Ayşe Hande Arpacı1, Berrin Işık2, Mustafa Arslan2, Ömer Hakan Emmez3, Özlem Erdem4, Metin Alkan2
1Anaesthesiology and Reanimation Specialist, Department of Oral and Maxillofacial Surgery, Faculty of Dentistry, Ankara University, Besevler, Ankara, (Turkey)
2Department of Anesthesiology & Reanimation, Faculty of Medicine, Gazi University,
Ankara, (Turkey)
3Department of Neurosurgery, Faculty of Medicine, Gazi University, Ankara, (Turkey)
4Department of Pathology, Faculty of Medicine, Gazi University, Ankara, (Turkey)
Correspondence: Mustafa Arslan, Department of Anesthesiology and Reanimation, Gazi University Medical Faculty, Ankara-06510 (Türkiye); Phone: 90 533 422 85 77; E-mail: mustarslan@gmail.com
ABSTRACT
Objective: Unintentional dural puncture with an epidural needle is common and indisputably one of the most important complications of epidural anesthesia. When no regression of the complaints are observed after conservative treatment, epidural blood patch (EBP) should be planned for the treatment of patients. However, EBP is not the optimal choise because the success rate is low and the risk of potential serious complications is high. Ankaferd Blood Stopper® (ABS), which is a medicinal plant extract, has been used as a antihemorrhagic agent against various types of bleeding. The purpose of this study is to investigate the effects of ABS administration, on fibrin formation and cerebro-spinal fluid (CSF) leakage after dural puncture in a rat model.
Methodology: The study was performed at Gazi University Experimental Research Center, Ankara, Turkey between May and July 2013. Eighteen rats were grouped randomly as control (Group C, n=6), dural punctured (Group DPC, n=6) and ABS application after dural puncture at the level of L4-L5 (Group A, n=6). Methylene blue was applied to the cervical level of the rat spine, in order to observe leakage from dural puncture in Group DPC, and Group A. Medulla spinalis tissues of the rats were evaluated histopathologically.
Results: CSF leakage was obtained in all rats in Group DPC (100%) that were dural punctured with spinal needle and given methylene blue. However, in the group of rats that ABS was performed before methylene blue (Group A) application, CSF leakage could be obtained in only half of the rats (50%), and in none of the rats in Group C. CSF leakage ratio in Group DPC was 100% and significantly higher than that in Group C (X2=5.178, p=0.002). Following Ankaferd administration, CSF leakage was identified in only 50% of rats in group A and this result was similar with achieved result in Group C (X2=1.778, p=0.182)
Conclusion: We opine that Ankaferd Blood Stopper® (ABS) is effective in preventing CSF leakage after post dural puncture and does not cause necrosis in rat medulla spinalis tissues.
Key words: Dural puncture; Ankaferd; CSF leakage; Epidural blood patch
Citation: Arpacı AH, Işık B, Arslan M, Emmez OH, Erdem O, Alkan M. Effects of Ankaferd® on controlling cerebrospinal fluid leakage after dural puncture in rats. Anaesth Pain & Intensive Care 2016;20(4):387-392
Received: 7 Jun 2016; Reviewed: 10, 15 Jun 2016; Corrected: 17 Jun 2016; Accepted: 4 Nov 2016
INTRODUCTION
Unintentional dural puncture with an epidural needle or catheter is common1-4 and indisputably one of the most important complications of epidural anesthesia. The leakage of cerebrospinal fluid (CSF) from the puncture hole results in a decreased volume of CSF, which has an adverse effect on the brain. This results with tension on the meninges, which causes headaches. Headaches are occasionally accompanied by serious morbidity, such as cranial nerve palsy, subdural hematoma or chronic headaches. Epidural blood patch (EBP) treatment should be planned for patients who have no reduction of the complaints following conservative treatment (bed rest, increased oral fluid intake, analgesic drugs and caffeine intake). The EBP forms a gelatinous tamponade, which prevents further leakage of spinal fluid, allowing the dura to undergo normal healing.5 On the other hand, EBP should not be taken into account as the excellent treatment choise because of its undesired effects.6
Ankaferd® [Ankaferd Blood Stopper (ABS), Ankaferd Health Products, Ltd., İstanbul, Turkey] is a traditional herbal extract that has been approved (Turkish Ministry of Health) for the management of bleedings during oral surgery procedures and external source hemorrhages.7-10 The substances that constitute the extract have some specific effects on endothelium, blood cells, angiogenesis, cellular reproduction, vascular dynamics, and intercellular interaction. ABS shows its efficacy on both fibrinogen and agglutinating proteins. Blood cells and blood proteins help ABS to provide cloth formation in the hemorrhagic region.10-12
ABS has been used as a hemostatic agent against various types of bleeding. Furthermore, it has been used successfully for the treatment or prevention of hemorrhage in dental patients with hemophilia.10 It has also been used as a topical agent for the prevention of postoperative intra-abdominal fibrosis, which has been associated to inflammatory responds in experimental studies, and various results have been obtained.7,13 Besides its homeostatic activity, Kocak et al.14) reported that ABS might also have anti-inflammatory effects.
