Jaya Sharma1, Shobha Purohit2, Mamta Sharma2, Manish Kumar1
1 Postgraduate Resident; 2Professor
SMS Medical College, JLN Marg, Jaipur, Rajasthan 302004 (India)
Correspondence: Dr. Jaya Sharma, Room No. 310, SMS Girl’s Hostel, SMS Medical College, JLN Marg, Jaipur, Rajasthan 302004 (India); Phone: +9529815146; E-mail: jaya.sharma1202@gmail.com
ABSTRACT
Hydrocephalus is a disorder of abnormal accumulation of cerebrospinal fluid (CSF) in the ventricular system. Accumulation of CSF occurs due to an imbalance between CSF production and absorption. In the newborn and infants, hydrocephalus almost entirely presents as an enlarged head resulting from the separation of cranial bone plates, so any newborn or infant with an enlarged head should undergo evaluation. Late reporting of cases makes it obvious that hydrocephalus may have become enlarged massively. We report a case of giant hydrocephalus in a 1 year old infant (head circumference of 86 cm) posted for ventriculoperitoneal shunt surgery with anesthetic consideration of difficult intubation because of such a large head size, positioning , age related pathophysiology and temperature regulation. The case was managed successfully with an uneventful hospital stay and a significant decrease in head size was noted from pre-operative 86 cm to 70 cm on fifteenth post-operative follow up day.
Key words: Hydrocephalus; Positioning; Craniotomy; Hydrocephalus / surgery; Ventriculoperitoneal Shunt
Citation: Sharma J, Purohit S, Sharma M, Kumar M. A case of massive hydrocephalus; perioperative challenges and literature review. Anaesth Pain & Intensive Care 2016;20(3):353-357
Received: 19 August 2016; Reviewed: 12 September 2016; Accepted: 15 September 2016
INTRODUCTION
Hydrocephalus is the consequences of many disease processes characterized by abnormal increase in cerebrospinal fluid (CSF) due to imbalance between CSF production, absorption or flow. Thus resulting in enlarged cerebral ventricles and usually present with increase in intracranial pressure (ICP).1 Hydrocephalus is a progressive disease so measuring of serial head circumference provides an effective means of progression of hydrocephalus.1 Rapid increase in size usually requires surgical interventions in the form of either shunt placement or endoscopic third ventriculostomy.1 Till date the largest reported case of hydrocephalus was an Indian child, who was 18 months old when her head grew to a circumference of 94 cm.2 Here, we present a case of 1 year old male patient with massive hydrocephalus (86 cm head circumference) and discuss the anesthetic consideration. We have searched thoroughly but as per our knowledge not a single case was reported at this age with such a large head circumference.
CASE REPORT
A one year old male child presented to neurosurgery department of SMS Medical College, with history of gradually progressive head size since birth. But patient was asymptomatic & able to take feed properly until a month ago when his mother noticed occasional vomiting. Patient was delivered via an uncomplicated cesarean section. Uneventful birth history was given by mother and the cry was normal at birth. Except for the large head size at time of birth; no further detail of exact cause of cesarean section; previous details of antenatal, natal and postnatal checkups could collected from the mother because of her rural background. On examination there was profoundly large head size, fontanelle was lax, positive sunset sign in bilateral eyes, enlarged and engorged scalp vein (Figure 1). CT of the brain showed bilateral massive enlargement of lateral ventricle, splaying of sutures & cortical thinning, barely any cortex was visible (Figure 2). The patient was scheduled to undergo placement of ventriculoperitoneal shunt. A thorough pre anesthetic examination was performed. Routine laboratory investigations including hemoglobin, total and differential leucocyte count, urinalysis, and urine microscopy, and x-ray chest, were within normal limits. A difficult airway was anticipated because of massively enlarged head size. Child was kept nil by mouth and clear liquids were allowed 4 h before surgery. Written informed consent was obtained from parents and child was taken inside operating room. Routine monitoring like pulse oximeter, electrocardiogram and blood pressure, were attached and inj fentanyl 1 μg/kg and inj glycopyrrolate 0.004 mg/kg was given. Child was preoxygenated with 100% oxygen for 5 min with the help of facemask and Jackson-Rees circuit. For induction attention was given to proper positioning of the patient for intubation. Head portion of OT table was tilted slightly downwards, a ramp was made up with rolled table cloth and placed below shoulders and upper chest to elevate patient’s body and a head ring was kept below the head of child to prevent it from falling sideways (Figure 3 & 4). Induction was done with inj thiopentone sodium 5 mg/kg intravenously. After checking ability to ventilate trachea was intubated with 4.5 mm uncuffed endotracheal tube with inj succinylcholine 1 mg/kg. Anesthesia was maintained with sevoflurane, nitrous oxide and oxygen and controlled respiration. For muscle relaxation inj atracurium 0.1 mg/kg given if required. Intraoperative vitals was stable throughout the procedure and the shunt was placed uneventfully. After the surgery, all sevoflurane was stopped and muscle relaxation was reversed. Suction was done and the tube was removed when the child was fully conscious. The child was shifted to post-operative recovery room for observation.
