Amjad Ali, FCPS*, Khan Muhammad Babar, FCPS**
*Assistant Professor, Department of Anaesthesiology, Bolan Medical College Quetta (Pakistan)
**Assistant Professor Department of Surgery, Bolan Medical College Quetta (Pakistan)
Correspondence: Dr. Amjad Ali, Assistant Professor, Department of Anaesthesiology, Bolan Medical College Quetta (Pakistan); Phone: 03337924149, 081-2853352; E-mail: dramjad_ali@hotmail.com; amjadalidr@gmail.com
Setup: Department of Anaesthesiology, Bolan Medical Complex Hospital, Quetta.
Design: Randomized, controlled trial.
Methodology: Sixty adult patients, 35–65 years of age, ASA physical status I or II, of either sex, undergoing elective open cholecystectomy were allocated randomly in this prospective, randomized, controlled study. Patients were randomly divided into two groups of 30 each, to receive either pregabalin 150 mg or celecoxib 200 orally 1 hour before surgery. Postoperative pain was assessed by a 10cm visual analogue scale, where 0 denoted no pain and 10 was taken as the worst imaginable pain. Sedation, postoperative nausea and/or vomiting and any other complication, were assessed periodically throughout the study time. Patients received inj. nalbuphine on demand during the postoperative period and nausea and vomiting were treated with metocloperamide 10 mg. Results were statistically analysed.
Results: Cumulative consumption of inj. nalbuphine over 24 hours was 13.5 ± 7.4 mg for pregabalin group, and 13 ±6.7mg for celecoxib group (P >0.05). No significant difference was found in frequency and severity of postoperative nausea and vomiting (PONV) during 24 hours (P > 0.05), although frequency of sedation was higher in pregabline group (P <0.05).
Conclusion: Preoperative single dose of pregabalin 150 mg provides no better pain relief when compared to celecoxib 200 mg, and high incidence of adverse effects were found in patients receiving pregabline before open cholycystectomy.
Keywords: Postoperative pain; Celecoxib; Pregabalin; Non-opioid analgesics; Open cholecystectomy.
Citation: Ali A, Babar KM. Comparison of preoperative dose of pregabalin with celecoxib for attenuation of postoperative pain after open cholecystectomy. Anaesth Pain & Intensive Care 2012; 16(2): 137-141
Pregabalin and its developmental predecessor gabapentin, were originally developed as spasmolytic agents and adjuncts for the management of generalized or partial epileptic seizures resistant to conventional therapies.7 Like gabapentin, pregabalin has a proven role in treating neuropathic pain and postoperative pain after breast surgery, spinal surgery, and laparoscopic cholecystectomy surgeries.8
Recent research indicates that cyclooxygenase-2 (COX-2) inhibitors, a selective class of non-steroidal anti-inflammatory drugs (NSAIDs), could play an important role in perioperative pain by reducing the inflammatory response, modulating peripheral nociceptors, and attenuating central sensitization.9 The COX-2 inhibitors provide effective pain control, and a lesser degree of platelet dysfunction and gastrointestinal toxicity compared to nonselective NSAIDs.10 Celecoxib, a highly specific drug has shown excellent efficacy in relieving inflammation and the associated pain,11 particularly postoperative pain.12
No trial has compared the efficacy of single-dose administration of pregabalin and celecoxib for attenuating postoperative pain after open cholecystectomy. The aim of the present study was to fill this void and to compare the analgesic effects of both drugs and their side-effects as a multimodal analgesia in this particular setting.
METHODOLOGY
This double blinded, randomized, controlled trial was [M1] conducted at Bolan Medical Complex Hospital, which is a tertiary care health facility at Quetta (Pakistan), from July 2010 to July 2011. Departmental ethical committee approval and written informed consent from all patients was obtained. The use of visual analogue scale (VAS) was explained to the patients during pre- anaesthesia consultation.
We included 60 adult patients of either sex, of ages 30–60 years, ASA physical status I or II, undergone open cholecystectomy under general anaesthesia. Patients with the following conditions were excluded from the study; kidney or liver dysfunction, asthma, coagulopathies, history of drug or alcohol abuse, history of chronic pain or routine intake of analgesics, uncontrolled medical disease like diabetes mellitus and hypertension, and intake of NSAIDS within 24 hours before surgery.
A sample size of twenty five by group was calculated to detect a significant difference of 2 cm VAS with power of 80% and significance level of 5 %. To take care of drop outs we enrolled thirty patients in each group. Sixty patients meeting the inclusion criteria were randomly assigned into two groups of thirty each, to receive either celecoxib 200 mg or pregabalin 150 mg, the doses suggested by previous studies. All the medications were identical, and administered orally 1 hour before the start of anaesthesia with sips of water by a staff nurse who was not involved in the study. [M2]
A standardized anaesthesia technique was used in both groups. Patients were induced with propofol 2 mg/kg, orotracheal intubation facilitated by atracurium 0.5 mg/ kg, nalbuphine 0.2 mg/kg was used for analgesia. Anaesthesia was maintained with isoflurane in 66% nitrous oxide and oxygen. All patients were monitored according to American Society of Anesthesiologist (ASA) recommendations. At the end of surgery, neuromuscular paralysis was reversed with neostigmine and glycopyrrolate. After satisfactory recovery, the patients were extubated and shifted to the post-anaesthesia care unit (PACU). In the PACU, nalbuphine 5 mg IV bolus was used as rescue analgesia, if pain score on VAS was four or more.
