Poonam S. Ghodki*, Shalini P. Sardesai**,
*Associate Professor; **Professor
Shrimati Kashibai Navale Medical College and General Hospital, Pune, Maharashtra, (India)
Correspondence: Dr. Poonam S. Ghodki, Department of Anesthesiology, Shrimati Kashibai Navale Medical College and General Hospital, Pune, Maharashtra, (India); E-mail: drpoonamghodki@gmail.com
ABSTRACT
Obstetric hemorrhage, a preventable condition, is one of the leading causes of death in developing countries. The role of anesthesiologist in the management of obstetric hemorrhage is very critical. The crux of management of obstetric hemorrhage is a consensual planning with formation of a multidisciplinary team and formulating a logistic protocol that can be disseminated locally for managing catastrophes. This review highlights the optimum preoperative care to assess the risk factors and isolate the patients prone to develop peripartum hemorrhage and also deals with the most recent guidelines and management protocols including conservative and interventional modes of treatment. Details of anticipated and unanticipated obstetric hemorrhage, fluid and blood transfusion strategies are discussed.
Key words: Hemorrhage, antepartum; Hemorrhage, intrapartum; Hemorrhage, postpartum; Obstetric hemorrhage; Multidisciplinary team; Blood transfusion.
Citation: Ghodki PS, Sardesai SP. Obstetric hemorrhage: anesthetic implications and management. Anaesth Pain & Intensive Care 2014;18(4):405-414
INTRODUCTION
Obstetric hemorrhage is one of the leading causes of maternal mortality in developing countries.1,2 Even in developed countries, it is a complication for which highest rate of substandard care and potential avoidance is observed.3 A study by World Health Organization (WHO) revealed that 25-30% of maternal deaths are due to peripartum hemorrhage globally.4 Advances in obstetrics including improvement in evaluation methods and introduction of non interventional radiological techniques of management have progressively decreased maternal morbidity and mortality.5,6 Unfortunately, few parturients sustain profound peripartum hemorrhage that overwhelms the compensatory mechanisms leading to poor outcomes.
Applying conventional definition of hemorrhage to peripartum hemorrhage may be misleading as blood loss up to 1000 ml is not uncommon during deliveries.7 Although no consensus exists on the definition of massive obstetric hemorrhage, presence of either of the following has been described:8
- Sudden blood loss > 1500 ml (25% of blood volume),
- Blood loss > 3000 ml in less than 3 hours (50% of blood volume)
- Blood loss of 150 ml/minute in 20 minutes (>50% of blood volume)
- Requirement of acute transfusion of > 4 units of packed red blood cells.
The clinical utility of these definitions cannot be validated in routine practice and the intervention and management should be tailored to individual patients.
Obstetric hemorrhage is challenging to anesthesia providers as it is usually sudden in onset, rapid and maybe life threatening.9 A multidisciplinary approach with a team consisting of obstetrician, anesthesiologist, neonatologist and hematologist is imperative for management of peripartum bleeding.
In the context of obstetric hemorrhage, it is important to know;
- How is obstetric hemorrhage different from other major causes of hemorrhage?
- What leads to obstetric hemorrhage i.e. the etiology; its classification and anesthetic implications?
- What is the role of anesthesiologist in obstetric hemorrhage including maternal and fetal resuscitation?
After acquiring a thorough knowledge of these, a strategy should be formulated based on national and international guidelines and a local protocol has to be finally explicated in every maternity unit.
HOW IS OBSTETRIC HEMORRHAGE DIFFERENT?
One of the major reasons behind high mortality after peripartum hemorrhage is the failure to recognize the presence of bleeding.10 This happens because of the following reasons:
a. Inability to recognize the risk factors
This occurs due to inappropriate clinical evaluation and inadequate investigation of the expectant mother. A high degree of suspicion is hence needed to identify the risk factors responsible for hemorrhage in the antenatal period.11 Most of the antepartum hemorrhages can be diagnosed by ultrasonography (USG) in the antenatal check up, which can be repeated at frequent intervals in case of suspicion.5 USG not only helps in confirming the cause of hemorrhage but may also diagnose the presence of concealed hemorrhage in an otherwise asymptomatic patient.
b. Difficulty in exact blood loss estimation
Blood loss estimation becomes difficult in peripartum hemorrhage due to dilution by amniotic fluid or the loss may not be revealed in cases of intrauterine bleeding, bleeding in peritoneal cavity or in retroperitoneal hemorrhages.12,13 Hence a high clinical experience is needed for estimating the blood loss; else inadequate volume replacement is inevitable.
c. Difficulty in early diagnosis
Early signs of shock like tachycardia and increased systemic vascular resistance are masked by the normal physiological changes of pregnancy.14 Hemodynamic collapse occurs only when almost 35-45% of circulating volume is lost.15 Thus there is a delay in intervention to control the bleeding.
d. High uteroplacental blood flow
The uteroplacental unit receives 12% of the cardiac output at term pregnancy i.e. 700 ml/min.7 Hence it forms a potential source of rapid bleeding, which if unabated may become life threatening.
Hemodynamic status should always be correlated to the blood loss and any discrepancy has to be communicated to the obstetrician without delay.
TYPES AND CAUSES OF OBSTETRIC HEMORRHAGE
Obstetric hemorrhage can be broadly classified as antepartum, intrapartum and postpartum hemorrhage.
