Baydaa H. Obaid 1, Manar D. Salman 2 , Nahlah F. Makki 3 , Ali A. Ahmed 4
Authors affiliations:
Background & objective: Hypothyroidism is a common disorder that affects metabolism and growth. Vitamin deficiencies, such as D3 and B12, are believed to exacerbate the symptoms of this condition. The mechanism explaining this effect is not precise, but it may be due to thyroid hormone receptors. This study used molecular docking techniques to analyze the relationship between vitamin D3 and B12 deficiency and hypothyroidism.
Methodology: The study included 90 women aged 35 and 55, divided into 45 hypothyroid patients and 45 control patients. Levels of thyroid hormones (T3 and T4), thyroid-stimulating hormone (TSH), calcium, vitamins D3 and B12 were determined. Molecular docking techniques study the interaction of thyroid hormone receptors (TRα and TRβ) with vitamins.
Results: The results showed a significant decrease in thyroid hormones, calcium, vitamin D3, and B12 levels, with increased TSH (p<0.01). The molecular docking results also showed a strong interaction between vitamin D3 and B12 with thyroid hormone receptors, indicating a possible regulatory role for gene expression, leading to an increase in the binding of thyroid hormone and its receptors. Additionally, the results of the ROC curve revealed 100% specificity and sensitivity for vitamin D3, vitamin B12, and calcium, and the cutoff points were (24.99, 214.5, and 8.26), respectively; these results indicate that these vitamins and calcium are ideal indicators for diagnosing the disease.
Conclusions: This study supports the hypothesis that vitamin D3 and vitamin B12 deficiency contribute to the worsening of hypothyroidism through their relationship with thyroid hormone receptors. Further studies are needed to understand the precise mechanisms of this relationship and the potential use of vitamin supplements as part of treatment strategies.Abbreviations: THR - thyroid hormone receptors; TSH - thyroid-stimulating hormone; HDL – high-density lipoprotein; TG - triglyceride; BMI - body mass index; SBP - systolic blood pressure; DBP - diastolic blood pressure Keywords: Hypothyroidism; Receptors; Molecular Docking Techniques; Thyroid hormone receptors; TRα; TRβ.
Citation: Baydaa H. Obaid BH, Salman MD, Makki NF, Ahmed AA. The role of vitamin D3 and B12 in thyroid health: in silico evaluation of potential binding to thyroid hormone receptors. Anaesth. pain intensive care 2026;30(2):215-223. DOI: 10.35975/apic.v30i2.3129
Received: January 09, 2026; Reviewed: January 12, 2026; Accepted: January 14, 2026
Thyroid hormone receptors (THRs), which are nuclear proteins that attach to thyroid hormones like thyroxine and triiodothyronine to control gene expression and affect metabolism, growth, and development, are primarily responsible for the activity of thyroid hormones.1 The thyroid hormone has two receptors. They are a subset of these nuclear receptors: Alpha receptor for thyroid hormone (TRα), primarily found in the neurological system, skeletal muscle, and heart, which has a significant impact on the development of the neurological system and heart rate regulation. The second receptor is a beta (TRβ) found in the brain, pituitary gland, liver, and kidneys, regulating metabolism, cholesterol levels, and hormonal balance.2 The most common cause of Hypothyroidism worldwide is too little iodine in the diet. Still, less common causes include radioactive iodine treatment, hypothalamic injury, certain medications, or thyroid surgery. Blood tests can confirm the diagnosis of Hypothyroidism, a disorder in which the thyroid gland does not produce enough thyroid hormones, resulting in symptoms such as fatigue, muscle aches, constipation, fatigue, depression, and weight gain.3,4 In Hypothyroidism, thyroid hormone receptors may change activity, quantity, and gene expression, and serum vitamin D can be evaluated as calcidiol levels.5 Two hydroxylation processes are involved: first, in the liver (25-hydroxy vitamin D or calcidiol), and subsequently, 25-hydroxy vitamin D is transformed in humans into active hormone (1, 25-dihydroxycholecalciferol or calcitriol) in the kidney.6 There are two primary vitamin D types: vitamin D2 (ergocalciferol) and vitamin D3 (cholecalciferol). The plant sterol ergosterol and fungi are the sources of vitamin D2 in supplements. In contrast, vitamin D3 is produced beneath the skin upon exposure to sunlight and is found in animal-derived foods.7 Low blood calcidiol (< 25 nmol/l) levels indicate this vitamin deficiency. Vitamin D prevents cancer and is crucial for maintaining healthy physical systems, including the immune system.8-10 Low serum vitamin D3 levels in hypothyroid patients may lead to musculoskeletal issues in these individuals. Two explanations exist for the low D3 levels in hypothyroid patients: the body may not properly activate D3 or poor absorption of vitamin D3 from the intestine.11 Some studies have found a link between D3 deficiency and autoimmune thyroid diseases like HT and Graves' disease, and low vitamin D3 levels related to thyroid malignancies.12 Vitamin B12, also called cobalamin, is necessary for producing red blood cells, cell metabolism, neuron function, and DNA synthesis—molecules inside cells containing genetic information. Meat, poultry, fish, and dairy products are among the foods that contain vitamin B12.13 Vitamin B12 insufficiency and autoimmune thyroid disorders are likely related since they are often accompanied by other autoimmune conditions, such as atrophic gastritis and/or pernicious anemia, which decrease vitamin B12 absorption. Patients with Hypothyroidism are more likely to have vitamin B12 deficiency as they age. In patients with hypothyroidism and vitamin B12 insufficiency, the symptoms of both conditions are typically present, including weakness, weariness, loss of feeling, itching, and poor memory retention.14,15 Vitamin B12 deficiency hinders the production of thyroid hormone, exacerbating hypothyroidism symptoms and Hashimoto thyroiditis.16 This study aims to investigate the relationship between vitamin D3 and B12 deficiency and Hypothyroidism in women in Baghdad. It analyzes the biochemical levels of these vitamins and evaluates their interaction with thyroid hormone receptors using molecular correlation techniques to understand their potential role in regulating thyroid functions.
2.1. Protein Preparation and Ligands
Thyroid hormone receptor alpha (TRα) and TRβ in humans were obtained from the protein data bank (PDB) by employing the PDB IDs (1NAV and 1XZX), respectively. Further, the docking experiment's target proteins are selected depending on their X-ray diffraction. Thyroid hormone receptor alpha and beta (TRβ) X-ray obtained crystallographic structures for molecular docking by removing all heteroatoms from the PDB, including ions and water, using the Chimera tool and the Discovery Studio 2021 Client.17 The chain selects specific protein-binding sites while avoiding others.
