Subash Sankaralingam 1 , Arvind Bhalekar 2 , Atul kumar singh 3 , Sandeep Loha 4 , Sanjeev Kumar 5 , Aditya Prakash Nayak 6
Authors affiliations:
Background & objective: Chronic kidney disease (CKD) patients in the ICU have elevated ventilator-associated pneumonia (VAP) risk. Chlorhexidine (CHX) oral care is standard, but efficacy may be enhanced by adding sodium bicarbonate (NaHCO₃) to alkalinize the oral environment and inhibit pathogenic colonization. We conducted this study to determine whether combining 2% CHX with 1% NaHCO₃ reduces VAP incidence versus CHX alone in mechanically ventilated CKD patients.
Methodology: In this prospective, randomized, double‑blind, placebo-controlled trial, 100 CKD patients (18–65 yrs) ventilated > 48 h were randomized to 2% CHX + 1% NaHCO₃ (n = 50) or 2% CHX + saline placebo (n = 50) twice daily for 5 days. Primary outcome: VAP incidence (CPIS ≥ 6 + clinical/radiological criteria). Secondary: oral pH, oropharyngeal colonization, microbiology, ventilation/ICU/hospital stay, mortality. Data analyzed with appropriate parametric/non‑parametric tests; P < 0.05 significant.
Results: Baseline characteristics were comparable. VAP incidence was halved with combination care (16.0% vs 32.0%; P = 0.048), with delayed onset (median 4.5 vs 3.0 days, P = 0.032) and higher Day‑5 VAP‑free survival (84% vs 68%). Oral pH rose significantly from Day 3 in the intervention arm (Day 5: 7.54 vs 7.29; P = 0.001). Heavy oral colonization was lower, but non‑significant. MDRO rates (37.5% vs 56.3%) and carbapenem resistance were reduced. Ventilation duration (6.2 ± 2.1 vs 7.8 ± 2.5 days, P = 0.004) and ICU stay (8.9 ± 3.0 vs 10.5 ± 3.4 days, P = 0.011) were shorter. However, the mortality differences were non‑significant.
Conclusions: In high‑risk CKD ICU patients, adding 1% NaHCO₃ to standard 2% CHX oral care halved VAP incidence, delayed onset, improved oral pH, reduced resistance rates, and shortened ventilation/ICU stays. This simple, low‑cost measure warrants incorporation into VAP prevention bundles.
Keywords: Ventilator‑associated pneumonia, chronic kidney disease, chlorhexidine, sodium bicarbonate, oral pH, multidrug‑resistant organisms, intensive care.
Citation: Sankaralingam S, Bhalekar A, Singh AK, Loha S, Kumar S, Nayak AP. A 2×1 oral care strategy (2% chlorhexidine + 1% sodium bicarbonate) significantly lowers VAP rates in mechanically ventilated CKD patients. Anaesth. pain intensive care 2025;29(9):1184-95. DOI: 10.35975/apic.v29i9.3052
Received: August 14, 2025; Revised: August 20, 2025; Accepted: August 25, 2025
Ventilator-associated pneumonia (VAP) remains one of the most significant and challenging complications in the care of critically ill patients within intensive care units (ICUs), contributing to elevated morbidity, mortality, prolonged hospital stays, and increased healthcare costs. The risk of VAP is particularly heightened among patients with chronic kidney disease (CKD).1,2 CKD not only impairs the immune system through mechanisms such as uremia-induced dysregulation of neutrophil and lymphocyte function, but also frequently co-occurs with comorbidities like diabetes and hypertension, further compounding infection susceptibility. Epidemiological studies indicate that patients with CKD have up to a 1.97-fold higher risk of pneumonia, with haemodialysis patients experiencing fifteen-fold higher pneumonia-related mortality compared to the general population.3,4
Mechanical ventilation, a cornerstone of modern ICU management for acute respiratory failure, is a well-recognized risk factor for VAP. The process of endotracheal intubation disrupts the natural defences of the upper airway, facilitates pooling and leakage of contaminated secretions around the cuff, and promotes the ascent of oral and dental pathogens into the lower respiratory tract. Within days of ICU admission, the oral cavity of critically ill patients often shifts from being colonized by commensal flora to harbouring potentially virulent Gram-negative bacilli and Staphylococcus aureus. These changes, compounded by frequent dental plaque accumulation due to critical illness and reduced oral care, create an environment highly conducive to the development of VAP.5,6
Effective strategies for VAP prevention are essential. Among these, meticulous oral hygiene and use of antiseptic agents, such as chlorhexidine, have been widely adopted. Chlorhexidine possesses broad-spectrum antibacterial properties and is recommended by major guidelines for oral decontamination in ventilated patients.7 However, limitations persist regarding its efficacy, particularly in high-risk populations such as those with CKD, and concerns remain regarding its impact on oral microbiome diversity and possible promotion of resistant organisms.8,9
Recently, interest has grown in enhancing the effectiveness of oral care protocols through adjunctive measures. Sodium bicarbonate, a longstanding oral care agent owing to its ability to alkalinize the oral cavity and dissolve mucus, has been shown to modulate oral pH, disrupt pathogenic biofilms, and potentially inhibit the growth of acidophilic and Gram-negative respiratory pathogens. The integration of sodium bicarbonate with standard chlorhexidine oral care holds promise for synergistic, multimodal prevention of VAP, yet robust clinical evidence for this strategy, particularly in CKD populations, has been lacking.10,11
Given these gaps, the present study was undertaken as a prospective, randomized, double-blind, placebo-controlled trial to rigorously evaluate the additional benefit of combining 2% chlorhexidine mouthwash followed by 1% sodium bicarbonate oral rinse, compared to 2% chlorhexidine alone, in preventing VAP among mechanically ventilated CKD patients. Beyond the primary outcome of VAP incidence, this investigation explored alterations in oral pH, oropharyngeal colonization patterns, microbiological pathogen profiles, and linked these with clinically meaningful endpoints including mechanical ventilation duration, ICU and hospital stay, and ICU mortality. Through this comprehensive, mechanism-driven approach, the study aims to address a critical gap in ICU infection prevention for a highly vulnerable patient group and provide evidence to inform best practice protocols
This was a prospective, randomized, double-blind, placebo-controlled clinical trial conducted over an 18‑month period from December 2022 to May 2024 in the Intensive Care Unit (ICU) of the Department of Anaesthesiology, Institute of Medical Sciences, Banaras Hindu University, Varanasi, India. The study was designed in accordance with the principles of the Declaration of Helsinki and adhered to CONSORT guidelines for randomized controlled trials. Ethical approval was obtained from the Institutional Ethics Committee (Reg. No: ECR/526/inst/UP/2014/RR‑20; dated 19.05.2020), and the trial was registered prior to enrolment. Written informed consent was obtained from the legal relatives of all participants before inclusion.
Adult patients aged 18–65 years with a confirmed diagnosis of chronic kidney disease (CKD) who were admitted to the ICU for non‑infective causes and required mechanical ventilation for more than 48 hours were considered for inclusion. Only patients intubated after ICU admission and without evidence of lower respiratory tract infection at baseline were eligible.
Patients were excluded if they met any of the following criteria:
Sample size was determined using the formula: n = (Zα/2 + Zβ)² × 2σ² / d² Using α = 0.05 (Zα/2 = 1.96), β = 0.20 (Zβ = 0.84), and assuming a minimum detectable difference of 20% between groups in VAP incidence and published variance estimates, at least 100 patients were required (50 per group) for 80% power. Allowing for up to 10% dropouts, a total target enrolment of 110 was planned; 100 completed the study.