- ANTEPARTUM HEMORRHAGE (APH)
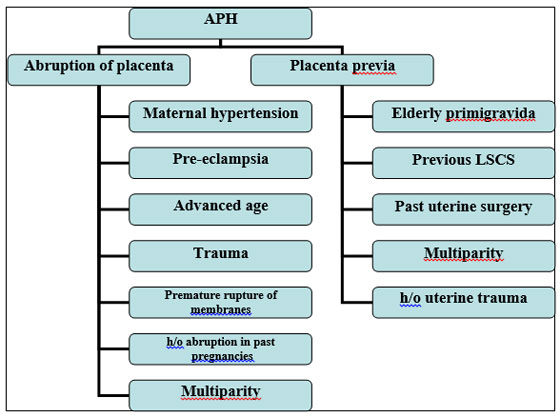
It is defined as hemorrhage that occurs after 24 weeks of gestation to before delivery.7 Although the incidence of APH is high, fortunately only a fraction of patients experience life threatening hemorrhage. It complicates 0.51 to 2.33% of pregnancies. APH represents a greater threat to fetus than mother and fetal compromise may precede maternal complications. Hence fetal demise occurs in 20-35% of cases.7,16 The causes and risk factors are elaborated in Figure 1.
Abruptio placentae:
Abruptio placentae is defined as a complete or partial separation of placenta from decidua basalis before delivery.17 Most of the cases of abruptio placentae originate from maternal hypertension and pre eclampsia. It may be revealed by per vaginal (PV) bleeding or may be concealed behind placenta (picture 1). It is associated with a perinatal mortality of up to 60%. Fetal compromise is likely because of the loss of placental surface area for materno-fetal exchange of oxygen and nutrients.18
Figure 1: Causes and risk factors of APH
Obstetric management:
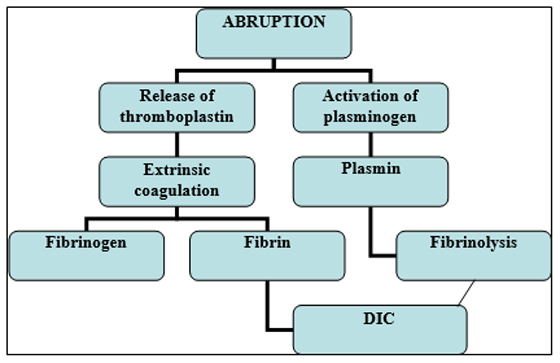
Definitive management for abruptio placenta is delivery of infant and placenta that in turn depends on weeks of gestation.19 Expectant management may be considered in preterm patients with no hemodynamic compromise and no concomitant coagulopathies. Abruptio placenta has to be expeditiously managed because of the inherent risk of disseminated intravascular coagulation (DIC) in pregnancy. (Figure 2) Concealed type has the highest incidence of underestimation of true blood loss and is the most common cause of coagulopathy.20
Figure 2: Abruptio placenta and DIC
Placenta previa:
Placenta previa is defined as the abnormal implantation of placenta in the lower uterine segment. It is further classified depending on its extension towards cervical os as marginal, partial or complete placenta previa (Picture 2). The incidence is around 0.4-0.8%.16
There is increased susceptibility in patients with prior uterine handling as the placenta gets implanted to the scarred area in uterus.21,22 Classical presentation of placenta previa is painless vaginal bleeding that may be overlooked by patient or the attending obstetrician.7 Timely ultrasonographic evaluation plays a pivotal role in early diagnosis5. It should be envisaged that per vaginal examination may lead to life threatening hemorrhage in patients having placenta previa. Nevertheless when the ultrasound report is inconclusive, PV examination may be warranted. A double set up is recommended for such high risk cases wherein the PV examination is carried out in the operating room in presence of anesthesiologist and neonatologist with full preparation for general anesthesia.7 Patient’s parts are painted and draped in lithotomy position for examination by obstetrician and the anesthesiologist is ready for intubation in case need of cesarean section arises.7,16
Obstetric management:
The goal of management is to delay delivery until fetal maturity and is dictated by the amount of bleeding. Tocolytics like magnesium sulfate, beta blockers, nifedipine and ritodrine are used with varying success rates.
Anesthetic plan in APH
Best anesthetic plan for abruptio placenta is general anesthesia with endotracheal tube, even in hemodynamiclly stable patients. This is because of the high propensity of post partum hemorrhage in patients of abruptio placenta.17 Rapid sequence induction has to be done considering all pregnant patients to be full stomach. The choice of induction agent strongly depends on the hemodynamic stability of the patient. Ketamine is the agent of choice in patients presenting with shock. Etomidate is another good alternative. However, thiopentone and propofol have to be used cautiously because of their deleterious effect on circulatory system. For maintenance, a volatile agent has to be added to prevent awareness with a concentration of not more than 0.5 MAC, else the chances of uterine atony and postpartum hemorrhage become high. Nitrous oxide should not be used for maintenance in case the indication for cesarean section is fetal distress. Pain has to be addressed well.23
Epidural analgesia can be offered to the patient as long as coagulation and volume status are acceptable.24
The type of anesthesia in placenta previa largely depends on hemodynamic stability of patient. Bonnar and colleagues determined that most of the anesthesiologists preferred neuraxial anesthesia over general anethesia in both elective and emergency situations.25 Spinal or epidural anesthesia may be considered in stable patients provided the possibility of placenta accreta has been ruled out.24 General anesthesia is administered in unstable patients with the above mentioned principles.26
INTRAPARTUM HEMORRHAGE (IPH)
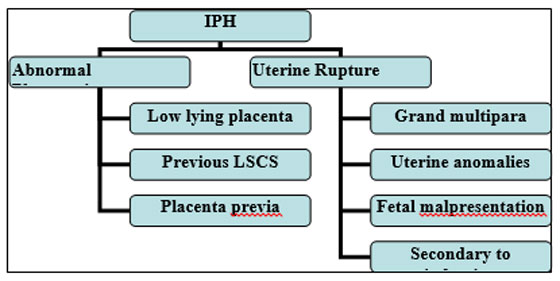
It occurs in 1 in 2000 deliveries. The most common causes are abnormal placentation and uterine rupture. The risk factors for each are depicted in Figure 3.
