Albinus Yunus Cobis 1 , Dewi Yulianti Bisri 2 , Iwan Abdul Rachman 3
Authors affiliations:
Cerebral abscess is a rare but serious intracranial infection, particularly in paediatric patients with cyanotic congenital heart disease (CCHD) such as Tetralogy of Fallot (TOF). In TOF, the presence of a right-to-left shunt facilitates haematogenous spread of bacteria to the brain. Chronic hypoxaemia leads to polycythaemia and increased blood viscosity, contributing to cerebral abscess formation.
A four-year-old boy presented with a right temporoparietal cerebral abscess in the context of uncorrected TOF. He complained of recurrent vomiting, fever, headache, and cyanosis, with oxygen saturation as low as 86%. CT scan revealed a 4.4×3.6×4.0 cm abscess with significant perilesional oedema. The patient underwent craniotomy and abscess drainage under general anesthesia. Induction was performed using a ketamine-propofol combination, while maintenance was achieved with 1% sevoflurane. The surgery proceeded uneventfully with successful drainage of 30 mL of purulent material.
The anesthetic strategy prioritised hemodynamic stability and adequate cerebral perfusion. The ketamine-propofol combination was selected for its ability to preserve systemic vascular resistance while avoiding a rise in ICP. Steroids were deliberately omitted in line with recent evidence suggesting increased risk of infectious complications. Postoperative analgesia was managed with a multimodal opioid-sparing regimen to allow prompt and reliable neurological assessment, which proved effective.
Conclusion: Careful anesthetic management using ketamine-propofol induction, controlled ventilation, and appropriate hydration proved successful in managing this complex case of cerebral abscess in TOF. Multidisciplinary coordination was pivotal, particularly in a resource-limited healthcare setting.
Keywords: Cerebral abscess; Neuroanesthesia; Tetralogy of Fallot.
Citation: Cobis AY, Bisri DY, Rachman IA.Neuroanesthetic management of cerebral abscess in Tetralogy of Fallot: balancing intracranial and cardiac risks. Anaesth. pain intensive care 2025;29(9):1321-26. DOI: 10.35975/apic.v29i9.3074
Received: August 23, 2025; Revised: October 10, 2025; Accepted: October 27, 2025
Cerebral abscess is an intracranial infection characterised by the accumulation of pus within the brain parenchyma and may result in severe neurological complications, particularly in paediatric patients. One of the main predisposing factors for this condition is cyanotic congenital heart disease (CCHD), notably Tetralogy of Fallot (TOF), which is the most common form of cyanotic heart defect. TOF induces a right-to-left shunt, bypassing pulmonary circulation and facilitating the haematogenous spread of microorganisms to the central nervous system. Chronic hypoxaemia also leads to polycythaemia and increased blood viscosity, which may trigger microthrombi formation and precipitate cerebral abscesses through septic infarction of the cerebral microparenchyma.1,2
Clinically, cerebral abscess in children often presents with headache, vomiting, and seizures, accompanied by signs of raised intracranial pressure. In patients with uncorrected TOF, additional findings such as cyanosis and digital clubbing due to chronic hypoxaemia are frequently observed.1
Globally, the prevalence of cerebral abscess in children is relatively low but remains higher in developing countries, accounting for approximately 8% of all intracranial lesions.3 In developed countries, the incidence is lower (1–2%) due to early detection and correction of congenital heart disease. In regions such as Asia and Africa, limited access to healthcare and delays in corrective interventions contribute to the continued incidence of cerebral abscess in TOF patients. Retrospective reports have indicated that 5–18.7% of patients with uncorrected CCHD are at risk of developing cerebral abscess during their lifetime, with TOF being the predominant aetiology.2,4
Neuroanesthetic management of paediatric patients with cerebral abscess and uncorrected TOF poses significant challenges to the anesthetist. The coexistence of raised intracranial pressure and haemodynamic instability due to the right-to-left shunt necessitates a meticulous and integrated anesthetic strategy. Risks such as oxygen desaturation, sudden cyanotic spells, and coagulopathy from polycythaemia must be actively addressed throughout the perioperative period. Ventilation strategies targeting appropriate PaCO₂ control, invasive haemodynamic monitoring, and safe induction protocols are critical to ensuring adequate cerebral perfusion without provoking cardiovascular instability.1,3
This case report aims to present a comprehensive neuroanesthetic approach in a four-year-old child with a cerebral abscess and uncorrected TOF. The anesthetic strategy focused on achieving a delicate balance between intracranial pressure control and cardiopulmonary stability during abscess drainage via craniotomy. The uniqueness of this case lies in the complex pathophysiological interplay and the pivotal role of multidisciplinary collaboration in achieving an optimal outcome. This report is intended to serve as a practical reference for clinicians, particularly in resource-limited settings, in managing similar cases safely and effectively.2,4
A four-year-old boy presented with complaints of persistent vomiting, fever, and severe headache that disrupted his sleep. The symptoms had been present for two weeks prior to hospital admission. The patient had a known history of congenital heart disease, specifically Tetralogy of Fallot (TOF), which had been diagnosed at the age of one but had remained uncorrected due to financial constraints. He was born at term via spontaneous vaginal delivery with a birth weight within normal limits. On presentation to the emergency department, he appeared moderately ill, with pallor and cyanosis noted. His Glasgow Coma Scale (GCS) was E4V5M6. Vital signs were: heart rate 110 bpm, respiratory rate 24 breaths per minute, temperature 38.9 °C, and oxygen saturation 86% on room air. Cardiac auscultation revealed a positive murmur, while physical examination of the extremities showed digital clubbing and acral cyanosis.