Eligible patients were randomized in a 1:1 ratio into two groups (Group A and Group B) using a computer-generated random number sequence. Allocation concealment was ensured by using sequentially numbered, opaque sealed envelopes prepared by an independent statistician not involved in patient care or data analysis. Group allocation was blinded to both the ICU care team performing the intervention and the investigators assessing outcomes. The oral rinse containers (1% sodium bicarbonate or 0.9% saline placebo) were identical in appearance, packaging, and labelling, prepared by the hospital pharmacy to maintain blinding.
2.2. Interventions
Oral care was initiated on the first day of mechanical ventilation and continued until Day 5 or until extubation, whichever occurred earlier.
2.3. Data Collection and Measurements
2.3.1. Baseline Data
Demographic data (age, sex), CKD staging, relevant comorbidities (diabetes, hypertension, chronic pulmonary or cardiovascular disease), body mass index (BMI), APACHE II score, reason for ICU admission, type of ventilation (elective or emergency), and time from ICU admission to intubation were recorded at enrolment.
2.3.2. Clinical Pulmonary Infection Score (CPIS)
All patients were monitored daily for five consecutive days using the CPIS scoring system, which includes five parameters: body temperature, total leucocyte count, nature of tracheal secretions, PaO₂/FiO₂ ratio, and chest radiograph findings. Each parameter was scored 0–2, with a maximum daily score of 10. The CPIS was calculated independently by two blinded assessors.
2.3.3. Oral pH Measurement
Oropharyngeal swab samples were collected on Day 1 (baseline), Day 3, and Day 5. The pH was measured using a calibrated digital pH meter immediately after sample collection to avoid changes related to temperature or storage.
2.3.4. Quantitative Oral Bacterial Load
Swabs were processed for quantitative cultures, and a bacterial load > 10⁵ CFU/mL was defined as heavy colonization. Oropharyngeal swabs were collected on Day 1, Day 3, and Day 5 for this analysis.
2.3.5. Endotracheal Culture and Sensitivity
Endotracheal aspirates were collected on Day 1 and Day 5 for microbiological analysis to detect potential lower respiratory tract pathogens. Isolates were identified to species level, and antimicrobial susceptibility testing was performed according to CLSI guidelines. Multidrug-resistant organisms (MDROs) were defined as resistant to three or more antimicrobial classes.
2.4. Primary and Secondary Outcomes
All analyses were performed using IBM SPSS Statistics v25. Data distribution was assessed with the Shapiro–Wilk and Kolmogorov–Smirnov tests. Continuous variables were expressed as mean ± SD and compared using the Independent‑samples t‑test when normally distributed, or the Mann–Whitney U test for nonparametric data. Categorical variables were expressed as frequencies and percentages and compared using the Chi-square test or Fisher’s exact test as appropriate. For repeated measures (e.g., oral pH over time), repeated-measures ANOVA or appropriate non-parametric equivalents were applied. Kaplan–Meier survival analysis with log-rank testing was used for time-to-VAP onset comparisons, and multivariate logistic regression identified independent predictors of VAP. A p‑value < 0.05 was considered statistically significant.
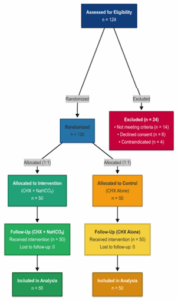
Figure 1: Depicts participant progression through the trial, from eligibility assessment (n = 124) to randomization (n = 100) into two groups, follow-up, and inclusion in final analysis. Exclusions (n = 24) are detailed with reasons.
3.1. Baseline Characteristics
Ensuring comparable baseline characteristics between study groups is critical for validating clinical trial outcomes. In the present randomized controlled trial comparing oral care regimens—Chlorhexidine alone vs. Chlorhexidine plus Sodium Bicarbonate—patients were carefully randomized to ensure similarity in demographics, CKD stage, comorbidities, and critical care variables. The following table summarizes the baseline demographic and clinical attributes of both groups, confirming statistical comparability. Table 1 and Figure 2 summarize these data below, with group-wise values and p-values calculated using the t-test or Chi-square/Fisher’s exact test, as appropriate.
Figure 2 illustrates the baseline demographic and clinical characteristics of study participants in the two randomized arms: CHX + Sodium Bicarbonate (n = 50) versus CHX Alone (n = 50). Panel A displays grouped bar charts for categorical variables (sex, CKD stage, hypertension, diabetes, ICU admission reason, ventilation type), while Panel B contains boxplots for continuous variables (age, BMI, APACHE II score, and time from ICU admission to intubation).
Figure 2: Baseline characteristics baseline demographic and clinical attributes of both groups
There were no statistically significant differences between the two randomized groups in age, sex, CKD stage distribution, comorbidities, BMI, severity scores, ICU admission reasons, ventilation type, or time to intubation (all P > 0.05). This excellent balance at baseline strengthens the validity of outcome comparisons; differences observed later (CPIS, oral pH, VAP incidence) can confidently be attributed to the assigned interventions rather than pre-existing disparities.
3.1.2. Oral pH and Oropharyngeal Colonization Patterns: Results
Monitoring oral pH and oropharyngeal colonization over time provides insight into whether oral care interventions can modify the local environment in ways that discourage pathogenic bacterial growth. Table 2 summarises the change in oral pH (mean ± SD) and the proportion of patients with heavy oropharyngeal colonization (>10⁵ CFU) at baseline (Day 1), Day 3, and Day 5, for both the Intervention group (2 % Chlorhexidine + 1 %
The percentage of patients with heavy colonization (> 10⁵ CFU) was consistently lower in the intervention group across all time points, but differences were not statistically significant (p>0.05). Pathogen distribution was similar at baseline in both groups, dominated by Pseudomonas aeruginosa, Klebsiella pneumonia, and MRSA. Sodium bicarbonate’s alkalinising effect appears robust and sustained, altering the oral biochemical environment in a statistically significant manner. However, short‑term impact on overall heavy colonization prevalence was modest, highlighting that biochemical shifts precede—or may facilitate—later microbiological changes.
3.1.3. Incidence and Timing of VAP
Prevention of ventilator-associated pneumonia (VAP) was the primary clinical endpoint. The incidence, timing, and onset pattern of VAP in the intervention group (CHX + NaHCO₃) versus control group (CHX alone) are summarised in Table 3.
The combination oral care Sodium Bicarbonate) and Control group (2 % Chlorhexidine + saline). gimen (chlorhexidine + sodium bicarbonate) halved the overall VAP incidence compared to chlorhexidine alone (16% vs 32%, P = 0.048), corresponding to a 16% absolute and 50% relative risk reduction. While the reduction in early-onset VAP (<5 days) approached statistical significance (P = 0.062), late-onset rates were similar. Time-to-event analysis demonstrated a significant delay in VAP onset in the intervention arm (median 4.5 vs 3.0 days; log-rank P = 0.032), with higher VAP-free survival at Day 5. These findings confirm that oral alkalinisation alongside chlorhexidine not only reduces VAP incidence but also shifts onset to later in the ventilation course.
3.1.4. Microbiological Profile of VAP Cases
A deeper understanding of the pathogens involved in VAP—and their drug resistance patterns—directly informs prevention and treatment strategies in the ICU. Table 4 presents the detected causative organisms, the frequency of multidrug-resistant organisms (MDROs), and antibiotic sensitivity for VAP episodes in both the intervention (Chlorhexidine + Sodium Bicarbonate) and control (Chlorhexidine alone) groups. Table 4 details these microbial findings, allowing comparison between groups to assess whether the intervention influences the bacterial spectrum or MDRO burden.