Primary outcomes were severity of postoperative pain and postoperative analgesic requirement. Secondary outcomes were incidence and severity of side-effects such as postoperative nausea and vomiting (PONV), headache, sedation, visual disturbances and respiratory depression if any. The outcomes were assessed by an anaesthetist blinded to group allocation.
Assessment of pain was done by a 10 cm VAS, marked 0 (no pain) at one end and 10 (the worst imaginable pain) on the other. Assessment of pain was done on arrival of patient to the PACU (0 hr) and then after 1 hr, 2hr, 4hr, 8hr, 16 hr, and 24 hr after operation. Pain scores at different time intervals for each patient were considered for statistical analysis. The severity of PONV was graded on a three-point ordinal scale (0 = no nausea or vomiting; 1 = nausea; and 2 = severe nausea with vomiting) and recorded in two phases of 12 hours each. Metocloperamide 10 mg IV was used in patients with PONV of grade 2. A four-point verbal scale (0 = none; 1 = slight; 2 = moderate and 3 = severe) was used to assess the sedation; patients with a sedation score of 2 or more were considered as sedated. Sedation was also recorded in two phases of 12 hours each. Respiratory depression was defined as respiratory rate ≤ 8 bpm and oxygen saturation <90% without oxygen supplementation. Occurrence of headache and visual disturbances was also noted.
SPSS 14.0 (SPSS Inc., Chicago, IL, USA) was used for statistical analysis. Descriptive statistics were used to describe the data. Patient characteristics were analysed with student’s t-test for continuous variables and chi square for categorical variables. The VAS pain scores were analysed with Mann–Whitney U-test[M3] ; frequency of side-effects were analysed with Fisher's exact test. P <0.05 was considered significant.[M4]
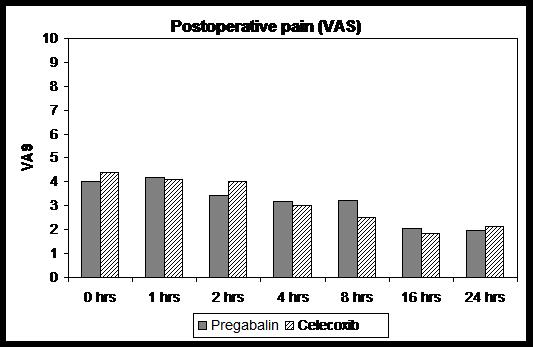
There was no difference in demographic characteristics among the groups with regard to age, weight, sex, duration of anaesthesia, and duration of surgery (Table 1). Postoperative pain scores were not significantly different in the pregabalin group when compared to celecoxib group (P>0.05) [M5] (Fig 1). Consumption of nalbuphine in each group was statistically not different; 13.5 ± 7.4 mg vs. 13 ± 6.7mg respectively (P >0.05).
No significant difference was found in frequency and severity of PONV at recovery and during next 24 hours (P > 0.05). Frequency and severity of sedation were higher in pregabline group (P <0.05) in first 12 hours but later on no significant differences were observed between the groups (Table 2). Number of patients requiring antiemetics for PONV was not significantly different (P > 0.05). Respiratory depression was not noted in any patient, but incidence of headache was higher in celecoxib group, whereas incidence of visual disturbance was higher in pregabalin group (P<0.05) (Table 2).
Prevention of post surgical pain early and late is an important challenge for anaesthesiologists.13 Although parenteral opioids remain the most widely used analgesics for the treatment of moderate to severe pain,14 adequate analgesia can only be achieved with associated risk of significant adverse events (e.g., nausea, vomiting, drowsiness, and respiratory depression).15 Postoperative pain involves multiple mechanisms; a multimodal analgesic regimen involving a combination of opioids, nonopioid analgesics, and regional analgesia has become the preferred choice.16 Concerns regarding the potential effects of nonselective NSAIDs on coagulation profile and the COX-2 inhibitors to increase prothrombotic complications after major surgery, lead investigators to use non-opioid analgesics, which are devoid of these side effects, as alternative analgesics.17
Preliminary studies using pregabalin have reported similar beneficial effects to the COX-2 inhibitors, so this was the base of our study. Pregabalin reduces the release of several neurotransmitters, including glutamate, noradrenaline, calcitonin gene-related peptide, (CGRP) and substance P. Modulation in neurotransmitter release is presumed to account for pregabalin analgesic actions.18,19
Our study could not confirm the hypothesis that pregabalin would demonstrate prolonged and additive analgesia. In a previously described study by Hill et al20, significant pain-relieving properties of pregabalin were demonstrated for patients in dental surgery with longer duration of analgesia when compared to ibuprofen. It was the first trial to report on the use of pregabalin for treating pain after surgery. They investigated the postoperative analgesic effect of pregabalin in a dose of 300 mg with ibuprofen 400 mg. Jokela et al21 observed that perioperative administration of pregabalin 300 mg before and after laparoscopic hysterectomy decreases oxycodone consumption, but is associated with an increased incidence of adverse effects. We observed no significant differences in postoperative nalbuphine consumption in both the groups, but our observations confirmed the findings of the study regarding incidence of adverse effects.22,23
Sedation scores in our study were high during first 12 hours in pregabalin group, a limiting factor for their use in day case set up. A recent meta-analysis24 confirmed the analgesic efficacy of the gabapentin compounds in the postoperative period but data regarding pregabalin is not convincing. In this regard a study by Paech and colleagues25 also supported our data, where they reported that a single preoperative dose of 100 mg pregabalin was ineffective in reducing acute postoperative pain or improving recovery after minor surgery involving only the uterus. Most of the trials used placebo for comparison; we used celecoxib to compare the analgesic effects of pregabalin.
