Lakesh K Anand, MD, FIMSA, FCCP, MNAMS, FCCS1, Manmeet Sandhu, MD2, Jasveer Singh, MD3, Sukanya Mitra, MD, MNAMS1
1Professor; 3Associate Professor
Department of Anesthesia and Intensive Care
Government Medical College and Hospital, Sector 32, Chandigarh (India)
2Attending Consultant, Fortis Hospital Mohali, Punjab (India)
Correspondence: Dr Lakesh K Anand, # 3110, Sector 48-B, Chandigarh, India-160047.
Phone: 911722673510, 919646121522; E-mail: lkanand11@gmail.com
ABSTRACT
Objective: Postoperative pain after laparoscopic cholecystectomy (LC) is the prevailing complaint and the primary reason for delayed discharge. Several studies have demonstrated the role of pregabalin in the postoperative pain management. However, there are limited studies, which evaluated the role of preoperative pregabalin in attenuating postoperative pain after LC. So, the present study was designed to evaluate the effect of a single oral dose of pregabalin to reduce postoperative pain and analgesic consumption after LC.
Methodology: The design of study was prospective, randomized and double blind in a tertiary-care hospital. Sixty consenting patients were randomly allocated into two group of thirty each to receive either a matching alprazolam 0.5 mg (placebo) or pregabalin 150 mg, orally 2 hours before surgery in a double-blind manner. The parameters assessed were; postoperative pain by VAS score, total analgesic consumption, hemodynamic parameters, sedation level, nausea and vomiting and dizziness at 0, 2, 4, 6, 12, 18, and 24 hours. Overall patient satisfaction with pain management was also assessed as a secondary outcome.
Results: Postoperative VAS scores for pain were reduced in pregabalin group; at 0, 2, 4 hours (p < 001) and 6, 12, 18 and 24 hours (p < 0.05). Analgesic consumption was also reduced in the study group (68.83 ± 37.36 vs. 175.87 ± 35.31, p < 0.001). The frequency of nausea and vomiting (p < 0.05 – < 0.001) was reduced in pregabalin group compared with placebo. The preoperative anxiety; postoperative hemodynamic parameters, and sedation scores were comparable in both of the groups. The patients in pregabalin group were more satisfied with overall pain management.
Conclusion: The results of our study show that a single dose of pregabalin 150 mg can effectively attenuate postoperative pain and reduce tramadol requirement as well as nausea and vomiting, without any untoward side-effects, and with higher satisfaction level of the patients.
Key words: Pregabalin; Anxiety; Cholecystectomy; Pain, Postoperative; Analgesia,
Citation: Anand LK, Sandhu M, Singh J, Mitra S. Evaluation of analgesic efficacy of pregabalin for postoperative pain relief after laparoscopic cholecystectomy: a double blind study. Anesth Pain & Intensive Care 2017;21(2):174─180
Received: 1 Aug 2016, Reviewed: 2, 3 Dec 2016, Corrected: 5 Dec 2016, Accepted: 28 Jun 2017
INTRODUCTION
Postoperative pain after laparoscopic cholecystectomy (LC) is the most prevailing complaint.1 Although, LC is assorted with less postoperative pain compared with the conventional open cholecystectomy, effective postoperative pain management remains a clinical challenge.2 Intense acute pain after LC might predict the development of chronic pain e.g., post laparoscopic cholecystectomy syndrome (PLCS).3
Pregabalin is a γ-aminobutyric acid (GABA) analogue that binds to α2 δ subunits of the voltage-gated calcium channels.4 The mechanisms of action of pregabalin is different from either opioids, nonsteroidal anti-inflammatory drugs or local anesthetics and it may be useful as an adjunct to control postsurgical pain.5 Pregabalin is rapidly absorbed orally with bioavailability of 90%, attains peak plasma levels within 30 min to 2 h and shows linear pharmacokinetics.6 It has an elimination half-life estimated to range from 5.5 to 6.7 hours, which is independent of the dose and frequency of administration.7
Pregabalin has been investigated in laparoscopic surgery patients for postoperative pain relief and analgesic consumption with variable results.8-11 Study using low-dose pregabaline shows limited analgesic benefit9 and those with higher dose in various combinations, found it to have an analgesic and opioid sparing effect but associated with an increased incidence of side effect.10,11 We hypothesised that preoperative treatment with pregabalin 150 mg would decrease postoperative pain and analgesic consumption and the incidence of side effects after LC.