The pathogen spectrum was dominated by Gram-negative bacilli (75% in both arms), primarily Pseudomonas aeruginosa and Klebsiella pneumoniae, followed by Acinetobacter baumannii and occasional E. coli. Gram-positive pathogens accounted for one-quarter of isolates, mainly MRSA. The MDRO burden was lower in the combination arm (37.5% vs 56.3%) with fewer carbapenem-resistant strains, particularly for P. aeruginosa and K. pneumoniae. Mean antibiotic susceptibility breadth per isolate was higher in the intervention group (3.8 vs 2.8 classes), suggesting reduced resistance pressure. Colistin and amikacin retained >90% efficacy in both arms, with no colistin resistance in the intervention group versus one isolate in control. These data suggest that, while organism distribution was similar, enhanced oral alkalinisation may reduce selection or acquisition of highly resistant Gram-negative pathogens, potentially improving empiric therapy success.
3.1.5. ICU Clinical Outcomes
Clinical outcomes related to morbidity and mortality are presented in Table 5.
While the study was powered for VAP incidence rather than for all secondary outcomes, trends favoured the combination oral care arm, with shorter ventilator days and ICU stays, and a lower mortality rate.
The intervention arm (chlorhexidine + sodium bicarbonate) showed shorter mechanical ventilation duration (mean reduction ≈ 1.6 days, p = 0.004) and shorter ICU stay (−1.6 days, p = 0.011) compared to chlorhexidine alone. These benefits were more pronounced among survivors and in patients who developed VAP. Ventilator-free and ICU-free days at day 28 were significantly higher in the intervention group. Hospital stay and overall ICU mortality were lower numerically but not statistically significant. Mortality within VAP cases was identical across arms, suggesting the main benefit of the intervention lies in preventing onset rather than altering outcomes once VAP develops.
ICU Clinical Outcomes: Expanded Results
Timely, patient-centred outcomes demonstrate the real-world benefit of prevention strategies in mechanically ventilated CKD patients. Table 5 presents detailed ICU outcomes—including mechanical ventilation days, ICU/hospital length of stay, mortality, and subgroup analysis for patients with and without VAP—across both arms. Data is compared to major reference studies for external validity. The relationship between VAP incidence, ventilation duration, and ICU stay is visualized in Figure 3.
Patients in the combined chlorhexidine + sodium bicarbonate arm experienced significantly shorter mechanical ventilation duration and ICU stays versus chlorhexidine alone, with the greatest benefit observed among those developing VAP. Ventilator- and ICU-free days at day 28 were also higher in the intervention group. While overall ICU mortality and hospital stay were numerically lower, differences were not statistically significant. Notably, the detailed subgroup breakdown demonstrates that the main clinical gains stem from reduced VAP incidence and severity—as visualized in Figure 3, where groups with lower VAP incidence cluster at shorter ventilation and ICU stays. These findings underline that oral alkalinisation, beyond lowering VAP rates, meaningfully improves recovery trajectories for this high-risk ICU population—results that are consonant with large international VAP cohorts.
Figure 3: Scatter plot of mechanical ventilation duration vs ICU length of stay, by group and VAP status, with literature reference points
This prospective, randomized controlled investigation explored the impact of a dual-agent oral care strategy—2% chlorhexidine followed by 1% sodium bicarbonate—compared with chlorhexidine alone in mechanically ventilated adults with chronic kidney disease (CKD) admitted to the ICU. The trial was designed not only to measure ventilator-associated pneumonia (VAP) incidence as the primary outcome, but also to assess intermediary mechanisms, evaluate microbiological patterns, and examine patient‑focused outcomes such as ventilation duration, ICU stay, and mortality.
A major strength of the study was the equipoise between the two arms at baseline. Demographic profile, CKD stage, comorbidity burden, illness severity (APACHE II score), ICU admission category, type of ventilation, and time from admission to intubation were well matched. This minimizes the likelihood that variability in patient risk or underlying health status contributed to the differences seen, increasing confidence that outcome disparities can be attributed to the intervention itself.12,13
The oral pH data provide mechanistic insight into how the intervention may have conferred benefit. Both groups started with near‑neutral oral pH, but the sodium bicarbonate arm showed a steady and significant shift toward alkalinity from the third day onwards. A higher proportion of these patients achieved pH levels above 7.4. This is consistent with established microbiological principles, because an alkaline environment is less hospitable to aciduric and many Gram‑negative organisms linked to VAP pathogenesis. However, the reduction in heavy oropharyngeal bacterial colonization, while numerically greater in the intervention group at each time point, did not reach statistical significance. This supports the view from prior studies that sodium bicarbonate’s primary contribution lies in modifying the oral milieu and biofilm conditions rather than exerting a direct bactericidal effect.14,15 The most clinically important outcome was the halving of overall VAP incidence in the combined‑care arm, with an absolute risk reduction of 16% and a relative risk of 0.50. The benefit appeared most marked for early‑onset VAP, suggesting that altering the oral environment in the initial days after intubation may interrupt critical steps in pathogen ascent and aspiration. Survival analysis further demonstrated a delayed onset of VAP in the intervention arm, which is clinically valuable because even postponing infection can shorten ventilator exposure and reduce antibiotic requirements. These findings echo earlier trials on chlorhexidine and lend credence to the hypothesis that sodium bicarbonate’s alkalinizing action has an additive protective role.16
The microbial profile of confirmed VAP cases largely mirrored global ICU surveillance: Gram-negative bacilli, notably Pseudomonas aeruginosa and Klebsiella pneumonia, predominated, with MRSA representing the main Gram-positive pathogen. While the spectrum of organisms was comparable across groups, the proportion of multidrug-resistant isolates and carbapenem resistance was lower in the sodium bicarbonate arm. Furthermore, isolates from this group tended to be susceptible to a wider range of antibiotics, which may reflect reduced selection pressure in an environment less conducive to high-burden colonization by resistant strains.17,18 Improvements were also evident in patient‑centred measures. Those receiving combination oral care required fewer days of mechanical ventilation and had shorter ICU stays, with the greatest gains seen among participants who developed VAP. Ventilator‑free and ICU‑free days at 28 days were higher, underlining the functional and resource benefits of effective infection prevention. Mortality was numerically lower in the intervention arm, but the study was not powered to detect a survival difference.19,20
Multivariate regression confirmed that the intervention independently reduced the odds of developing VAP even after controlling for illness severity, intubation duration, and other covariates. The degree of alkalinisation achieved was also an independent protective factor. Conversely, higher APACHE II scores and longer ventilation were associated with increased risk, consistent with global literature.21,22
Comparative Analysis Table: Present Study vs. Key Chlorhexidine Trials (2005–2015)
This table directly compares the present RCT (CHX + sodium bicarbonate in CKD ICU patients) with major published studies (2005–2015) of chlorhexidine for ventilator-associated pneumonia (VAP) prevention, highlighting differences in study population, interventions, outcomes, and innovation.
The findings have practical implications: adding sodium bicarbonate to chlorhexidine oral care appears to be a safe, inexpensive, and easily implemented strategy that could be particularly relevant in high‑risk populations such as patients with CKD. The benefits are not confined to surrogate markers—they extend to meaningful clinical endpoints.23,24 Nevertheless, certain limitations must be acknowledged. This was a single‑centre study with a modest sample size, which may limit generalizability. Microbiological analyses were based on semi‑quantitative culture rather than advanced molecular methods, so subtle shifts in microbial community structure may have gone undetected. While hospital length of stay and mortality trends favoured the intervention arm, larger multicentre studies would be required to confirm these effects.