Initial laboratory investigations revealed: haemoglobin 19.9 g/dL, haematocrit 58.9%, erythrocyte count 7.49 ×10⁶/µL, leukocyte count 13.8 ×10³/µL, activated partial thromboplastin time (APTT) 13.4 seconds, prothrombin time (PT) 29.3 seconds, sodium 132.7 mEq/L, potassium 3.8 mEq/L, and chloride 99.5 mEq/L. Non-contrast cranial CT scan demonstrated a cerebral abscess measuring approximately 4.4 × 3.6 × 4.0 cm in the right temporoparietal subcortical region, accompanied by diffuse vasogenic oedema extending to the right temporo-occipital-parietal lobes.
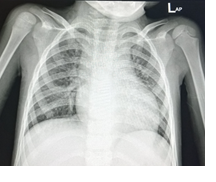
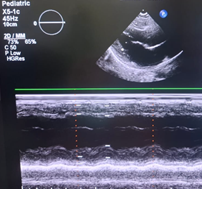
Figure 1: Chest X-ray and echocardiogram findings
Echocardiography findings indicated preserved left ventricular systolic function (ejection fraction 58%, fractional shortening 30.2%), with end-diastolic volume (EDV) 77.3 mL, end-systolic volume (ESV) 32.5 mL, and left ventricular mass 96.8 g. Chest radiography revealed cardiomegaly with a cardiothoracic ratio (CTR) > 55%, along with bilateral paracardiac and perihilar infiltrates. Impression: suspected bilateral bronchopneumonia and cardiomegaly. The final diagnosis was cerebral abscess with uncorrected Tetralogy of Fallot.
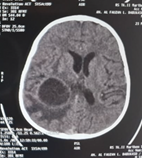
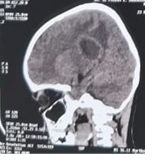
Figure 2: CT scan images
Surgical intervention was planned under general anesthesia, and the patient was categorised as ASA Physical Status Class III due to significant systemic disease (uncorrected TOF). A functioning 20-gauge intravenous line had been secured prior to transfer from the ward. Preoperative fasting protocols were followed, and maintenance fluid requirements were appropriately met with intravenous crystalloids.
In the preoperative holding area, the patient received intravenous premedication with midazolam 1 mg and low-dose ketamine 5 mg to achieve anxiolysis and preserve haemodynamic stability, alongside supplemental oxygen. Upon entering the operating room, standard monitoring was established, including non-invasive blood pressure, oxygen saturation, and electrocardiography (ECG). Anesthesia was carefully induced via titrated administration of ketamine 15 mg (1 mg/kg) and propofol 15 mg (1 mg/kg), with the addition of fentanyl 25 mcg and atracurium 8 mg to achieve adequate depth of anesthesia, considering both cardiovascular and neurological factors. The airway was managed with adequate preoxygenation followed by endotracheal intubation using a 5.5-mm cuffed tube.
Anesthesia was maintained using oxygen and 1 vol% sevoflurane, with intermittent administration of analgesics and neuromuscular relaxants. Ventilation was carefully controlled to maintain a balance between cerebral and pulmonary physiology. The neurosurgical procedure lasted two hours, during which haemodynamics and oxygenation remained stable. Approximately 30 mL of purulent fluid was evacuated from the abscess. Intraoperative fluid management was conducted cautiously and precisely.
At the end of surgery, the patient resumed spontaneous respiration and was extubated uneventfully in the operating room. Postoperatively, he was monitored in the Intensive Care Unit (ICU) and received oxygen therapy via nasal cannula at 2–3 L/min. His postoperative parameters were: blood pressure 90/65 mmHg, heart rate 90 bpm, and SpO₂ 87%. Multimodal analgesia was initiated, consisting of paracetamol 200 mg four times daily and metamizole 200 mg three times daily. Cardiac therapy was continued with propranolol 3 mg three times daily, alongside intravenous ceftriaxone 700 mg twice daily, which had been started in the preoperative period.
