Umesh Kumar Kannojia1, Rajesh Kumar Meena2, Anil Kumar Paswan3,
Kavita Meena1, Shashi Prakash2, Sandeep Loha2
1Senior Resident; 2Assistant Professor; 3Associate Professor
Department of Anesthesiology, Institute Of Medical Sciences, Banaras Hindu University, Varanasi, Uttar Pradesh 221005, (India)
Correspondence: Dr Rajesh Kumar Meena, Assistant Professor, Department of Anesthesiology, Institute Of Medical Sciences, Banaras Hindu University, Aurobindo Colony, Banaras Hindu University Campus, Varanasi, Uttar Pradesh 221005, (India); E-mail:
drrajaiims86@gmail.com
ABSTRACT
Introduction: Caudal epidural block is one of the most popular, reliable, and safe techniques in pediatric patients that can provide analgesia for a variety of supra- and infra-umblical surgical procedures. This study aimed to compare the efficacy of dexmedetomidine–bupivacaine, fentanyl-bupivacaine mixture and bupivacaine alone on duration of postoperative analgesia, sedation, emergence agitation, duration of sensory and motor block, hemodynamic stability and side effects.
Methodology: After approval from ethical committee 90 pediatric patients of age 2-7 y were enrolled. The children were randomly allocated to three equal groups of 30 each using a computer generated randomization list. Group BD (n=30) received 1 µg/kg dexmedetomidine with 0.5 ml/kg bupivacaine 0.25%; Group BF (n=30) received 1 µg/kg fentanyl with 0.5 ml/kg bupivacaine 0.25%, and Group B (n=30) received 0.5 ml/kg of bupivacaine 0.25% for caudal epidural analgesia.
Caudal block was given after induction of general anesthesia for urogenital surgery. General anesthesia was maintained with sevoflurane at a concentration adjusted to maintain BIS at 40-60. Hemodynamic parameters, Pediatric Anaesthesia Emergence Delirium (PAED) score, Richmond agitation sedation scale (RASS), and Children’s Hospital of Eastern Ontario Pain Scale (CHEOPS) were recorded immediate postoperatively and then regularly every hour for the next 12 hours.
Results: PAED score was less in group BD than group B and BF from baseline. RASS Score was less in Group BD than Group BF from base line to 12 h except at 240 min and Group BF is less than Group B from base line to 12 h. Group BD was less than B from base line to 12 h. CHEOP score was less in Group BD than Group BF and Group B from base line to 12 h.
Conclusion: Dexmedetomidine (1 µg/kg) added to bupivacaine in caudal block increases the duration of postoperative analgesia, provides arousable sedation, and decreases emergence delirium with stable hemodynamics and minimal side effects in pediatric patients.
Key words: Bupivacaine; Caudal epidural block; Dexmedetomidine; Emergence agitation; Fentanyl; Postoperative analgesia
Citation: Kannojia UK, Meena RK, Paswan AK, Meena K, Prakash S, Loha S. Comparison of caudal dexmedetomidine and fentanyl combined with bupivacaine in pediatric patients undergoing urogenital surgery. Anaesth Pain & Intensive Care 2017;21(2):204-211
Received: 19 Dec 2016, Reviewed: 20 Dec 2016, 28 Jan 2017, Corrected: 11 Feb, 13 Jun 2017, Accepted: 15 Jun 2017
INTRODUCTION
Caudal epidural block can be used safely in pediatric population as it can provide analgesia for a various supra-umbilical and infra-umbilical surgical procedures. The main disadvantage of caudal block is the shorter duration of analgesia after a single injection.1
Various drugs have been added to local anesthetic solutions to prolong the duration of caudal analgesia such as epinephrine, opioids, ketamine, and α2 agonists.2 α-adrenoceptor agonists have relevant physiological properties: these produce sedation and analgesia, reduce plasma catecholamine concentration, attenuate the stress responses of the surgery and prevent shivering via α2 adrenoceptors in the central nervous system.2 Dexmedetomidine is a selective α2 agonist with sedative and analgesic properties. It has a α2/ α1 selectivity ratio of 1600:1, which is eight times more than clonidine (200:1).3
Dexmedetomidine has no preservatives, and no additives or chemical stabilizers. In the absence of data for caudal dexmedetomidine in children, we adopted a cautious study design by using a low dose of dexmedetomidine 1µg/kg. The selected caudal dose of dexmedetomidine was based on previous studies reported in adults. It produces conscious sedation, in which patients becomes calm, cooperative as well as responsive when aroused, and then back to sleep when not stimulated. Traditional sedatives causes confusion but it has not been described for dexmedetomidine as it does not depend primarily on activation of the gamma aminobutyric acid system.4
Fentanyl, a lipophilic opioid, is added frequently to local anesthetics. Fentanyl has been suggested as the opioid least likely to cause respiratory depression when given extradurally, because of its high lipid solubility.5 Studies in adults report improved and/or prolonged analgesia following the addition of fentanyl to lumbar extradural bupivacaine for lower abdominal procedures,6 cesarean section7 and pain relief in labour.4 In a study of children undergoing lower abdominal surgery, the addition of fentanyl to a single caudal injection of bupivacaine provided analgesia for more than 24 h without effect on respiration.8
Our study aimed to compare the efficacy of mixture of dexmedetomidine –bupivacaine, fentanyl-bupivacaine and bupivacaine alone on duration of postoperative analgesia, sedation, emergence agitation, duration of sensory and motor block, hemodynamic stability and side effects.