In this study, we evaluated the efficacy of single oral dose of pregabalin in patients undergoing LC for postoperative pain relief and analgesic consumption as primary outcomes. Secondary outcomes were assessment of preoperative anxiety, postoperative hemodynamic parameters, sedation, nausea and vomiting and overall satisfaction with pain management.
METHODOLOGY
After the approval of the Institutional Ethics Committee and written informed consent from 60 patients who had an American Society of Anesthesiologists (ASA) physical status of I and II, aged 20-60 years, scheduled for elective LC, were included.
Patients were excluded if they had a history of hepatic or renal insufficiency, uncontrolled medical disease (diabetes mellitus and hypertension), psychiatric disorders, chronic pain and on analgesic medications, known allergy to study drug or body mass index (BMI) > 35 kg.m-2
The study design was prospective, randomized, double blind and placebo controlled. Patients meeting the inclusion criteria during the preanesthetic evaluation were explained about visual analogue scale (VAS) for pain (where 0 represents no pain and 100 mm the worst imaginable pain) and similarly visual analogue scale for anxiety (VAS-A).12 Using a computer generated random numbers table, patients were randomly allocated to Group A (Alprazolam n = 30) or Group P (Pregabalin n = 30), to receive either a matching alprazolam 0.5 mg as placebo or pregabalin 150 mg. Double blinding was ensured by using sequentially numbered, coded, and sealed envelopes containing placebo or study drug were administered orally, 2 hour before the expected time of surgery with sips of water.
In the operating room anxiety was evaluated using a VAS-A score,12 standard monitors of continuous ECG, heart rate (HR), oxygen saturation (SpO2), non-invasive blood pressure (NIBP) and end-tidal CO2 (EtCO2) (Aestiva 5; Datex-Ohmeda, Madison, WI, USA) were placed before induction of general anesthesia. After obtaining intravenous (IV) access, glycopyrrolate 0.2 mg IV was administered. Standard anesthesia technique was used in all patients. After preoxygenation, morphine 0.1 mg/kg and propofol 2.0-2.5 mg/kg were given. After checking for ability to achieve adequate mask ventilation, vecuronium 0.1 mg/kg was administered to facilitate laryngoscopy and tracheal intubation. A close circle system was connected and ventilator was adjusted to achieve effective oxygenation and ventilation using volume-controlled positive pressure ventilation with 60% nitrous oxide and isoflurane (1-2% end-tidal) in 40% oxygen using fresh gas flow of 2 lit/min, tidal volume of 8 to 12 ml/kg, respiratory rate 12 to 16 breaths/min and inspiratory to expiratory time (I:E) ratio of 1:2. Cardiovascular parameters were maintained within 20% of the base line values by supplemental intraoperative analgesia with injection morphine 1-2 mg IV and adjusting isoflurane (1-2% end-tidal) as indicated. Intra-operative continuous ECG, HR, NIBP, SpO2 and EtCO2 were monitored continually and recorded every five minutes till the end of surgery and the duration of surgery was noted.
At the end approximately 15 min before the completion of surgery all patients were given inj. ondansetron 6 mg IV, to reduce emesis. Residual neuromuscular block was antagonized with mixture of neostigmine (0.05 mg/kg) and glycopyrrolate (0.01 mg/kg) at the end of surgery. After satisfactory recovery, the patients were extubated and shifted to the post anesthesia care unit (PACU). In the PACU patients were observed for 2 hours and monitored with ECG, NIBP, SpO2 and zero hour (0) was taken when patients were conscious and responding to verbal commands. Postoperative analgesia was provided with inj tramadol hydrochloride 1 mg/kg IV and was given at any point of time with VAS score for pain greater than 30. If required, inj ondansetron (6 mg) was given IV for nausea and vomiting. Total analgesic consumption was recorded for 24 hours.