2.2. Preparation of Ligands
Open Babel software and PubChem were used to retrieve the 3D chemical structures in SDF format, and PubChem was used to convert the compounds to PDB format.18 Vitamins D3 and B12 were used in all docking research stages and retrieved via PubChem17.
2.3. Molecular Docking
Molecular docking studies were performed using the AutoDock Vina software on the PyRx platform. Ligands and protein-receptor structures were prepared using the AutoDock tool, and all files were converted to PDBAT format. Blind docking was applied by selecting grid boxes to cover the entire receptor surface. For thyroid hormone receptor alpha, the center of the grid box was set at the following coordinates: vitamin D3: X = 54.5034, Y = 59.3040, Z = 45.9163), B12: X = 57.4195, Y = 62.4676, Z = 51.8841, while beta thyroid hormone receptor, the coordinates: D3:X = 45.7751, Y = 55.2060, Z = 59.9333), B12: X = 39.0471, Y = 53.4503, Z = 57.8232. The docking results were visualized and analyzed using Discovery Studio Visualizer 3.0.18
2.4. Participants and Assay
This case-control study included 45 hypothyroid and 45 healthy female participants aged 35–55 years. Samples were collected at the City of Medicine / Educational Laboratories between November 2024 and March 2025. Endocrinology specialists confirmed the diagnosis. A structured questionnaire was used to record systolic and diastolic blood pressure, weight, and height. Exclusion criteria included chronic liver or kidney disease, rheumatoid arthritis, thyroid cancer, obesity, and pregnancy. Ethical approval was obtained from the
Institutional Review Board of the City of Medicine, by the Declaration of Helsinki. All participants provided written informed consent.
Eight milliliters of venous blood were collected from all participants in the early morning into clean gel tubes. Hemolyzed samples were excluded. After clotting, samples were centrifuged at 3500 rpm for 10 minutes, and serum was aliquoted into five Eppendorf tubes and stored at -80 °C. Serum levels of T3, T4, TSH, vitamin D3, and vitamin B12 were measured using ELISA kits (BioMerieux, France), while lipid and calcium levels were assessed spectrophotometrically.
2.4. Statistical analysis
Data were analyzed using SPSS version 25 (IBM, Chicago, IL). Descriptive statistics (count, mean, standard deviation) were applied, and comparisons were made using Student’s t-test. A p-value ≤ 0.05 was considered statistically significant. ROC curve analysis was used to assess the diagnostic value of vitamin D3, B12, and calcium levels in hypothyroidism.19
3.1. Molecular Docking study (Thyroid hormone receptors TRα and TRβ)
Table 1 displayed the results of a molecular docking study of vitamin D3 and vitamin B12 with two types of thyroid hormone receptors: TRα and TRβ.
Table 2 shows how the ligand interacts with the fourteen-amino-acid chain A of the thyroid hormone receptor alpha (TRα).
In Table 3 shows how the ligand interacts with the 18-amino-acid chain A of the thyroid hormone receptor alpha (TR α). The results in Table 4 display how the ligand interacts with the eleven amino acid chain X of the thyroid hormone receptor beta (TRβ).
The output in Table 5 indicates the interaction of the ligand with chain X of the thyroid hormone receptor beta (TRβ), which has eighteen twenty amino acids.
3.2. Clinical study
The findings in Table 6 shows the clinical characteristics and comparative analysis between hypothyroid patients and healthy controls across a range of biomarkers and physiological parameters, using t-test.
The outputs of the ROC curve were displayed in Table 7, indicate that vitamin D3, vitamin B12, and calcium have 100% specificity and sensitivity in Figure 3.
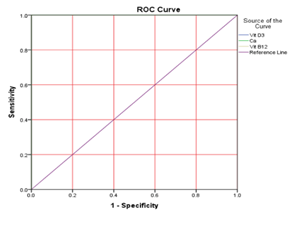
Figure 3: Receiver operating characteristic curves of measured vitamin D3, B12, and calcium in differentiating between hypothyroid patients
4.1. Molecular Docking study (Thyroid hormone receptors TRα and TRβ)
Thyroid hormone receptor alpha (TRα) is primarily expressed in the brain, heart, and bone, whereas TRβ1 is found in the liver, kidney, and thyroid, and TRβ2 in the retina, cochlea, and pituitary gland.20 Mutations in these receptors can alter serum thyroid hormone levels. TRα is a key docking target for vitamins D3 and B12, as shown in Table 1, with stronger receptor–ligand interactions indicated by more negative binding affinity values. Molecular docking employs root mean square deviation (RMSD) to assess protein structural changes. The upper bound RMSD compares each atom to itself without considering symmetry, while the lower bound RMSD matches each atom to the nearest of the same element type.21,22
Similarly, TRβ docking studies show stronger ligand-receptor interactions with higher negative affinity values, evaluated using the same RMSD criteria. Docking analysis revealed three main types of interactions between vitamin D3 and thyroid hormone receptor alpha (TRα) chain A: van der Waals, Pi-Alkyl, and Alkyl bonds. The binding affinity was strong, with low binding energy (−6.9 kcal/mol), indicating a stable interaction. The RMSD value between the docked and
experimental ligands was 0.0 Å, confirming the reliability of the docking results. Using PyRx software, virtual screening identified optimal binding of vitamin D3 to active sites on TRα, as detailed in Table 2 and visualized in Figure 1. Docking analysis showed that vitamin B12 interacts with thyroid hormone receptor alpha (TRα) chain A through conventional hydrogen bonds and van der Waals forces. The interaction demonstrated good stability, with a low binding energy of −6.8 kcal/mol. The RMSD value between the docked and experimental ligands was 0.0 Å, confirming docking accuracy. Using PyRx software, vitamin B12 was virtually screened and found to bind effectively to active sites on TRα, as detailed in Table 2 and illustrated in the Figure. 1B.
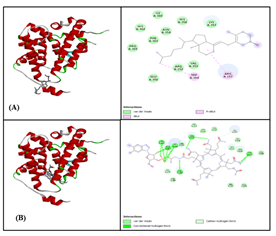
Figure 1: Thyroid hormone receptor alpha (TRɑ) with A. Vitamin D3 B. Vitamin B12
Vitamin D3 showed strong binding to thyroid hormone receptor beta (TRβ) chain X through three types of hydrogen bond interactions: conventional, alkyl, and van der Waals. The binding affinity was high, with a low energy value of −7.3 kcal/mol. The RMSD between docked and experimental ligands was 0.0 Å, indicating accurate docking. Virtual screening using PyRx confirmed optimal interaction of vitamin D3 within the active sites of TRβ, as shown in Table 4 and visualized in Figure 2A. Vitamin B12 interacted with thyroid hormone receptor beta (TRβ) chain X through five types of bonds: conventional hydrogen bonds, van der Waals, Pi-cation, alkyl, and unfavorable donor–donor interactions. The binding affinity was strong, with a low energy of −7.1 kcal/mol. The RMSD value between docked and experimental ligands was 0.0 Å, confirming the validity of the docking results. PyRx-based virtual screening identified optimal binding of vitamin B12 to active sites on TRβ, as detailed in Table 5 and shown in Figure 2B. The study demonstrated strong binding affinities between vitamin D3 and thyroid hormone receptors TRα and TRβ (−6.9 and −7.3 kcal/mol, respectively), supporting the association between low vitamin D3 levels and hypothyroidism.