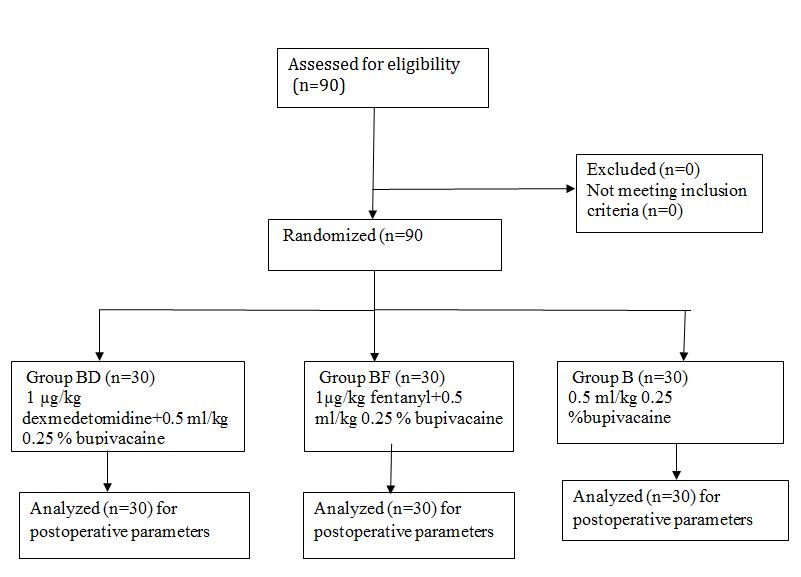
Figure 1: CONSORT flow diagram
METHODOLOGY
This prospective randomized double blind controlled study was conducted in the our department of anesthesiology, from Oct 2015 to June 2016. After approval from ethical committee 90 pediatric patients of age 2-7 y were enrolled. Written and informed consent was taken from parents, the procedure was explained and caudal blocks were given after induction of general anesthesia for urogenital surgery. Patients who did not consent, patients with psychiatric illness, or infection at the site of block, or bleeding abnormality, sensitivity to any drug, bronchial asthma, cardiovascular disease, or spinal deformity were excluded from the study (Figure 1).
Children were premedicated 5 min before surgery with midazolam 0·2 mg/kg by nasal spray. Standard monitors were attached. Anesthesia was induced with sevoflurane in nitrous oxide and oxygen. After placement of a 22G IV cannula, neuromuscular blocking agent was given and trachea was intubated with appropriate size endotracheal tube. Anesthesia was maintained with sevoflurane, nitrous oxide and oxygen.
During surgery, all children received lactated Ringer’s solution 6 ml/kg/h. In postoperative period dextrose plus half normal saline solution was given at a rate of 4 ml/kg. Preparation and infusion of study drug was done by anesthesiologist who was blinded to the study. After induction of anesthesia, a caudal block was performed in lateral decubitus position using a 23G short beveled needle under aseptic precautions. The children were randomly allocated to three equal groups of 30 using a computer generated randomization list.