As far as is known, there are no reports concerning the effect of ABS in the treatment of post-dural puncture cerebrospinal fluid leakage (PDP-CSF-L). We hypothesized that ABS could be used in the treatment of PDP-CSF-L and that the anti-inflammatory and blood stopper elements of it could be present experimental study was designed to investigate the effect of intradurally administered ABS on treating CSF leakage on a rat model.
METHODOLOGY
The study protocol was approved by the Gazi University Animal Ethics Committee, Ankara, Turkey and performed according to the guidelines of the Faculty of Medicine, Research Committee of Gazi University between May and July 2013. In the study, 18 male Wistar Albino rats were used. The rats were fed solid rat chow and water, ad libitum and were maintained under climate controlled conditions (12 h light/dark cycles, 22 ± 3°C).
The animals were weighed (370–420 g) and anesthetized with intramuscular injection of 100 mg/kg ketamine (Ketalar®, Parke Davis Eczacıbaşı, Istanbul, Turkey). Animals were randomly assigned to one of the three groups; control group (Group C), dural puncture control group (Group DPC), and dural puncture plus ABS group (Group A).
The rats were placed in prone position; a midline incision was made along the spinous process of the lumbar area. After dissecting the fascia and paraspinal muscles, the spinous processes were removed. Laminectomy was performed with a drill through L3-L5 vertebral levels. The dura mater was punctured with 27 Gauge needle in Group DPC and in Group A. 5 ml’s of ABS/serum physiologic solution (1/4) was administered to the peridural tissue in Group A.
A second midline incision was performed from occiput to cervical vertebra. Occipitocervical membrane was exposed. Finally, rats were injected intrathecally at the level of occipitocervical junction through cisterna magna with 10 µL of methylene blue via a syringe connected with the punctured needle in Group DPC and Group A (Figure 1).
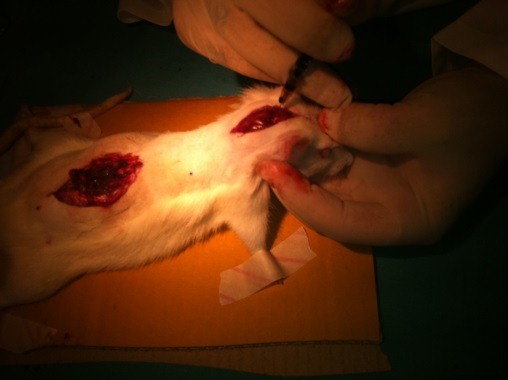
Figure 1: Incision in the midline of the lumbar area and intradural methylene blue application from the occipitocervical junction through cisterna magna in Group DPC and Group A.
The above mentioned procedure was performed to all test subjects by the same surgeon in a similar manner. L4-L5 spine level, which is the dural puncture field in Group DPC (Figure 2) and Group A (Figure 3), was evaluated 30 min after dural puncture as in terms of dural leakage in all three groups.