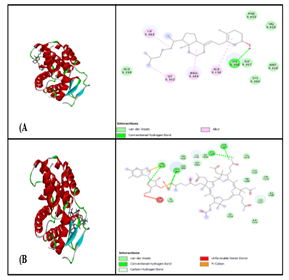
Figure 2: Thyroid hormone receptor alpha (TRβ) with A. Vitamin D3 B. Vitamin B12
This deficiency is commonly observed in hypothyroid patients and may result from impaired vitamin D metabolism or shared autoimmune mechanisms.23 Similarly, vitamin B12 exhibited good binding affinity with TRα and TRβ (−6.8 and −7.1 kcal/mol, respectively), consistent with previous reports of reduced serum B12 levels in individuals with hypothyroidism. This reduction is likely attributed to impaired gastrointestinal absorption and digestion associated with hypothyroid states. Furthermore, vitamin B12 deficiency may exacerbate symptoms such as fatigue and neuropathy.24-26 These findings suggest that vitamins D3 and B12 have regulatory roles at the molecular level in thyroid function. However, the precise physiological mechanisms underlying these interactions remain unclear and warrant further investigation.
4.2. Clinical study
This study adds to the growing evidence linking vitamins D3 and B12 to hypothyroidism. A significant increase in systolic and diastolic blood pressure was observed in hypothyroid patients compared to controls (p < 0.01), consistent with the known cardiovascular effects of thyroid hormone deficiency, which include reduced heart rate and increased vascular resistance.27-30 Additionally, BMI was significantly elevated in patients (35.28 ± 3.08), reflecting the well-established association between hypothyroidism and obesity.31,32 Vitamin D3, B12, and calcium levels were significantly reduced in hypothyroid patients. Hypothyroidism impairs metabolism, which may minimize intestinal vitamin D absorption, contributing to a calcium imbalance. These findings align with previous studies highlighting the interconnected role of thyroid function and vitamin D/calcium homeostasis.33,34 Vitamin B12 deficiency is also common in hypothyroid patients, often linked to autoimmune conditions like atrophic gastritis, which impairs absorption. The deficiency tends to worsen with age, as previously reported.35,36 The study also found significantly higher triglyceride levels and reduced HDL (good cholesterol) in hypothyroid patients (P < 0.01), suggesting an increased risk of cardiovascular disease due to impaired lipid metabolism.37 These changes are attributed to decreased thyroid hormone levels, which slow down fat breakdown and liver cholesterol clearance. As expected, serum T3 and T4 levels were markedly lower, while TSH levels were significantly elevated in hypothyroid patients (P < 0.01), confirming primary hypothyroidism and reflecting a disruption in the hypothalamic–pituitary–thyroid axis.38 Finally, ROC curve analysis demonstrated that vitamin D3, vitamin B12, and calcium levels had 100% sensitivity and specificity, suggesting their potential utility as reliable biomarkers for hypothyroidism diagnosis. The respective cut-off values were 24.99 ng/mL, 214.5 pg/mL, and 8.26 mg/dL (Table 7; Figure 3).
These findings reinforce the hypothesis that micronutrients such as vitamin D3, vitamin B12, and calcium play a critical role in the pathogenesis of hypothyroidism. The observed associations align with previous studies suggesting a strong link between these nutrients and thyroid dysfunction.39 Therefore, routine monitoring and appropriate correction of
deficiencies through diet or supplementation under medical supervision may benefit individuals with hypothyroidism.
This study demonstrated that patients with hypothyroidism had significantly lower levels of vitamin D3 and B12 compared to healthy controls, suggesting a potential link between these deficiencies and thyroid dysfunction. Molecular docking analysis revealed strong interactions between both vitamins and thyroid hormone receptors (TRα and TRβ), implying a possible regulatory role at the molecular level. These findings underscore the importance of evaluating vitamin D3 and B12 levels in hypothyroid patients as part of clinical assessment to support early intervention and prevent disease progression.
6. Data availability
The numerical data generated and analyzed during this study are available from the corresponding author upon reasonable request.
7. Acknowledgement
The authors would like to thank all the staff at the City of Medicine / Educational Laboratories for their valuable support and cooperation during the study.
8. Conflict of interest
The authors declare that they have no competing financial or non-financial interests.
9. Authors’ contribution
All authors contributed equally to the design, execution of the study, and preparation of the manuscript.
Authors affiliations:
- Baydaa H. Obaid, Research and Technology of Environment, Water and Renewable Energy, Scientific Research Commission, Ministry of Higher Education and Scientific Research, Baghdad, Iraq; Email: Baidaa.hamad1105@csw.uobaghdad.edu.iq 2. Manar D. Salman, Research and Technology of Environment, Water and Renewable Energy, Scientific Research Commission, Ministry of Higher Education and Scientific Research, Baghdad, Iraq; Email: manar.dawood3@gmail.com 3. Nahlah F. Makki, Chemistry Department, Science, University of Kufa, Al Najaf, Iraq; Email: nahla.almutawall@uokufa.edu.iq 4. Ali A. Ahmed, Ministry of Education, Rusafa Second Directorate of Education, Baghdad, Iraq; Email: ali.alhussaini@student.uokufa.edu.iq
ABSTRACT
Background & objective: Hypothyroidism is a common disorder that affects metabolism and growth. Vitamin deficiencies, such as D3 and B12, are believed to exacerbate the symptoms of this condition. The mechanism explaining this effect is not precise, but it may be due to thyroid hormone receptors. This study used molecular docking techniques to analyze the relationship between vitamin D3 and B12 deficiency and hypothyroidism.
Methodology: The study included 90 women aged 35 and 55, divided into 45 hypothyroid patients and 45 control patients. Levels of thyroid hormones (T3 and T4), thyroid-stimulating hormone (TSH), calcium, vitamins D3 and B12 were determined. Molecular docking techniques study the interaction of thyroid hormone receptors (TRα and TRβ) with vitamins.