Group BD: (n=30) received 0.5 ml/kg bupivacaine 0.25% plus 1 µg/kg dexmedetomidine
Group BF: (n=30) received 0.5 ml/kg bupivacaine 0.25% plus 1µg/kg fentanyl
Group B: (n=30) received 0.5 ml/kg bupivacaine 0.25% only
Surgical intervention was started 5 to 10 min after the injection. Intraoperatively muscle relaxant, fentanyl were given and use of these drugs was recorded. Patients were monitored for heart rate, ECG, end-tidal carbon dioxide, arterial oxygen saturation and non-invasive blood pressure. Sevoflurane concentration was adjusted to maintain BIS between 40 and 60. At the beginning of skin closure, sevoflurane and nitrous oxide were discontinued. After the operation, the duration of surgery and the time from discontinuation of anesthesia to spontaneous eye opening (recovery time) were noted. After removal of the endotracheal tube, patients were taken to the recovery room and observed for 4 h. Then, the patients
| Pediatric Anesthesia Emergence Delirium (PAED) score used for emergence agitation | |
| 1. The child makes eye contact with the care giver.
2. Child’s action purpose full. 3. The Child’s is aware of his/her surroundings. 4. Child is restless. 5. Child is inconsolable. |
|
| Richmond agitation sedation scale (RASS) | |
| Target RAS | RASS description |
| +4 | Combative, violent, danger to staff |
| +3 | Pulls or removes tube(s) or catheter; aggressive |
| +2 | Frequent non-purposeful movement, fights ventilator |
| +1 | Anxious, apprehensive but not aggressive |
| 0 | Alert and calm |
| -1 | Awakens to voice ( eye opening/contact) > 10 seconds |
| -2 | Light sedation, briefly awakens to voice ( eye opening/contact ) < 10 seconds |
| -3 | Moderate sedation, movement or eye opening, no eye contact |
| -4 | Deep sedation, no response to voice but movement or eye opening to physical stimulation |
| -5 | Unarousable , no response to voice or physical stimulation |
Box 1: PAED scale and RASS (Richmond agitation sedation scale)
were discharged from the recovery room to the ward, where they were monitored for another 8 to 10 h.
In infants and smaller children’s behavioral and physiologic signs indicates pain reactivity and pain related stress. The analgesic effect of the caudal block was evaluated using the Children’s Hospital of Eastern Ontario Pain Scale (CHEOPS) and sedation was assessed using Richmond agitation sedation scale (RASS) 15min after extubation (Box 1). The first at baseline, 15 min, 30 min, 45 min, 60 min,… 12 h. Analgesic requirement and time were also recorded. When CHEOP scale > 6, paracetamol 10 mg/kg orally was given. Pediatric Anaesthesia Emergence Delirium (PAED) score, RASS and CHEOPS were monitored postoperatively. Respiratory depression was defined as a decrease in SpO2 to < 93% requiring supplementary oxygen. An intra-operative increase in blood pressure or heart rate by >15% was defined as insufficient analgesia, and was treated with rescue opioid (fentanyl; 1µg/kg). An intra-operative decrease of blood pressure or heart rate > 20% from baseline was defined as hypotension or bradycardia, and was treated by fluid bolus, or ephedrine and atropine as necessary.
Statistical analysis: The number of patients required in each group was determined after a power calculation according to data obtained from previous studies. The calculation of sample size revealed that at least 29 patients in each group would be required for appropriate study with an α0.05 and a power of 0.8. Data are presented as mean or median with range or 95% confidence interval (CI) as appropriate. Student’s t-test or the Mann–Whitney U-test were used as appropriate. Categorical data are presented as absolute numbers (%) and were analysed with x2 analysis. Fisher’s exact test was used as appropriate. P < 0.05 was considered statistically significant.
RESULTS
Demographic data of patients in all of the three groups is presented in Table 1.
Table 1: Demographic data
| Variable | Group BD
( n=30) |
Group BF
( n=30) |
Group B
(n=30) |
p-value |
| Age (y) | 3.683 ± 2.3131 | 3.633 ± 1.9205 | 4.065 ± 1.8962 | 0.668 |
| Weight (kg) | 11.4 ± 3.3 | 12 ± 3.9 | 11.8 ± 3.7 | 0.228 |
| Gender (M/F) | 24/6 | 22/8 | 25/6 | 0.749 |
| Duration of anesthesia (min) | 68.4 ± 17.6 | 71.3 ± 17.8 | 69.5 ± 17.4 | 0.660 |
Demographic profile was comparable and nonsignificant. Figure 2 shows mean heart rate in three groups at different time interval.