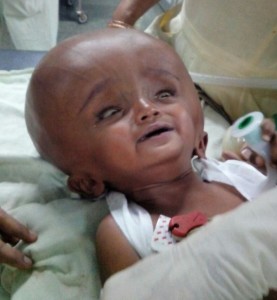
Figure 1: Infant with giant hydrocephalus and sunset sign in both eyes
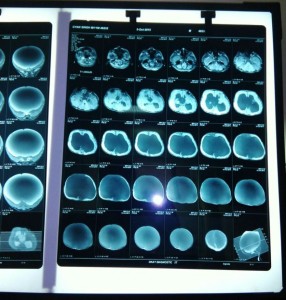
Figure 2: Preoperative CT demonstrates enlarged ventricles & thinning of cortical tissue
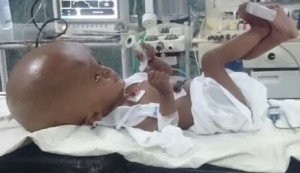
Figure 3: Infant with giant hydrocephalus
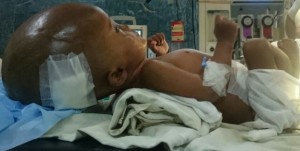
Figure 4: Infant positioning with a role towel under the chest
DISCUSSION
CSF is primarily produced by choroid plexus with partial contribution of ependymal capillaries and brain interstitial fluid. Production occurs at the rate of 0.3 to 0.6 ml/min with about 500-600 ml/day.3 Production rate is related to cerebral blood flow that is higher in infants & children in comparison to adults (90-100 ml/100 g/min vs. 50 ml/100 g/min).4-5 CSF circulates from site of secretion to site of absorption (arachnoid villi) and any resistance in CSF circulation pathway and absorption or increased rate of production leads to development of hydrocephalus. An increasing volume of CSF results in ventricular enlargement and later on rise in ICP.3-5
Causes of hydrocephalus may be classified into congenital and acquired ones, and the lesion may be of communicating or non-communicating type.6,7 Congenital hydrocephalus can be genetic or may be commonly associated with neural tube defects like spina bifida, myelomeningocele & Arnold-Chiari or Dandy-Walker malformation, vascular malformations, arachnoid cysts, syndromes such as X-linked hydrocephalus and trisomies 13, 18, 9, and 9p, and other CNS structural abnormalities.6,7 In our case it was the congenital hydrocephalus but the cause was unknown at the time of surgery. So preoperative history and perioperative anesthetic plan should be formulated by keeping these possibilities of associated neurologic abnormalities and comorbidities in mind.