Postoperatively patients were assessed by blinded observer at 0, 2, 4, 6, 12, 18, and 24 hours after the completion of surgery. Patients were asked to quantify the pain from the operative site with visual analogue scale (0-100). At every evaluation of postoperative pain; heart rate, blood pressure, sedation, nausea and vomiting and any other side effect like dizziness and blurring of vision was assessed and recorded. Sedation was assessed by using the 4 point sedation (somnolence) score.12: 0 = No sedation: Patient awake and oriented, 1 = Mild sedation: aroused by verbal stimuli; oriented, 2 = Moderate sedation: aroused by physical stimuli; oriented when awakened, 3 = Marked sedation: aroused by physical stimuli; disoriented when awakened. The severity of postoperative nausea and vomiting (PONV) was assessed by 4 point scale: 0= no nausea or vomiting, 1= slight nausea resolving without treatment, 2= slight nausea and /or vomiting resolves on treatment, 3= nausea and/or vomiting not resolving on treatment. Total tramadol consumption was recorded at the end of 24 hours in each patient. After 24 hours overall patient satisfaction with pain management was assessed using a VAS (0-100 mm) where zero represents very dissatisfied and 100 very satisfied. A primary outcome of the study was postoperative pain relief and analgesic consumption in patient undergoing LC. Secondary outcomes were assessment of preoperative anxiety, postoperative hemodynamics (Pulse rate and mean arterial pressure [MAP]), sedation, nausea and vomiting and overall satisfaction with pain management.
Our sample size was based on a pilot study and on the results of previous study8 in which the postoperative VAS scores after preoperative administration of pregabalin 150 mg would be 30 mm when compared with 45 mm in the placebo group with a standard deviation of 20 mm. For the results to be of statistical significance with α= 0.05 and β=0.80, our sample size came out to be 28 patients in each group. To allow for potential dropout, we recruited 30 patients per group
All the descriptive data was expressed as mean ± SD unless otherwise stated. All continuous data was tested for normal distribution. Student´s t test was used for comparison of means of continuous variables for normally distributed data; otherwise Mann-Whitney U test was used. ANOVA was used for variable differences within groups. Categorical data was analyzed using chi square test analysis or Fisher´s exact test, as considered appropriate. Mann- Whitney test was used to compare the preoperative VAS scores for anxiety, postoperative pain and postoperative drowsiness. Significance was determined at p < 0.05.
RESULTS
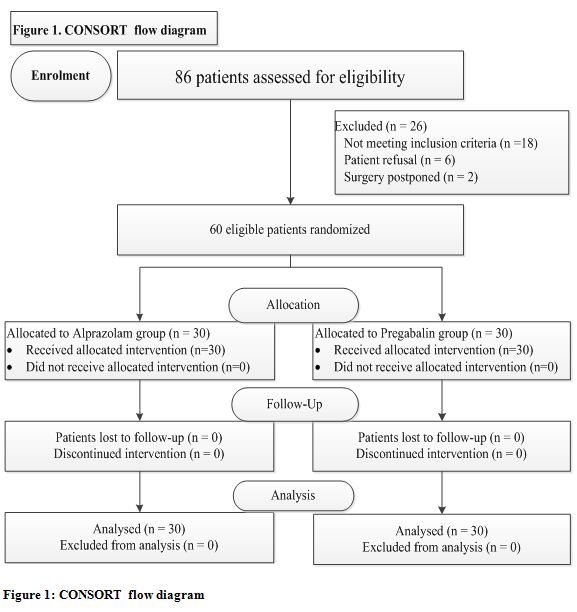
Eighty six patients were assessed for eligibility in the study. Twenty six were not randomized and not studied, which left 60 patients (30 patients per group) for data analysis depicted in Consort flow diagram (Figure 1).