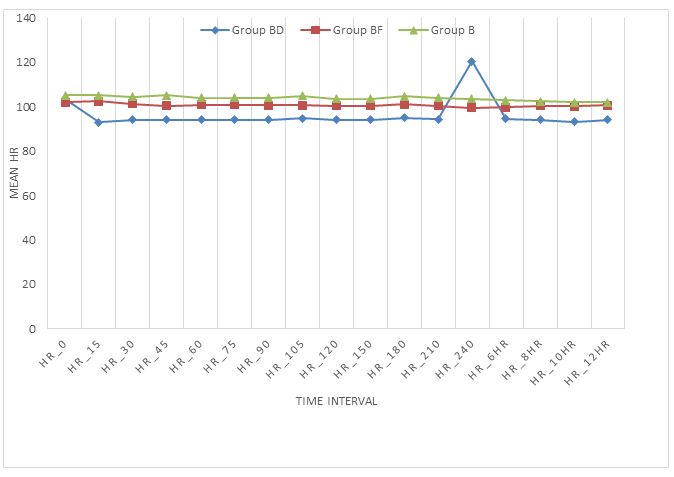
Figure 2: Mean heart rates in three groups at different time intervals
Figure 3 shows mean systolic blood pressure in three groups at different time interval. Mean systolic blood pressure is lower in Group BD than Group BF and B .Group B shows higher mean systolic blood pressure than other two group, Group BD and Group BF and this difference is statistically significant (p < 0.05).
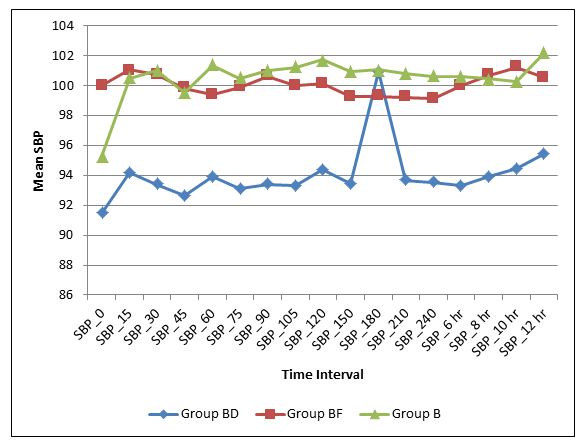
Figure 3: Mean systolic blood pressure in three groups at different time interval
Figure 4 depicts mean diastolic blood pressure in three groups at different time interval. Mean diastolic blood pressure is lower in Group BD than Group BF and Group B. Group B shows higher mean diastolic blood pressure than other two groups, Group BD and Group BF are statistically significant (p < 0.05)
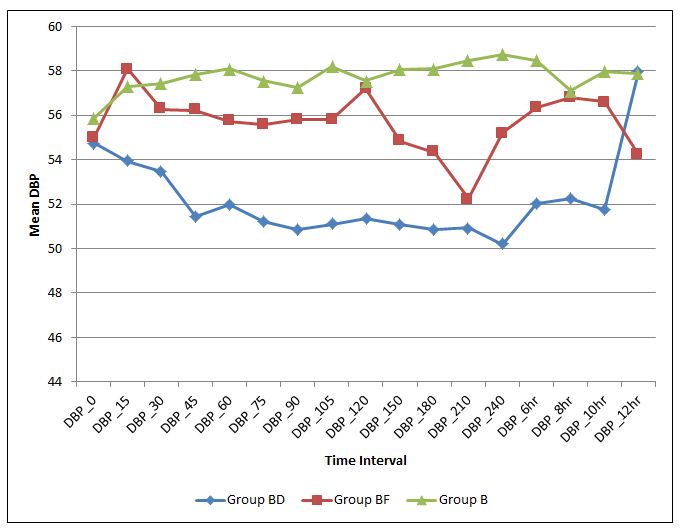
Figure 4: Mean diastolic blood pressure in three groups at different time interval
Table 2: Statistical comparison of mean PAED SCORE at different time intervals in different groups
| Variables | Group BD vs. Group BF | Group BD vs. Group B | Group BF vs. Group B |
| PAED 0 min | 0.000* | 0.004* | 1.000 |
| PAED 15min | 0.002* | 0.020* | 1.000 |
| PAED 30min | 0.188 | 0.696 | 1.000 |
| PAED 45min | 0.321 | 1.000 | 0.951 |
| PAED 60min | 1.000 | 1.000 | 1.000 |
| PAED 75min | 0.053 | 0.005* | 1.000 |
| PAED 90min | 1.000 | 0.052 | 0.342 |
| PAED 105min | 0.089 | 0.001* | 0.494 |
| PAED 120min | 0.005* | 0.002* | 1.000 |
| PAED 150min | 0.057 | 0.014* | 1.000 |
| PAED 180 min | 0.023* | 0.006* | 1.000 |
| PAED 210min | 0.054 | 0.019* | 1.000 |
| PAED 240min | 0.008* | 0.090 | 1.000 |
| PAED 6 h | 0.022* | 0.108 | 1.000 |
| PAED 8 h | 0.400 | 0.197 | 1.000 |
| PAED 10 h | 1.000 | 0.377 | 1.000 |
| PAED 12 h | 1.000 | 0.243 | 0.435 |
Table 2 shows statistical comparison of postoperative mean PAED score at different time interval between the three groups. The corresponding mean differences in various groups were comparable and statistically significant (p < 0.05). Group BD vs. Group BF was significant only at baseline, 15 min, 75 min, 120 min, 180 min, 210 min and 6 h (p < 0.05).