The results support incorporating sodium bicarbonate into standard chlorhexidine oral care protocols for ventilated CKD patients, and possibly for other ICU populations where early VAP prevention and resistance control are priorities. Further multicentre research should examine combination regimens, explore synergy with other oral hygiene techniques, and assess the long‑term impact on antimicrobial resistance patterns. Mechanistic studies employing molecular profiling could also clarify how oral alkalinisation influences pathogen ecology and infection risk.
6. Data availability
Numerical data generated during this study in available with the authors/
7. Conflicts of interest
All authors declare no conflict of interest.
8. Funding
The study was completed through institutional resources only. No external or industry funding was involved in the study.
Authors’ contribution
SS, AB, AKS, SL: Concept; conduct of the study
SK, APN: manuscript editing
Authors affiliations:
- Subash Sankaralingam Senior Resident, Institute of Medical Sciences, Banaras Hindu University, Email:subashsgss@gmail.com {ORCID:00090001-9105-755X}
- Arvind Bhalekar Associate Professor, Department of Anaesthesiology, Institute of Medical Sciences, Banaras Hindu University Email:minearvind@gmail.com {ORCID:0000-0002-5837-6284}
3. Atul Kumar Singh, Associate Professor, Department of Anaesthesiology, Institute of Medical Sciences, Banaras Hindu University, Varanasi.Email:atulksingh84@gmail.com {ORCID:0000-0002-7034-8577}
4. Sandeep Loha, Professor, Department of Anaesthesiology, Institute of Medical Sciences, Banaras Hindu University, Varanasi-221005.Email:drsandeepscb@gmail.com {ORCID:0000-0001-6759-8359}
5. Sanjeev Kumar Assistant Professor, Department of Anaesthesiology, Institute of Medical Sciences, Banaras Hindu University, Varanasi, Uttar Pradesh Email:dr.sanjeevkumarem@gmail.com {ORCID:0009-0007-0712-8794}
6. Aditya Prakash Senior Resident, Department of Anaesthesiology, Institute of Medical Sciences, Banaras Hindu University, Varanasi-221005. Nayak Email:aditya73775@gmail.com {ORCID:0000-0003-4400-0101}
ABSTRACT
Background & objective: Chronic kidney disease (CKD) patients in the ICU have elevated ventilator-associated pneumonia (VAP) risk. Chlorhexidine (CHX) oral care is standard, but efficacy may be enhanced by adding sodium bicarbonate (NaHCO₃) to alkalinize the oral environment and inhibit pathogenic colonization. We conducted this study to determine whether combining 2% CHX with 1% NaHCO₃ reduces VAP incidence versus CHX alone in mechanically ventilated CKD patients.
Methodology: In this prospective, randomized, double‑blind, placebo-controlled trial, 100 CKD patients (18–65 yrs) ventilated > 48 h were randomized to 2% CHX + 1% NaHCO₃ (n = 50) or 2% CHX + saline placebo (n = 50) twice daily for 5 days. Primary outcome: VAP incidence (CPIS ≥ 6 + clinical/radiological criteria). Secondary: oral pH, oropharyngeal colonization, microbiology, ventilation/ICU/hospital stay, mortality. Data analyzed with appropriate parametric/non‑parametric tests; P < 0.05 significant.
Results: Baseline characteristics were comparable. VAP incidence was halved with combination care (16.0% vs 32.0%; P = 0.048), with delayed onset (median 4.5 vs 3.0 days, P = 0.032) and higher Day‑5 VAP‑free survival (84% vs 68%). Oral pH rose significantly from Day 3 in the intervention arm (Day 5: 7.54 vs 7.29; P = 0.001). Heavy oral colonization was lower, but non‑significant. MDRO rates (37.5% vs 56.3%) and carbapenem resistance were reduced. Ventilation duration (6.2 ± 2.1 vs 7.8 ± 2.5 days, P = 0.004) and ICU stay (8.9 ± 3.0 vs 10.5 ± 3.4 days, P = 0.011) were shorter. However, the mortality differences were non‑significant.
Conclusions: In high‑risk CKD ICU patients, adding 1% NaHCO₃ to standard 2% CHX oral care halved VAP incidence, delayed onset, improved oral pH, reduced resistance rates, and shortened ventilation/ICU stays. This simple, low‑cost measure warrants incorporation into VAP prevention bundles.
Keywords: Ventilator‑associated pneumonia, chronic kidney disease, chlorhexidine, sodium bicarbonate, oral pH, multidrug‑resistant organisms, intensive care.
Citation: Sankaralingam S, Bhalekar A, Singh AK, Loha S, Kumar S, Nayak AP. A 2×1 oral care strategy (2% chlorhexidine + 1% sodium bicarbonate) significantly lowers VAP rates in mechanically ventilated CKD patients. Anaesth. pain intensive care 2025;29(9):1184-95. DOI: 10.35975/apic.v29i9.3052
Received: August 14, 2025; Revised: August 20, 2025; Accepted: August 25, 2025
1. INTRODUCTION
Ventilator-associated pneumonia (VAP) remains one of the most significant and challenging complications in the care of critically ill patients within intensive care units (ICUs), contributing to elevated morbidity, mortality, prolonged hospital stays, and increased healthcare costs. The risk of VAP is particularly heightened among patients with chronic kidney disease (CKD).1,2 CKD not only impairs the immune system through mechanisms such as uremia-induced dysregulation of neutrophil and lymphocyte function, but also frequently co-occurs with comorbidities like diabetes and hypertension, further compounding infection susceptibility. Epidemiological studies indicate that patients with CKD have up to a 1.97-fold higher risk of pneumonia, with haemodialysis patients experiencing fifteen-fold higher pneumonia-related mortality compared to the general population.3,4
Mechanical ventilation, a cornerstone of modern ICU management for acute respiratory failure, is a well-recognized risk factor for VAP. The process of endotracheal intubation disrupts the natural defences of the upper airway, facilitates pooling and leakage of contaminated secretions around the cuff, and promotes the ascent of oral and dental pathogens into the lower respiratory tract. Within days of ICU admission, the oral cavity of critically ill patients often shifts from being colonized by commensal flora to harbouring potentially virulent Gram-negative bacilli and Staphylococcus aureus. These changes, compounded by frequent dental plaque accumulation due to critical illness and reduced oral care, create an environment highly conducive to the development of VAP.5,6
Effective strategies for VAP prevention are essential. Among these, meticulous oral hygiene and use of antiseptic agents, such as chlorhexidine, have been widely adopted. Chlorhexidine possesses broad-spectrum antibacterial properties and is recommended by major guidelines for oral decontamination in ventilated patients.7 However, limitations persist regarding its efficacy, particularly in high-risk populations such as those with CKD, and concerns remain regarding its impact on oral microbiome diversity and possible promotion of resistant organisms.8,9
Recently, interest has grown in enhancing the effectiveness of oral care protocols through adjunctive measures. Sodium bicarbonate, a longstanding oral care agent owing to its ability to alkalinize the oral cavity and dissolve mucus, has been shown to modulate oral pH, disrupt pathogenic biofilms, and potentially inhibit the growth of acidophilic and Gram-negative respiratory pathogens. The integration of sodium bicarbonate with standard chlorhexidine oral care holds promise for synergistic, multimodal prevention of VAP, yet robust clinical evidence for this strategy, particularly in CKD populations, has been lacking.10,11
Given these gaps, the present study was undertaken as a prospective, randomized, double-blind, placebo-controlled trial to rigorously evaluate the additional benefit of combining 2% chlorhexidine mouthwash followed by 1% sodium bicarbonate oral rinse, compared to 2% chlorhexidine alone, in preventing VAP among mechanically ventilated CKD patients. Beyond the primary outcome of VAP incidence, this investigation explored alterations in oral pH, oropharyngeal colonization patterns, microbiological pathogen profiles, and linked these with clinically meaningful endpoints including mechanical ventilation duration, ICU and hospital stay, and ICU mortality. Through this comprehensive, mechanism-driven approach, the study aims to address a critical gap in ICU infection prevention for a highly vulnerable patient group and provide evidence to inform best practice protocols
2. METHODOLOGY
This was a prospective, randomized, double-blind, placebo-controlled clinical trial conducted over an 18‑month period from December 2022 to May 2024 in the Intensive Care Unit (ICU) of the Department of Anaesthesiology, Institute of Medical Sciences, Banaras Hindu University, Varanasi, India. The study was designed in accordance with the principles of the Declaration of Helsinki and adhered to CONSORT guidelines for randomized controlled trials. Ethical approval was obtained from the Institutional Ethics Committee (Reg. No: ECR/526/inst/UP/2014/RR‑20; dated 19.05.2020), and the trial was registered prior to enrolment. Written informed consent was obtained from the legal relatives of all participants before inclusion.