Figure 3: Causes and risk factors of IPH
Abnormal placentation:
It is defined as abnormal attachment of placenta to uterine wall.27 Depending on the degree of myometrial invasion it is further classified as placenta accreta, increta and percreta (Picture 3).
Obstetric management:
Antenatal USG confirms diagnosis and allows preparation for delivery.28Once the diagnosis is confirmed, following protocol has to be followed-
- Proper pre operative counselling
- Planned cesarean section with consent and preparation of obstetric hysterectomy.29
- Anticipation of blood loss and arrangement of blood and blood products.28
- Intra operative blood salvage can be considered.30-32
- Pre operative internal iliac artery balloon tamponade or embolisation.33-35
Uterine rupture:
Although rare, uterine rupture is the most life threatening emergencies in obstetrics; associated with high maternal and perinatal morbidity and mortality.36 It can either present in the form of scar dehiscence or complete uterine wall rupture.37
Obstetric management:
Fetal delivery with repair of ruptured uterine wall is the definitive treatment. Uterine and internal iliac arterial ligation may be done in anticipated cases.38-40 In intractable blood loss, as a last resort, obstetric hysterectomy is advocated without wasting time in decision making.41,42
POSTPARTUM HEMORRHAGE (PPH)
WHO has defined PPH as blood loss of >500 ml in first 24 hours or a 10% decline in hemoglobin from admission to post partum period. PPH occurs in around 10% deliveries.43 It can be either:
Primary – Blood loss in first 24 hours or
Secondary – Blood loss after 24 hours to 6 weeks post delivery.7,16
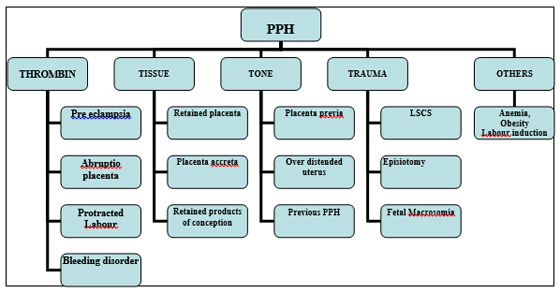
Risk factors: Briefed as 4Ts44-46 (Figure 4)
Figure 4: Risk factors for PPH
Uterine atony:
Atonic uterus is responsible for 80% of PPH. It is defined as lack of efficient uterine contractility after placental separation.45,46
Early contemplation and administration of uterotonics (fig 4) is of paramount importance to prevent atony of uterus.
Table 1: Uterotonic drugs
|
Drug |
Description |
| OXYTOCIN47,48 |
|
| ERGOMETRINE49 |
|
| PROSTAGLANDIN F2α AGONIST50,51 (Carboprost) |
|
| PROSTAGLANDIN E1 ANALOGUE52(Misoprostol) |
|
Obstetric management:
If uterotonic drugs fail to contract the uterus following invasive measures are considered-
1. Bimanual compression of uterus.7,
2. Uterine massage16
3. Uterine compression suture- B Lynch suture.53
4. Surgical iliac or uterine artery ligation.54-56
5. And if all the above fail, obstetric hysterectomy should be done as a life saving procedure. While the obstetrician shows his dexterity in surgical skills, anesthesiologist too should deftly manage fluid and volume resuscitation concomitantly.
Obstetric trauma:
Obstetric trauma may occur during operative delivery for e.g. during cesarean section or episiotomy.44
Management depends on the type of hematoma. Small pelvic hematoma generally has no hemodynamic compromise and can be managed conservatively. In case of large hematoma, however, surgical exploration followed by evacuation may be demanded. In severe cases, vessel ligations may be warranted.45Optimal care has to be taken to prevent infection and septicemia. When uterine inversion is the precipitating factor for trauma, immediate replacement of uterus even prior to removal of placenta is considered as best management. This remedy requires general anesthesia with good relaxation aided by volatile agents.16,46
Coagulopathy:
Other than obstetric causes of coagulopathy in pregnancy (abruption placenta, pre eclampsia), bleeding disorders should also be kept in mind as an important differential diagnosis.7 PPH may be the first indication in patients suffering from von Willebrand disease that is characterized by deficiency of vWF (von Willebrand factor).57A patient with menorrhagia when screened in antenatal clinic may be timely diagnosed for such types of bleeding diathesis.
Treatment includes administration of tranexamic acid, DDAVP (desmopressin), when refractory; the coagulation factor needs to be specifically replaced. In a patient diagnosed as vWD, epidural anesthesia may be safely performed if vWF > 50 IU/dl. 16,57
Other causes of PPH like retained placenta and products of conception require manual removal under general anesthesia with inhalational agents. These procedures are quite painful and hence adequate analgesia should be provided.
THE ROLE OF ANESTHESIOLOGIST
The anesthesiologists have thorough knowledge of normal physiological changes in pregnancy and hence their role is crucial in the management of obstetric hemorrhage. Multiple tasks have to be carried out quickly and with expertise in the event of massive hemorrhage.58 Hence timely and effective communication between the obstetrician and anesthesiologist is adjuratory.