Results: The results showed a significant decrease in thyroid hormones, calcium, vitamin D3, and B12 levels, with increased TSH (p<0.01). The molecular docking results also showed a strong interaction between vitamin D3 and B12 with thyroid hormone receptors, indicating a possible regulatory role for gene expression, leading to an increase in the binding of thyroid hormone and its receptors. Additionally, the results of the ROC curve revealed 100% specificity and sensitivity for vitamin D3, vitamin B12, and calcium, and the cutoff points were (24.99, 214.5, and 8.26), respectively; these results indicate that these vitamins and calcium are ideal indicators for diagnosing the disease.
Conclusions: This study supports the hypothesis that vitamin D3 and vitamin B12 deficiency contribute to the worsening of hypothyroidism through their relationship with thyroid hormone receptors. Further studies are needed to understand the precise mechanisms of this relationship and the potential use of vitamin supplements as part of treatment strategies.Abbreviations: THR - thyroid hormone receptors; TSH - thyroid-stimulating hormone; HDL – high-density lipoprotein; TG - triglyceride; BMI - body mass index; SBP - systolic blood pressure; DBP - diastolic blood pressure Keywords: Hypothyroidism; Receptors; Molecular Docking Techniques; Thyroid hormone receptors; TRα; TRβ.
Citation: Baydaa H. Obaid BH, Salman MD, Makki NF, Ahmed AA. The role of vitamin D3 and B12 in thyroid health: in silico evaluation of potential binding to thyroid hormone receptors. Anaesth. pain intensive care 2026;30(2):215-223. DOI: 10.35975/apic.v30i2.3129
Received: January 09, 2026; Reviewed: January 12, 2026; Accepted: January 14, 2026
1. INTRODUCTION
Thyroid hormone receptors (THRs), which are nuclear proteins that attach to thyroid hormones like thyroxine and triiodothyronine to control gene expression and affect metabolism, growth, and development, are primarily responsible for the activity of thyroid hormones.1 The thyroid hormone has two receptors. They are a subset of these nuclear receptors: Alpha receptor for thyroid hormone (TRα), primarily found in the neurological system, skeletal muscle, and heart, which has a significant impact on the development of the neurological system and heart rate regulation. The second receptor is a beta (TRβ) found in the brain, pituitary gland, liver, and kidneys, regulating metabolism, cholesterol levels, and hormonal balance.2 The most common cause of Hypothyroidism worldwide is too little iodine in the diet. Still, less common causes include radioactive iodine treatment, hypothalamic injury, certain medications, or thyroid surgery. Blood tests can confirm the diagnosis of Hypothyroidism, a disorder in which the thyroid gland does not produce enough thyroid hormones, resulting in symptoms such as fatigue, muscle aches, constipation, fatigue, depression, and weight gain.3,4 In Hypothyroidism, thyroid hormone receptors may change activity, quantity, and gene expression, and serum vitamin D can be evaluated as calcidiol levels.5 Two hydroxylation processes are involved: first, in the liver (25-hydroxy vitamin D or calcidiol), and subsequently, 25-hydroxy vitamin D is transformed in humans into active hormone (1, 25-dihydroxycholecalciferol or calcitriol) in the kidney.6 There are two primary vitamin D types: vitamin D2 (ergocalciferol) and vitamin D3 (cholecalciferol). The plant sterol ergosterol and fungi are the sources of vitamin D2 in supplements. In contrast, vitamin D3 is produced beneath the skin upon exposure to sunlight and is found in animal-derived foods.7 Low blood calcidiol (< 25 nmol/l) levels indicate this vitamin deficiency. Vitamin D prevents cancer and is crucial for maintaining healthy physical systems, including the immune system.8-10 Low serum vitamin D3 levels in hypothyroid patients may lead to musculoskeletal issues in these individuals. Two explanations exist for the low D3 levels in hypothyroid patients: the body may not properly activate D3 or poor absorption of vitamin D3 from the intestine.11 Some studies have found a link between D3 deficiency and autoimmune thyroid diseases like HT and Graves' disease, and low vitamin D3 levels related to thyroid malignancies.12 Vitamin B12, also called cobalamin, is necessary for producing red blood cells, cell metabolism, neuron function, and DNA synthesis—molecules inside cells containing genetic information. Meat, poultry, fish, and dairy products are among the foods that contain vitamin B12.13 Vitamin B12 insufficiency and autoimmune thyroid disorders are likely related since they are often accompanied by other autoimmune conditions, such as atrophic gastritis and/or pernicious anemia, which decrease vitamin B12 absorption. Patients with Hypothyroidism are more likely to have vitamin B12 deficiency as they age. In patients with hypothyroidism and vitamin B12 insufficiency, the symptoms of both conditions are typically present, including weakness, weariness, loss of feeling, itching, and poor memory retention.14,15 Vitamin B12 deficiency hinders the production of thyroid hormone, exacerbating hypothyroidism symptoms and Hashimoto thyroiditis.16 This study aims to investigate the relationship between vitamin D3 and B12 deficiency and Hypothyroidism in women in Baghdad. It analyzes the biochemical levels of these vitamins and evaluates their interaction with thyroid hormone receptors using molecular correlation techniques to understand their potential role in regulating thyroid functions.
2. METHODOLOGY
2.1. Protein Preparation and Ligands
Thyroid hormone receptor alpha (TRα) and TRβ in humans were obtained from the protein data bank (PDB) by employing the PDB IDs (1NAV and 1XZX), respectively. Further, the docking experiment's target proteins are selected depending on their X-ray diffraction. Thyroid hormone receptor alpha and beta (TRβ) X-ray obtained crystallographic structures for molecular docking by removing all heteroatoms from the PDB, including ions and water, using the Chimera tool and the Discovery Studio 2021 Client.17 The chain selects specific protein-binding sites while avoiding others.
2.2. Preparation of Ligands
Open Babel software and PubChem were used to retrieve the 3D chemical structures in SDF format, and PubChem was used to convert the compounds to PDB format.18 Vitamins D3 and B12 were used in all docking research stages and retrieved via PubChem17.
2.3. Molecular Docking
Molecular docking studies were performed using the AutoDock Vina software on the PyRx platform. Ligands and protein-receptor structures were prepared using the AutoDock tool, and all files were converted to PDBAT format. Blind docking was applied by selecting grid boxes to cover the entire receptor surface. For thyroid hormone receptor alpha, the center of the grid box was set at the following coordinates: vitamin D3: X = 54.5034, Y = 59.3040, Z = 45.9163), B12: X = 57.4195, Y = 62.4676, Z = 51.8841, while beta thyroid hormone receptor, the coordinates: D3:X = 45.7751, Y = 55.2060, Z = 59.9333), B12: X = 39.0471, Y = 53.4503, Z = 57.8232. The docking results were visualized and analyzed using Discovery Studio Visualizer 3.0.18
2.4. Participants and Assay
This case-control study included 45 hypothyroid and 45 healthy female participants aged 35–55 years. Samples were collected at the City of Medicine / Educational Laboratories between November 2024 and March 2025. Endocrinology specialists confirmed the diagnosis. A structured questionnaire was used to record systolic and diastolic blood pressure, weight, and height. Exclusion criteria included chronic liver or kidney disease, rheumatoid arthritis, thyroid cancer, obesity, and pregnancy. Ethical approval was obtained from the
Institutional Review Board of the City of Medicine, by the Declaration of Helsinki. All participants provided written informed consent.