The classical NSAIDS e.g., diclofenic, ketorolac and naproxen, nonselective at COX-1 and COX-2 receptors, can produce significant opioid-sparing effects; but producing their side effects (e.g., bleeding complications, renal dysfunction, gastrointestinal distress).26,27 Peters et al28 used COX-2 inhibitors as a part of multimodal analgesic regimen in patients undergoing total joint arthroplasty procedures and reported early mobilization, shortened length of stay, and improved pain control with less opioid use. We selected celecoxib for the same reasons; good analgesia with minimal risk of bleeding in open cholycystectomy, which is associated with more tissue trauma compared to laparoscopic cholecystectomy. Placebo-controlled studies evaluating the effects of preoperative administration of a COX-2 inhibitor reported no significant increase in intraoperative bleeding, the likelihood of a reoperation due to hematoma formation (29) or serious cardiovascular complications.29,30 Analogous to celecoxib, pregabalin reduced postoperative opioid usage compared with a placebo. The optimal dosing regimen remains unresolved, many trials have mentioned that it can be started at an effective dose of 150 mg/day preoperatively,31 the dose and timing of pregabalin used in the present study. Reuben et al used a larger dose of 150 mg given on two separate occasions where as we used single dose preoperatively; Chang et al used 300mg of pregabalin in their study.32,33 The established side effects of pregabalin are somnolence, dizziness, diplopia, and confusion.23 We particularly wished to avoid these side effects in our population where postoperative care is suboptimal. Nevertheless, we found that these and similar effects would affect discharge time and patient satisfaction adversely, particularly in public hospitals where few nurses are available to care for a large number of patients.
Limitations of the study: The primary limitation of this study was the lack of a placebo group to compare the opioid sparing effects of the trial drugs, as neither drug was found to reduce opioid-related side effects in the postoperative period. More clinical trials for perioperative pain management are needed till an optimum regimen is finally established.
Cochrance Database Sys Rev 2006;18(4):CD006229.
2. Syrakos T, Antonitsis P, Zacharakis E, Takis A, Manuousari A, Bakogiannis K, et al. Small incision (Mini-Laparotomy) versus laparoscopic cholecystectomy: A retrospective study in a University Hospital. Langenbecks Arch Surg 2004;389(3):172-177.
3. Berggren U, Gordh T, Grame D, Haglund U, Rastad J, Arvidsson D. Laparoscopic versus open cholecystectomy hospitalization, sick leave, analgesia and trauma responses. Br J Surg 1994;81(9):1362-1365.
4. Gottschalk A, Smith DS. New concepts in acute pain therapy: preemptive analgesia. Am Fam Physician 2001;63:1979–84.
5. White PF, Kehlet H. Postoperative pain management and patient outcome: time to return to work! Anesth Analg 2007;104:487–9 [Editorial]
6. Ittichaikulthol W, Virankabutra T, Kunopart M, Khamhom W, Putarawuthichai P, Rungphet S. Effects of Pregabalin on Postoperative Morphine Consumption and Pain after Abdominal Hysterectomy with/without Salphingo-oophorectomy:A randomized, Double-Blind Trial. J Med Assoc Thai 2009;92(10):1318-23
7. Bryans JS, Wustrow DJ. 3-substituted GABA analogs with central nervous system activity: a review. Med Res Rev 1999;19:149–77.
8. Agarwal A, Guatum S, Gupta D, Agarwal S, Singh PK, Singh U. Evaluation of a single preoperative dose of pregabalin for attenuation of postoperative pain after laparoscopic cholecystectomy. Br J Anaesth 2008;101(5):700-704
9. Reuben SS. Update on the role of nonsteroidal anti-inflammatory drugs and coxibs in the management of acute pain. Curr Opin Anaesthesiol 2007;20:440-450
10. Akaraviputh T, Leelouhapong C, Lohsiriwat V, Aroonpruksakul S.Efficacy of perioperative parecoxib injection on postoperative pain relief after laparoscopic cholecystectomy: A prospective randomized study. World J Gastroenterol 2009;15(16):2005-8.
11. Mateos JL. Selective inhibitors of cyclooxygenase-2 (COX-2), celecoxib and parecoxib: a systematic review. Drugs Today (Barc);2010 Feb;46 Suppl A:1-25.
12. Derry S, Barden J, McQuay HJ, Moore RA: Single dose oral celecoxib for acute postoperative pain in adults. Cochrane Database Syst Rev 2008;(4):CD004233
13. Kehlet H, Jensen TS, Woolf CJ. Persistent postsurgical pain: risk factors and prevention. Lancet 2006;367:1618–25.
14. Walder B, Schafer M, Henzi I, Tramer MR. Efficacy and safety of patient-controlled opioid analgesia for acute postoperative pain. A quantitative systematic review. Acta Anaesthesiol Scand 2001;45:795– 804.
15. Wheeler M, Oderda GM, Ashburn MA, Lipman AG. Adverse events associated with postoperative opioid analgesia: a systematic review. Clin J Pain 2002;3:159–80.
16. De Cosmo G, Aceto P, Gualtieri E, Congedo E. Analgesia in thoracic surgery: review. Minerva Anestesiol 2008;75:393–400.
19. Dooley DJ, Donovan CM, Pugsley TA. Stimulus-dependent modulation of [3H] norepinephrine release from rat neocortical slices by gabapentin and pregabalin. J Pharmacol Exp Ther 2000;295:1086–93.