Figure 1: CONSORT flow diagram
There was no statistically significant difference amongst the groups of patients with regard to age, sex, ASA physical status, body weight, height, BMI, surgical time and anesthesia time [p > 0.05) (Table 1).
Table 1: Demographic data and timing
| Variable | Group A
(N=30) |
Group P
(N=30) |
p- value |
| Age (y) | 35.43 ± 8.51 | 33.57 ± 7.42 | > 0.05 |
| Sex ( M/ F) | 25/5 | 26/4 | > 0.05 |
| ASA* I/II | 22/8 | 19/11 | > 0.05 |
| Weight (Kg) | 60.87 ± 9.92 | 64.07 ± 10. | > 0.05 |
| Height (cm) | 160.2 ± 5.3 | 161.5 ± 6.9 | > 0.05 |
| BMI† (kg/m-2) | 23.72 ± 4.2 | 24.64 ± 4.3 | > 0.05 |
| Surgical time (min) | 61.93 ± 14.4 | 58.0 ± 14.2 | > 0.05 |
| Anesthesia time (min) | 84.73 ± 14.2 | 75.8 ± 14.3 | > 0.05 |
Data are presented as the mean ± standard deviation, unless otherwise denoted.
*ASA=American Society of Anesthesiologists; †BMI=body mass index
The preoperative anxiety assessed by VAS-A score and intraoperative morphine consumption was comparable (p > 0.05) (Table 2).
Table 2: Comparison of preoperative anxiety VAS-A analgesic consumption, patient satisfaction with pain management between the groups
| Variable | Group A
(N=30) |
Group P
(N=30) |
p-value |
| Preoperative anxiety VAS-A* | 29.90 ± 3.38 | 30.97 ± 5.14 | > 0.05 |
| Morphine (mg) intraoperative | 5.83 ± 1.04 | 5.85 ± 1.10 | > 0.05 |
| Tramadol (mg/24 h) | 175.87 ± 35.31 | 68.83 ± 37.36* | < 0.001 |
| Overall satisfaction with pain management at 24 h | 26.93 ± 8.53 | 80.20 ± 10.48* | < 0.001 |
Data are presented as the mean ± standard deviation, *VAS-A =visual analogue scale for anxiety
The pregabalin group had a significantly reduced VAS scores for postoperative pain. The VAS scores differed statistically significant at 0, 2, 4 hours (p < 001) and thereafter at 6, 12, 18 and 24 hours (p < 0.05) which were lower in pregabalin group compared with placebo (Table 3). Postoperative tramadol consumption was reduced (68.83 ± 37.36 vs. 175.87 ± 35.31) in pregabalin and placebo group respectively (p < 0.001) (Table 2). Three patients in placebo group required rescue analgesic (morphine 1.5 mg) bolus in addition to tramadol.
Table 3: Postoperative pain VAS* scores
| Time points | Group A
(N=30) |
Group P
(N=30) |
p-value |
| 0 h | 44.87 ± 17.85 | 8.17 ± 9.78 | < 0.001 |
| 2 h | 52.60 ± 14.12 | 18.77 ± 14.38 | < 0.001 |
| 4 h | 37.23 ± 13.98 | 22.33 ± 14.96 | < 0.001 |
| 6 h | 39.53 ± 15.27 | 29.83 ± 15.20 | < 0.05 |
| 12 h | 37.93 ± 11.84 | 31.33 ± 14.07 | < 0.05 |
| 18 h | 35.93 ± 12.74 | 29.33 ± 14.17 | < 0.05 |
| 24 h | 40.53 ± 15.36 | 35.90 ± 12.96 | < 0.05 |
Data are presented as the mean ± standard deviation
*VAS =visual analogue scale
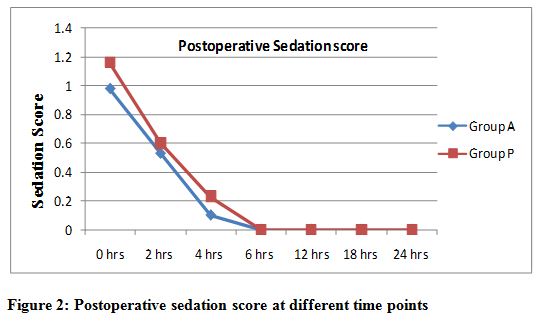
The sedation score as measured by somnolence sedation scale at zero time points recorded less in the placebo group (0.98 ± 0.41) compared to pregabalin group (1.18 ± 0.37), but was statistically insignificant (p=0.067) which decreased gradually in due course of time (p > 0.05). No patient had a sedation score of 3, and all patients were free of sedation at 6 hours and thereafter (Figure 2). Two patient in placebo and 6 patients in pregabalin group had headache and dizziness; three patients complained of visual disturbances in pregabalin group while none in placebo.