Pre anesthetic evaluation:
The involvement of anesthesiologist from before the delivery of patient is fundamental. One needs to proficiently recognize the patients with risk factors (vide supra) and isolate and prepare them accordingly. Investigations requiring special attention are hemoglobin levels, coagulation studies, blood grouping and cross matching.6 Availability of whole blood and blood products have to be confirmed before institution of anesthesia in identified patients. Prevalence of chronic anemia being a concern in developing countries, it has to be separately dealt with and treated with oral or parenteral iron.59Recombinant human erythropoietin may be required in refractory cases.60 Coagulaopathies (thrombocytopenia, coagulation factor deficiencies) can be diagnosed by doing a thromboelastogram (TEG) preoperatively.61TEG is also helpful in patients who are on low molecular weight heparin and to guide safety for central neuraxial blockade. It is pertinent for the anesthesiologist to go through the USG reports for any missed out findings.
Assessment of intravascular depletion:
The extent of bleeding is almost always underestimated in obstetric patients.62Signs suggestive of hypovolemia in APH patients should be monitored carefully:62,63
- Hypotension
- Heart rate > 120 beats/minute
- Urine output < 0.5 ml/kg/minute
- Capillary refill time < 5 seconds
Irrespective of the degree of hypovolemic shock, fluid therapy is best guided by continual assessment of maternal vital signs, urine output, hemoglobin and acid base balance.62-64
Interventional radiology suite:
With the advances in treatment modalities, placement of arterial catheters and occlusion balloons before delivery is currently the choice whenever major hemorrhage is suspected.65This is a well recognized and less radical approach that avoids peripartum hysterectomy and preserves fertility. In contemplated cases, indwelling epidural catheters may prove beneficial.66,67In advanced centers where facility for shifting the patient from operating room to radiology suite is available, the patient may be transferred by attending anesthesiologist once hemodynamically stable.67In the interventional radiology suite, embolisaiotn is performed under fluoroscopic guidance. Occasionally, angiographic occlusion balloon catheters (in hypogastric/common iliac arteries) may be needed.
Intraoperative role:
Care for an actively bleeding mother should follow an algorithm that has rapid and successive sequences of medical and surgical approaches to stem bleeding. It is vital that junior anesthesiologists and obstetricians do not perceive the calling of senior colleagues as involving ‘loss of face’. At the same time, senior staff must be in receptive mode to concerns expressed by their juniors. Hence, CALL FOR HELP, as apt communication is the major pillar in the management of obstetric hemorrhage.58,68
Resuscitation:
The urgency and measures taken to resuscitate and arrest hemorrhage need to be tailored to the degree of shock.68,69
Maternal resuscitation:68,70
(Adapted and modified from RCOG Green top guidelines no. 52, 2009)
Minor bleeding (Up to 100-1000 ml blood loss, no s/o clinical shock)
- Gain intravenous access ( 14 G cannula x1)
- Commence crystalloid infusion.
Protocol for resuscitation
Protocol to be followed:
In massive hemorrhage (> 1000ml/ continuing blood loss, or patient in clinical shock) mothers need active resuscitation. Following factors need to be addressed;
- Assess airway
- Assess breathing
- Evaluate circulation
- Oxygen by mask @ 10-15 litres/minute
- Intravenous access – 14 G cannula x 2, central venous cannulation in collapsed patient not only provides a vascular access but may also aid in monitoring the central venous pressure and guide fluid resuscitation.
- Flat position
- Keep the mother warm using appropriate available measures.
- Until blood is available, infuse up to 3.5 litres of crystalloid (RL 2 litres, avoid hypertonic solutions) and/or colloid (1-2 litres) as rapidly as possible. The fluid should be adequately WARMED.
- Special blood filters should NOT be used as they slow the rate of infusion.
- Recombinant factor VIIa therapy should be based on coagulation studies.
Monitoring:
Clinical signs of shock have to be continuously monitored like color of the patient, pallor, urine output, temperature and capillary refill time. Invasive blood pressure monitoring may be done for fragile patients.68,69All the parameters recorded have to be documented well on a flow chart for further references.
Fluid and blood replacement therapy:
The cornerstones of resuscitation in peripartum hemorrhage are restoration of both blood volume and oxygen carrying capacity.68-70 All delivery units, especially small units without the blood bank facility, should maintain a supply of O Rh negative blood, as this might offer the only means of restoring oxygen carrying capacity within an acceptable timescale. In addition, the Confidential Enquiry into Maternal and Child Health recommends that women with known risk factors for obstetric hemorrhage should not be delivered in a hospital without a blood bank on site.68
The clinical picture is the main determinant for the need of blood transfusion and time should not be wasted for laboratory results. Volume replacement must be undertaken on the basis that blood loss is often grossly underestimated. Compatible blood in the form of red cell concentrate is the best fluid to replace major blood loss and should be transfused whenever deemed necessary.68,69
A 2006 guideline from the British Committee for Standards in Hematology summarizes the main therapeutic goal of management of massive blood loss is to maintain:68
- Hemoglobin > 8 g%
- Platelet count > 75 x 109 /liter
- Prothrombin time (PT) < 1.5 x mean control
- Activated prothrombin time (APTT) < 1.5 x mean control
- Fibrinogen > 1.0 g/l
Choice of fluid/blood/blood products:
There is controversy as to the appropriate fluids for volume resuscitation.70 The nature of fluid infused is of less importance than rapid administration and warming of the infusion.70,71
Table 2: Fluids for volume resuscitation
|
IV Fluid |
Use |
| Crystalloid | Up to 2 lit of Hartmann’s solution |
| Colloid | Up to 1-2 lit colloid until blood arrives |
| Blood | CrossmatchedIf crossmatched blood unavailable, give uncrossmatched group-specific blood OR ‘O-negative’ blood |
| Fresh frozen plasma (FFP) | 4 units for every 6 units of red cells or PT/APTT > 1.5 x mean control ( 12-15 ml/kg or total 1 lit) |
| Platelet concentrates | If platelet < 50 x 10 9 |
| Cryoprecipitate | If fibrinogen < 1 g/l |
In cases of massive hemorrhage (80% blood volume loss), large volume of fluid replacement fluids leads to clotting factors deficiency making patient prone to develop DIC.20 The challenge of replacing with blood components lies in the fact that in the event of active and ongoing bleeding, there is always going to be a short delay in arrival of laboratotry reports. Hence, in face of relentless bleeding empirical treatment with 1 liter of FFP and 10 units of cryoprecipitate (2 packs) can be given, while awaiting coagulation studies.72 Such empirical use of FFP and cryoprecipitate is in line with the recommendations in the British Committee for Standards in Hematology guideline.68 A replacement therapy guided by ROTEM may always be helpful in saving time.61
Subsequent management after blood transfusion:
The increasingly important issues in blood transfusion are adverse effects associated with transfusion, including potential infection and transmission of prions, rising costs and the possible future problems of availability.68 Patients should be monitored carefully for diagnosing the complications (fever, hyperkalemia, hypocalcemia, citrate toxicity etc) seen after massive blood transfusion. Equal attention should be given to rebound hypercoagulation and thromboembolism that follows blood transfusion especially in pregnancy which is a hypercoagulable state by itself.63 This can be prevented by usage of graduated compression stockings and pharmacological thromboprophylaxis.68,70
Intrauterine fetal resuscitation:
There is a constant threat to the fetus in obstetric hemorrhage as the uteroplacental circulation is at risk because of the following reasons-7,16
- Compensatory selective vasoconstriction: This occurs as a result of diversion of blood from less vital maternal organs (skin, gut, muscles, uteroplacental unit) to maintain circulation of vital organs.