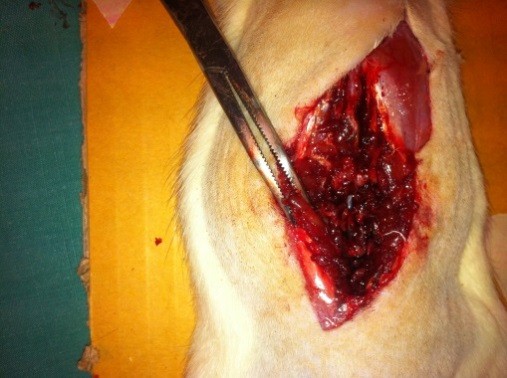
Figure 2: Macroscopic evaluation of the rat in Group DPC
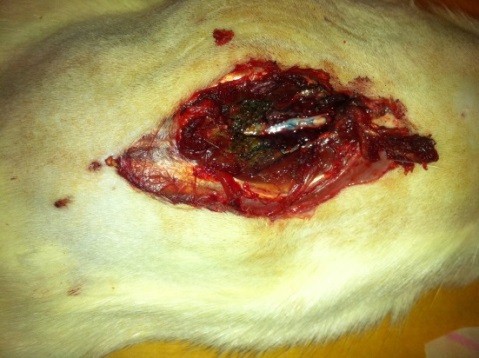
Figure 3: Macroscopic evaluation of the rat in Group A
After bolus doses of ketamine, rats were sacrificed. Spines were removed en bloc, the spinal cords were completely dissected and methylene blue distribution was investigated and compared. To determine whether acute needle puncture induced a significant local inflammatory response, hematoxylin and eosin (H&E) staining was performed on the lumbar spinal cords. The rat spines were fixated with 10% formalin, decalcified, and paraffin embedded. Tissues were sliced at 4 µm thickness and stained with H&E. H&E stained sections were selected from the L4-L5 spinal cord of the rat to analyze the inflammation in the spinal dorsal horn (Figures 4-6).
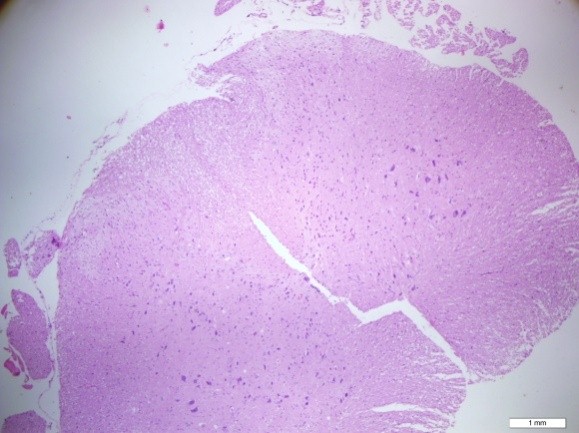
Figure 4: H&E staining of the lumbar spinal cord (Group C)
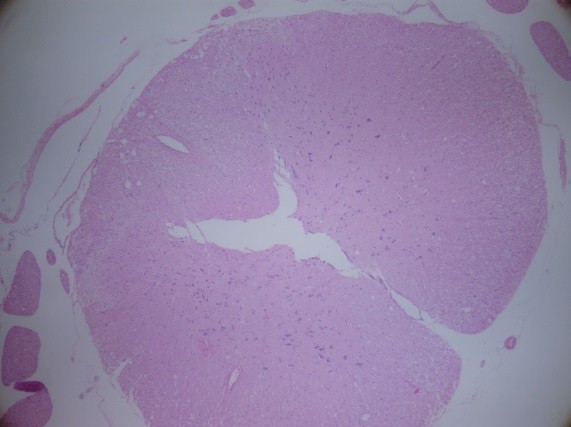
Figure 5: H&E staining of the lumbar spinal cord (Group DPC)
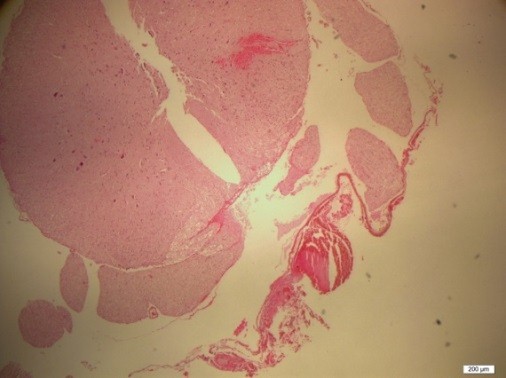
Figure 6: H&E staining of the lumbar spinal cord (Group A)
Statistical analysis:
Statistical Package for Social Sciences (SPSS, Chicago, IL, USA) 20.0 program was used for statistical analysis. CSF leakage was compared using Chi-square and Fisher’s exact tests. Statistical significance was set at a P value < 0.05.
RESULTS
There was no difference in the mean weights of the three groups when compared.
CSF leakage was obtained in all rats in Group DPC (100%) that were dural punctured with spinal needle and given methylene blue. However, in the group of rats that ABS was performed before methylene blue (Group A) application, CSF leakage could be obtained in only half of the rats (50%), and in none of the rats in Group C. CSF leakage ratio in Group DPC was 100% and significantly higher than that in Group C (X2 = 5.178, p = 0.002). Following Ankaferd administration, CSF leakage was identified in only 50% of rats in group A and this result was similar with achieved result in Group C (X2 = 1.778, p = 0.182).