Figure 2: Postoperative sedation score at different time points
Postoperatively all the patients in both of the groups were hemodynamically stable. Pulse rate and mean arterial pressure (MAP) measured at different time points were comparable in both the groups (p > 0.05).
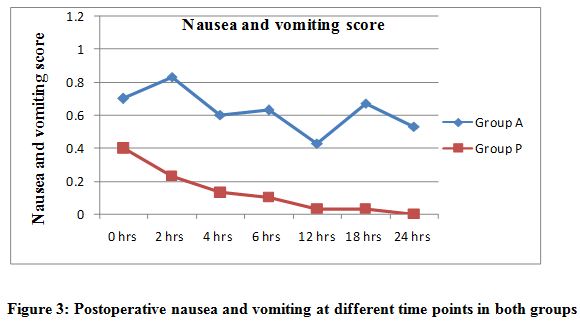
The placebo group recorded more patients with increased severity of nausea and vomiting than the pregabalin group (p < 0.001) except at zero four hours. Thirteen patient in placebo group had a score of 2 and required antiemetic, whereas 12 patients had a score of 1 and none required antiemetic in pregabalin group (Figure 3).
Figure 3: Postoperative nausea and vomiting at different time points in both groups
Overall satisfaction with pain management as assessed by VAS score was (80.20 ± 10.48 vs. 26.93 ± 8.53) in pregabalin and placebo group respectively (p < 0.001) (Table 2). The patients in pregabalin group were more satisfied with overall pain management as compared to placebo.
DISCUSSION
The results of our study demonstrated that preoperative pregabalin (150 mg) was effective in reducing postoperative pain along with postoperative tramadol consumption and PONV in patients undergoing LC. In addition, the overall satisfaction with pain management was better in pregabalin group. No significant differences existed between the two groups with respect to preoperative anxiety; postoperative hemodynamic, sedation.
Pain after LC is definitely significantly decreased compared with the open cholecystectomy, but pain control after LC still remains a clinical challenge. Optimum postoperative pain relief is important as it may reduce postoperative complications and encourage early discharge from hospital. The ideal method for pain relief should be simple to perform, inexpensive, and causes minimal morbidity.14 Pregabalin has some proven role in the control of anxiety and postoperative pain either singly or in combination with other antinociceptive drug for synergistic effects; various clinical studies with the drugs for postoperative analgesia and analgesic consumption have shown promising results.5,8,10
In the present study, pregabalin demonstrated that reductions in the VAS score for pain and tramadol consumption at different time points were found to be highly significant when compared with the placebo group. Several clinical studies have compared pregabalin for postoperative pain and analgesic consumption with conflicting results.8-11,15-19 Some studies showed no significant effect on the VAS score for postoperative pain.9,11,17,18 Other studies8,10,15,16,19 observed significantly reduced VAS score for postoperative pain in pregabalin group as compared with the controlled group; similar results were observed in the present study using single dose pregabaline 150 mg preoperatively. In agreement with ours, the study conducted by Agarwal A et al.8 to compare 150 mg of pregabalin in a randomized placebo controlled, double-blind study for post LC pain control, the pain scores were significantly lesser in pregabalin group without any increased side effects. To determine the efficacy of two different doses by Jokela R et al.10 with pregabalin 75 mg or 150 mg, compared with diazepam 5 mg, concluded that analgesia was better with pregabalin 150 mg. Further, studies was conducted by Balaban F et al.15 and Singh TH et al.16 in a placebo-controlled manner with two different doses 150 mg, and 300 mg of pregabalin for pain scores and postoperative fentanyl consumption in patients after LC. These studies show that pregabalin 150 mg was an effective method for reducing postoperative pain and analgesic requirement without any untoward effect. In contrast, Chang et al.11 failed to confirm these results using two perioperative doses of 300 mg pregabalin in a placebo controlled study.