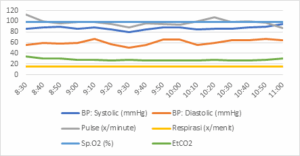
Figure 3: Haemodynamics during surgery
Cerebral abscess in a paediatric patient with uncorrected Tetralogy of Fallot (TOF) represents a high-risk dual pathology necessitating meticulous anesthetic planning. Children with cyanotic congenital heart disease (CCHD), such as TOF, are particularly prone to cerebral abscess formation. Studies report that 5–20% of patients with uncorrected TOF will develop cerebral abscess during their lifetime, with TOF accounting for the majority of these cases.5,6 Chronic hypoxaemia and right-to-left shunting enable unfiltered, unoxygenated blood to bypass the pulmonary circulation, allowing pathogens direct access to the cerebral vasculature.7,8 Sustained hypoxaemia contributes to polycythaemia and hyperviscosity, predisposing to microthrombus formation and focal cerebral ischaemia, key precursors to infection.8,9 This combination leads to severe neurological infection with reported mortality rates of up to 10% in recent literature.10
In this case, a 4-year-old presented with vomiting, headache, and fever lasting two weeks. CT imaging revealed a large abscess in the right temporoparietal region with surrounding oedema, indicative of raised intracranial pressure (ICP). Clinical features of TOF, including cyanosis, digital clubbing, systolic murmur, and oxygen saturation of 86% on room air, were evident. Echocardiography confirmed preserved left ventricular function (EF 58%) but a high risk for acute hypoxic episodes ("tet spells"). Laboratory findings showed haemoglobin of 19.9 g/dL and haematocrit of 58.9%, consistent with polycythaemia secondary to chronic hypoxaemia. This increased the risk of thrombosis; thus, preoperative hydration with crystalloids was optimised to prevent haemoconcentration.9 Correction of electrolyte imbalances and adequate hydration during fasting were also crucial.8
Ceftriaxone therapy was initiated for both treatment and endocarditis prophylaxis. Steroids were withheld in this patient, in line with recent evidence questioning their benefit in paediatric cerebral abscess. Several paediatric reviews and clinical audits highlight potential drawbacks, including reduced antibiotic penetration into the abscess capsule, impaired encapsulation, and suppression of local immune response, all of which may increase the risk of abscess rupture or secondary infection.11 Current guidelines from the European Society of Clinical Microbiology and Infectious Diseases recommend steroids only in cases of impending cerebral herniation or significant mass effect.12
To avoid ICP elevation and exaggerated sympathetic response, a combination of midazolam and low-dose ketamine was selected for premedication. Midazolam provided anxiolysis with minimal cardiovascular depression, while ketamine preserved systemic vascular resistance (SVR) and heart rate. Ketamine also prevented SVR drops that could exacerbate the right-to-left shunt.8,9 Supplemental oxygen via nasal cannula improved pre-induction saturation to 93–95%, consistent with earlier reports.
Intraoperatively, standard monitoring (ECG, NIBP, SpO₂, EtCO₂) was established. Due to known inaccuracy of pulse oximetry in TOF, arterial blood gas analysis was employed for accurate assessment.8 A 20G intravenous line was verified patent and free of air to avoid paradoxical embolism.9 The head was elevated at 15° to facilitate cerebral venous drainage. A urinary catheter was inserted to assess volume status and cardiac output.
Anesthetic induction was titrated using ketamine 1 mg/kg and propofol 1 mg/kg, balancing haemodynamic stability and intracranial dynamics. Ketamine, an NMDA receptor antagonist, has a rapid onset (30–60 seconds) and short duration (10–15 minutes), attributed to redistribution.13,14 It maintains SVR by stimulating the sympathetic nervous system, resulting in modest increases in heart rate, blood pressure, and cardiac output, beneficial in hypotension, prone patients including those with intracranial pathology.13,14
Propofol, via GABA-A modulation, induces neuronal hyperpolarisation, leading to hypnosis, reduced cerebral metabolism, and decreased cerebral blood flow. It has a rapid onset (30 seconds) and short duration (5–10 minutes), primarily due to redistribution. Its ICP-lowering effect stems from reduced cerebral metabolic rate, though caution is warranted due to its vasodilatory hypotensive potential.15
The ketamine-propofol combination synergises both agents pharmacological profiles, mitigating individual side effects. Clinically, it reduces the risk of hypotension compared to propofol alone. A recent systematic review also demonstrated that ketamine does not raise ICP in intubated, hyperventilated children, and may even significantly lower ICP while maintaining adequate cerebral perfusion pressure (CPP).14 Prospective studies confirm ketamine’s safety in paediatric neuroanesthesia, particularly in traumatic brain injury with invasive ICP monitoring.13,14
Fentanyl 1 µg/kg was administered to blunt the sympathetic response to laryngoscopy. Atracurium was used as the neuromuscular blocker due to its minimal effect on SVR and organ-independent metabolism.9
Anesthesia was maintained with 1 vol% sevoflurane in 100% oxygen, supplemented by intermittent fentanyl. Low-dose sevoflurane has minimal haemodynamic effects and may improve pulmonary blood flow when used judiciously.8,9 Mild hyperventilation (EtCO₂ 30–32 mmHg) was targeted to reduce ICP through cerebral vasoconstriction, without significantly compromising cerebral perfusion.8 Maximum oxygenation was ensured, and factors increasing pulmonary vascular resistance (PVR) hypothermia, acidosis, pain were avoided.
Fluid therapy was conservative with crystalloids, aiming for normovolaemia. Dehydration, which could worsen blood viscosity, was strictly avoided. No significant blood loss occurred and transfusion was not required.
Transient hypotension during abscess manipulation was managed with ketamine boluses and phenylephrine 5 µg/kg, which increased SVR and improved oxygenation.8,9 Ephedrine was also administered as needed.