At L4-5 spinal cord level, inflammation was not observed in any of the groups and groups were similar as in terms of inflammation (Figures 4-6).
DISCUSSION
Post-dural puncture cerebrospinal fluid leakagessociated with significant morbidity and may result in poor outcomes. Therapeutic difficulties may delay the appropriate management of the leaks.1-3,15-17 The epidural blood patch is considered as an effective method in treating PDP headache.4,5 Initially, Gormley18 reported placing 2-3 ml autologous blood in the epidural space for PDP headache with a 100% success rate. Despite Gormley’s success rate, an initial large volume (20 ml) lumbar EBP has an efficacy closer to 60%, and success may increase to over 90% if a second EBP is administered.18-22 Physiologic mechanisms by which the large volume EBP is effective include a physical “patch” effect, in which injected blood directly forms a seal over the dural leak and a “pressure” effect, in which increased epidural pressure is transmitted to the CSF space. The required volume of blood by which these effects are properly achieved has been a matter of continued debate.4,5,19-21
On the other hand, EBP is not an excellent method because of slightly lower success rate and potentially serious complications like persistent back pain, radicular pain, epidural abscess, seizures, permanent paraparesis, facial palsy, cauda equine syndrome, spinal subdural hematoma and arachnoiditis.23-27
Many techniques and methods, which can assist in decreasing the complications of PDP, have been proposed.2,4,5 Various materials have been investigated to prevent or reduce the severity of PDP related headache. For such purposes, intravenous or oral caffeine, sumatriptan, epidural saline, dextran 40, opioids and fibrin glue have been tried with variable success rates. Unfortunately, despite continuous advances and research, no ideal method to prevent or reduce PDP related headache has been reported up to this day.28
We have developed a PDP model in rats to evaluate of the administration of epidural ABS in controlling PDP-CSF-L and to compare the efficacy of application of epidural ABS for restoring the PDP-CSF-L. Our results confirm the efficacy of epidural administration of ABS to manage CSF leakage after dural puncture.
In a rat model, which colon surgery was performed, some adhesive materials were reported, which can be used to repair anastomotic leaks. For example fibrin glue; a biological adhesive material made from concentrated fibrinogen. It is a water-resistant cover and can thus constitute a physical barrier around the leakage area.29-35 Kanellos et al.31 reported that the application of fibrin glue around a sutured anastomosis reduced the rate of anastomotic leaks and strengthened the anastomosis. Similarly, ABS is efficient in controlling bleeding after gastrointestinal hepatojejunostomy or solitary rectal ulcer.32-36
ABS is a new hemostatic agent comprising the following plant extracts: Urtica dioica (0.06 mg/ml), Vitis vinifera (0.08 mg/ml), Glycyrrhiza glabra (0.07 mg/ml), Alpinia officinarum (0.07 mg/ml), and Thymus vulgaris (0.05 mg/ml). Each of these plant extracts have effects on the endothelium, blood cells, angiogenesis, cellular proliferation, vascular dynamics, and cell mediators. Although the basic mechanism of action of ABS remains unclear, it appears to cause the formation of an encapsulated protein network representing focal points for vital erythrocyte aggregation. The ABS induced protein network that forms with blood cells, particularly erythrocytes, covers the primary and secondary hemostatic system without disturbing individual coagulation factors. ABS also upregulates the GATA/FOG transcription system, affecting erythroid functions and urotensin II. These data have been obtained via MALDI-TOF proteomic molecular analysis, cytometric array, transcription analysis, and scanning electron microscopy examination in in vitro and in vivo research settings.32-37 Specifically, the blood clot forming mechanism of ABS varies from the normal blood clot mechanism. In addition, ABS has effects on new bone formation, endothelium, blood cells, angiogenesis, cellular proliferation, vascular dynamics, cellular mediators and/or bacteria.38-43
In our research, inflammation was not observed in rats, which ABS was administered. We have found no previous reports existing in the literature reporting the effectiveness of ABS in controlling CSF leakage in patients or experimental animals with PDP-CCF-L. In summary, we experienced controlling PDP-CCF-L with topical application of ABS in rats with comparative experimental research. Therefore we would like to present our initial experience in managing this difficult situation and the evaluation with macroscopy and histopathology.