- Absent autoregulatory capacity: Reduction in maternal blood pressure decreases the uterine blood flow.
Fetal resuscitation can be achieved by following-
- Maintaining uteroplacental circulation will also ensure fetal well being. Hence maternal resuscitation is of paramount importance.
- Monitoring by
Electronic fetal heart rate monitor
NICHD guidelines advocate use of fetal scalp pH, fetal scalp lactate and fetal pulse oximetry for adequacy of fetal circulation.73 - Improving placental perfusion by using appropriate oxytocin regimen. In case of fetal distress or hyperstimulation, oxytocin drip has to be discontinued.
BLOOD CONSERVATION STRATEGIES:74
Autologous blood transfusion:
In pregnanacy, concerns have been raised regarding placental insufficiency, whether the woman will make up her hemoglobin before delivery and whether the collected units will be sufficient in the event of major obstetric hemorrhage.75 Hence, the use of pre-autologous blood deposit is not recommended in pregnancy (exception being a mother who is Jehovah’s witness)76.
Intra operative cell salvage:
This technique is commonly being used in cardiac, orthopedic and vascular surgeries with relative reduction of blood transfusion by 39%.77 The use of cell salvage in peripartum hemorrhage is still controversial. The potential difficulty in bleeding mothers is the effective removal of amniotic fluid and degree of contamination with fetal red cells. Still, several bodies based on current evidence have endorsed cell salvage in obstetrics. Intraoperative cell salvage has an undisputed role in patients who refuse blood or blood component transfusion (Jehovah’s Witness) and in patients where massive blood loss is anticipated (placenta accreta, percreta)77
Recombinant factor VIIa (rFVIIa) therapy:
rFVIIa was developed for patients with hemophilia and the first report of its use in peripartum hemorrhage was released in 2001.78 A 2007 review identified case reports of 65 mothers treated with rFVIIa, with controversial results as 30 out of 65 patients had to undergo peripartum hysterectomy. It is hence suggested that rFVIIa may be used as an adjuvant to standard pharmacological and surgical treatment under the guidance of a hematologist. The recommended dose is 90 mcg/kg, repeated within 15-30 minutes. Empirical use of rFVIIa is not advisable as it is not relevant to use this drug in low fibrinogen levels and in thrombocytopenia.78,79 The pre requisite for administration of rFVIIa is fibrinogen level> 1 g/l and platelet count > 20×109.
Use of antifibrinolytics:
Although evidence is conflicting, there is a consensus that antifibrinolytic agents (tranexamic acid) seldom, if ever, have a place in the management of obstetric hemorrhage.68
CONCLUSION
The role of anesthesiologist in the management of obstetric hemorrhage is crucial. It should be envisaged that the various guidelines available for management should be incorporated into a local protocol for each maternity unit. Above all, a multidisciplinary approach with consensual planning catalyses the management even in crisis situation.
REFERENCES [Free full text] [PubMed]
- Carroli G, Cuesta C, Abalos E, Gulmezoglu AM. Epidemiology of postpartum haemorrhage: a systematic review. Best Pract Res Clin Obstet Gynaecol. 2008;22:999-1012. [PubMed] doi: 10.1016/j.bpobgyn.2008.08.004.
- Chhabra S, Sirohi R. Trends in maternal mortality due to haemorrhage: two decades of Indian rural observations. J Obstet Gynaecol. 2004;24(1):40–43. [PubMed] [Free full text]
- Bouvier-Colle MH, Ould El Joud D, Varnoux N, Goffinet F, Alexander S, Bayoumeu F, et al. Evaluation of the quality of care for severe obstetrical haemorrhage in three French regions. BJOG 2001;108:898–903 [PubMed]
- Khan KS, Wojdyla D, Say L, Gulmezoglu AM, Van Look PFA. WHO analysis of causes of maternal death: a systematic review. The Lancet 2006;367:1066-1074. [PubMed] [Free full text]
- Lam G, Kuller J, McMahon M. Use of magnetic resonance imaging and ultrasound in the antenatal diagnosis of placenta accreta. J Soc Gynecol Investig 2002;9:37–40. [PubMed]
- Graham WJ, Bell JS, Bullough CW. Brouwere V, Lerberghe W. Can skilled attendance at delivery reduce maternal mortality in developing countries? Safe motherhood strategies: A review of the evidence. Studies in Health Organisational Services and Policy.2001:97–129.