Thirty millilitres of purulent material were successfully drained. Neither mannitol nor hypertonic saline was required, as ICP was adequately controlled. Extubation was performed while the patient was fully awake, with precautions to avoid coughing or laryngospasm. Post-extubation oxygen saturation remained stable at 94% with nasal oxygen.
The patient was transferred to the ICU for intensive monitoring. Oxygen was continued via nasal cannula. Multimodal analgesia (paracetamol and metamizole) was maintained, and propranolol was continued to prevent “tet spells”. Opioids were avoided to allow accurate neurological assessment. There were no signs of progressive cerebral oedema, and corticosteroids remained unnecessary. Head elevation and volume status were closely monitored.
Uncorrected TOF patients are prone to haemodynamic instability, reduced SVR, and increased PVR, which may exacerbate right-to-left shunting. Additional challenges include managing ICP without disrupting systemic circulation, and preventing cyanotic crises triggered by pain, hypovolaemia, or stress. Agents such as ketamine and ephedrine are particularly valuable for their haemodynamic profiles.
Neuroanesthetic management in children with cerebral abscess and uncorrected Tetralogy of Fallot (TOF) requires an individualised and comprehensive approach, balancing cardiovascular stability with intracranial pressure control. The ketamine-propofol combination proved effective in maintaining haemodynamic equilibrium while suppressing sympathetic responses that could exacerbate right-to-left shunting. The decision to avoid corticosteroids was based on current evidence suggesting a potential for adverse infectious outcomes without proven benefit in this context.
An integrated anesthetic strategy comprising preoperative hydration, invasive and non-invasive haemodynamic monitoring, and controlled ventilation targeting precise PaCO₂ levels was pivotal in achieving perioperative success. This allowed effective ICP control without inducing systemic instability or hypoxaemia. Postoperative multimodal analgesia further facilitated optimal recovery, enabling accurate neurological assessment and preventing cyanotic episodes.
This case underscores the vital importance of multidisciplinary collaboration among anesthesia, neurosurgery, cardiology, and intensive care teams in managing paediatric patients with the complex combination of cerebral abscess and TOF. It also provides practical guidance for anesthetists working in resource-limited settings, enabling safe and effective care for similarly challenging cases.
DYB: Supervision, manuscript review, guidance in study design.
IAR: Supervision, critical revision of manuscript, guidance in study design.
Authors affiliations:
- Albinus Yunus Cobis, MD (Anest), MPH, Department of Anesthesiology & Intensive Care, Jayapura Regional General Hospital, Papua, Indonesia; Email: albinuscobis@gmail.com; {ORCID:0009-0005-5261-6565}
- Dewi Yulianti Bisri, MD (Anest), MPH, PhD, Department of Anesthesiology & Intensive Care, Faculty of Medicine, Padjadjaran University / Dr. Hasan Sadikin Central General Hospital, Bandung, Indonesia; Email: dewi.yulianti@unpad.ac.id; {ORCID:0000-0002-7637-9710}
- Iwan Abdul Rachman, MD (Anest), M.PH, Department of Anesthesiology & Intensive Care, Faculty of Medicine, Padjadjaran University / Dr. Hasan Sadikin Central General Hospital, Bandung, Indonesia; Email: i.abdulrachman@yahoo.com;{ORCID:0000-0002-7584-2371}
ABSTRACT
Cerebral abscess is a rare but serious intracranial infection, particularly in paediatric patients with cyanotic congenital heart disease (CCHD) such as Tetralogy of Fallot (TOF). In TOF, the presence of a right-to-left shunt facilitates haematogenous spread of bacteria to the brain. Chronic hypoxaemia leads to polycythaemia and increased blood viscosity, contributing to cerebral abscess formation.
A four-year-old boy presented with a right temporoparietal cerebral abscess in the context of uncorrected TOF. He complained of recurrent vomiting, fever, headache, and cyanosis, with oxygen saturation as low as 86%. CT scan revealed a 4.4×3.6×4.0 cm abscess with significant perilesional oedema. The patient underwent craniotomy and abscess drainage under general anesthesia. Induction was performed using a ketamine-propofol combination, while maintenance was achieved with 1% sevoflurane. The surgery proceeded uneventfully with successful drainage of 30 mL of purulent material.
The anesthetic strategy prioritised hemodynamic stability and adequate cerebral perfusion. The ketamine-propofol combination was selected for its ability to preserve systemic vascular resistance while avoiding a rise in ICP. Steroids were deliberately omitted in line with recent evidence suggesting increased risk of infectious complications. Postoperative analgesia was managed with a multimodal opioid-sparing regimen to allow prompt and reliable neurological assessment, which proved effective.
Conclusion: Careful anesthetic management using ketamine-propofol induction, controlled ventilation, and appropriate hydration proved successful in managing this complex case of cerebral abscess in TOF. Multidisciplinary coordination was pivotal, particularly in a resource-limited healthcare setting.
Keywords: Cerebral abscess; Neuroanesthesia; Tetralogy of Fallot.