Eight milliliters of venous blood were collected from all participants in the early morning into clean gel tubes. Hemolyzed samples were excluded. After clotting, samples were centrifuged at 3500 rpm for 10 minutes, and serum was aliquoted into five Eppendorf tubes and stored at -80 °C. Serum levels of T3, T4, TSH, vitamin D3, and vitamin B12 were measured using ELISA kits (BioMerieux, France), while lipid and calcium levels were assessed spectrophotometrically.
2.4. Statistical analysis
Data were analyzed using SPSS version 25 (IBM, Chicago, IL). Descriptive statistics (count, mean, standard deviation) were applied, and comparisons were made using Student’s t-test. A p-value ≤ 0.05 was considered statistically significant. ROC curve analysis was used to assess the diagnostic value of vitamin D3, B12, and calcium levels in hypothyroidism.19
3. RESULTS
3.1. Molecular Docking study (Thyroid hormone receptors TRα and TRβ)
Table 1 displayed the results of a molecular docking study of vitamin D3 and vitamin B12 with two types of thyroid hormone receptors: TRα and TRβ.
| Table 1: Interaction analysis of the ligand with thyroid hormone receptors | ||||||
| Type of receptors | Docking term for ligands | Binding Affinity
(kcal/mol) |
RMSD
/UB |
RMSD /LB | Material name | |
| TRα | 1NAV_5280795_UFF_E= 694.85 | -6.9 | 0.0 | 0.0 | Vitamin D3 | |
| 1NAV_126480146_UFF_E= 3935.18 | -6.8 | 0.0 | 0.0 | Vitamin B12 | ||
| TRβ | 1XZX_5280795_UFF_E= 694.85 | -7.3 | 0.0 | 0.0 | Vitamin D3 | |
| 1XZX_126480146_UFF_E= 4338.56 | -7.1 | 0.0 | 0.0 | Vitamin B12 | ||
Table 2 shows how the ligand interacts with the fourteen-amino-acid chain A of the thyroid hormone receptor alpha (TRα).
| Table 2: Interaction profile of Vitamin D3 with thyroid hormone receptor alpha (TRα): involved amino acids, their locations, number, and types of bonds. | ||
| Amino acids | Location inside the chain | Type of Bonds |
| VAL | 353 | Van der Waals |
| ARG | 152 | |
| GLU | 350 | |
| PRO | 365 | |
| PHE | 363 | |
| ASN | 354 | |
| HIS | 358 | |
| LYS | 357 | |
| HIS | 362 | |
| ILE | 360 | |
| TRP | 364 | Pi-Alkyl |
| ARG | 157 | |
| TRP | 364 | Alkyl |
| ARG | 157 | |
In Table 3 shows how the ligand interacts with the 18-amino-acid chain A of the thyroid hormone receptor alpha (TR α). The results in Table 4 display how the ligand interacts with the eleven amino acid chain X of the thyroid hormone receptor beta (TRβ).
| Table 3: Interaction profile of Vitamin B12 with thyroid hormone receptor alpha (TRα): involved amino acids, their locations, number, and types of bonds | ||
| Amino acids | Location inside the chain | Type of Bonds |
| VAL | 265 | Carbon hydrogen bond |
| SER | 307 | Conventional hydrogen bond |
| ALA | 180 | |
| ARG | 188 | |
| GLN | 187 | |
| LYS | 186 | |
| ASP | 313 | |
| VAL | 316 | Van der Waals |
| HIS | 175 | |
| ASP | 312 | |
| ALA | 308 | |
| GLU | 270 | |
| PRO | 269 | |
| LEU | 306 | |
| LEU | 311 | |
| ASN | 310 | |
| GLY | 278 | |
| ARG | 189 | |
| Table 4: Interaction profile of Vitamin D3 with thyroid hormone receptor beta (TRβ): interacting amino acids, their positions, number of interactions, and types of bonds | ||
| Amino acids | Location inside the chain | Type of Bonds |
| PHE | 459 | Van der Waals |
| VAL | 458 | |
| GLY | 307 | |
| GLU | 299 | |
| MET | 310 | |
| CYS | 309 | |
| ILE | 302 | Alkyl |
| PRO | 384 | |
| ALA | 436 | |
| ILE | 303 | |
| LYS | 306 | Conventional hydrogen bond |
The output in Table 5 indicates the interaction of the ligand with chain X of the thyroid hormone receptor beta (TRβ), which has eighteen twenty amino acids.
| Table 5: Interaction profile of Vitamin B12 with thyroid hormone receptor beta (TRβ): interacting amino acids, their positions, number of interactions, and bond types | ||
| Amino acids | Location inside the chain | Type of Bonds |
| LEU | 360 | Carbon hydrogen bond |
| ASN | 364 | |
| ASP | 367 | Conventional hydrogen bond |
| LEU | 365 | |
| ASN | 364 | |
| ARG | 410 | |
| VAL | 319 | Van der Waals |
| SER | 361 | |
| ASP | 366 | |
| PRO | 323 | |
| HIS | 413 | |
| VAL | 370 | |
| ASN | 233 | |
| TRP | 239 | |
| ARG | 320 | |
| GLY | 236 | |
| SER | 237 | |
| GLN | 241 | |
| ARG | 410 | Pi-Cation |
| HIS | 412 | Unfavourable Donor- Donor |
3.2. Clinical study
The findings in Table 6 shows the clinical characteristics and comparative analysis between hypothyroid patients and healthy controls across a range of biomarkers and physiological parameters, using t-test.