Table 3: Statistical comparison of mean RASS at different time intervals in different groups
| Variables | Group BD vs. Group BF | Group BD vs. Group B | Group BF vs. Group B |
| RASS 0 min | 0.115 | 1.000 | 0.058 |
| RASS 15 min | 0.005* | 0.858 | 0.095 |
| RASS 30 min | 0.000* | 0.289 | 0.002* |
| RASS 45 min | 0.023* | 0.265 | 0.919 |
| RASS 60 min | 0.068 | 1.000 | 0.439 |
| RASS 75 min | 0.008* | 0.370 | 0.358 |
| RASS 90 min | 0.042* | 0.263 | 1.000 |
| RASS 105 min | 0.000* | 0.019 | 0.155 |
| RASS 120 min | 0.040* | 0.652 | 0.578 |
| RASS 150 min | 0.061 | 0.690 | 0.728 |
| RASS 180 min | 0.147 | 0.287 | 1.000 |
| RASS 210 min | 0.569 | 0.502 | 1.000 |
| RASS 240 min | 0.130 | 0.069 | 1.000 |
| RASS 6 h | 0.036* | 0.222 | 1.000 |
| RASS 8 h | 0.000* | 0.073 | 0.120 |
| RASS 10 h | 0.000* | 0.045* | 0.236 |
| RASS 12 h | 0.000* | 0.024* | 0.060 |
Table 3 shows statistical comparison of postoperative mean RASS scale at different time interval between three groups. The corresponding mean differences in various groups were comparable and statistically significant (p < 0.05) .Group BD vs. Group BF was significant except at base line, 60 min, 150 min and 180 min (p > 0.05). Group BD vs. Group B was significant at 10 h and 12 h. Group BF vs. Group B was statistically not significant (p > 0.05).
Table 4: showing of mean CHEOP SCORE among different groups at different time intervals
| Variables | Group BD vs. Group BF | Group BD vs. Group B | Group BF vs. Group B |
| CHEOPS 0 min | 0.001* | 0.009* | 1.000 |
| CHEOPS 15 min | 0.015* | 0.040* | 1.000 |
| CHEOPS 30 min | 0.000 | 0.005* | 0.135 |
| CHEOPS 45 min | 0.000* | 0.016* | 0.063 |
| CHEOPS 60 min | 0.000* | 0.008* | 0.048* |
| CHEOPS 75 min | 0.000* | 0.000* | 0.087 |
| CHEOPS 90 min | 0.000* | 0.020* | 0.235 |
| CHEOPS 105 min | 0.000* | 0.000* | 0.572 |
| CHEOPS 120 min | 0.000* | 0.002* | 0.005* |
| CHEOPS 150 min | 0.000* | 0.044* | 0.034* |
| CHEOPS 180 min | 0.000* | 0.989 | 0.000 |
| CHEOPS 210 min | 0.000* | 1.000 | 0.001 |
| CHEOPS 240 min | 0.000* | 0.004 | 0.007 |
| CHEOPS 6 h | 0.000* | 0.192 | 0.000 |
| CHEOPS 8 h | 0.000* | 0.702 | 0.000 |
| CHEOPS 10 h | 0.000* | 1.000 | 0.000 |
| CHEOPS 12 h | 0.000* | 1.000 | 1.000 |
Table 4 shows statistical comparison of mean CHEOPS score postoperatively. The corresponding mean differences in various groups were comparable and statistically significant (p < 0.05). Group BD vs. Group BF was significant from base line to 12 h. Group BD vs. Group B was significant from base line to 150 min and 240 min. Group BF vs. Group B was statistically significant (p < 0.05) except at baseline, 15 min, 30 min, 45 min, 75 min, 90 min, 105 min, 12 h.