- Mayer D, Spielman FJ, Bell EA. Antepartum and postpartum hemorrhage. In: Chestnut DH, editor. Obstetric anesthesia. Principles and practice. 3rd edition. Philadelphia: Elsevier Mosby; 2004: 662–82.
- Rath WH. Post partum hemorrhage- Update on problems of definitions and diagnosis. Acta Obstet Gynecol Scand 2011:90(5);421-8. [PubMed] doi: 10.1111/j.1600-0412.2011.01107.x.
- Thomas TA, Cooper GM: Deaths associated with anaesthesia, in Lewis G (ed): Why Mothers Die 1997-1999. Report from The Confidential Enquiries into Maternal Deaths in the United Kingdom. London, RCOG 2001, 134-149
- Hawkins JL, Koonin LM, Palmer SK, Gibbs CP. Anesthesia-related deaths during obstetric delivery in the United States, 1979-1990. Anesthesiology 1997:86;277-284. [PubMed]
- Larsson C, Saltvedt S, Wiklund I, Skogvoll E, Schei B. Estimation of blood loss after cesarean section and vaginal delivery has low validity with a tendency to exaggeration. Acta Obstet Gynecol Scand 2006:85;1448-1452. [PubMed]
- Sullivan I, Faulds J, Ralph C: Contamination of salvaged maternal blood by amniotic fluid and fetal red cells during elective Caesarean section. Br J Anaesth 2008:101;225-229. [PubMed] [Free full text]
- Patel A, Goudar SS, Geller SE, Kodkany BS, Edlavitch SA, Wagh K, et al. Drape estimation vs. visual assessment for estimating postpartum hemorrhage. Int J Gynecol Obstet. 2006;93:220–224. [PubMed]
- Henrich W, Surbek D, Kainer F, Grottke O, Hopp H, Kiesewetter H, et al: Diagnosis and treatment of peripartum bleeding. J Perinat Med 2008:36;467-478. [PubMed]
- Maslovitz S, Barkai G, Lessing JB, Ziv A, Many A. Improved accuracy of postpartum blood loss estimation as assessed by simulation. Acta Obstet Gynecol Scand 2008:87;929-934. [PubMed] doi: 10.1080/00016340802317794.
- Camann WR, Biehl DH. Antepartum and postpartum hemorrhage. In: Hughes SC, Levinson G, Rosen MA, editors. Shnider and Levinson’s anesthesia for obstetrics. 4th edition. Philadelphia: Lippincott Williams & Wilkins; 2002:361–71.
- Calleja-Agius J, Custo R, Brincat M, et al. Placental abruption and placenta praevia. Eur Clin Obstet Gynaecol 2006;2:121-7. [Online]
- Oyelese Y, Ananth CV. Placental abruption. Obstet Gynecol 2006;108:1005-16. [PubMed]
- Prendiville WJ, Elbourne D, McDonald S. Active versus expectant management in the third stage of labour [review]. Cochrane Database Syst Rev 2000;3.
- Moloney WC, Phillips LL, Pritchard JA, Ratnof OD, Reid DE, Schnieder CL. Management of the obstetrical patient with hemorrhage due to an acute or subacute defibrination syndrome. Blood.1959:14;1354-1367. [Online]
- Chattopadhyay SK, Kharif H, Sherbeeni MM. Placenta previa and accreta after previous caesarean section. Eur J Obstet Gynecol Reprod Biol 1993;52:151–6. [PubMed]
- Clark SL, Koonings PP, Phelan JP. Placenta previa/accreta and prior cesarean section. Obstet Gynecol 1985;66:89–92. [PubMed]
- Bhavani-Shankar K, Lynch EP, Datta S: Airway changes during Cesarean hysterectomy. Can J Anesth 2000:47;338-341. [PubMed]
- Chestnut DH, Redick LF: Continuous epidural anesthesia for elective cesarean hysterectomy. South Med J , 1985:78;1168-1169, 1173 [PubMed]
- Bonnar J. Massive obstetric haemorrhage. Baillieres Best Pract Res Clin Obstet Gynaecol. 2000:14(1);1-18. [PubMed]
- Parekh N, Husaini SW, Russell IF: Caesarean section for placenta praevia: A retrospective study of anaesthetic management. Br J Anaesth 2000:84;725-730. [PubMed]
- Bauer ST, Bonanno C. Abnormal placentation. Semin Perinatol 2009;33:88-95.Hudon L, Belfort MA, Broome DR: Diagnosis and management of placenta percreta: A review. Obstet Gynecol Surv 1998:53;509-517. [PubMed]
- Weiniger CF, Elram T, Ginosar Y, Mankuta D, Weissman C, Ezra Y. Anaesthetic management of placenta accreta: Use of a pre-operative high and low suspicion classification. Anesthesia 2005:60;1079-1084. [PubMed]
- Chestnut DH, Dewan DM, Redick LF, Caton D, Spielman FJ. Anesthetic management for obstetric hysterectomy: A multi-institutional study. Anesthesiology 1989:70;607-610. [PubMed]
- Catling S, Joels L: Cell salvage in obstetrics: The time has come. BJOG 2005:112;131-132. [PubMed]
- Camann W: Cell salvage during cesarean delivery: Is it safe and valuable? Maybe, maybe not! Int J Obstet Anesth 1999:8;75-76. [PubMed]
- Allam J, Cox M, Yentis SM: Cell salvage in obstetrics. Int J Obstet Anesth 2008:17;37-45. [PubMed]
- Dabelea VG, Schultze PM, McDuffie RS. Intrauterine balloon tamponade in the management of postpartum haemorrhage. Am J Perinatol. 2007 Jun;24(6):359-64. [PubMed]