Citation: Cobis AY, Bisri DY, Rachman IA.Neuroanesthetic management of cerebral abscess in Tetralogy of Fallot: balancing intracranial and cardiac risks. Anaesth. pain intensive care 2025;29(9):1321-26. DOI: 10.35975/apic.v29i9.3074
Received: August 23, 2025; Revised: October 10, 2025; Accepted: October 27, 2025
1. INTRODUCTION
Cerebral abscess is an intracranial infection characterised by the accumulation of pus within the brain parenchyma and may result in severe neurological complications, particularly in paediatric patients. One of the main predisposing factors for this condition is cyanotic congenital heart disease (CCHD), notably Tetralogy of Fallot (TOF), which is the most common form of cyanotic heart defect. TOF induces a right-to-left shunt, bypassing pulmonary circulation and facilitating the haematogenous spread of microorganisms to the central nervous system. Chronic hypoxaemia also leads to polycythaemia and increased blood viscosity, which may trigger microthrombi formation and precipitate cerebral abscesses through septic infarction of the cerebral microparenchyma.1,2
Clinically, cerebral abscess in children often presents with headache, vomiting, and seizures, accompanied by signs of raised intracranial pressure. In patients with uncorrected TOF, additional findings such as cyanosis and digital clubbing due to chronic hypoxaemia are frequently observed.1
Globally, the prevalence of cerebral abscess in children is relatively low but remains higher in developing countries, accounting for approximately 8% of all intracranial lesions.3 In developed countries, the incidence is lower (1–2%) due to early detection and correction of congenital heart disease. In regions such as Asia and Africa, limited access to healthcare and delays in corrective interventions contribute to the continued incidence of cerebral abscess in TOF patients. Retrospective reports have indicated that 5–18.7% of patients with uncorrected CCHD are at risk of developing cerebral abscess during their lifetime, with TOF being the predominant aetiology.2,4
Neuroanesthetic management of paediatric patients with cerebral abscess and uncorrected TOF poses significant challenges to the anesthetist. The coexistence of raised intracranial pressure and haemodynamic instability due to the right-to-left shunt necessitates a meticulous and integrated anesthetic strategy. Risks such as oxygen desaturation, sudden cyanotic spells, and coagulopathy from polycythaemia must be actively addressed throughout the perioperative period. Ventilation strategies targeting appropriate PaCO₂ control, invasive haemodynamic monitoring, and safe induction protocols are critical to ensuring adequate cerebral perfusion without provoking cardiovascular instability.1,3
This case report aims to present a comprehensive neuroanesthetic approach in a four-year-old child with a cerebral abscess and uncorrected TOF. The anesthetic strategy focused on achieving a delicate balance between intracranial pressure control and cardiopulmonary stability during abscess drainage via craniotomy. The uniqueness of this case lies in the complex pathophysiological interplay and the pivotal role of multidisciplinary collaboration in achieving an optimal outcome. This report is intended to serve as a practical reference for clinicians, particularly in resource-limited settings, in managing similar cases safely and effectively.2,4
2. CASE REPORT
A four-year-old boy presented with complaints of persistent vomiting, fever, and severe headache that disrupted his sleep. The symptoms had been present for two weeks prior to hospital admission. The patient had a known history of congenital heart disease, specifically Tetralogy of Fallot (TOF), which had been diagnosed at the age of one but had remained uncorrected due to financial constraints. He was born at term via spontaneous vaginal delivery with a birth weight within normal limits. On presentation to the emergency department, he appeared moderately ill, with pallor and cyanosis noted. His Glasgow Coma Scale (GCS) was E4V5M6. Vital signs were: heart rate 110 bpm, respiratory rate 24 breaths per minute, temperature 38.9 °C, and oxygen saturation 86% on room air. Cardiac auscultation revealed a positive murmur, while physical examination of the extremities showed digital clubbing and acral cyanosis.
Initial laboratory investigations revealed: haemoglobin 19.9 g/dL, haematocrit 58.9%, erythrocyte count 7.49 ×10⁶/µL, leukocyte count 13.8 ×10³/µL, activated partial thromboplastin time (APTT) 13.4 seconds, prothrombin time (PT) 29.3 seconds, sodium 132.7 mEq/L, potassium 3.8 mEq/L, and chloride 99.5 mEq/L. Non-contrast cranial CT scan demonstrated a cerebral abscess measuring approximately 4.4 × 3.6 × 4.0 cm in the right temporoparietal subcortical region, accompanied by diffuse vasogenic oedema extending to the right temporo-occipital-parietal lobes.


Figure 1: Chest X-ray and echocardiogram findings
Echocardiography findings indicated preserved left ventricular systolic function (ejection fraction 58%, fractional shortening 30.2%), with end-diastolic volume (EDV) 77.3 mL, end-systolic volume (ESV) 32.5 mL, and left ventricular mass 96.8 g. Chest radiography revealed cardiomegaly with a cardiothoracic ratio (CTR) > 55%, along with bilateral paracardiac and perihilar infiltrates. Impression: suspected bilateral bronchopneumonia and cardiomegaly. The final diagnosis was cerebral abscess with uncorrected Tetralogy of Fallot.