Conclusion: Inflammation properties did not show any differences between groups but new research should be performed to evaluate the long-term effects of ABS on neural tissue.
Conflict of interest: None declared by the authors.
Authors’ Contribution:
AHA: Main author to write the article, Concept, conduction of the study work and manuscript editing
BI & MA: Manuscript preparation
ÖHE: Conducted the experimental study
ÖE: Histopathological evaluation
MA: Collection of data
REFERENCES
- Choi PT, Galinski SE, Takeuchi L, Lucas S,Tamayo C, Jadad AR. PDPH is a common complication of neuraxial blockade in parturients: a meta-analysis of obstetrical studies. Can J Anaesth. 2003 May;5(5):460-9 [PubMed]
- Van de Velde M, Schepers R, Berends N, Vandermeersch E, De Buck F. Ten years of experience with accidental dural puncture and post-dural puncture headache in a tertiary obstetric anaesthesia department. Int J Obstet Anesth. 2008 Oct;17(4):329-35. doi: 10.1016/j.ijoa.2007.04.009 [PubMed] [Free full text]
- Gleeson C, Reynolds F. Accidental dural puncture rates in UK obstetric practice. Int J Obstet Anesth. 1998 Oct;7(4):242-6. [PubMed]
- Sprigge JS, Harper SJ. Accidental dural puncture and post dural puncture headache in obstetric anaesthesia: presentation and management: A 23-year survey in a district hospital. Anaesthesia. 2008 Jan;63(1):36-43.[PubMed] [Free full text]
- Di Giovanni AJ, Galbert MW, Wahle WM. Epidural injection of autologous blood for post-lumbar-puncture headache-II. additional clinical experiences and laboratory investigation. Anesth Analg. 1972 Mar-Apr;51(2):226-32.[PubMed]
- Riley CA, Spiegel JE. Complications following large-volume epidural blood patches for postdural puncture headache. Lumbar subdural hematoma and arachnoiditis: initial cause or final effect? J Clin Anesth. 2009 Aug;21(2):355-9 doi: 10.1016/j.jclinane.2008.08.028. .[PubMed]
- Behcet AL, Kilic H, Zengin S, Güler M,Taysi S, Yıldırım C, et al. Efficiency of Ankaferd Blood Stopper used in bleeding control on intraabdominal adhesions formed postoperatively. Clin Appl Thromb Hemost. 2014 Jul;20(5):524-9.[PubMed]
- Altunhan H, Annagür A, Tokgöz H, Çalişkan U, Örs R. Persistent nasal bleeding due to nasal CPAP application in 2 premature newborns successfully treated with topical ‘Ankaferd blood stopper’. Clin Appl Thromb Hemost 2011; 17: E181-2. [Free full text]
- Çakarer S, Eyüpoğlu E, Günes ÇÖ, Küseoğlu G,Berberoğlu HK, Keskin C.. Evaluation of the hemostatic effects of Ankaferd blood stopper during dental extractions in patients on antithrombotic therapy. Clin Appl Thromb Hemost. 2013 Jan-Feb;19(1):96‑ doi: 10.1177/1076029611435836. .[PubMed]
- Kazancıoğlu HO, Cakır O, Ak G, Zulfikar B. The Effectiveness of a New Hemostatic Agent (Ankaferd Blood Stopper) for the Control of Bleeding following Tooth Extraction in Hemophilia: A Controlled Clinical Trial. Turk J Haematol. 2013 Mar; 30(1):19-24 doi: 10.4274/tjh.2012.0036.[PubMed] [Free full text]
- Huri E, Haznedaroglu IC, Akgul T, Astarci M,Ustun H, Germiyanoulu C.. Biphasic effects of ankaferd blood stopper on renal tubular apoptosis in the rat partial nephrectomy model representing distinct levels of hemorrhage. Saudi Med J. 2010 Aug;31(8):864-8.[PubMed]
- Uz B, Guven GS, Isik A, Kuyumcu ME, Bektas O, Eliacik E, et al. Long‑Term Sustained Hemorrhage Due to Bone Marrow Biopsy Successfully Treated With Topical Ankaferd Hemostat in a Bleeding‑Prone Patient With Secondary Amyloidosis. Clin Appl Thromb Hemost. 2013; 19: 338-40. [Free full text]
