SPSS for Windows, version 21.0; SPSS Inc; Chicago IL). Descriptive frequencies were expressed using the mean (SD) and differences between the means of continuous variables were compared using the unpaired t-test, and those of the co-morbidities and the categorical variables were compared with the Mann-Whitney U-test and the chi-square test. A p-value of < 0.5 was considered statistically significant.
RESULTS
Complete follow up and data collection was feasible in 33 patients with co-existing extracranial injuries (Group 1) and in 47 patients with diagnosis of only head injury (Group 2). The most frequently reported extracranial injuries in Group 1 were limb fractures (45.5%), thoracic injuries (33.33%), maxillofacial injuries (21.2%) and blunt trauma abdomen (12.1%). Distribution patterns of head injury were similar in both the groups with nearly half of patients with an extradural hematoma and the remaining half with a contusion and subdural hematoma.
Only 22 patients in Group 1 and 28 in Group 2 underwent an operative intervention (Figure 1).
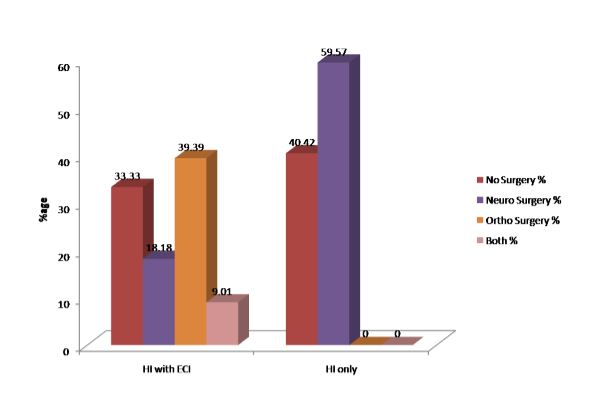 Figure 1: Operative interventions performed
Figure 1: Operative interventions performed
Demographic and clinical characteristics between the two groups are summarized in Table 1. Outcome parameters (mortality, morbidity and neurological recovery) are summarized in Table 2. Severity of head injury, as mild, moderate and severe, was comparable between the two groups
Table 1: Demographic and clinical characteristics
| Patient characteristics | Group 1
( HI+EC injuries) N= 33 |
Group 2
(HI) N= 47 |
P value |
| Age (in years) | 34.03 ± 9.9 | 34.7 ± 10.1 | 0.77
|
| Incidence of co-morbidities (%)
a) None b) Single c) Multiple |
93.93 3.03 3.03 |
91.48 6.38 2.12 |
0.70 |
| Major Trauma and the Injury Severity Score
ISS (mean± SD) Major Trauma (ISS>15) |
19.73 ± 7.73 23/33 |
12.09 ± 5.06 20/47 |
0.00 |
| SOFA scores | 4.45 ± 3.11 | 3.96 ± 2.45 | 0.44 |
| Severity of head injury
Mild (GCS 13-15) Moderate (GCS 9-12) Severe (GCS ≤ 8) |
6/33* 6/33 21/33 |
14/47** 16/47 17/47 |
please give p values |
*Four out of six had normal GCS. **Four out of 14 had normal GCS.
Table 2: Outcome parameters; mortality, morbidity and neurological recovery (Data given as mean ± SD)
| Patient characteristics | Group 1
( HI+EC injuries) N= 33 |
Group 2
(HI) N= 47 |
P value |
| Duration of hospital stay (days) | 11.58 ± 8.3 | 9.72 ± 10.1 | 0.38 |
| Duration of ICU stay (days) | 1.76 ± 3.6 | 0.49 ± 1.8 | 0.045 |
| Duration of mechanical ventilation (days) | 2.52 ± 2.6 | 1.5 ± 1.9 | .06 |
| Mortality | 3/33 | 6/47 | .609 |
| GCS Scores
GCS on admission GCS on discharge ΔGCS scores |
9.06 ± 3.4 14.30 ± 1.08 5.17 ± 3.08 |
9.72 ± 3.4 13.88 ± 1.9 3.80 ± 3.42 |
0.39 0.29 0.086 |
Δ GCS in Group 1(n = 33): Analyzed for only 26 patients. Calculation was not done for the remaining 7 patients as four patients had GCS of 15, one patient had no change in GCS (remained at a value of 14); two had severe TBI (GCS < 8) but again had a change in GCS of less than two (both of them eventually expired).