Intracranial hypertension almost always accompanies hydrocephalus in infants and children which is the most important anesthetic consideration.1 Normal range of ICP in infant is 1.5-6 mmHg while in adult it is higher (10-15 mmHg).8 Autoregulatory reserve is decreased in infants due to lower values of mean arterial pressure, cerebral blood flow and cerebral perfusion pressure (CPP) (CPP: 40-60 mmHg for infants, 50-70 mmHg for adults).9,10 Flexibility of infant’s skull leads to enlargement of cranial compartment if hydrocephalus progresses slowly; once the upper limit of enlargement reaches ICP begins to rise, and acute increase in cranial volume may cause hernaiation.9 Increased ICP leads to nonspecific symptoms in infants such as irritability, poor feeding, vomiting, lethargy and bulging fontanelle, downward gazing eyes.9 Repeated episodes of vomiting, signs of intravascular volume contraction and infant’s altered mental status calls for arterial blood gas analysis before surgery.11 Measures should be taken to avoid rise in ICP i.e. slight head-up tilt, avoidance of hypercarbia and hypoxia, correction of hypotension and hypertension, administration of osmotic agents and diuretics, avoidance of PEEP to prevent venous engorgement.1
Positional changes (head flexion, head down), behavioral change (crying) and physiologic change (hypercarbia) can affect ICP so the child should be kept in head-up position with minimal possible agitating maneuvers.1 Pediatric airway is often considered more challenging than in adults because of anatomical differences (larger tongue, cephalad larynx, angulated vocal cord, omega shape epiglottis, narrowed subglottic region) and physiological differences (rapid desaturation during periods of apnea, more chances of upper airway obstruction under anesthesia).12 Additionally in congenital hydrocephalus, macrocephaly may distort the normal anatomy of the skull, making airway management even more difficult. Perioperative plan must be framed by anesthesiologist based on difficult airway management as enlarged occiput forces neck in extreme flexion and large forehead may obscure view of laryngoscopy so elevating the body with folded sheet or pillow is necessary to facilitate intubation.13 Rolling of head, especially after induction of anesthesia is to be avoided by keeping head rest. Our patient’s head circumference was 86 cm, which was approximately 1.85 times for a one year-old. This resulted in a challenging combination for intubation. Prop-up positioning of the patient with the towel sheets allowed us to get optimized view of laryngoscopy. Difficult airway cart and alternative management plan were kept ready. Balance must be maintained between promoting sedation by pharmacological means and minimizing risk of hypoventilation as increased PaCO2 causes further rise in ICP.1 Method of induction should be determined by circumstances of case and effect of Anesthetic agent’s on neuro-physiology.1 Intravenous induction should be preferred if child has signs of raised ICP, stupor or delayed gastric emptying, as it reduces cerebral blood flow and ICP.1,14 Inhalational induction can be an acceptable alternative with non-irritant volatile anesthetics like sevoflurane in children with no intravenous access.1 Maintenance of anesthesia can be done by a combination of volatile anesthetics, muscle relaxants and opioids intraoperatively.
Temperature regulation and maintenance of normoglycemia should be kept in mind as infants are more prone for hypothermia and hypoglycemia and surgical procedure of ventriculoperitoneal shunt requires exposure from head to abdomen, which further adds in issues of hypothermia.15 Core temperature monitoring should be done along with measures like maintenance of operating room temperature, covering of infant’s body with cotton pads or warm air blankets, use of warm intravenous fluids. Normal saline should be used preferably for maintenance fluid as hyperosmolar nature may help in attenuating cerebral edema.14 Initial skin incision and tunneling of the shunt are most stimulating part of surgery, increase in heart rate and ICP during this period can be managed by either increasing depth of anesthesia or using short acting opioids such as fentanyl or remifentanil.16,17 On initial cannulation of ventricle, BP may drop abruptly as brainstem pressure is relieved suddenly. During ventriculostomies, serious dysrhythmias, especially bradycardias may occur, apart from using atropine, simply alerting the surgeon of the bradycardia and pausing may resolve problem many times.15 Look for the signs of pneumothorax as trocar is placed subcutaneously to allow tunneling. Removal of large quantity of cerebrospinal fluid (CSF) may cause hemodynamic and electrolyte disturbances that needs to be corrected perioperatively and rapid drainage of CSF may lead to serious arrhythmias or even cardiac arrest; precautions should be taken to avoid this.18
Postoperatively, the infant can be at risk of respiratory problems and vomiting aspiration because of poor airway control due to neurological deficit, so these infants should be kept under close observation.6 Risk of chronic neurological deficit exist postoperatively. This is attributed not only to failure rate of shunts but also to known fact that hydrocephalus causes ischemia and hypoxia by stretching and compressing the periventricular tissue, and chronically it may result in altered neuronal activity, axonal degeneration, neurogliosis, altered transport, metabolic impairment and at last cellular death.6
CONCLUSION
Infants with hydrocephalus presenting late, usually offer multiple challenges during anesthesia, but a well-coordinated plan of action results in optimization of issues and reduced peri-operative morbidity and mortality with improved outcome.
Conflict of interest: None declared by the authors
Authors’ contribution: JS, SP, MS, MK: Preparation of manuscript; Final proof check
REFERENCES
- Diu MW, Mancuso TJ. Pediatric diseases. In: Hines RL, Marschall KE, editors. Stoelting’s Anesthesia & Co-existing Disease, 2nd South Asia: Reed Elsevier India Private Limited; 2014. P. 611-13.