- Cömert M, Karakaya K, Barut F, Karadeniz Çakmak G,Uçan HB, Gültekin FA, et al. Does intraabdominal use of Ankaferd Blood Stopper cause increased intraperitoneal adhesions? Ulus Travma Acil Cerrahi Derg. 2010 Sep;16(5):383-9.[PubMed]
- Koçak E, Akbal E, Taş A, Köklü S, Karaca G, Can M, et al. Anti-inflammatory efficiency of Ankaferd blood stopper in experimental distal colitis model. Saudi J Gastroenterol. 2013 May-Jun;19(3):126-30 doi: 10.4103/1319-3767.111955.. [PubMed] [Free full text]
- Zeidan A, Farhat O, Maaliki H, Baeaka A. Does postdural puncture headache left untreated lead to subdural hematoma? Case report and review of the literature. Int J Obstet Anesth. 2006 Jan;15(1): 50-8. [PubMed]
- Vos PE, de Boer WA, Wurzer JA, van Glin J. Subdural hematoma after lumbar puncture: two case reports and review of the literature. Clin Neurol Neurosurg. 1991; 93:127-32. [PubMed]
- Sidebotham D, Willoughby E, Schug S. Late recurrence of postdural puncture headache. Reg Anesth. 1997 Jul-Aug;22(4):382–4. [PubMed]
- Gormley JB. Treatment of post-spinal headache. Anesthesiology 1960; 21: 565–6.
- Safa-Tisseront V, Thormann F, Malassiné P, Henry M, Riou B, Coriat P, et al. Effectiveness of epidural blood patch in the management of post-dural puncture headache. Anesthesiology. 2001 Aug; 95(2):334–9. [PubMed] [Free full text]
- Taivainen T, Pitkänen M, Tuominen M, Rosenberg PH. Efficacy of epidural blood patch for postdural puncture headache. Acta Anaesthesiol Scand. 1993;37:702–5. [PubMed]
- Duffy PJ. Crosby ET. The epidural blood patch. Resolving the controversies Can J Anaesth. 1999 Sep;46(9):878–86 .[PubMed]
- Sanders JC, Gandhoke R, Moro M. Lumbar epidural blood patch to treat a large, symptomatic postsurgical cerebrospinal fluid leak of 5 weeks duration in a 3-year-old. Anesth Analg. 2004 Mar;98(3):629-31. [PubMed]
- Sudlow C, Warlow C. Epidural blood patching for preventing and treating post-dural puncture headache. Cochrane Database Syst Rev. 2002;(2):CD001791. [PubMed]
- Sudlow C, Warlow C. Posture and fluids for preventing post-dural puncture headache. Cochrane Database Syst Rev. 2002;(2):CD001790. [PubMed]
- Kardash K, Morrow F, Béïque F. Seizures after epidural blood patch with undiagnosed subdural hematoma. Reg Anesth Pain Med. 2002 Jul-Aug;27(4):433-6 [PubMed]
- Diaz JH. Permanent paraparesis and cauda equina syndrome after epidural blood patch for postdural puncture headache. Anesthesiology. 2002 Jun;96(6):1515-7.[PubMed] [Free full text]
- Perez M, Olmos M, Garrido FJ. Facial nerve paralysis after epidural blood Reg Anesth. 1993 May-Jun;18(3):196-8. [PubMed]
- Turnbull DK, Shepherd DB. Post-dural puncture headache: pathogenesis, prevention and treatment. Br J Anaesth. 2003 Nov;91(5):718-29. [PubMed] [Free full text]
- Senol M, Altintas MM, Cevık A, Altuntas YE, Barisik NO, Bildik N, et al. The effect of fibrin glue on the intensity of colonic anastomosis in the presence and absence of peritonitis: an experimental randomized controlled trial on rats. ISRN Surg. 2013;2013:521413. doi: 10.1155/2013/521413. [PubMed] [Free full text]
- Ayhan B, Erikoglu M, Tavli SS, Toy H. A comparison of the application of fibrin glue and adhesive film for repair of anastomotic leaks in the rat. Clin Invest Med. 2012 Aug 4;35(4):E216-22. [PubMed]