20. Hill CM, Balkenohl M, Thomas DW, Walker R, Mathé H, Murray G. Pregabalin in patients with postoperative dental pain. Eur J Pain 2001;5:119 –24.
21. Jokela R, Ahonen J, Tallgren M, Haanpaa M, Korttila K. A randomized controlled trial of perioperative administration of pregabalin for pain after laparoscopic hysterectomy. Pain 2008;134:106–12.
22. Jokela R, Ahonen J, Tallgren M, Haanpää M, Korttila K. Premedication with pregabalin 75 or 150 mg with ibuprofen to control pain after day-case gynaecological laparoscopic surgery. Br J Anaesth 2008;100:834–40.
23. Mikkelsen S, Hilsted KL, Andersen PJ, Hjortso NC, Englgaard TP, Jorgensen DG, et al. The effect of gabapentin on post-operative pain following tonsillectomy in adults. Acta Anaesthesiol Scand 2006;50:809–15.
24. Zhang J, Ho KY, Wana Y. Efficacy of pregabalin in acute postoperative pain: a meta- analysis. Br. J. Anaesth. 2011;106 (4):454-462.
25. Paech MJ, Goy R, Chua S, Scott K, Christmas T, Doherty DA. A randomized, placebo- controlled trial of preoperative oral pregabalin for postoperative pain relief after minor gynaecological surgery. Anesth Analg 2007;105:1449–53.
26. Outer AJ, Fredman B, White PF. Controversies in the perioperative use of nonsteroidal antiinflammatory drugs. Anesth Analg 1994;79:1178–90.
27. Ott E, Nussmeier NA, Duke PC, Feneck RO, Alston RP, Snabes MC, et al. Efficacy and safety of the cyclooxygenase 2 inhibitors parecoxib and valdecoxib in patients undergoing coronary artery bypass surgery. J Thorac Cardiovasc Surg 2003;125:1481-92.
28. Peters CL, Shirley B, Erickson J. The effect of a new multimodal perioperative anesthetic regimen on postoperative pain, side effects, rehabilitation, and length of hospital stay after total joint arthroplasty. J Arthroplasty 2006;21:132–8.
29. Reuben SS, Connelly NR. Postoperative analgesic effects of celecoxib or rofecoxib after spinal fusion surgery. Anesth Analg 2000;91:1221–5.
30. Moiniche S, Kehlet H, Dahl JB. A qualitative and quantitative systematic review of preemptive analgesia for postoperative pain relief: the role of timing of analgesia. Anesthesiology 2002;96:725–41.
31. Ben-Menachem E. Pregabalin pharmacology and its relevance to clinical practice. Epilepsia 2004;45:13–8.
32. Reuben SS, Buvanendran A, Kroin JS, Raghunathan K. The analgesic efficacy of celecoxib, pregabalin, and their combination for spinal fusion surgery. Anesth Analg 2006;103:271–7.
33. Chang SH, Lee HW, Kim HK, Kim SH, Kim DK. An evaluation of perioperative pregabalin for prevention and attenuation of postoperative shoulder pain after laparoscopic cholecystectomy. Anesth Analg 2009;109:1284-1286.
Table 1: Patients demographics and clinical data
* Nalbuphine 5mg IV bolus used as rescue analgesia
Data presented as mean±SD unless specified otherwise.
Table 2: Complications in postoperative period
The data presented as numbers with percentages and the data was analyzed by Fisher’s Exact Test. No significant difference was observed for PONV, P > 0.05. Signficance was present for sedation, visual disturbance, headache and headache plus visual disturbance, P <0.05
Figure 1: Mean pain scores on VAS in two groups (Mann Whitney U test)
*Assistant Professor, Department of Anaesthesiology, Bolan Medical College Quetta (Pakistan)
**Assistant Professor Department of Surgery, Bolan Medical College Quetta (Pakistan)
Correspondence: Dr. Amjad Ali, Assistant Professor, Department of Anaesthesiology, Bolan Medical College Quetta (Pakistan); Phone: 03337924149, 081-2853352; E-mail: dramjad_ali@hotmail.com; amjadalidr@gmail.com
ABSTRACT
Objective: To evaluate the efficacy of a single preoperative dose of pregabalin in comparison to celecoxib for attenuating postoperative pain after open cholecystectomy.Setup: Department of Anaesthesiology, Bolan Medical Complex Hospital, Quetta.
Design: Randomized, controlled trial.
Methodology: Sixty adult patients, 35–65 years of age, ASA physical status I or II, of either sex, undergoing elective open cholecystectomy were allocated randomly in this prospective, randomized, controlled study. Patients were randomly divided into two groups of 30 each, to receive either pregabalin 150 mg or celecoxib 200 orally 1 hour before surgery. Postoperative pain was assessed by a 10cm visual analogue scale, where 0 denoted no pain and 10 was taken as the worst imaginable pain. Sedation, postoperative nausea and/or vomiting and any other complication, were assessed periodically throughout the study time. Patients received inj. nalbuphine on demand during the postoperative period and nausea and vomiting were treated with metocloperamide 10 mg. Results were statistically analysed.
Results: Cumulative consumption of inj. nalbuphine over 24 hours was 13.5 ± 7.4 mg for pregabalin group, and 13 ±6.7mg for celecoxib group (P >0.05). No significant difference was found in frequency and severity of postoperative nausea and vomiting (PONV) during 24 hours (P > 0.05), although frequency of sedation was higher in pregabline group (P <0.05).