Figure 2: CT scan images
Surgical intervention was planned under general anesthesia, and the patient was categorised as ASA Physical Status Class III due to significant systemic disease (uncorrected TOF). A functioning 20-gauge intravenous line had been secured prior to transfer from the ward. Preoperative fasting protocols were followed, and maintenance fluid requirements were appropriately met with intravenous crystalloids.
In the preoperative holding area, the patient received intravenous premedication with midazolam 1 mg and low-dose ketamine 5 mg to achieve anxiolysis and preserve haemodynamic stability, alongside supplemental oxygen. Upon entering the operating room, standard monitoring was established, including non-invasive blood pressure, oxygen saturation, and electrocardiography (ECG). Anesthesia was carefully induced via titrated administration of ketamine 15 mg (1 mg/kg) and propofol 15 mg (1 mg/kg), with the addition of fentanyl 25 mcg and atracurium 8 mg to achieve adequate depth of anesthesia, considering both cardiovascular and neurological factors. The airway was managed with adequate preoxygenation followed by endotracheal intubation using a 5.5-mm cuffed tube.
Anesthesia was maintained using oxygen and 1 vol% sevoflurane, with intermittent administration of analgesics and neuromuscular relaxants. Ventilation was carefully controlled to maintain a balance between cerebral and pulmonary physiology. The neurosurgical procedure lasted two hours, during which haemodynamics and oxygenation remained stable. Approximately 30 mL of purulent fluid was evacuated from the abscess. Intraoperative fluid management was conducted cautiously and precisely.
At the end of surgery, the patient resumed spontaneous respiration and was extubated uneventfully in the operating room. Postoperatively, he was monitored in the Intensive Care Unit (ICU) and received oxygen therapy via nasal cannula at 2–3 L/min. His postoperative parameters were: blood pressure 90/65 mmHg, heart rate 90 bpm, and SpO₂ 87%. Multimodal analgesia was initiated, consisting of paracetamol 200 mg four times daily and metamizole 200 mg three times daily. Cardiac therapy was continued with propranolol 3 mg three times daily, alongside intravenous ceftriaxone 700 mg twice daily, which had been started in the preoperative period.

Figure 3: Haemodynamics during surgery
3. DISCUSSION
Cerebral abscess in a paediatric patient with uncorrected Tetralogy of Fallot (TOF) represents a high-risk dual pathology necessitating meticulous anesthetic planning. Children with cyanotic congenital heart disease (CCHD), such as TOF, are particularly prone to cerebral abscess formation. Studies report that 5–20% of patients with uncorrected TOF will develop cerebral abscess during their lifetime, with TOF accounting for the majority of these cases.5,6 Chronic hypoxaemia and right-to-left shunting enable unfiltered, unoxygenated blood to bypass the pulmonary circulation, allowing pathogens direct access to the cerebral vasculature.7,8 Sustained hypoxaemia contributes to polycythaemia and hyperviscosity, predisposing to microthrombus formation and focal cerebral ischaemia, key precursors to infection.8,9 This combination leads to severe neurological infection with reported mortality rates of up to 10% in recent literature.10
In this case, a 4-year-old presented with vomiting, headache, and fever lasting two weeks. CT imaging revealed a large abscess in the right temporoparietal region with surrounding oedema, indicative of raised intracranial pressure (ICP). Clinical features of TOF, including cyanosis, digital clubbing, systolic murmur, and oxygen saturation of 86% on room air, were evident. Echocardiography confirmed preserved left ventricular function (EF 58%) but a high risk for acute hypoxic episodes ("tet spells"). Laboratory findings showed haemoglobin of 19.9 g/dL and haematocrit of 58.9%, consistent with polycythaemia secondary to chronic hypoxaemia. This increased the risk of thrombosis; thus, preoperative hydration with crystalloids was optimised to prevent haemoconcentration.9 Correction of electrolyte imbalances and adequate hydration during fasting were also crucial.8
Ceftriaxone therapy was initiated for both treatment and endocarditis prophylaxis. Steroids were withheld in this patient, in line with recent evidence questioning their benefit in paediatric cerebral abscess. Several paediatric reviews and clinical audits highlight potential drawbacks, including reduced antibiotic penetration into the abscess capsule, impaired encapsulation, and suppression of local immune response, all of which may increase the risk of abscess rupture or secondary infection.11 Current guidelines from the European Society of Clinical Microbiology and Infectious Diseases recommend steroids only in cases of impending cerebral herniation or significant mass effect.12
To avoid ICP elevation and exaggerated sympathetic response, a combination of midazolam and low-dose ketamine was selected for premedication. Midazolam provided anxiolysis with minimal cardiovascular depression, while ketamine preserved systemic vascular resistance (SVR) and heart rate. Ketamine also prevented SVR drops that could exacerbate the right-to-left shunt.8,9 Supplemental oxygen via nasal cannula improved pre-induction saturation to 93–95%, consistent with earlier reports.
Intraoperatively, standard monitoring (ECG, NIBP, SpO₂, EtCO₂) was established. Due to known inaccuracy of pulse oximetry in TOF, arterial blood gas analysis was employed for accurate assessment.8 A 20G intravenous line was verified patent and free of air to avoid paradoxical embolism.9 The head was elevated at 15° to facilitate cerebral venous drainage. A urinary catheter was inserted to assess volume status and cardiac output.