- Δ GCS of ≤ 4 was recorded in 5 patients
- Δ GCS of 5-8 was recorded in 17 patients
- Δ GCS of ≥ 8 was recorded in 4 patients
Δ GCS in Group 2 (n = 47): Analyzed for only 41 patients. Calculation was not done for the remaining 6 patients, as four had a GCS of 15 and two with severe TBI had a change of less than 2 (both eventually expired)
- Δ GCS of ≤ 4 was recorded in 8 patients
- Δ GCS of 5-8 was recorded in 28 patients
- Δ GCS of ≥ 8 was recorded in 5 patients
Linear regression analyses of age, co-morbidities, SOFA and ISS scores with ΔGCS revealed that out of all the above mentioned parameters only the SOFA and ISS scores correlate significantly with Δ GCS (p < 0.01). Kaplan-Meier survival analysis was done to observe the effect of extra-cranial injuries on survival in patients with TBI. “Accordingly there was no statistically significant difference between groups p > 0/05.” (Figure 2).
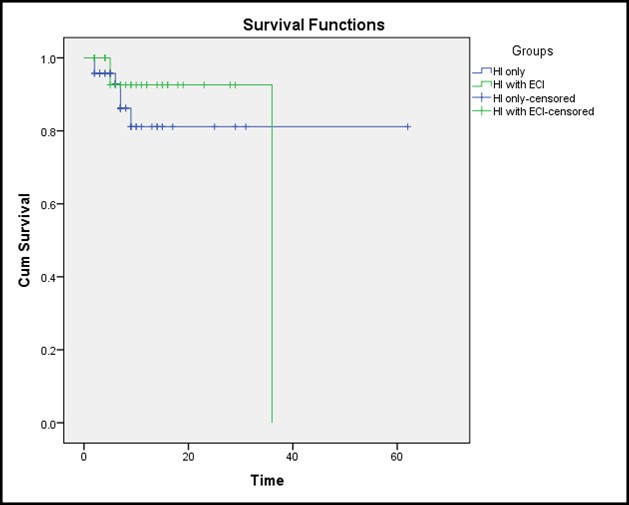
Figure 2: The Kaplan-Meier survival curves for the two groups.
DISCUSSION
The groups differed significantly only in the duration of ICU stay and the ISS scores which were significantly higher in patients with extracranial injuries. However, survival was independent of presence or absence of an extracranial injury. These results are supported by a previous study by Baltas et al.10 who reported that polytrauma in hemodynamically stable patients did not influence patient mortality. Severity of head injury was the main determinant of survival. Extracranial injuries were shown to have a synergistic effect on morbidity. In our study the severity of head injury and the GCS scores were comparable between the two groups and so the mortality was comparable. The prolonged duration of ICU stay in patients with extracranial injuries can be explained on the basis of statistically significant higher ISS scores in this subset.
Results of our study are also similar to that of Sarrafzadeh et al.11 who studied the occurrence of secondary insults and the influence of extracranial injuries in patients with severe head injury and reported a comparable outcome which was independent of the presence of extracranial lesions in patients with AIS ≤ 5. The authors had measured intracranial pressure, mean arterial blood pressure, cerebral perfusion pressure, end-tidal CO2, brain tissue PO2 and jugular bulb oxyhemoglobin saturation.11 In our study, we monitored clinical status of the patient using GCS scores.
Lefering et al.4 analyzed data of 21,356 cases from ‘The Trauma Registry of the German Society for Trauma Surgery’ and concluded that the increase in mortality with concomitant injuries was dependent on the injury severity. Patients with head injuries and torso and/or extremities (TEI) but the same AIS severity level had different survival scores. Absolute increase in mortality rate reported with TEI of grade 4 and grade 5 was 5% and 15% respectively. But the authors did not elaborate whether concomitant severe TEI increase early or late mortality in patients with head trauma. Organ failure, sepsis, and hospital stay were found to be more dependent on TEI severity. Intubation free days were equally affected by both types of injuries if severity level was at least AIS grade 3.
This was a pilot study and one of the main limitation was the small sample size. Thus we did not compare the groups regarding different levels of AIS.