No significant respiratory depression was reported in this study, none of the children had a drop in SpO2 value of < 95% and it was statistically not significant. This compares our finding with previous studies that α2 agonists have no clinical respiratory effect.
DISCUSSION
In our study we found that dexmedetomidine (1 µg/kg) added to bupivacaine (0.5 ml) in caudal block increased the duration of postoperative analgesia. Patients in Group BD had lower CHEOPS score as well as less use of paracetamol over 24 h. It also decreased emergence delirium.
Observations made in our study are similar to El-Hennawy et al.9 (2009) in this study, a comparison of the analgesic effects and side-effects of dexmedetomidine and clonidine added to bupivacaine was done in pediatric patients undergoing lower abdominal surgeries. Post study it was concluded that addition of dexmedetomidine or clonidine to caudal bupivacaine significantly promoted analgesia with no significant advantage of dexmedetomidine over clonidine and without an increase in incidence of side-effects. Further study conducted by Saadawy et al.10 (2009) to evaluate the characteristics of emergence, objective pain score, sedation score and quality of caudal dexmedetomidine with bupivacaine in providing pain relief over a 24-h period concluded that caudal dexmedetomidine was a promising adjunct to provide excellent analgesia without side effects over a 24-h period. It has the advantage of keeping the patients calm for a prolonged time. Furthermore Xiang et al.11 (2013) supplemented caudal bupivacaine with dexmedetomidine and concluded that the addition of dexmedetomidine to caudal bupivacaine reduced the response to hernia sac traction, and prolonged the duration of postoperative analgesia in children undergoing inguinal hernia repair.
Solanki SL et al.14 administered intrathecal dexmedetomidine and clonidine with bupivacaine in trauma patients and studied the onset and duration of sensory and motor blockade, severity of postoperative pain, time to first rescue analgesia and total analgesic requirement for 24 hours. No significant difference was noted in the time of onset of block but the duration of sensory and motor blockade was prolonged with clonidine and dexmedetomidine, compared with control group. Duration of analgesia was prolonged and lower postoperative pain scores were noted with dexmedetomidine compared with clonidine, the latter being longer than control group (p = 0.0001). In a further study by Campbell et al.,12 single caudal injection of bupivacaine–fentanyl mixture was administered and analgesic efficacy and safety was evaluated. In a prospective, controlled, triple blinded study of 34 children, aged 1-11 year undergoing urological surgery. There were no differences in pain and sedation scores with the use of bupivacaine plus epinephrine with or without addition of fentanyl. The incidence of side effects did not differ between groups and no side effect required treatment.
Ganesh et al.13 evaluated the efficacy of adding fentanyl to epidural bupivacaine in infants up to 6 months of age after a thoracotomy. Parameters that were evaluated up to 24 h postoperatively were pain, amount of analgesic rescues, time to first rescue, pain scores, behavior scores (five-item behavior score), and complications including respiratory depression, oxygen requirement, vomiting, and urinary retention. It was concluded that addition of epidural fentanyl to bupivacaine provided improved post-thoracotomy analgesia without increased side effects, compared with bupivacaine alone.
Study conducted by Gaitini et al.15 on stress response suggests that addition of fentanyl 1 μg/kg to bupivacaine in the caudal epidural block in children has no influence on plasma levels of epinephrine and norepenphrine and nor does it improve the analgesic intensity of the caudal block. However our study shows that fentanyl-bupivacaine group had better analgesia than bupivacaine alone.
CONCLUSION
We conclude that dexmedetomidine (1 µg/kg) added to bupivacaine (0.5 ml) in caudal block increased the duration of postoperative analgesia. Provided arousable sedation, also decreased emergence delirium with stable hemodynamics and minimal side effects in pediatric patients. So it can be termed as a suitable additive for caudal block in children.