Conclusion: Preoperative single dose of pregabalin 150 mg provides no better pain relief when compared to celecoxib 200 mg, and high incidence of adverse effects were found in patients receiving pregabline before open cholycystectomy.
Keywords: Postoperative pain; Celecoxib; Pregabalin; Non-opioid analgesics; Open cholecystectomy.
Citation: Ali A, Babar KM. Comparison of preoperative dose of pregabalin with celecoxib for attenuation of postoperative pain after open cholecystectomy. Anaesth Pain & Intensive Care 2012; 16(2): 137-141
INTRODUCTION
Historically, open cholecystectomy has been performed for the diseased gall bladder for more than 100 years,1 but with the introduction of laparoscopy into general surgical practice in 1990,2 laparoscopic cholecystectomy is now considered as the first option for cholelithiasis.3 Unfortunately, a lack of facilities and expertise in laparoscopic techniques has restricted the operative options solely to open cholecystectomy in the Balochistan province of Pakistan. It has been suggested that pain is inadequately treated in more than fifty percent of the surgical patients.4 The goal in optimizing postoperative pain management should be to reduce pain, improve the quality of the patient's recovery, and facilitate early resumption of normal activities of daily living.5 The increased cost on prolonged indoor treatment has compelled anaesthesiologists to use multiple approaches to achieve these goals. Postoperative pain may result from diverse aetiologies and mechanisms; therefore, its management may be difficult to achieve with the use of one approach. Multimodal analgesia regimen, with a combination of opioid and non-opioid analgesics, is expected to enhance analgesic efficacy and reduce total opioid requirements and the side effects.6Pregabalin and its developmental predecessor gabapentin, were originally developed as spasmolytic agents and adjuncts for the management of generalized or partial epileptic seizures resistant to conventional therapies.7 Like gabapentin, pregabalin has a proven role in treating neuropathic pain and postoperative pain after breast surgery, spinal surgery, and laparoscopic cholecystectomy surgeries.8
Recent research indicates that cyclooxygenase-2 (COX-2) inhibitors, a selective class of non-steroidal anti-inflammatory drugs (NSAIDs), could play an important role in perioperative pain by reducing the inflammatory response, modulating peripheral nociceptors, and attenuating central sensitization.9 The COX-2 inhibitors provide effective pain control, and a lesser degree of platelet dysfunction and gastrointestinal toxicity compared to nonselective NSAIDs.10 Celecoxib, a highly specific drug has shown excellent efficacy in relieving inflammation and the associated pain,11 particularly postoperative pain.12
No trial has compared the efficacy of single-dose administration of pregabalin and celecoxib for attenuating postoperative pain after open cholecystectomy. The aim of the present study was to fill this void and to compare the analgesic effects of both drugs and their side-effects as a multimodal analgesia in this particular setting.
METHODOLOGY
This double blinded, randomized, controlled trial was [M1] conducted at Bolan Medical Complex Hospital, which is a tertiary care health facility at Quetta (Pakistan), from July 2010 to July 2011. Departmental ethical committee approval and written informed consent from all patients was obtained. The use of visual analogue scale (VAS) was explained to the patients during pre- anaesthesia consultation.
We included 60 adult patients of either sex, of ages 30–60 years, ASA physical status I or II, undergone open cholecystectomy under general anaesthesia. Patients with the following conditions were excluded from the study; kidney or liver dysfunction, asthma, coagulopathies, history of drug or alcohol abuse, history of chronic pain or routine intake of analgesics, uncontrolled medical disease like diabetes mellitus and hypertension, and intake of NSAIDS within 24 hours before surgery.
A sample size of twenty five by group was calculated to detect a significant difference of 2 cm VAS with power of 80% and significance level of 5 %. To take care of drop outs we enrolled thirty patients in each group. Sixty patients meeting the inclusion criteria were randomly assigned into two groups of thirty each, to receive either celecoxib 200 mg or pregabalin 150 mg, the doses suggested by previous studies. All the medications were identical, and administered orally 1 hour before the start of anaesthesia with sips of water by a staff nurse who was not involved in the study. [M2]
A standardized anaesthesia technique was used in both groups. Patients were induced with propofol 2 mg/kg, orotracheal intubation facilitated by atracurium 0.5 mg/ kg, nalbuphine 0.2 mg/kg was used for analgesia. Anaesthesia was maintained with isoflurane in 66% nitrous oxide and oxygen. All patients were monitored according to American Society of Anesthesiologist (ASA) recommendations. At the end of surgery, neuromuscular paralysis was reversed with neostigmine and glycopyrrolate. After satisfactory recovery, the patients were extubated and shifted to the post-anaesthesia care unit (PACU). In the PACU, nalbuphine 5 mg IV bolus was used as rescue analgesia, if pain score on VAS was four or more.
Primary outcomes were severity of postoperative pain and postoperative analgesic requirement. Secondary outcomes were incidence and severity of side-effects such as postoperative nausea and vomiting (PONV), headache, sedation, visual disturbances and respiratory depression if any. The outcomes were assessed by an anaesthetist blinded to group allocation.
Assessment of pain was done by a 10 cm VAS, marked 0 (no pain) at one end and 10 (the worst imaginable pain) on the other. Assessment of pain was done on arrival of patient to the PACU (0 hr) and then after 1 hr, 2hr, 4hr, 8hr, 16 hr, and 24 hr after operation. Pain scores at different time intervals for each patient were considered for statistical analysis. The severity of PONV was graded on a three-point ordinal scale (0 = no nausea or vomiting; 1 = nausea; and 2 = severe nausea with vomiting) and recorded in two phases of 12 hours each. Metocloperamide 10 mg IV was used in patients with PONV of grade 2. A four-point verbal scale (0 = none; 1 = slight; 2 = moderate and 3 = severe) was used to assess the sedation; patients with a sedation score of 2 or more were considered as sedated. Sedation was also recorded in two phases of 12 hours each. Respiratory depression was defined as respiratory rate ≤ 8 bpm and oxygen saturation <90% without oxygen supplementation. Occurrence of headache and visual disturbances was also noted.