- Sproule MW, Bendomir AM, Grant KA, Reid AW.Embolisation of massive bleeding following hysterectomy, despite internal iliac artery ligation. Br J Obstet Gynaecol.1994;101:908–9. [PubMed]
- Brown BJ, Heaston DK, Poulson AM, Gabert HA, Mineau DE, Miller FJ Jr. Uncontrollable postpartum bleeding: a new approach to hemostasis through angiographic arterial embolization. Obstet Gynecol 1979;54:361–5. [PubMed]
- Miller DA, Goodwin TM, Gherman RB, Paul RH. Intrapartum rupture of the unscarred uterus. Obstet Gynecol 1997;89:671–3. [PubMed]
- Ofir K, Sheiner E, Levy A, Katz M, Mazor M. Uterine rupture: differences between a scarred and an unscarred uterus. Am J Obstet Gynecol 2004;191:425–9. [PubMed]
- Joshi VM, Otiv SR, MajumberR, Nikam YA, Shrivastava M.Internal iliac artery ligation for arresting postpartum haemorrhage. BJOG 2007;114:356–61. [PubMed]
- O’Leary JA. Uterine artery ligation in the control of postcesarean hemorrhage. J Reprod Med 1995;40:189–93. [PubMed]
- Evans S, McShane P. The efficacy of internal iliac artery ligation in obstetric hemorrhage. Surg Gynecol Obstet 1985;160:250–3. [PubMed]
- Habek D, Becarevic R. Emergency peripartum hysterectomy in a tertiary obstetric center: 8-year evaluation. Fetal Diagn Ther 2007;22:139–42 [PubMed]
- Glaze S, Ekwalanga P, Roberts G, Lange I, Birch C, Rosengarten A, et al. Peripartum hysterectomy: 1999 to 2006. Obstet Gynecol 2008;111:732-8. [PubMed]
- You WB, Zahn CM. Postpartum haemorrhage: abnormally adherent placenta, uterine inversion, and puerperal hematomas. Clin Obstet Gynecol 2006;49:184-97 [PubMed]
- Combs CA, Murphy EL, Laros RK Jr. Factors associated with postpartum hemorrhage with vaginal birth. Obstet Gynecol 1991;77:69–76. [PubMed]
- Magann EF, Evans S, Hutchinson M, Collins R, Howard BC, Morrison JC. Postpartum hemorrhage after vaginal birth: an analysis of risk factors. Southern Med J.2005;98(4):419–422. [PubMed] [Free full text]
- Tsu V. Postpartum haemorrhage in Zimbabwe: a risk factor analysis. Br J Obstet Gynaecol. 1993;100:327–333. [PubMed]
- Thomas JS, Koh SH, Cooper GM: Haemodynamic effects of oxytocin given as i.v. bolus or infusion on women undergoing Caesarean section. Br J Anaesth , 2007:98;116-119. [PubMed]
- Svanstrom MC, Biber B, Hanes M, Johansson G, Näslund U, Bålfors EM. Signs of myocardial ischaemia after injection of oxytocin: A randomized double-blind comparison of oxytocin and methylergometrine during Caesarean section. Br J Anaesth 2008:100;683-689. [PubMed]
- Ribbing M, Reinecke H, Breithardt G, Kerber S. Acute anterior wall infarct in a 31-year-old patient after administration of methylergometrin for peripartal vaginal hemorrhage. Herz 2001:26;489-493. [PubMed]
- Hayashi RH, Castillo MS, Noah ML. Management of severe postpartum hemorrhage with a prostaglandin F2 alpha analogue. Obstet Gynecol 1984;63:806–8. [PubMed]
- Harber CR, Levy DM, Chidambaram S, Macpherson MB. Life-threatening bronchospasm after intramuscular carboprost for postpartum haemorrhage. BJOG2007;114:366-8. [PubMed]
- Goffinet F, Haddad B, Carbonne B, Sebban E, Papiernik E, Cabrol D. Practical use of sulprostone in the treatment of hemorrhages during delivery. J Gynecol Obstet Biol Reprod 1995;24:209–16. [PubMed]
- B-Lynch C, Coker A, Lawal AH, Abu J, Cowen MJ. The B-Lynch surgical technique for the control of massive postpartum hemorrhage: an alternative to hysterectomy
- ? Five cases reported. Br J Obstet Gynaecol 1997;104:372–85. [PubMed]
- Deux JF, Bazot M, LeBlanche AF, Tassart M, Khalil A, Berkane N, et al. Is selective embolization of uterine arteries a safe alternative to hysterectomy in patients with postpartum hemorrhage? AJR Am J Roentgenol 2001;177:145–9. [PubMed] [Free full text]
- Hong TM, Tseng HS, Lee RC, Wang JH, Chang CY. Uterine artery embolization: an effective treatment for intractable obstetric haemorrhage. Clin Radiol 2004;59:96–101. [PubMed]
- Soncini E, Pelicelli A, Larini P, Marcato C, Monaco D, Grignaffini A. Uterine artery embolization in the treatment and prevention of postpartum hemorrhage. Int J Gynaecol Obstet 2007;96:181–5. [PubMed]
- James AH, Kouides PA, Abdul-Kadir R, Edlund M, Federici AB, Halimeh S, et al. Von Willebrand disease and other bleeding disorders in women: consensus on diagnosis and management from an international expert panel. Am J Obstet Gynecol 2009;201:12e1-8. [PubMed] doi: 10.1016/j.ajog.2009.04.024.