Conflict of interest: None
Author’s contribution: UKK- conduction of study
RKM- concept of study and manuscript editing
AKP- concept of study
KM- data collection
SP- manuscript editing
SL- statistical analysis
REFERENCES
- Lloyd-Thomas AR. Pain management in paediatric patients. Br J Anaesth 1990; 64: 85–104 [PubMed]
- Kanazi GE, Aouad MT, Jabbour- Khoury SI, Al Jazzar MD, et al. Effect of low-dose dexmedetomidine or clonidine on the characteristics of bupivacaine spinal block. Acta Anaesthesiol Scand 2006;50(2):222-7 [PubMed] [Free full text] DOI: 1111/j.1399-6576.2006.00919.x
- Coursin DB, Maccioli GA. Dexmedetomidine. Curr Opin Crit Care 2001;7:221–6. [PubMed] [Free full text] DOI: 10.1111/bcp.13293
- Celleno D, Capogna G. Epidural fentanyl plus bupivacaine 0.125 per cent for labour: analgesic effects. Can J Anaesth 1988;35:375-8. [PubMed] [Free full text] DOI: 10.1007/BF03010859
- Morgan M. The rational use of intrathecal and extradural opioids. Br J Anaesth 1989;63:165-88 [PubMed]
- Rucci FS, Cardamone M, Migliori P. Fentanyl and bupivacaine mixtures for extradural blockade. Br J Anaesth 1985;57:275-84 [PubMed]
- Paech M J, Westmore MD, Speirs HM. A double-blind comparison of epidural bupivacaine and bupivacaine fentanyl for Caesarean section. Anaesth Intensive Care 1990;18:22-30 [PubMed]
- Moine P, Ecoffey C. Bupivacaine caudal block with fentanyl in children. Anesthesiology 1989;71:A1017
- El-Hennawy AM, Abd-Elwahab AM, Abd- Elmaksoud AM, El-Ozairy HS and Boulis SR. Addition of clonidine or dexmedetomidine to bupivacaine prolongs caudal analgesia in children. Br J Anaesth 2009;103(2):268–74 [PubMed] [Free full text] DOI: 1093/bja/aep159
- Saadawy I, Boker A, Elshahawy MA, Almazrooa A, Melibary S, Abdellatif AA, Afifi W. Effect of dexmedetomidine on the characteristics of bupivacaine in a caudal block in pediatrics. Acta Anaesthesiol Scand 2009;53:251–256 [PubMed] [Free full text] doi: 10.1111/j.1399-6576.2008.01818.x.
- Xiang Q, Huang DY, Zhao YL, Wang GH, Liu YX, Zhong L, Luo T. Caudal dexmedetomidine combined with bupivacaine inhibit the response to hernial sac traction in children undergoing inguinal hernia repair. Br J Anaesth. 2013 Mar;110(3):420-4 [PubMed] [Free full text] doi: 10.1093/bja/aes385.
- Campbell FA, Yentis SM, Fear DW, Bissonnette B. Analgesic efficacy and safety of a caudal bupivacaine-fentanyl mixture in children. Can J Anaesth 1992;39 (7):661-4 [PubMed] [Free full text] DOI: 10.1007/BF03008226
- Ganesh A, Adzick NS, Foster T, Cucchiaro G. Efficacy of addition of fentanyl to epidural bupivacaine on postoperative analgesia after thoracotomy for lung resection in infants. Anesthesiology 2008 Nov;109(5):890-4 [PubMed] [Free full text] doi: 10.1097/ALN.0b013e31818aa6cb.
- Solanki SL, Bharti N, Batra YK, Jain A, Kumar P, Nikhar SA. The analgesic effect of intrathecal dexmedetomidine or clonidine, with bupivacaine, in trauma patients undergoing lower limb surgery: a randomised, double-blind study. Anaesth Intensive Care 2013 Jan;41(1):51-6 [PubMed] [Free full text]
- Gaitini L. A., Somri M., Vaida S. J., Yanovski B., Mogilner G., Sabo E., et al. Does the addition of fentanyl to bupivacaine in caudal epidural block have an effect on the plasma level of catecholamines in children? Anesth Analg 2000;90:1029–33 [PubMed] [Free full text] DOI: 10.1097/00000539-200005000-00006




 Facebook
Facebook Twitter
Twitter GooglePlus
GooglePlus Youtube
Youtube