SPSS 14.0 (SPSS Inc., Chicago, IL, USA) was used for statistical analysis. Descriptive statistics were used to describe the data. Patient characteristics were analysed with student’s t-test for continuous variables and chi square for categorical variables. The VAS pain scores were analysed with Mann–Whitney U-test[M3] ; frequency of side-effects were analysed with Fisher's exact test. P <0.05 was considered significant.[M4]
RESULTS
A total of 60 patients were included in these clinical trails, 6 patients were dropped due to absence of designated nurses on those particular days and these subjects were replaced by new study patients to make the number in both groups equal. Subjects received study medication after randomization and the study was completed in twenty four of them.There was no difference in demographic characteristics among the groups with regard to age, weight, sex, duration of anaesthesia, and duration of surgery (Table 1). Postoperative pain scores were not significantly different in the pregabalin group when compared to celecoxib group (P>0.05) [M5] (Fig 1). Consumption of nalbuphine in each group was statistically not different; 13.5 ± 7.4 mg vs. 13 ± 6.7mg respectively (P >0.05).
No significant difference was found in frequency and severity of PONV at recovery and during next 24 hours (P > 0.05). Frequency and severity of sedation were higher in pregabline group (P <0.05) in first 12 hours but later on no significant differences were observed between the groups (Table 2). Number of patients requiring antiemetics for PONV was not significantly different (P > 0.05). Respiratory depression was not noted in any patient, but incidence of headache was higher in celecoxib group, whereas incidence of visual disturbance was higher in pregabalin group (P<0.05) (Table 2).
DISCUSSION
The results of our study demonstrated that no significant effect was made on postoperative nalbuphine analgesia by pregabline as compared to celecoxib given one hour before surgery in open cholycystectomy. Patients receiving pregabalin were more likely to experience side effects such as feeling light-headed or drowsy.Prevention of post surgical pain early and late is an important challenge for anaesthesiologists.13 Although parenteral opioids remain the most widely used analgesics for the treatment of moderate to severe pain,14 adequate analgesia can only be achieved with associated risk of significant adverse events (e.g., nausea, vomiting, drowsiness, and respiratory depression).15 Postoperative pain involves multiple mechanisms; a multimodal analgesic regimen involving a combination of opioids, nonopioid analgesics, and regional analgesia has become the preferred choice.16 Concerns regarding the potential effects of nonselective NSAIDs on coagulation profile and the COX-2 inhibitors to increase prothrombotic complications after major surgery, lead investigators to use non-opioid analgesics, which are devoid of these side effects, as alternative analgesics.17
Preliminary studies using pregabalin have reported similar beneficial effects to the COX-2 inhibitors, so this was the base of our study. Pregabalin reduces the release of several neurotransmitters, including glutamate, noradrenaline, calcitonin gene-related peptide, (CGRP) and substance P. Modulation in neurotransmitter release is presumed to account for pregabalin analgesic actions.18,19
Our study could not confirm the hypothesis that pregabalin would demonstrate prolonged and additive analgesia. In a previously described study by Hill et al20, significant pain-relieving properties of pregabalin were demonstrated for patients in dental surgery with longer duration of analgesia when compared to ibuprofen. It was the first trial to report on the use of pregabalin for treating pain after surgery. They investigated the postoperative analgesic effect of pregabalin in a dose of 300 mg with ibuprofen 400 mg. Jokela et al21 observed that perioperative administration of pregabalin 300 mg before and after laparoscopic hysterectomy decreases oxycodone consumption, but is associated with an increased incidence of adverse effects. We observed no significant differences in postoperative nalbuphine consumption in both the groups, but our observations confirmed the findings of the study regarding incidence of adverse effects.22,23
Sedation scores in our study were high during first 12 hours in pregabalin group, a limiting factor for their use in day case set up. A recent meta-analysis24 confirmed the analgesic efficacy of the gabapentin compounds in the postoperative period but data regarding pregabalin is not convincing. In this regard a study by Paech and colleagues25 also supported our data, where they reported that a single preoperative dose of 100 mg pregabalin was ineffective in reducing acute postoperative pain or improving recovery after minor surgery involving only the uterus. Most of the trials used placebo for comparison; we used celecoxib to compare the analgesic effects of pregabalin.