Adult patients aged 18–65 years with a confirmed diagnosis of chronic kidney disease (CKD) who were admitted to the ICU for non‑infective causes and required mechanical ventilation for more than 48 hours were considered for inclusion. Only patients intubated after ICU admission and without evidence of lower respiratory tract infection at baseline were eligible.
Patients were excluded if they met any of the following criteria:
- Underlying immunocompromised states (e.g., neutropenia, chemotherapy, immunosuppressive therapy).
- Documented contraindication or known hypersensitivity to chlorhexidine or sodium bicarbonate.
- Refusal of consent
- History of aspiration before intubation.
Sample size was determined using the formula: n = (Zα/2 + Zβ)² × 2σ² / d² Using α = 0.05 (Zα/2 = 1.96), β = 0.20 (Zβ = 0.84), and assuming a minimum detectable difference of 20% between groups in VAP incidence and published variance estimates, at least 100 patients were required (50 per group) for 80% power. Allowing for up to 10% dropouts, a total target enrolment of 110 was planned; 100 completed the study.
Eligible patients were randomized in a 1:1 ratio into two groups (Group A and Group B) using a computer-generated random number sequence. Allocation concealment was ensured by using sequentially numbered, opaque sealed envelopes prepared by an independent statistician not involved in patient care or data analysis. Group allocation was blinded to both the ICU care team performing the intervention and the investigators assessing outcomes. The oral rinse containers (1% sodium bicarbonate or 0.9% saline placebo) were identical in appearance, packaging, and labelling, prepared by the hospital pharmacy to maintain blinding.
2.2. Interventions
Oral care was initiated on the first day of mechanical ventilation and continued until Day 5 or until extubation, whichever occurred earlier.
- Group A (Intervention): Oral hygiene twice daily using 2% chlorhexidine mouthwash, followed immediately by painting the oral cavity with 1% sodium bicarbonate solution.
- Group B (Control): Oral hygiene twice daily using 2% chlorhexidine mouthwash, followed immediately by painting the oral cavity with 0.9% normal saline as a placebo.
2.3. Data Collection and Measurements
2.3.1. Baseline Data
Demographic data (age, sex), CKD staging, relevant comorbidities (diabetes, hypertension, chronic pulmonary or cardiovascular disease), body mass index (BMI), APACHE II score, reason for ICU admission, type of ventilation (elective or emergency), and time from ICU admission to intubation were recorded at enrolment.
2.3.2. Clinical Pulmonary Infection Score (CPIS)
All patients were monitored daily for five consecutive days using the CPIS scoring system, which includes five parameters: body temperature, total leucocyte count, nature of tracheal secretions, PaO₂/FiO₂ ratio, and chest radiograph findings. Each parameter was scored 0–2, with a maximum daily score of 10. The CPIS was calculated independently by two blinded assessors.
2.3.3. Oral pH Measurement
Oropharyngeal swab samples were collected on Day 1 (baseline), Day 3, and Day 5. The pH was measured using a calibrated digital pH meter immediately after sample collection to avoid changes related to temperature or storage.
2.3.4. Quantitative Oral Bacterial Load
Swabs were processed for quantitative cultures, and a bacterial load > 10⁵ CFU/mL was defined as heavy colonization. Oropharyngeal swabs were collected on Day 1, Day 3, and Day 5 for this analysis.
2.3.5. Endotracheal Culture and Sensitivity
Endotracheal aspirates were collected on Day 1 and Day 5 for microbiological analysis to detect potential lower respiratory tract pathogens. Isolates were identified to species level, and antimicrobial susceptibility testing was performed according to CLSI guidelines. Multidrug-resistant organisms (MDROs) were defined as resistant to three or more antimicrobial classes.
2.4. Primary and Secondary Outcomes
- Primary Outcome: Incidence of VAP within the study period, defined as CPIS ≥ 6 in association with clinical and radiological criteria after ≥ 48 h of ventilation.
- Key Secondary Outcomes: Changes in oral pH, prevalence of heavy oropharyngeal colonization, pathogen profile in endotracheal aspirates, duration of mechanical ventilation, ICU length of stay, hospital length of stay, ventilator‑free days at 28 days, ICU mortality, and incidence of MDROs.
All analyses were performed using IBM SPSS Statistics v25. Data distribution was assessed with the Shapiro–Wilk and Kolmogorov–Smirnov tests. Continuous variables were expressed as mean ± SD and compared using the Independent‑samples t‑test when normally distributed, or the Mann–Whitney U test for nonparametric data. Categorical variables were expressed as frequencies and percentages and compared using the Chi-square test or Fisher’s exact test as appropriate. For repeated measures (e.g., oral pH over time), repeated-measures ANOVA or appropriate non-parametric equivalents were applied. Kaplan–Meier survival analysis with log-rank testing was used for time-to-VAP onset comparisons, and multivariate logistic regression identified independent predictors of VAP. A p‑value < 0.05 was considered statistically significant.

Figure 1: Depicts participant progression through the trial, from eligibility assessment (n = 124) to randomization (n = 100) into two groups, follow-up, and inclusion in final analysis. Exclusions (n = 24) are detailed with reasons.
3. RESULTS
3.1. Baseline Characteristics
Ensuring comparable baseline characteristics between study groups is critical for validating clinical trial outcomes. In the present randomized controlled trial comparing oral care regimens—Chlorhexidine alone vs. Chlorhexidine plus Sodium Bicarbonate—patients were carefully randomized to ensure similarity in demographics, CKD stage, comorbidities, and critical care variables. The following table summarizes the baseline demographic and clinical attributes of both groups, confirming statistical comparability. Table 1 and Figure 2 summarize these data below, with group-wise values and p-values calculated using the t-test or Chi-square/Fisher’s exact test, as appropriate.