Recently, a systematic review conducted by Mishriky BM et al.19 observed that pregabalin tested (75-150, and 300 mg) as single or multiple doses resulted in reduction of postoperative pain and analgesic requirement at 24 hours after surgery. Specifically results showed that, there were no significant differences in postoperative pain outcomes with pregabalin 100-300 mg between single preoperative dosing regimens and those including additional doses repeated postoperatively. These results suggest that for postoperative pain outcomes and analgesic requirement, there is no significant benefit of repeated and higher doses of pregabalin compared with a single preoperative dose.
Postoperatively at 24 hours after surgery tramadol consumption was significantly less in pregabalin group than the placebo, additionally rescue boluses of morphine (1.5 mg) required in placebo group. These findings are supported by studies of Agarwal et al8 and Balaban F et al.15 that pregabalin reduces the opioid consumption after LC. Preoperative anxiety is a usual component of the surgical experience, and increased levels of anxiety may cause increased postoperative pain and subsequent analgesic requirment.20 Pregabalin also has been used for its anxiolytic and analgesic effects by Spreng UJ et al.21 they concluded that single oral dose of pregabalin (150 mg) reduced post-operative pain and morphine consumption and lower preoperative anxiety, without increased incidence of untoward side effects. To palliate the effect of preoperative anxiety on postoperative pain we used the alprazolam (0.5 mg) as placebo; the preoperative anxiety score assessed by VAS-A was comparable in both the groups.
In our study the sedation score at zero hour was more in pregabalin group patients as compared to placebo, but was statistically insignificant which decreased gradually and at 2 hours, and all patients were free of sedation at 6 hours and thereafter. Two patient in placebo and 6 patients in pregabalin group had headache and dizziness, three patients complained of visual disturbances while none in placebo. Gajraj NM et al.6 reviewed the pharmacology of pregabalin and found that it is associated with common side effects like somnolence and dizziness. The side-effects of pregabalin are dose dependent, mild to moderate and are usually transient. Single dose of pregabalin 150 mg is unlikely to cause sedation, headache, dizziness, or visual disturbances as observed by Agarwal A et al.8 and Balaban F et al.15 However, studies evaluating higher and multiple doses of pregabalin are associated with increased sedation, headache, dizziness, or visual disturbances.10,18
PONV is a usual complication of surgery and anesthesia. Although it is rarely fatal, it is unpleasant, associated with patient discomfort, and dissatisfaction with perioperative management. The nausea and vomiting score were minimal in the pregabalin group patients as compared with the placebo; the same had been reported by White PF et al.18 and Ittichaikulthol W et al.22 This may be explained on the association of greater VAS score in the placebo group with increased demand of rescue opioid analgesics.
The hemodynamic (pulse rate and MAP) parameters in both the groups were comparable and statistically not significant. Our findings are consistent with that of the independent studies conducted by Kholi M et al,23 and Ruben SS et al,24 affirming the findings that pregabalin has no action on blood pressure or heart rate. However, study conducted by Gupta K et al.25 the role of pregabalin in attenuating hemodynamic response to laryngoscopy and intubation has been highlighted; which may due to the increased sedation rather than the actual inhibition of sympatho-adrenal axis.