The classical NSAIDS e.g., diclofenic, ketorolac and naproxen, nonselective at COX-1 and COX-2 receptors, can produce significant opioid-sparing effects; but producing their side effects (e.g., bleeding complications, renal dysfunction, gastrointestinal distress).26,27 Peters et al28 used COX-2 inhibitors as a part of multimodal analgesic regimen in patients undergoing total joint arthroplasty procedures and reported early mobilization, shortened length of stay, and improved pain control with less opioid use. We selected celecoxib for the same reasons; good analgesia with minimal risk of bleeding in open cholycystectomy, which is associated with more tissue trauma compared to laparoscopic cholecystectomy. Placebo-controlled studies evaluating the effects of preoperative administration of a COX-2 inhibitor reported no significant increase in intraoperative bleeding, the likelihood of a reoperation due to hematoma formation (29) or serious cardiovascular complications.29,30 Analogous to celecoxib, pregabalin reduced postoperative opioid usage compared with a placebo. The optimal dosing regimen remains unresolved, many trials have mentioned that it can be started at an effective dose of 150 mg/day preoperatively,31 the dose and timing of pregabalin used in the present study. Reuben et al used a larger dose of 150 mg given on two separate occasions where as we used single dose preoperatively; Chang et al used 300mg of pregabalin in their study.32,33 The established side effects of pregabalin are somnolence, dizziness, diplopia, and confusion.23 We particularly wished to avoid these side effects in our population where postoperative care is suboptimal. Nevertheless, we found that these and similar effects would affect discharge time and patient satisfaction adversely, particularly in public hospitals where few nurses are available to care for a large number of patients.
Limitations of the study: The primary limitation of this study was the lack of a placebo group to compare the opioid sparing effects of the trial drugs, as neither drug was found to reduce opioid-related side effects in the postoperative period. More clinical trials for perioperative pain management are needed till an optimum regimen is finally established.
CONCLUSION
In patients who underwent open cholycystectomy, preoperative single dose of pregabalin 150 mg and celecoxib 200 mg oral administration used as an adjuvant, resulted in equivalent pain scores, but frequency of adverse effects observed was more in pregabalin group.ACKNOWLEDGMENTS
The authors gratefully acknowledge the assistance of our nursing staff, Dr. Nasir for expert statistical advice, and Mr Khadim Ali for computer formatting.REFERENCES
1. Keus F, De Jong JA, Gooszen HG, Van Laarhoven CJ. Laparoscopic versus small incision cholecystectomy for patients with symptomatic cholecystolithiasis..Cochrance Database Sys Rev 2006;18(4):CD006229.
2. Syrakos T, Antonitsis P, Zacharakis E, Takis A, Manuousari A, Bakogiannis K, et al. Small incision (Mini-Laparotomy) versus laparoscopic cholecystectomy: A retrospective study in a University Hospital. Langenbecks Arch Surg 2004;389(3):172-177.
3. Berggren U, Gordh T, Grame D, Haglund U, Rastad J, Arvidsson D. Laparoscopic versus open cholecystectomy hospitalization, sick leave, analgesia and trauma responses. Br J Surg 1994;81(9):1362-1365.
4. Gottschalk A, Smith DS. New concepts in acute pain therapy: preemptive analgesia. Am Fam Physician 2001;63:1979–84.
5. White PF, Kehlet H. Postoperative pain management and patient outcome: time to return to work! Anesth Analg 2007;104:487–9 [Editorial]
6. Ittichaikulthol W, Virankabutra T, Kunopart M, Khamhom W, Putarawuthichai P, Rungphet S. Effects of Pregabalin on Postoperative Morphine Consumption and Pain after Abdominal Hysterectomy with/without Salphingo-oophorectomy:A randomized, Double-Blind Trial. J Med Assoc Thai 2009;92(10):1318-23
7. Bryans JS, Wustrow DJ. 3-substituted GABA analogs with central nervous system activity: a review. Med Res Rev 1999;19:149–77.
8. Agarwal A, Guatum S, Gupta D, Agarwal S, Singh PK, Singh U. Evaluation of a single preoperative dose of pregabalin for attenuation of postoperative pain after laparoscopic cholecystectomy. Br J Anaesth 2008;101(5):700-704
9. Reuben SS. Update on the role of nonsteroidal anti-inflammatory drugs and coxibs in the management of acute pain. Curr Opin Anaesthesiol 2007;20:440-450
10. Akaraviputh T, Leelouhapong C, Lohsiriwat V, Aroonpruksakul S.Efficacy of perioperative parecoxib injection on postoperative pain relief after laparoscopic cholecystectomy: A prospective randomized study. World J Gastroenterol 2009;15(16):2005-8.
11. Mateos JL. Selective inhibitors of cyclooxygenase-2 (COX-2), celecoxib and parecoxib: a systematic review. Drugs Today (Barc);2010 Feb;46 Suppl A:1-25.
12. Derry S, Barden J, McQuay HJ, Moore RA: Single dose oral celecoxib for acute postoperative pain in adults. Cochrane Database Syst Rev 2008;(4):CD004233
13. Kehlet H, Jensen TS, Woolf CJ. Persistent postsurgical pain: risk factors and prevention. Lancet 2006;367:1618–25.
14. Walder B, Schafer M, Henzi I, Tramer MR. Efficacy and safety of patient-controlled opioid analgesia for acute postoperative pain. A quantitative systematic review. Acta Anaesthesiol Scand 2001;45:795– 804.
15. Wheeler M, Oderda GM, Ashburn MA, Lipman AG. Adverse events associated with postoperative opioid analgesia: a systematic review. Clin J Pain 2002;3:159–80.
16. De Cosmo G, Aceto P, Gualtieri E, Congedo E. Analgesia in thoracic surgery: review. Minerva Anestesiol 2008;75:393–400.
19. Dooley DJ, Donovan CM, Pugsley TA. Stimulus-dependent modulation of [3H] norepinephrine release from rat neocortical slices by gabapentin and pregabalin. J Pharmacol Exp Ther 2000;295:1086–93.
20. Hill CM, Balkenohl M, Thomas DW, Walker R, Mathé H, Murray G. Pregabalin in patients with postoperative dental pain. Eur J Pain 2001;5:119 –24.