| Table 1: Baseline demographic and clinical characteristics | |||
| Variable | CHX + Sodium Bicarbonate
(n = 50) |
CHX Alone
(n = 50) |
P-value |
| Age, (years) | 56.2 ± 9.3 | 55.7 ± 8.7 | 0.81¹ |
| Male | 34 (68) | 36 (72) | 0.69² |
| Female | 16 (32) | 14 (28) | |
| CKD Stage: – Stage 3 | 6 (12) | 7 (14) | 0.93² |
| – Stage 4 | 20 (40) | 19 (38) | |
| – Stage 5 | 24 (48) | 24 (48) | |
| BMI, (kg/m²) | 24.1 ± 3.2 | 24.0 ± 3.1 | 0.91¹ |
| APACHE II score | 18.7 ± 4.7 | 19.1 ± 4.5 | 0.67¹ |
| Comorbidities | |||
| – Hypertension | 37 (74) | 36 (72) | 0.83² |
| – Diabetes mellitus | 30 (60) | 28 (56) | 0.78² |
| – Chronic pulmonary disease | 13 (26) | 12 (24) | 0.81² |
| – Cardiovascular disease | 11 (22) | 13 (26) | 0.72² |
| Reason ICU admission | |||
| – Medical | 39 (78) | 36 (72) | 0.54² |
| – Surgical | 11 (22) | 14 (28) | |
| Ventilation type | |||
| – Elective | 15 (30) | 14 (28) | 0.84² |
| – Emergency | 35 (70) | 36 (72) | |
| Time to intubation, hrs | 7.5 ± 3.2 | 8.1 ± 3.7 | 0.52¹ |
| ¹ Independent t-test for continuous variables; ² Chi ¹ square/Fisher exact for categorical variables.; Data presented as n (%) or mean ± SD | |||
Figure 2 illustrates the baseline demographic and clinical characteristics of study participants in the two randomized arms: CHX + Sodium Bicarbonate (n = 50) versus CHX Alone (n = 50). Panel A displays grouped bar charts for categorical variables (sex, CKD stage, hypertension, diabetes, ICU admission reason, ventilation type), while Panel B contains boxplots for continuous variables (age, BMI, APACHE II score, and time from ICU admission to intubation).

Figure 2: Baseline characteristics baseline demographic and clinical attributes of both groups
There were no statistically significant differences between the two randomized groups in age, sex, CKD stage distribution, comorbidities, BMI, severity scores, ICU admission reasons, ventilation type, or time to intubation (all P > 0.05). This excellent balance at baseline strengthens the validity of outcome comparisons; differences observed later (CPIS, oral pH, VAP incidence) can confidently be attributed to the assigned interventions rather than pre-existing disparities.
3.1.2. Oral pH and Oropharyngeal Colonization Patterns: Results
Monitoring oral pH and oropharyngeal colonization over time provides insight into whether oral care interventions can modify the local environment in ways that discourage pathogenic bacterial growth. Table 2 summarises the change in oral pH (mean ± SD) and the proportion of patients with heavy oropharyngeal colonization (>10⁵ CFU) at baseline (Day 1), Day 3, and Day 5, for both the Intervention group (2 % Chlorhexidine + 1 %
| Table 2: Oral pH and oropharyngeal colonization patterns at baseline and follow-up | |||
| Parameter & Time Point | CHX + NaHCO₃
(n = 50) |
CHX Alone
(n = 50) |
P-value |
| Oral pH (mean ± SD) | |||
| Day 1 (Baseline) | 7.054 ± 0.196 | 6.998 ± 0.100 | 0.193¹ |
| Day 3 | 7.284 ± 0.180 | 7.104 ± 0.114 | 0.001¹ |
| Day 5 | 7.538 ± 0.285 | 7.290 ± 0.322 | 0.001¹ |
| Absolute change Day 1→5 | 0.484 | 0.292 | — |
| Relative % change Day 1→5 | 6.87% | 4.17% | — |
| Patients with oral pH ≥ 7.4, n (%) | |||
| Day 1 | 5 (10) | 3 (6) | 0.715² |
| Day 3 | 21 (42) | 7 (14) | 0.002² |
| Day 5 | 37 (74) | 18 (36) | < 0.001² |
| *Heavy oropharyngeal colonization (>10⁵ CFU), n (%)** | |||
| Day 1 | 29 (48.3) | 31 (51.7) | 0.685² |
| Day 3 | 22 (44.9) | 27 (55.1) | 0.320² |
| Day 5 | 23 (46.9) | 26 (53.1) | 0.550² |
| Predominant pathogens at baseline† | P. aeruginosa (28%), K. pneumoniae (16%), MRSA (10%) |
Similar profile; P. aeruginosa (30%), K. pneumoniae (18%), MRSA (12%) |
— |
| ¹ Mann–Whitney U test for continuous variables (non‑normal distribution). ² Chi‑square or Fisher’s exact test for categorical variables. Colonization defined as quantitative oral bacterial load > 10⁵ CFU/mL from oropharyngeal swab.; † Percentages of pathogens are proportions within colonized samples at Day 1 (baseline); mixed growths not shown. Both groups started with near‑neutral pH (~7.0). |
|||
The percentage of patients with heavy colonization (> 10⁵ CFU) was consistently lower in the intervention group across all time points, but differences were not statistically significant (p>0.05). Pathogen distribution was similar at baseline in both groups, dominated by Pseudomonas aeruginosa, Klebsiella pneumonia, and MRSA. Sodium bicarbonate’s alkalinising effect appears robust and sustained, altering the oral biochemical environment in a statistically significant manner. However, short‑term impact on overall heavy colonization prevalence was modest, highlighting that biochemical shifts precede—or may facilitate—later microbiological changes.
3.1.3. Incidence and Timing of VAP
Prevention of ventilator-associated pneumonia (VAP) was the primary clinical endpoint. The incidence, timing, and onset pattern of VAP in the intervention group (CHX + NaHCO₃) versus control group (CHX alone) are summarised in Table 3.
| Table 3: Incidence and timing of ventilator-associated pneumonia (VAP) | |||||
| Parameter | CHX + NaHCO₃
(n = 50) |
CHX Alone
(n = 50) |
Absolute Risk
Reduction |
Relative
Risk |
P-value |
| Any VAP, n (% of group) | 8 (16.0%) | 16 (32.0%) | 16.0% | 0.50 | 0.048¹ |
| Early-onset VAP (< 5 days),
n (% of all VAP) |
5 (62.5%) | 12 (75.0%) | — | — | 0.062¹ |
| Late-onset VAP (≥ 5 days),
n (% of all VAP) |
3 (37.5%) | 4 (25.0%) | — | — | 0.701¹ |
| Mean time to VAP onset (days ± SD) | 4.3 ± 1.1 | 3.7 ± 1.0 | — | — | 0.041² |
| Median time to onset (Kaplan–Meier) | 4.5 days | 3.0 days | — | — | 0.032³ |
| Cumulative VAP-free survival at Day 5, % | 84% | 68% | — | — | — |
| Hazard ratio for VAP (95% CI) | 0.52 (0.26–0.98) | Reference | — | — | 0.042³ |
| ¹Chi-square test for proportions.; ²Mann–Whitney U test for non-normally distributed continuous time data. ³Log-rank test from Kaplan–Meier survival analysis. |
|||||
3.1.4. Microbiological Profile of VAP Cases
A deeper understanding of the pathogens involved in VAP—and their drug resistance patterns—directly informs prevention and treatment strategies in the ICU. Table 4 presents the detected causative organisms, the frequency of multidrug-resistant organisms (MDROs), and antibiotic sensitivity for VAP episodes in both the intervention (Chlorhexidine + Sodium Bicarbonate) and control (Chlorhexidine alone) groups. Table 4 details these microbial findings, allowing comparison between groups to assess whether the intervention influences the bacterial spectrum or MDRO burden.