| Table 6: Clinical characteristics and comparative analysis of hypothyroid patients with healthy subjects. | |||||||
| Parameter | Controls | Patients | t-test | df | P value | ||
| SBP | 12.02±0.10 | 13.33±0.86 | -10.1 | 88 | 0.001* | ||
| DBP | 8.04±0.14 | 9.09±0.50 | -13.5 | 88 | 0.001* | ||
| BMI | 25.99±1.89 | 35.28±3.08 | -17.2 | 88 | 0.001* | ||
| Vit D3 (ng/ml) | 42.22±10.0 | 14.27±4.84 | 16.86 | 88 | 0.001* | ||
| Vit B12(pg/ml) | 410.9±83.9 | 145.0±25.0 | 20.35 | 88 | 0.001* | ||
| Ca (mg/dl) | 9.57±0.53 | 6.92±1.11 | 14.45 | 88 | 0.001* | ||
| TG (mg/dl) | 107.5±22.1 | 209.2±49.2 | -12.6 | 88 | 0.001* | ||
| HDL(mg/dl) | 47.27±8.62 | 34.38±3.19 | 9.41 | 88 | 0.001* | ||
| T3 (ng/dl) | 1.01±0.17 | 0.75±0.03 | 11.88 | 88 | 0.001* | ||
| T4 (Ug/dl) | 7.64±7.79 | 4.56±0.32 | 2.65 | 88 | 0.001* | ||
| TSH (uIU/ml) | 1.54±0.65 | 11.65±10.01 | -6.78 | 88 | 0.001* | ||
| * Highly significant (P < 0.01) | |||||||
The outputs of the ROC curve were displayed in Table 7, indicate that vitamin D3, vitamin B12, and calcium have 100% specificity and sensitivity in Figure 3.
| Table 7: ROC curve analysis results for vitamin D3, vitamin B12, and calcium | |||||
| Test Result Variable(s) | AUC | p value | %
Sensitivity |
%
specificity |
Cutoff point |
| Vit D3 | 1.00 | 0.000 | 100 | 100 | 24.99 |
| Vit B12 | 1.00 | 0.000 | 100 | 100 | 214.5 |
| Ca | 1.00 | 0.000 | 100 | 100 | 8.26 |

Figure 3: Receiver operating characteristic curves of measured vitamin D3, B12, and calcium in differentiating between hypothyroid patients
4. DISCUSSION
4.1. Molecular Docking study (Thyroid hormone receptors TRα and TRβ)
Thyroid hormone receptor alpha (TRα) is primarily expressed in the brain, heart, and bone, whereas TRβ1 is found in the liver, kidney, and thyroid, and TRβ2 in the retina, cochlea, and pituitary gland.20 Mutations in these receptors can alter serum thyroid hormone levels. TRα is a key docking target for vitamins D3 and B12, as shown in Table 1, with stronger receptor–ligand interactions indicated by more negative binding affinity values. Molecular docking employs root mean square deviation (RMSD) to assess protein structural changes. The upper bound RMSD compares each atom to itself without considering symmetry, while the lower bound RMSD matches each atom to the nearest of the same element type.21,22
Similarly, TRβ docking studies show stronger ligand-receptor interactions with higher negative affinity values, evaluated using the same RMSD criteria. Docking analysis revealed three main types of interactions between vitamin D3 and thyroid hormone receptor alpha (TRα) chain A: van der Waals, Pi-Alkyl, and Alkyl bonds. The binding affinity was strong, with low binding energy (−6.9 kcal/mol), indicating a stable interaction. The RMSD value between the docked and
experimental ligands was 0.0 Å, confirming the reliability of the docking results. Using PyRx software, virtual screening identified optimal binding of vitamin D3 to active sites on TRα, as detailed in Table 2 and visualized in Figure 1. Docking analysis showed that vitamin B12 interacts with thyroid hormone receptor alpha (TRα) chain A through conventional hydrogen bonds and van der Waals forces. The interaction demonstrated good stability, with a low binding energy of −6.8 kcal/mol. The RMSD value between the docked and experimental ligands was 0.0 Å, confirming docking accuracy. Using PyRx software, vitamin B12 was virtually screened and found to bind effectively to active sites on TRα, as detailed in Table 2 and illustrated in the Figure. 1B.

Figure 1: Thyroid hormone receptor alpha (TRɑ) with A. Vitamin D3 B. Vitamin B12
Vitamin D3 showed strong binding to thyroid hormone receptor beta (TRβ) chain X through three types of hydrogen bond interactions: conventional, alkyl, and van der Waals. The binding affinity was high, with a low energy value of −7.3 kcal/mol. The RMSD between docked and experimental ligands was 0.0 Å, indicating accurate docking. Virtual screening using PyRx confirmed optimal interaction of vitamin D3 within the active sites of TRβ, as shown in Table 4 and visualized in Figure 2A. Vitamin B12 interacted with thyroid hormone receptor beta (TRβ) chain X through five types of bonds: conventional hydrogen bonds, van der Waals, Pi-cation, alkyl, and unfavorable donor–donor interactions. The binding affinity was strong, with a low energy of −7.1 kcal/mol. The RMSD value between docked and experimental ligands was 0.0 Å, confirming the validity of the docking results. PyRx-based virtual screening identified optimal binding of vitamin B12 to active sites on TRβ, as detailed in Table 5 and shown in Figure 2B. The study demonstrated strong binding affinities between vitamin D3 and thyroid hormone receptors TRα and TRβ (−6.9 and −7.3 kcal/mol, respectively), supporting the association between low vitamin D3 levels and hypothyroidism.

Figure 2: Thyroid hormone receptor alpha (TRβ) with A. Vitamin D3 B. Vitamin B12
This deficiency is commonly observed in hypothyroid patients and may result from impaired vitamin D metabolism or shared autoimmune mechanisms.23 Similarly, vitamin B12 exhibited good binding affinity with TRα and TRβ (−6.8 and −7.1 kcal/mol, respectively), consistent with previous reports of reduced serum B12 levels in individuals with hypothyroidism. This reduction is likely attributed to impaired gastrointestinal absorption and digestion associated with hypothyroid states. Furthermore, vitamin B12 deficiency may exacerbate symptoms such as fatigue and neuropathy.24-26 These findings suggest that vitamins D3 and B12 have regulatory roles at the molecular level in thyroid function. However, the precise physiological mechanisms underlying these interactions remain unclear and warrant further investigation.