21. Jokela R, Ahonen J, Tallgren M, Haanpaa M, Korttila K. A randomized controlled trial of perioperative administration of pregabalin for pain after laparoscopic hysterectomy. Pain 2008;134:106–12.
22. Jokela R, Ahonen J, Tallgren M, Haanpää M, Korttila K. Premedication with pregabalin 75 or 150 mg with ibuprofen to control pain after day-case gynaecological laparoscopic surgery. Br J Anaesth 2008;100:834–40.
23. Mikkelsen S, Hilsted KL, Andersen PJ, Hjortso NC, Englgaard TP, Jorgensen DG, et al. The effect of gabapentin on post-operative pain following tonsillectomy in adults. Acta Anaesthesiol Scand 2006;50:809–15.
24. Zhang J, Ho KY, Wana Y. Efficacy of pregabalin in acute postoperative pain: a meta- analysis. Br. J. Anaesth. 2011;106 (4):454-462.
25. Paech MJ, Goy R, Chua S, Scott K, Christmas T, Doherty DA. A randomized, placebo- controlled trial of preoperative oral pregabalin for postoperative pain relief after minor gynaecological surgery. Anesth Analg 2007;105:1449–53.
26. Outer AJ, Fredman B, White PF. Controversies in the perioperative use of nonsteroidal antiinflammatory drugs. Anesth Analg 1994;79:1178–90.
27. Ott E, Nussmeier NA, Duke PC, Feneck RO, Alston RP, Snabes MC, et al. Efficacy and safety of the cyclooxygenase 2 inhibitors parecoxib and valdecoxib in patients undergoing coronary artery bypass surgery. J Thorac Cardiovasc Surg 2003;125:1481-92.
28. Peters CL, Shirley B, Erickson J. The effect of a new multimodal perioperative anesthetic regimen on postoperative pain, side effects, rehabilitation, and length of hospital stay after total joint arthroplasty. J Arthroplasty 2006;21:132–8.
29. Reuben SS, Connelly NR. Postoperative analgesic effects of celecoxib or rofecoxib after spinal fusion surgery. Anesth Analg 2000;91:1221–5.
30. Moiniche S, Kehlet H, Dahl JB. A qualitative and quantitative systematic review of preemptive analgesia for postoperative pain relief: the role of timing of analgesia. Anesthesiology 2002;96:725–41.
31. Ben-Menachem E. Pregabalin pharmacology and its relevance to clinical practice. Epilepsia 2004;45:13–8.
32. Reuben SS, Buvanendran A, Kroin JS, Raghunathan K. The analgesic efficacy of celecoxib, pregabalin, and their combination for spinal fusion surgery. Anesth Analg 2006;103:271–7.
33. Chang SH, Lee HW, Kim HK, Kim SH, Kim DK. An evaluation of perioperative pregabalin for prevention and attenuation of postoperative shoulder pain after laparoscopic cholecystectomy. Anesth Analg 2009;109:1284-1286.
Table 1: Patients demographics and clinical data
| Variables | Pregabalin
n=30 |
Celecoxibe
n=30 |
P value |
| Age | 47.87 ± 9.52 | 48.33 ± 10.61 | P > 0.05 |
| Gender [n(%)] | |||
| Male | 10 (33.33%) | 6 (20 % ) | P > 0.05 |
| Female | 20 (66.7 % ) | 24 ( 80 % ) | |
| Weight | 61.81 ± 8.35 | 66.8 ± 11.4 | P> 0.05 |
| Duration of anesthesia ( Hrs) | 1.27 ± .161 | 1.36 ± 0.28 | P> 0.05 |
| Baseline Blood Pressure | |||
| Systolic | 118 ± 11.45 | 117 ± 11.3 | P > 0.05 |
| Diastolic | 71 ± 11.37 | 71 ± 11.84 | |
| Rescue analgesia consumed in 24 hours*
|
13.5 ± 7.4 | 13± 6.75 | P >0.05 |
* Nalbuphine 5mg IV bolus used as rescue analgesia
Data presented as mean±SD unless specified otherwise.
Table 2: Complications in postoperative period
| Pregabalin
N=30 |
Celecoxib
N=30 |
P value | |
| PONV (in PACU) | |||
| Nausea | 8 (26 %) | 7 (3.33 %) | P >0.05 |
| Vomiting | 6 (20 %) | 5 (16.6 %) | |
| PONV first 12 hours | |||
| Nausea | 9 (30 %) | 7 (23.3 %) | P > 0.05 |
| Vomiting | 7 (23 %) | 3 (10 %) | |
| PONV next 12 hours | P > 0.05 | ||
| Nausea | 5 (16 %) | 3 (10 %) | |
| Vomiting | 3 (10 %) | 2 (6.6%) | |
| Total Anti emetic used | 16 (53.7 % ) | 10 ( 30 % ) | |
| Sedation first 12 hours | |||
| None Mild Moderate |
8 (26.6 %) 10 (33.3 %) 9 (30%) |
12 (40) 12 (40 %) 6 (20 % ) |
P < 0.05 |
| Sever | 3 (10 % ) | nil | |
| Sedation next 12 hours | |||
| None | 17 ( 56 %) | 23 (76.6%) | P > 0.05 |
| Mild | 8 (%) | 5 (16.6%) | |
| Moderate | 5 (%) | 2 (6.6%) | |
| Sever | 0 | 0 | |
| Visual disturbance | 6 (20 %) | 0 | P < 0.05 |
| Headache | 1 (3 %) | 5 (16 %) | |
| Headache+ Visual disturbance | 1 (3 %) | 0 |