- Kanellos I, Christoforidis E, Kanellos D, Pramateftakis MG, Sakkas L, Betsis D, et al. The healing of colon anastomosis covered with fibrin glue after early postoperative intraperitoneal chemotherapy. Tech Coloproctol. 2006 Jul;10(2):115-20. [PubMed]
- Bilgili H, Kosar A, Kurt M, Onal JK, GokerHm Captug O, et al. Hemostatic efficacy of Ankaferd Blood Stopper in a swine bleeding model. Med Princ Pract. 2009;18(3): 165-9. doi: 10.1159/000204344 [PubMed] [Free full text]
- Goker H, Haznedaroglu IC, Ercetin S, Kerazii S, Akman U, Ozturk Y, et al. Haemostatic actions of the folkloric medicinal plant extract, Ankaferd blood stopper. J Int Med Res. 2008 Jan-Feb;36(1):163-70. [PubMed]
- Baykul T, Alanoglu EG, Kocer G. Use of Ankaferd Blood Stopper as a hemostatic agent: a clinical experience. J Contemp Dent Pract. 2010 Jan 1;11(1):E088-94. .[PubMed]
- Cipil H, Kosar A, Kaya A, Uz B, Haznedaroglu IC, Goker H, et al. In vivo hemostatic effect of the medicinal plant extract Ankaferd blood stopper in rats pretreated with warfarin. Clin Appl Thromb Hemost. 2009 May-Jun;15(3):270-6. doi: 10.1177/1076029608329578. .[PubMed]
- Kurt M, Akdogan M, Onal IK, Kekilli M, Arhan M, Shorbagi A, et al. Endoscopic topical application of Ankaferd Blood Stopper for neoplastic gastrointestinal bleeding: A retrospective analysis. Dig Liver Dis. 2010 Mar;42(3):196-9. doi: 10.1016/j.dld.2009.05.006 [PubMed]
- Beyazit Y, Kekilli M, Haznedaroglu IC, Kayacetin E, Basaranoglu M. Ankaferd hemostat in the management of gastrointestinal hemorrhages. World J Gastroenterol. 2011 Sep 21;17(35):3962-70. doi: 10.3748/wjg.v17.i35.3962. [PubMed] [Free full text]
- Goker H, Haznedaroglu IC, Ercetin S, Kerazii S, Akman U, Ozturk Y, et al. Haemostatic actions of the folkloric medicinal plant extract Ankaferd Blood Stopper. J Int Med Res. 2008 Jan-Feb;36(1):163-70. [PubMed]
- Akalin C, Kuru S, Barlas AM, Kismet K,Kaptanoglu B, Demir A, et al . Beneficial effects of Ankaferd Blood Stopper on dermal wound healing: An experimental study. Int Wound J. 2014 Feb;11(1):64-8. doi: 10.1111/j.1742-481X.2012.01063. [PubMed]
- Tasdelen Fisgin N, Tanriverdi Cayci Y, Coban AY, Ozatli D,Tanyel E, Durupinar B, et al. Antimicrobial activity of plant extract Ankaferd Blood Stopper. Fitoterapia. 2009 Jan;80(1):48-50. doi: 10.1016/j.fitote.2008.09.006 [PubMed]
- Işler SC, Demircan S, Cakarer S, Cebi Z,Keskin C, Soluk M, et al. Effects of folk medicinal plant extract Ankaferd Blood Stopper on early bone healing. J Appl Oral Sci. 2010 Jul-Aug;18(4):409-14. [PubMed] [Free full text]
- Sheela ML, Ramakrishna MK, Salimath BP. Angiogenic and proliferative effects of the cytokine VEGF in Ehrlich ascites tumor cells is inhibited by Glycyrrhiza glabra. Int Immunopharmacol. 2006 Mar;6(3):494-8. [PubMed]
- Beyazit Y, Kurt M, Kekilli M, Goker H, Haznedaroglu IC. Evaluation of hemostatic effects of Ankaferd as an alternative medicine. Altern Med Rev. 2010 Dec;15(4):329-36. [PubMed] [Free full text]
FIGURES

Figure 1: Incision in the midline of the lumbar area and intradural methylene blue application from the occipitocervical junction through cisterna magna in Group DPC and Group A.

Figure 2: Macroscopic evaluation of the rat in Group DPC

Figure 3: Macroscopic evaluation of the rat in Group A

Figure 4: Hematoxylin&Eosin Staining of the Lumbar Spinal Cord (Group C)

Figure 5: Hematoxylin&Eosin Staining of the Lumbar Spinal Cord (Group DPC)

Figure 6: Hematoxylin&Eosin Staining of the Lumbar Spinal Cord (Group A)




 Facebook
Facebook Twitter
Twitter GooglePlus
GooglePlus Youtube
Youtube