4.2. Clinical study
This study adds to the growing evidence linking vitamins D3 and B12 to hypothyroidism. A significant increase in systolic and diastolic blood pressure was observed in hypothyroid patients compared to controls (p < 0.01), consistent with the known cardiovascular effects of thyroid hormone deficiency, which include reduced heart rate and increased vascular resistance.27-30 Additionally, BMI was significantly elevated in patients (35.28 ± 3.08), reflecting the well-established association between hypothyroidism and obesity.31,32 Vitamin D3, B12, and calcium levels were significantly reduced in hypothyroid patients. Hypothyroidism impairs metabolism, which may minimize intestinal vitamin D absorption, contributing to a calcium imbalance. These findings align with previous studies highlighting the interconnected role of thyroid function and vitamin D/calcium homeostasis.33,34 Vitamin B12 deficiency is also common in hypothyroid patients, often linked to autoimmune conditions like atrophic gastritis, which impairs absorption. The deficiency tends to worsen with age, as previously reported.35,36 The study also found significantly higher triglyceride levels and reduced HDL (good cholesterol) in hypothyroid patients (P < 0.01), suggesting an increased risk of cardiovascular disease due to impaired lipid metabolism.37 These changes are attributed to decreased thyroid hormone levels, which slow down fat breakdown and liver cholesterol clearance. As expected, serum T3 and T4 levels were markedly lower, while TSH levels were significantly elevated in hypothyroid patients (P < 0.01), confirming primary hypothyroidism and reflecting a disruption in the hypothalamic–pituitary–thyroid axis.38 Finally, ROC curve analysis demonstrated that vitamin D3, vitamin B12, and calcium levels had 100% sensitivity and specificity, suggesting their potential utility as reliable biomarkers for hypothyroidism diagnosis. The respective cut-off values were 24.99 ng/mL, 214.5 pg/mL, and 8.26 mg/dL (Table 7; Figure 3).
These findings reinforce the hypothesis that micronutrients such as vitamin D3, vitamin B12, and calcium play a critical role in the pathogenesis of hypothyroidism. The observed associations align with previous studies suggesting a strong link between these nutrients and thyroid dysfunction.39 Therefore, routine monitoring and appropriate correction of
deficiencies through diet or supplementation under medical supervision may benefit individuals with hypothyroidism.
5. CONCLUSION
This study demonstrated that patients with hypothyroidism had significantly lower levels of vitamin D3 and B12 compared to healthy controls, suggesting a potential link between these deficiencies and thyroid dysfunction. Molecular docking analysis revealed strong interactions between both vitamins and thyroid hormone receptors (TRα and TRβ), implying a possible regulatory role at the molecular level. These findings underscore the importance of evaluating vitamin D3 and B12 levels in hypothyroid patients as part of clinical assessment to support early intervention and prevent disease progression.
6. Data availability
The numerical data generated and analyzed during this study are available from the corresponding author upon reasonable request.
7. Acknowledgement
The authors would like to thank all the staff at the City of Medicine / Educational Laboratories for their valuable support and cooperation during the study.
8. Conflict of interest
The authors declare that they have no competing financial or non-financial interests.
9. Authors’ contribution
All authors contributed equally to the design, execution of the study, and preparation of the manuscript.
10. REFERENCES
- Brtko J. Thyroid hormone and thyroid hormone nuclear receptors: history and present state of art. Endocrine Regulations. 2021;55(2):103-19. DOI: 10.2478/enr-2021-0012
- Rehman G, Kumari N, Bano F, Tyagi RK. Thyroid hormone receptor beta: relevance in human health and diseases. Endocrine and Metabolic Science. 2023;13:100144. DOI: 10.1016/j.endmts.2023.100144
- Bath SC. Thyroid function and iodine intake: global recommendations and relevant dietary trends. Nature Reviews Endocrinology. 2024 Aug;20(8):474-86. DOI: 10.1038/s41574-024-00983-z
- Sultana S, Jahan MA, Ahmed SM, Sultana R, Ara FF. Calcium Imbalance and Hypothyroidism: Exploring the Relationship. KYAMC Journal. 2024 Jun 25;14(04):194-7. DOI: 10.3329/kyamcj.v14i04.70094
- Babić Leko M, Jureško I, Rozić I, Pleić N, Gunjača I, Zemunik T. Vitamin D and the thyroid: a critical review of the current evidence. International journal of molecular sciences. 2023;24(4):3586. DOI: 10.3390/ijms24043586
- Ashok T, Palyam V, Azam AT, Odeyinka O, Alhashimi R, Thoota S, Sange I. Relationship between vitamin D and thyroid: an enigma. Cureus. 2022;14(1). DOI: 7759/cureus.21069
- Eldosouky HF, Saudi WS, Parash MT, Shimmi SC. Hypothyroidism and its effect on serum vitamin D and iron among adult female: A review from Middle East perspective. Journal of Research in Clinical Medicine. 2022;10(1):1. DOI: 34172/jrcm.2022.001
- Pérez-Castrillon JL, Usategui-Martín R, Pludowski P. Treatment of vitamin D deficiency with calcifediol: efficacy and safety profile and predictability of efficacy. Nutrients. 2022 May 5;14(9):1943. DOI: 10.3390/nu14091943
- Appunni S, Rubens M, Ramamoorthy V, Saxena A, Tonse R, Veledar E, McGranaghan P. Association between vitamin D deficiency and hypothyroidism: results from the National Health and Nutrition Examination Survey (NHANES) 2007–2012. BMC endocrine disorders. 2021 Nov;21(1):224. DOI: 10.1186/s12902-021-00897-1
- Sîrbe C, Rednic S, Grama A, Pop TL. An update on the effects of vitamin D on the immune system and autoimmune diseases. International journal of molecular sciences. 2022 Aug 29;23(17):9784. DOI: 10.3390/ijms23179784
- Vernia F, Valvano M, Longo S, Cesaro N, Viscido A, Latella G. Vitamin D in inflammatory bowel diseases. Mechanisms of action and therapeutic implications. Nutrients. 2022 Jan 9;14(2):269. https://doi: 10.3390/nu14020269
- Babić Leko M, Jureško I, Rozić I, Pleić N, Gunjača I, Zemunik T. Vitamin D and the thyroid: a critical review of the current evidence. International journal of molecular sciences. 2023;24(4):3586. DOI: 10.3390/ijms24043586
- Orgler E, Dabsch S, Malfertheiner P, Schulz C. Autoimmune gastritis: update and new perspectives in therapeutic management. Current Treatment Options in Gastroenterology. 2023;21(1):64-77. DOI: 10.3390/ijms18091949
- Halczuk K, Kaźmierczak-Barańska J, Karwowski BT, Karmańska A, Cieślak M. Vitamin B12—Multifaceted in vivo functions and in vitro applications. Nutrients. 2023 Jun 13;15(12):2734. DOI: 10.3390/ nu15122734
- Gupta R, Choudhary S, Chatterjee T. A study on vitamin B12 levels in hypothyroid patients presenting to a tertiary care teaching hospital. Cureus. 2023;15(8). DOI: 10.7759/cureus.44197
- Benites-Zapata VA, Ignacio-Cconchoy FL, Ulloque-Badaracco JR, Hernandez-Bustamante EA, Alarcón-Braga EA, Al-kassab-Córdova A, Herrera-Añazco P. Vitamin B12 levels in thyroid disorders: A systematic review and meta-analysis. Frontiers in Endocrinology.2023;14:1070592. DOI: 10.3389/fendo.2023.1070592
- Trott O, Olson AJ. AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. Journal of Computational Chemistry. 2010;31(2):455-61. DOI: 10.1002/jcc.21334
- O'Boyle NM, Banck M, James CA, Morley C, Vandermeersch T, Hutchison GR. Open Babel: An open chemical toolbox. Journal of cheminformatics. 2011 Oct 7;3(1):33. DOI: 10.1186/1758-2946-3-33
- Gogola-Mruk J, Sirek A, Kumor I, Wojtaszek G, Roszak K, Kulig K, Ptak A. Relationship between thyroid dysfunction and ovarian cancer. Biomolecules. 2025;15(6):870. http ://doi.org/10.3390/biom1506 0870
- Sandler B, Webb P, Apriletti JW, Huber BR, Togashi M, Lima ST, Juric S, Nilsson S, Wagner R, Fletterick RJ, Baxter JD. Thyroxine-thyroid hormone receptor interactions. Journal of Biological Chemistry. 2004;279(53):55801-8.DOI: 1074/jbc .M410124200
- Dahll LK, Westbye AB, Vinorum K, Sejersted Y, Barøy T, Thorsby PM, Hammerstad SS. Clinical and biochemical characteristics of untreated adult patients with resistance to thyroid hormone alpha. Journal of the Endocrine Society. 2023(8):bvad089. DOI: 10.1210/jendso/bvad089
- Biswas P, Toh KC, Ye Y. A distributed SDP approach for large-scale noisy anchor-free graph realization with applications to molecular conformation. SIAM Journal on Scientific Computing. 2008;30(3):1251-77. DOI: 10.1137/060670233.