Various prognostic models for death at 14 days and for death or severe disability at time interval of six months have been proposed and validated in patients with traumatic brain injury.12 Use of computer based models for prediction of outcome is advocated in patients with polytrauma as it facilitates therapeutic decision making. It optimizes the use of limited health care resources and therapeutic interventions for those predicted to have a good outcome and helps in critical decision making and optimization of health care resources. In the Medical Research Council (MCR) CRASH Trial, age, Glasgow coma scale (GCS), pupil reactivity and the presence of major extracranial injury were included as prognostic indicators but the strength of a predictor in evaluating outcome was found to vary according to residence of the patient in either high or low-middle income country.12 Results of the study concluded that it was inappropriate to extrapolate from models of high income countries to developing countries. In our study, we find that presence of extracranial injuries do not influence the outcome of patients with head injuries
CONCLUSION
Presence of extracranial injuries increase the ISS scores and duration of stay in the ICU. But if secondary insults like hypoxia, hypercapnia / hypocapnia, hypotension, hyperpyrexia, hypoglycemia / hyperglycemia and intracranial hypertension are avoided, then presence of extracranial injuries do not influence the outcome of patients with head injuries. However, further studies on a large number of patients are needed to analyze the influence of extracranial injuries in patients at different AIS severity levels.
Conflict of interest: None declared by the authors
Authors’ contribution:
VD & NA: Conduction of study and manuscript writing
HB & PK: Concept
TS: Manuscript writing
VG: Statistical analysis
SD: Recruiting orthopedic cases
PS: Recruiting neurotrauma cases
REFERENCES
- Hofman K, Primack A, Keusch G, Hrynkow S. Addressing the growing burden of trauma and injury in low- and middle-income countries. Am J Public Health 2005 Jan;95(1):13-7.[PubMed][Free full text]
- Gennarelli TA, Spielman GM, Langfitt TW, Gildenberg PL, Harrington T, Jane JA, et al.: Influence of the type of intracranial lesion on outcome from severe head injury. J Neurosurg 1982 Jan;56:26-32. [PubMed]
- Choksey M, Crockard HA, Sandilands M: Acute traumatic intracerebral haematomas: determinants of outcome in a retrospective series of 202 cases. Br J Neurosurg 1993;7(6):611-622. [PubMed]
- Lefering R, Paffrath T, Linker R, Bouillon B, Neugebauer EA; Deutsche Gesellschaft für Unfallchirurgie/German Society for Trauma Surgery. Head injury and outcome–what influence do concomitant injuries have? J Trauma 2008 Nov;65(5):1036-43. [PubMed] doi: 10.1097/TA.0b013e318184ee48
- Gururaj G. Epidemiology of traumatic brain injuries: Indian scenario. Neurol Res 2002 Jan;24(1):24-8. [PubMed]
- Kortbeek JB, Al Turki SA, Ali J, Antoine JA, Bouillon B, Brasel K, et al. Advanced trauma life support, 8th edition, the evidence for change. J Trauma 2008 Jun;64(6):1638-50.[PubMed]
- Sagher O. Treatment guidelines from the Brain Trauma Foundation. J Neurosurg 2013 Nov;119(5):1246. [PubMed] doi: 10.3171/2013.4.JNS13634.
- The Abbreviated Injury Scale-1990, Update 1998. Des Plaines, IL: 2008.
- Vincent JL, de Mendonça A, Cantraine F, Moreno R, Takala J, Suter PM, et al. Use of SOFA score to assess the incidence of organ dysfunction/organ failure in intensive care units: results of a multicenter, prospective study. Workimg group on ‘sepsis related problems’ of the European Society of Intensive Care Medicine. Crit Care Med 1998 Nov;26(11):1793–1800. [PubMed]
- Baltas I, Gerogiannis N, Sakellariou P, Matamis D, Prassas A, Fylaktakis M. Outcome in severely head injured patients with and without multiple trauma. J Neurosurg Sci 1998 Jun;42(2):85– 88.[PubMed]
- Sarrafzadeh AS, Peltonen EE, Kaisers U, Ku¨chler I, Lanksch WR, Unterberg AW. Secondary insults in severe head injury – Do multiply injured patients do worse? Crit Care Med 2001 Jun;29(6):1116 –1123. [PubMed]
- MCR CRASH Trial Collaborators, Perel PA, Arango M, Clayton T, Edwards P, Komolafe E, et al. Predicting outcome after traumatic brain injury: practical prognostic models based on large cohort of international patients. BMJ 2008 Feb 23;336(7641):425-9. [PubMed] [Free full text] doi: 10.1136/bmj.39461.643438.25.




 Facebook
Facebook Twitter
Twitter GooglePlus
GooglePlus Youtube
Youtube