- Gallos G, Redai I, Smiley R. The role of the anesthesiologist in management of obstetric hemorrhage. Semin Perinatol 2009;33:116-23. [PubMed] doi: 10.1053/j.semperi.2008.12.005.
- The Royal College of Obstetricians and Gynaecologists (RCOG). Blood transfusion in obstetrics: Green top guidelines no.47. London: RCOG; 2008 [https://www.rcog.org.uk/en/guidelines-research-services/guidelines/gtg47/]
- Braga j, Marques R, Branco A, Goncalves J, Lobato L, Pimentel JP, et al. Maternal and perinatal implications of the use of human recombinant erythropoietin. Acta Obstet Gynecol Scand 1996;75:449-53. [PubMed]
- Karlsson O, Jeppson A, Hellgren M. Major obstetric hemoorhage: monitoring with thromboelastogram, laboratory analysis or both? Int J Obstet Anesth. 2014;23(1):10-7 [PubMed] doi: 10.1016/j.ijoa.2013.07.003
- Bose P, Regan F, Paterson-Brown S. Improving the accuracy of estimated blood loss at obstetric haemorrhage using clinical reconstructions. BJOG 2006;113:919-24. [PubMed]
- The Royal College of Obstetricians and Gynaecologists (RCOG). Antepartum hemorrhage: Green top guidelines no.63. London:RCOG; 2011. Available at https://www.rcog.org.uk/en/guidelines-research-services/guidelines/gtg63/
- Wise A, Clark V: Strategies to manage major obstetric haemorrhage. Curr Opin Anaesthesiol 2008;21:281-287. [PubMed] doi: 10.1097/ACO.0b013e3282f8e257.
- Mok M, Heidemann B, Dundas K, Gillespie I, Clark V. Interventional radiology in women with suspected placenta accreta undergoing caesarean section. Int J Obstet Anesth 2008:17;255-261. [PubMed] doi: 10.1016/j.ijoa.2007.11.010.
- O’Rourke N, McElrath T, Baum R, Camann W, Tuomala R, Stuebe A, et al: Cesarean delivery in the interventional radiology suite: A novel approach to obstetric hemostasis. Anesth Analg 2007:104;1193-1194. [PubMed]
- Mitty HA, Sterling KM, Alvarez M, Gendler R. Obstetric hemorrhage: prophylactic and emergency arterial catheterization and embolotherapy. Radiology 1993;188:183–7. [PubMed] DOI: http://dx.doi.org/10.1148/radiology.188.1.8511294
- The Royal College of Obstetricians and Gynaecologists (RCOG). Postpartum hemorrhage: Prevention and Management (Green-top Guidelines No. 52). London: RCOG;2009. Available on https://www.rcog.org.uk/en/guidelines-research-services/guidelines/gtg52/ (Accessed in December 2014)
- Reyal F, SibonyO, Oury JF, Luton D, Bang J, Blot P. Criteria for transfusion in severe postpartum hemorrhage: analysis of practice and risk factors. Eur J Obstet Gynecol Reprod Biol 2004;112:61–4. [PubMed]
- Jansen AJ, van Rhenen DJ, Steegers EA, Duvekot JJ. Postpartum hemorrhage and transfusion of blood and blood components. Obstet Gynecol Surv 2005;60:663–71 [PubMed]
- Padmanabhan A, Schwartz J, Spitalnik SL. Transfusion therapy in postpartum haemorrhage. Semin Perinatol 2009;33:124-7. [PubMed] doi: 10.1053/j.semperi.2009.01.002.
- Gonzalez EA, Moore FA, Holcomb JB, Miller CC, Kozar RA, Todd SR, et al. Fresh frozen plasma should be given earlier to patients requiring massive transfusion. J Trauma 2007;62:112–9. [PubMed]
- Macones GA, Hankins GD, Spong CY, Hauth J, Moore T. The 2008 National Institute of Child Health and Human Development workshop report on electronic fetal monitoring: update on definitions, interpretation and reaerch guidelines. Obstet Gynecol 2008;112:661-6. [PubMed] doi: 10.1097/AOG.0b013e3181841395
- Catling S: Blood conservation techniques in obstetrics: A UK perspective. Int J Obstet Anesth 2007:16;241-249. [PubMed]
- Estella NM, Berry DL, Baker BW, Wali AT, Belfort MA. Normovolemic hemodilution before cesarean hysterectomy for placenta percreta. Obstet Gynecol. 1997:90;669-670. [PubMed]
- Nagy CJ, Wheeler AS, Archer TL. Acute normovolemic hemodilution, intraoperative cell salvage and PulseCO hemodynamic monitoring in a Jehovah’s Witness with placenta percreta. Int J Obstet Anesth 2008:17;159-163. [PubMed]
- Fong J, Gurewitsch ED, Kang HJ, Kump L, Mack PF.An analysis of transfusion practice and the role of intraoperative red blood cell salvage during cesarean delivery. Anesth Analg 2007;104(3):666–72. [PubMed]
- Franchini M, Lippi G, Franchi M. The use of rFVIIa in obstetric and gynaecological haemorrhage. BJOG 2007;114:8–15. [PubMed]
- Bouwmeester FW, Jonkhoff AR, Verheijen R. Successful treatment of life threatening postpartum hemorrhage with recombinant activated factor VII. Obstet Gynecol 2003;101:1174–6. [PubMed]




 Facebook
Facebook Twitter
Twitter GooglePlus
GooglePlus Youtube
Youtube