Anesthetic induction was titrated using ketamine 1 mg/kg and propofol 1 mg/kg, balancing haemodynamic stability and intracranial dynamics. Ketamine, an NMDA receptor antagonist, has a rapid onset (30–60 seconds) and short duration (10–15 minutes), attributed to redistribution.13,14 It maintains SVR by stimulating the sympathetic nervous system, resulting in modest increases in heart rate, blood pressure, and cardiac output, beneficial in hypotension, prone patients including those with intracranial pathology.13,14
Propofol, via GABA-A modulation, induces neuronal hyperpolarisation, leading to hypnosis, reduced cerebral metabolism, and decreased cerebral blood flow. It has a rapid onset (30 seconds) and short duration (5–10 minutes), primarily due to redistribution. Its ICP-lowering effect stems from reduced cerebral metabolic rate, though caution is warranted due to its vasodilatory hypotensive potential.15
The ketamine-propofol combination synergises both agents pharmacological profiles, mitigating individual side effects. Clinically, it reduces the risk of hypotension compared to propofol alone. A recent systematic review also demonstrated that ketamine does not raise ICP in intubated, hyperventilated children, and may even significantly lower ICP while maintaining adequate cerebral perfusion pressure (CPP).14 Prospective studies confirm ketamine’s safety in paediatric neuroanesthesia, particularly in traumatic brain injury with invasive ICP monitoring.13,14
Fentanyl 1 µg/kg was administered to blunt the sympathetic response to laryngoscopy. Atracurium was used as the neuromuscular blocker due to its minimal effect on SVR and organ-independent metabolism.9
Anesthesia was maintained with 1 vol% sevoflurane in 100% oxygen, supplemented by intermittent fentanyl. Low-dose sevoflurane has minimal haemodynamic effects and may improve pulmonary blood flow when used judiciously.8,9 Mild hyperventilation (EtCO₂ 30–32 mmHg) was targeted to reduce ICP through cerebral vasoconstriction, without significantly compromising cerebral perfusion.8 Maximum oxygenation was ensured, and factors increasing pulmonary vascular resistance (PVR) hypothermia, acidosis, pain were avoided.
Fluid therapy was conservative with crystalloids, aiming for normovolaemia. Dehydration, which could worsen blood viscosity, was strictly avoided. No significant blood loss occurred and transfusion was not required.
Transient hypotension during abscess manipulation was managed with ketamine boluses and phenylephrine 5 µg/kg, which increased SVR and improved oxygenation.8,9 Ephedrine was also administered as needed.
Thirty millilitres of purulent material were successfully drained. Neither mannitol nor hypertonic saline was required, as ICP was adequately controlled. Extubation was performed while the patient was fully awake, with precautions to avoid coughing or laryngospasm. Post-extubation oxygen saturation remained stable at 94% with nasal oxygen.
The patient was transferred to the ICU for intensive monitoring. Oxygen was continued via nasal cannula. Multimodal analgesia (paracetamol and metamizole) was maintained, and propranolol was continued to prevent “tet spells”. Opioids were avoided to allow accurate neurological assessment. There were no signs of progressive cerebral oedema, and corticosteroids remained unnecessary. Head elevation and volume status were closely monitored.
Uncorrected TOF patients are prone to haemodynamic instability, reduced SVR, and increased PVR, which may exacerbate right-to-left shunting. Additional challenges include managing ICP without disrupting systemic circulation, and preventing cyanotic crises triggered by pain, hypovolaemia, or stress. Agents such as ketamine and ephedrine are particularly valuable for their haemodynamic profiles.
| Table 1: Summary of Anesthetic Strategies | |
| Phase | Strategy |
| Preoperative | IV rehydration, electrolyte correction, ceftriaxone, midazolam + ketamine |
| Induction | Ketamine + Propofol, fentanyl, atracurium, preoxygenation |
| Maintenance | Sevoflurane 1%, controlled ventilation (EtCO₂ 30–32 mmHg), intermittent fentanyl |
| Haemodynamics | Ephedrine to maintain SVR, avoid hypothermia, maintain normovolaemia |
| Extubation | Performed awake, oxygen via nasal cannula 2–3 L/min |
| Postoperative | ICU care, multimodal analgesia, propranolol, continued antibiotics |
4. CONCLUSION
Neuroanesthetic management in children with cerebral abscess and uncorrected Tetralogy of Fallot (TOF) requires an individualised and comprehensive approach, balancing cardiovascular stability with intracranial pressure control. The ketamine-propofol combination proved effective in maintaining haemodynamic equilibrium while suppressing sympathetic responses that could exacerbate right-to-left shunting. The decision to avoid corticosteroids was based on current evidence suggesting a potential for adverse infectious outcomes without proven benefit in this context.
An integrated anesthetic strategy comprising preoperative hydration, invasive and non-invasive haemodynamic monitoring, and controlled ventilation targeting precise PaCO₂ levels was pivotal in achieving perioperative success. This allowed effective ICP control without inducing systemic instability or hypoxaemia. Postoperative multimodal analgesia further facilitated optimal recovery, enabling accurate neurological assessment and preventing cyanotic episodes.