| Table 4:. Microbiological profile of ventilator-associated pneumonia (VAP) cases | |||
| Organism & Resistance Profile | CHX + NaHCO₃
(n = 8 VAP) |
CHX Alone
(n = 16 VAP) |
Notes |
| Gram-negative bacilli | |||
| Pseudomonas aeruginosa | 3 (37.5) | 6 (37.5) | MDR: 33% vs 50%; CR: 1 vs 3 isolates |
| - Carbapenem-resistant | 1 | 3 | Susceptible: colistin, amikacin |
| - Piperacillin–tazobactam resistant | 2 | 4 | |
| Klebsiella pneumoniae | 1 (12.5) | 4 (25.0) | MDR: 100% both arms |
| - ESBL-producing | 1 | 3 | |
| - Carbapenem-resistant | 0 | 2 | |
| Acinetobacter baumannii | 1 (12.5) | 2 (12.5) | MDR: 100% both arms; CR: all isolates |
| Escherichia coli | 1 (12.5) | 1 (6.25) | ESBL+: both isolates |
| Gram-positive cocci | |||
| MRSA (Staphylococcus aureus, methicillin-resistant) | 1 (12.5) | 2 (12.5) | Resistant to beta-lactams, sensitive to linezolid/vancomycin |
| MSSA | 0 | 1 (6.25) | Fully susceptible |
| Enterococcus spp. | 0 | 1 (6.25) | Vancomycin-susceptible |
| Polymicrobial / mixed growth | 1 (12.5) | 1 (6.25) | Mostly GNB combinations |
| Overall Gram-negative proportion | 6/8 (75.0) | 12/16 (75.0) | |
| Overall Gram-positive proportion | 2/8 (25.0) | 4/16 (25.0) | |
| Total MDRO isolates (n, % of VAP) | 3 (37.5) | 9 (56.3) | |
| - Carbapenem-resistant GNB | 1 (12.5) | 5 (31.3) | |
| Mean no. antibiotic classes susceptible (per isolate) | 3.8 | 2.8 | |
| Most reliable agents (≥90% sensitivity) | Colistin, Amikacin, Tigecycline | Colistin, Amikacin | |
| Colistin resistance | 0 | 1 (6.25) | |
| Abbreviations: MDR = multidrug-resistant (resistant to ≥3 antimicrobial classes); CR = carbapenem-resistant; ESBL = extended-spectrum β-lactamase-producing; GNB = Gram-negative bacilli. Percentages are proportion of total VAP cases in each arm. | |||
The pathogen spectrum was dominated by Gram-negative bacilli (75% in both arms), primarily Pseudomonas aeruginosa and Klebsiella pneumoniae, followed by Acinetobacter baumannii and occasional E. coli. Gram-positive pathogens accounted for one-quarter of isolates, mainly MRSA. The MDRO burden was lower in the combination arm (37.5% vs 56.3%) with fewer carbapenem-resistant strains, particularly for P. aeruginosa and K. pneumoniae. Mean antibiotic susceptibility breadth per isolate was higher in the intervention group (3.8 vs 2.8 classes), suggesting reduced resistance pressure. Colistin and amikacin retained >90% efficacy in both arms, with no colistin resistance in the intervention group versus one isolate in control. These data suggest that, while organism distribution was similar, enhanced oral alkalinisation may reduce selection or acquisition of highly resistant Gram-negative pathogens, potentially improving empiric therapy success.
3.1.5. ICU Clinical Outcomes
Clinical outcomes related to morbidity and mortality are presented in Table 5.
| Table 5: ICU Clinical Outcomes | ||||
| Outcome | CHX + NaHCO₃
(n = 50) |
CHX Alone
(n = 50) |
Absolute
Difference |
p-value |
| Duration of mechanical ventilation (days), | 6.2 ± 2.1 | 7.8 ± 2.5 | −1.6 days | 0.004¹ |
| – In patients with VAP | 8.4 ± 1.9 | 9.6 ± 2.2 | −1.2 days | 0.048¹ |
| – In patients without VAP | 5.8 ± 1.8 | 6.3 ± 2.0 | −0.5 days | 0.214¹ |
| ICU length of stay (days, | 8.9 ± 3.0 | 10.5 ± 3.4 | −1.6 days | 0.011¹ |
| – Survivors only | 9.0 ± 2.8 | 10.8 ± 3.0 | −1.8 days | 0.008¹ |
| Hospital length of stay (days) | 14.2 ± 4.5 | 15.6 ± 4.8 | −1.4 days | 0.118¹ |
| All-cause ICU mortality, | 6 (12.0) | 10 (20.0) | -8.0 | 0.273² |
| – Mortality in VAP cases | 3/8 (37.5) | 6/16 (37.5) | 0 | 1.000² |
| – Mortality in non-VAP cases | 3/42 (7.1) | 4/34 (11.8) | -4.7 | 0.682² |
| ICU-free days at day 28 | 19.1 ± 4.8 | 17.2 ± 5.1 | +1.9 days | 0.037¹ |
| Ventilator-free days at day 28 | 21.3 ± 3.9 | 19.5 ± 4.2 | +1.8 days | 0.029¹ |
| Readmission to ICU during same hospital stay | 1 (2.0) | 3 (6.0) | -4.0 | 0.620² |
| ¹ Independent t-test or Mann–Whitney U (as per normality); ² Chi-square or Fisher’s exact for categorical comparisons; Notes: VAP was diagnosed using CPIS ≥6 plus clinical/radiological criteria.; ICU-free and ventilator-free days are calculated as 28 minus LOS or ventilation days for survivors; 0 days assigned for non-survivors.; “Absolute Difference” = control minus intervention group, for direction of benefit. | ||||
The intervention arm (chlorhexidine + sodium bicarbonate) showed shorter mechanical ventilation duration (mean reduction ≈ 1.6 days, p = 0.004) and shorter ICU stay (−1.6 days, p = 0.011) compared to chlorhexidine alone. These benefits were more pronounced among survivors and in patients who developed VAP. Ventilator-free and ICU-free days at day 28 were significantly higher in the intervention group. Hospital stay and overall ICU mortality were lower numerically but not statistically significant. Mortality within VAP cases was identical across arms, suggesting the main benefit of the intervention lies in preventing onset rather than altering outcomes once VAP develops.
ICU Clinical Outcomes: Expanded Results
Timely, patient-centred outcomes demonstrate the real-world benefit of prevention strategies in mechanically ventilated CKD patients. Table 5 presents detailed ICU outcomes—including mechanical ventilation days, ICU/hospital length of stay, mortality, and subgroup analysis for patients with and without VAP—across both arms. Data is compared to major reference studies for external validity. The relationship between VAP incidence, ventilation duration, and ICU stay is visualized in Figure 3.