Present study using pregabalin 150 mg demonstrated lower VAS score compared to the placebo group and studies evaluating higher and multiple doses of pregabalin are associated with increased sedation and side effects.15,16 Considering the above concepts and discussion, pregabalin 150 mg may be a worthy option for postoperative pain in patient undergoing LC with negligible side effects.
LIMITATIONS
Our study has several limitations. Firstly, we did not evaluate the dose-response or the effect of continuation of therapy beyond 24 hours. Secondly, in addition to the resting pain, pain on movement and other tools for assessment of quality of analgesia should been included. Further studies are suggested in these areas.
CONCLUSION
Present study showed that a single oral dose of pregabalin 150 mg in patients undergoing laparoscopic cholecystectomy was effective in reducing postoperative pain, tramadol consumption, as well as nausea and vomiting, without any untoward side-effects.
Conflicts of interest: Not declared
Author contribution: All authors took equal part in concept, conduction of the study work and manuscript writing / editing
REFERENCES
- Lau H, Brooks DC. Predictive factors for unanticipated admissions after ambulatory laparoscopic cholecystectomy. Arch Surg. 2001 Oct;136(10):1150-3. [PubMed] [Free full text]
- Bisgaard T. Analgesic treatment after laparoscopic cholecystectomy:a critical assessment of the evidence. Anesthesiology. 2006 Apr;104(4):835-46. [PubMed] [Free full text]
- Bisgaard T, Rosenberg J, Kehlet H. From acute to chronic pain after laparoscopic cholecystectomy: a prospective follow-up analysis. Scand J Gastroenterol. 2005 Nov;40(11):1358-64. [PubMed]
- Ha KY, Carragee E, Cheng I, Kwon SE, Kim YH. Pregabalin as a neuroprotector after spinal cord injury in rats: biochemical analysis and effect on glial cells. J Korean Med Sci. 2011 Mar;26(3):404-11. doi: 10.3346/jkms.2011.26.3.404 [PubMed] [Free full text]
- Gilron I. Gabapentin and pregabalin for chronic neuropathic and early postsurgical pain: current evidence and future directions. Curr Opin Anaesthesiol. 2007 Oct;20(5):456–472. [PubMed]
- Gajraj NM. Pregabalin: its pharmacology and use in pain management. Anesth Analg. 2007 Dec;105(6):1805-15. [PubMed]
- Frampton JE, Scott LJ. Pregabalin: In the treatment of painful diabetic peripheral neuropathy. Drugs. 2004;64(24):2813-20. [PubMed]
- Agarwal A, Gautam S, Gupta D, Agarwal S, Singh PK, Singh U. Evaluation of a single dose of pregabalin for attenuation of postoperative pain after laparoscopic cholecystectomy. Br J Anesth. 2008 Nov;101(5):700-4. doi: 10.1093/bja/aen244 [PubMed] [Free full text]
- Peng PW, Li C, Farcas E, Haley A, Wong W, Bender J, et al. Use of low-dose pregabalin in patients undergoing laparoscopic cholecystectomy. Br J Anaesth. 2010 Aug;105(2):155-61. doi: 10.1093/bja/aeq116 [PubMed] [Free full text]
- Jokela R, Ahonen J, Tallgren M, Haanpaa M, Korttila K. A randomized controlled trial of perioperative administration of pregabalin for pain after laparoscopic hysterectomy. Pain. 2008 Jan;134(1-2):106-12. [PubMed]
- Chang SH, Lee HW, Kim HK, Kim SH, Kim DK. An evaluation of perioperative pregabalin for prevention and attenuation of postoperative shoulder pain after laparoscopic cholecystectomy. Anesth Analg. 2009 Oct;109(4):1284-6. doi: 10.1213/ane.0b013e3181b4874d [PubMed]