- Udas S. Indian child Roona Begum recovering after hydrocephalus surgery [Online]. August 28, 2013 [cited 19 Sept 2016]; Available from: URL:http://edition.cnn.com/2013/08/28/world/asia/india-child-hydrocephalus/index.html
- Johanson CE, Duncan JA 3rd, Klinge PM, Brinker T, Stopa EG, Silverberg GD. Multiplicity of cerebrospinal fluid functions: New challenges in health and disease. Cerebrospinal Fluid Res. 2008;5:10. doi: 10.1186/1743-8454-5-10. [PubMed] [Free full text]
- Kennedy C, Sokoloff L. An adaptation of the nitrous oxide method to the study of the cerebral circulation in children; normal values for cerebral blood flow and cerebral metabolic rate in childhood. J Clin Invest. 1957;36:1130‑ [PubMed] [Free full text]
- Chiron C, Raynaud C, Maziére B, Zilbovicius M, Laflamme L, Masure MC, et al. Changes in regional cerebral blood flow during brain maturation in children and adolescents. J Nucl Med. 1992;33:696‑ [PubMed] [Free full text]
- McAllister JP 2nd. Pathophysiology of congenital and neonatal hydrocephalus. Semin Fetal Neonatal Med. 2012;17:285-94. doi: 10.1016/j.siny.2012.06.004. [PubMed]
- Beni-Adani L, Biani N, Ben-Sirah L, Constantini S. The occurrence of obstructive vs absorptive hydrocephalus in newborns and infants: relevance to treatment choices. Childs Nerv Syst. 2006;22:1543-63. [PubMed]
- Dunn LT. Raised Intracranial pressure. Neurol Neurosurg Psychiatry. 2002;73(Suppl 1):i23-i27. [PubMed] [Free full text]
- Vavilala MS, Sulpicio, S.G. Anesthesia for Neurosurgery. In: Cote CJ, Lerman, J, and Todres, ID, editors. A Practice of Anesthesia for Infants and Children. 4th ed. Philadelphia: Saunders Elsevier 2009. p. 713-37.
- Kochanek PM, Carney N, Adelson PD, Ashwal S, Bell MJ, Bratton S, et al. Guidelines for the acute medical management of severe traumatic brain injury in infants, children, and adolescents–second edition. Pediatr Crit Care Med. 2012 Jan;13 Suppl 1:S1-82. doi: 10.1097/PCC.0b013e31823f435c. [PubMed]
- Singla D, Mangla M. Perioperative management of a infant with huge hydrocephalus: a case report. Pediatric Anesthesia and Critical Care Journal 2016;4(1):24-27. [Free full text]
- Cote CJ. Pediatric Anesthesia. In: Miller RD, editor. Miller’s Anesthesia. 8th ed. Philadelphia: Saunders Elsevier; 2015. P. 2757-96.
- Hamid RK, Newfield P. Pediatric neuroanesthesia. Hydrocephalus. Anesthesiol Clin North America 2001;19:207-18. [PubMed]
- Hamid NA, Hamid RKA, Newfield P. Pediatric neuroanesthesia. In: Newfield P, Cottrell JE, editors. Handbook of Neuroanesthesia, 5th ed. Philadelphia: Wolters Kluwer Lippincott Williams & Wilkins; 2012. P. 257-79.
- Rath GP, Dash HH. Anaesthesia for neurosurgical procedures in paediatric patients. Indian J Anaesth. 2012;56:50210. [PubMed] [Free full text]
- Rath GP, Prabhakar H, Bithal PK, Dash HH, Narang KS, Kalaivani M. Effects of butorphanol and fentanyl on cerebral pressures and cardiovascular hemodynamics during tunneling phase for ventriculoperitoneal shunt insertion. Middle East J Anesthesiol. 2008;19:1041–53 [PubMed]
- Prabhakar H, Rath GP, Bithal PK, Chouhan RS. Intracranial pressure and haemodynamic changes during the tunnelling phase of ventriculoperitoneal shunt insertion. Eur J Anaesthesiol. 2005;22:947-50. [PubMed]
- Alfery DD, Shapiro HM, Gagnon RL. Cardiac arrest following rapid drainage of cerebrospinal fluid in a patient with hydrocephalus. Anesthesiology. 1980;52:443-4. [PubMed] [Free full text]




 Facebook
Facebook Twitter
Twitter GooglePlus
GooglePlus Youtube
Youtube