This case underscores the vital importance of multidisciplinary collaboration among anesthesia, neurosurgery, cardiology, and intensive care teams in managing paediatric patients with the complex combination of cerebral abscess and TOF. It also provides practical guidance for anesthetists working in resource-limited settings, enabling safe and effective care for similarly challenging cases.
- Ethical Considerations
- Conflict of Interest
- 7. Authors contribution
DYB: Supervision, manuscript review, guidance in study design.
IAR: Supervision, critical revision of manuscript, guidance in study design.
8. REFERENCES
- Mishra SS, Das S, Senapati SB, Parida DK. Challenges in anesthetic management of patients with cyanotic congenital heart disease undergoing neurosurgery. J Clin Anesth. 2016;34:354–60. DOI: 1016/j.jclinane.2016.05.012.
- Prasad GL, Rajesh A, Radhakrishnan M. Brain abscess in cyanotic congenital heart disease: still a challenge in developing countries. Pediatr Neurosurg. 2019;54(5):305–11. DOI: 1159/000500764 .
- Brouwer MC, Tunkel AR, McKhann GM 2nd, van de Beek D. Brain abscess. N Engl J Med. 2014;371(5):447–56. DOI: 1056/NEJMra1301635.
- Al-Anazi K, Al-Jasser A, Yassin M. Anesthetic management of brain abscess with cyanotic congenital heart disease: Two case reports. Saudi J Anaesth. 2023;17(1):71–74.
- Dwivedi P, Batra YK, Bharti N, Panda NB. Anesthetic management of an 8-year-old with uncorrected TOF and multiple brain abscesses. Anesth Essays Res. 2020;14(2):349–51.
- Marulasiddappa V, Raghavavendra BS. Anesthesia for a Rare Case of Uncorrected Pentalogy of Fallot Undergoing Craniotomy and Drainage of Brain Abscess. J Clin Diagn Res. 2015 UD1–2. PMCID: PMC4573017 DOI: 7860/JCDR/2015/13650.6149
- Lakhani M, Memon RS, Khan F. Brain abscess: a rare complication in a child with tetralogy of Fallot. IDCases. 2020;22:e00954. https://doi.org/10.1016/j.idcr.2020.e00954
- Mameli, C., Genoni, T., Madia, C., Doneda, C., Penagini, F., & Zuccotti, G. (2019). Brain abscess in pediatric age: a review. Child's nervous system : ChNS : official journal of the International Society for Pediatric Neurosurgery, 35(7), 1117–1128. https://doi.org/10.1007/s00381-019-04182-4
- Junghare SW, Desurkar V. Congenital heart diseases and anaesthesia. Indian J Anaesth. 2017;61(9):744–752. https://doi.org/10.4103/ija.IJA_415_17
- Mazandi VM, Lang SS, Rahman RK, Nishisaki A, Beaulieu F, Zhang B, et al (2023). Co-administration of Ketamine in Pediatric Patients with Neurologic Conditions at Risk for Intracranial Hypertension. Neurocritical care, 38(2), 242–253. PMCID: PMC10267668 DOI: 1007/s12028-022-01611-2
- Simjian, T., Muskens, I. S., Lamba, N., Yunusa, I., Wong, K., Veronneau, R., et al. (2018). Dexamethasone Administration and Mortality in Patients with Brain Abscess: A Systematic Review and Meta-Analysis. World neurosurgery, 115, 257–263. https://doi.org/10.1016/j.wneu.2018.04.130
- Bodilsen, J., D'Alessandris, Q. G., Humphreys, H., Iro, M. A., Klein, M., Last, K., et al (2024). European society of Clinical Microbiology and Infectious Diseases guidelines on diagnosis and treatment of brain abscess in children and adults. Clinical microbiology and infection : the official publication of the European Society of Clinical Microbiology and Infectious Diseases, 30(1), 66–89. https://doi.org/10.1016/j.cmi.2023.08.016
- Laws JC, Vance EH, Betters KA, Anderson JJ, Fleishman S, Bonfield CM, et al. (2023). Acute Effects of Ketamine on Intracranial Pressure in Children With Severe Traumatic Brain Injury. Critical care medicine, 51(5), 563–572. https://doi.org/10.1097/CCM.0000000000005806
- Wang X, Ding X, Tong Y, Zong J, Zhao X, Ren H, et al. Ketamine does not increase intracranial pressure compared with opioids: meta-analysis of randomized controlled trials. J Anesth, 2014;28(6):821–827. DOI: 1007/s00540-014-1845-3
- Schwenk ES, Viscusi ER, Buvanendran A, Hurley RW, Wasan AD, Narouze S, et al. Consensus guidelines on the use of intravenous ketamine infusions for acute pain management from the American Society of Regional Anesthesia and Pain Medicine, the American Academy of Pain Medicine, and the American Society of Anesthesiologists. Reg Anesth Pain Med. 2018;43(5):456-466. doi:10.1097/AAP.0000000000000806