- Czarnywojtek A, Florek E, Pietrończyk K, Sawicka-Gutaj N, Ruchała M, Ronen O, Nixon IJ, Shaha AR, Rodrigo JP, Tufano RP, Zafereo M. The role of vitamin D in autoimmune thyroid diseases: a narrative review. Journal of Clinical Medicine. 2023 Feb 11;12(4):1452. DOI: 10.3390/jcm12041452
- Aktaş HŞ. Vitamin B12 and vitamin D levels in patients with autoimmune hypothyroidism and their correlation with anti-thyroid peroxidase antibodies. Medical principles and practice. 2020;29(4):364-70.370. DOI: 10.1159/000505094
- Kacharava T, Giorgadze E, Janjgava S, Lomtadze N, Taboridze I. Correlation between vitamin B12 deficiency and autoimmune thyroid diseases. Endocrine, Metabolic & Immune Disorders-Drug Targets (Formerly Current Drug Targets-Immune, Endocrine & Metabolic Disorders). 2023 Jan 1;23(1):86-94. DOI: 10.2174/1871530322666220627145635
- Kacharava T, Giorgadze E, Janjgava S, Lomtadze N, Taboridze I. Correlation between vitamin B12 deficiency and autoimmune thyroid diseases. Endocrine, Metabolic & Immune Disorders-Drug Targets (Formerly Current Drug Targets-Immune, Endocrine & Metabolic Disorders). 2023;23(1):86-94. http://doi.org/10.2174/1871530322666220826160757
- Olanrewaju OA, Asghar R, Makwana S, Yahya M, Kumar N, Khawar MH, Ahmed A, Islam T, Kumari K, Shadmani S, Ali M. Thyroid and its ripple effect: impact on cardiac structure, function, and outcomes. Cureus. 2024;16(1).http://doi.org/10.7759/cureus.51574
- Rabadia JP, Thite VS, Desai BK, Bera RG, Patel S. Cardiovascular System, Its Functions and Disorders. InCardioprotective Plants 2024 Aug 21 (pp. 1-34). http://doi.org/10.1007/978-981-97-4627-9_1
- Suonsyrjä N, Metso S, Moilanen E, Mustonen J, Jaatinen P, Pörsti I. Haemodynamics of hyperthyroidism: increased cardiac work and findings related to vasodilatation. European thyroid journal. 2024;13(5). http://doi.org/10.1530/ETJ-24-0090
- Schettini IV, Rios DR, Figueiredo RC. Effect of different classes of antihypertensive drugs on arterial stiffness. Current hypertension reports. 2023 ;25(5):61-70.
- Walczak K, Sieminska L. Obesity and thyroid axis. International journal of environmental research and public health. 2021;18(18):9434. http://doi.org/10.3390/ijerph18189434
- Ekinci I. Metabolic Syndrome In Thyroid Disease. InMetabolic Syndrome: A Comprehensive Update with New Insights 2025 (pp. 402-420). Bentham Science Publishers. https://doi10.2174/9789815322 1321250101
- Sittitrai P, Ruenmarkkaew D, Klibngern H, Ariyanon T, Hanprasertpong N, Boonyaprapa S, Sreesawat M. Perioperative versus postoperative calcium and vitamin D supplementation to prevent symptomatic hypocalcemia after total thyroidectomy: a randomized placebo controlled trial. International Journal of Surgery. 2023;109(1):13-20. http://doi.org/10.1097/JS9.0000000000000192
- Safari S, Rafraf M, Malekian M, Molani-Gol R, Asghari-Jafarabadi M, Mobasseri M. Effects of vitamin D supplementation on metabolic parameters, serum irisin and obesity values in women with subclinical hypothyroidism: A double-blind randomized controlled trial. Frontiers in Endocrinology.2023;14:1306470.4-170 . https://doi. org /10.3389/fendo.2023.1306470
- Jouda J, Ibrahim RK, Herez MS. Vitamin D 3, parathyroid hormones, and calcium levels in patients with hypothyroidism and chronic kidney disease and the relationship between them. EurAsian Journal of Biosciences. 2020;14(1).
- Ruscio M, Guard G, Piedrahita G, D’Adamo CR. The relationship between gastrointestinal health, micronutrient concentrations, and autoimmunity: a focus on the thyroid. Nutrients. 2022;14(17):3572. DOI: 10.3390 /nu141735 72
- Makki NF. Effect of Warfarin on cholesterol, LDL, HDL and TG levels in coronary artery patients. Journal of Pharmaceutical Sciences and Research. 2018 ;10(2):257-9. DOI: 10.13140 /RG.2.2 .197 47.71208
- Brown ED, Obeng-Gyasi B, Hall JE, Shekhar S. The thyroid hormone axis and female reproduction. International Journal of Molecular Sciences. 2023 ;24(12):9815. DOI: 10.3390/ijms24129815
- Shulhai AM, Rotondo R, Petraroli M, Patianna V, Predieri B, Iughetti L, Esposito S, Street ME. The role of nutrition on thyroid function. Nutrients. 2024;16(15):2496. https:// doi.org/ 10.3390/ nu16152496.