- Facco E, Stellini E, Bacci C, Manani G, Pavan C, Cavallin F, et al. Validation of visual analogue scale for anxiety (VAS-A) in preanesthesia evaluation. Minerva Anestesiol. 2013 Dec;79(12):1389-95. [PubMed]
- Dougherty TB, Porche VH, Thall PF. Maximum tolerated dose of nalmefene in patients receiving epidural fentanyl and dilute bupivacaine for postoperative analgesia. Anesthesiology, 2000;92:1010-6. [PubMed] [Free full text]
- Casey KL. Concepts of pain mechanisms: the contribution of functional imaging of the human brain. Prog Brain Res. 2000;129:277-87. [PubMed]
- Balaban F, Yagar S, Ozgok A, Koc M, Gullapoglu H. A randomized, placebo-controlled study of pregabalin for postoperative pain intensity after laparoscopic cholecystectomy. J Clin Anesth, 2012 May;24(3):175-8. doi: 10.1016/j.jclinane.2011.06.027 [PubMed]
- Singh TH, Thokchom R, Rajkumar G, Singh YAK, Meitei AJ, Singh NR, et al. Pregabalin for post-cholecystectomy pain relief- A study on the response of two different doses. Int J Health Sci Res. 2014;4:159-68. [Free full text]
- Peach MJ, Goy R, Chua S, Scoot K, Christmas T, Doherty DA. A randomized, placebo-controlled trial of preoperative oral pregabalin for postoperative pain relief after minor gynaecological surgery. Anaesth Analg. 2007 Nov;105(5):1449-53. [PubMed]
- White PF, Tufanogullari B, Taylor J, Klein K. The effects of pregabalin on preoperative anxiety and sedation levels: a dose ranging study. Anaesth Analg. 2009 Apr;108(4):1440-45. doi: 10.1213/ane.0b013e31818d40ce. [PubMed]
- Mishriky BM, Waldron NH, Habib AS. Impact of pregabalin on acute and persistent postoperative pain: a systematic review and meta-analysis. Br J Anaesth. 2015 Jan;114(1):10-31. doi: 10.1093/bja/aeu293. [PubMed] [Free full text]
- Vaughn F, Wichowski H, Bosworth G. Does preoperative anxiety level predict postoperative pain? AORN J. 2007 Mar;85(3):589-604. [PubMed]
- Spreng UJ, Dahl V, Raeder J. Effect of a single dose of pregabalin on post-operative pain and pre-operative anxiety in patients undergoing discectomy. Acta Anaesthesiol Scand. 2011 May;55(5):571-6. doi: 10.1111/j.1399-6576.2011.02410.x [PubMed]
- Ittichaikulthol W, Virankabutra T, Kunopart M, Khamhom W, Putarawuthichai P, Rungphet S. Effects of pregabalin on postoperative morphine consumption and pain after abdominal hysterectomy with/without salphingo-oophorectomy: a randomized, double-blind trial. J Med Assoc Thai. 2009 Oct;92(10):1318-23. [PubMed]
- Kohli M, Murali T, Gupta R, Khan P, Bogra J. Optimization of subarachnoid block by oral pregabalin for hysterectomy. J Anaesthesiol Clin Pharmacol. 2011 Jan;27(1):101-5. [PubMed] [Free full text]
- Reuben SS, Buvanendran A, Kroin JS, Raghunathan K. The analgesic efficacy of celecoxib, pregabalin, and their combination in spinal fusion surgery. Anaesth Analg. 2006 Nov;103(5):1271-7. [PubMed]
- Gupta K, Bansal P, Gupta PK, Singh YP. Pregabalin premedication – A new treatment option for hemodynamic stability during general anesthesia: A prospective study. Anesth Essays Res. 2011 Jan-Jun;5(1):57-62. doi: 10.4103/0259-1162.84192 [PubMed] [Free full text]
 This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.© Anaesthesia, Pain & Intensive Care, 2010-2013



 Facebook
Facebook Twitter
Twitter GooglePlus
GooglePlus Youtube
Youtube