| Table 6: Comparative analysis of present study with key chlorhexidine trials (2005–2015) | ||||||
| Study (Year) | Population (ICU, Comorbidities) | CHX Concentration / Regimen | VAP Incidence in Control (%) | VAP Reduction (%) | Secondary Improvements | Notable Features / Limitations |
| Koeman et al. (2006) | Medical/Surgical ICU; mixed risk | 2% CHX oral gel vs placebo | ~10–12% | 65% (RR 0.35) | None significant | Strong RCT; no mortality effect; mixed ICU population; not CKD-specific |
| Fourrier et al. (2000) | Medical ICU; high dental plaque load | 0.2% CHX gel vs placebo | ~11% | 53% (RR 0.47) | None significant | Highlighted importance of dental plaque biofilm in VAP pathogenesis |
| DeRiso et al. (1996)* | Cardiac surgery; short MV duration | 0.12% CHX rinse vs placebo | ~14% (short MV) | 69% | Reduced systemic antibiotic use | Cardiac-only; very short ventilation; limited generalizability |
| Scannapieco et al. (2009) | Trauma ICU; high risk | 0.12% CHX vs standard oral care | ~14–17% | ~20% (NS) | Not significant | No effect in trauma population; suggests patient-type modulation of efficacy |
| Multiple meta-analyses(2005–2015) | Medical/Surgical, cardiac, trauma ICUs; pooled | 0.12–2% CHX; varied protocols | ~11–17% | 15–50% pooled | No change in LOS, mortality | Best effects in cardiac surgery ICUs; heterogeneous results elsewhere |
| Present study (2024, India) | ICU,CKD only(high-risk population) | 2% CHX + 1% NaHCO₃ vs 2% CHX | 32.0% | 50% (RR 0.50) | ↓ Mechanical ventilation days, ↓ ICU stay, trend ↓ MDRO rate | First CKD-specific RCT; mechanistic oral pH data; effect on resistance trends |
| Abbreviations: CHX = chlorhexidine; NaHCO₃ = sodium bicarbonate; MV = mechanical ventilation; RCT = randomized controlled trial; NS = not statistically significant; LOS = length of stay; MDRO = multidrug-resistant organism; RR = relative risk. *DeRiso et al. (1996) included for historic context despite being pre‑2005. |
||||||

Figure 3: Scatter plot of mechanical ventilation duration vs ICU length of stay, by group and VAP status, with literature reference points
4. DISCUSSION
This prospective, randomized controlled investigation explored the impact of a dual-agent oral care strategy—2% chlorhexidine followed by 1% sodium bicarbonate—compared with chlorhexidine alone in mechanically ventilated adults with chronic kidney disease (CKD) admitted to the ICU. The trial was designed not only to measure ventilator-associated pneumonia (VAP) incidence as the primary outcome, but also to assess intermediary mechanisms, evaluate microbiological patterns, and examine patient‑focused outcomes such as ventilation duration, ICU stay, and mortality.
A major strength of the study was the equipoise between the two arms at baseline. Demographic profile, CKD stage, comorbidity burden, illness severity (APACHE II score), ICU admission category, type of ventilation, and time from admission to intubation were well matched. This minimizes the likelihood that variability in patient risk or underlying health status contributed to the differences seen, increasing confidence that outcome disparities can be attributed to the intervention itself.12,13
The oral pH data provide mechanistic insight into how the intervention may have conferred benefit. Both groups started with near‑neutral oral pH, but the sodium bicarbonate arm showed a steady and significant shift toward alkalinity from the third day onwards. A higher proportion of these patients achieved pH levels above 7.4. This is consistent with established microbiological principles, because an alkaline environment is less hospitable to aciduric and many Gram‑negative organisms linked to VAP pathogenesis. However, the reduction in heavy oropharyngeal bacterial colonization, while numerically greater in the intervention group at each time point, did not reach statistical significance. This supports the view from prior studies that sodium bicarbonate’s primary contribution lies in modifying the oral milieu and biofilm conditions rather than exerting a direct bactericidal effect.14,15 The most clinically important outcome was the halving of overall VAP incidence in the combined‑care arm, with an absolute risk reduction of 16% and a relative risk of 0.50. The benefit appeared most marked for early‑onset VAP, suggesting that altering the oral environment in the initial days after intubation may interrupt critical steps in pathogen ascent and aspiration. Survival analysis further demonstrated a delayed onset of VAP in the intervention arm, which is clinically valuable because even postponing infection can shorten ventilator exposure and reduce antibiotic requirements. These findings echo earlier trials on chlorhexidine and lend credence to the hypothesis that sodium bicarbonate’s alkalinizing action has an additive protective role.16
The microbial profile of confirmed VAP cases largely mirrored global ICU surveillance: Gram-negative bacilli, notably Pseudomonas aeruginosa and Klebsiella pneumonia, predominated, with MRSA representing the main Gram-positive pathogen. While the spectrum of organisms was comparable across groups, the proportion of multidrug-resistant isolates and carbapenem resistance was lower in the sodium bicarbonate arm. Furthermore, isolates from this group tended to be susceptible to a wider range of antibiotics, which may reflect reduced selection pressure in an environment less conducive to high-burden colonization by resistant strains.17,18 Improvements were also evident in patient‑centred measures. Those receiving combination oral care required fewer days of mechanical ventilation and had shorter ICU stays, with the greatest gains seen among participants who developed VAP. Ventilator‑free and ICU‑free days at 28 days were higher, underlining the functional and resource benefits of effective infection prevention. Mortality was numerically lower in the intervention arm, but the study was not powered to detect a survival difference.19,20
Multivariate regression confirmed that the intervention independently reduced the odds of developing VAP even after controlling for illness severity, intubation duration, and other covariates. The degree of alkalinisation achieved was also an independent protective factor. Conversely, higher APACHE II scores and longer ventilation were associated with increased risk, consistent with global literature.21,22
Comparative Analysis Table: Present Study vs. Key Chlorhexidine Trials (2005–2015)
This table directly compares the present RCT (CHX + sodium bicarbonate in CKD ICU patients) with major published studies (2005–2015) of chlorhexidine for ventilator-associated pneumonia (VAP) prevention, highlighting differences in study population, interventions, outcomes, and innovation.
The findings have practical implications: adding sodium bicarbonate to chlorhexidine oral care appears to be a safe, inexpensive, and easily implemented strategy that could be particularly relevant in high‑risk populations such as patients with CKD. The benefits are not confined to surrogate markers—they extend to meaningful clinical endpoints.23,24 Nevertheless, certain limitations must be acknowledged. This was a single‑centre study with a modest sample size, which may limit generalizability. Microbiological analyses were based on semi‑quantitative culture rather than advanced molecular methods, so subtle shifts in microbial community structure may have gone undetected. While hospital length of stay and mortality trends favoured the intervention arm, larger multicentre studies would be required to confirm these effects.
5. CONCLUSION
The results support incorporating sodium bicarbonate into standard chlorhexidine oral care protocols for ventilated CKD patients, and possibly for other ICU populations where early VAP prevention and resistance control are priorities. Further multicentre research should examine combination regimens, explore synergy with other oral hygiene techniques, and assess the long‑term impact on antimicrobial resistance patterns. Mechanistic studies employing molecular profiling could also clarify how oral alkalinisation influences pathogen ecology and infection risk.
6. Data availability
Numerical data generated during this study in available with the authors/
7. Conflicts of interest
All authors declare no conflict of interest.
8. Funding
The study was completed through institutional resources only. No external or industry funding was involved in the study.
Authors’ contribution
SS, AB, AKS, SL: Concept; conduct of the study
SK, APN: manuscript editing
9. REFERENCES
- Koeman M, van der Ven AJ, Hak E, Joore HC, Kaasjager K, de Smet AG, et al. Oral decontamination with chlorhexidine reduces the incidence of ventilator-associated pneumonia. Am J Respir Crit Care Med.;173(12):1348-55. [PubMed] DOI: 1164/rccm.200505-820OC
- Fourrier F, Cau-Pottier E, Boutigny H, Roussel-Delvallez M, Jourdain M, Chopin C. Effects of dental plaque antiseptic decontamination on bacterial colonization and nosocomial infections in critically ill patients. Intensive Care Med. 2000;26(9):1239-47. [PubMed] DOI: 1007/s001340000585
- DeRiso AJ, Ladowski JS, Dillon TA, Justice JW, Peterson AC. Chlorhexidine gluconate 0.12% oral rinse reduces the incidence of total nosocomial respiratory infection and nonprophylactic systemic antibiotic use in patients undergoing heart surgery. Chest.;109(6):1556-61. [PubMed] DOI: 1378/chest.109.6.1556
- Scannapieco FA, Yu J, Raghavendran K, Vacanti A, Owens S, Wood K, Mylotte JM. A randomized controlled trial of chlorhexidine gluconate on oral bacterial pathogens in mechanically ventilated patients. Crit Care. 2009;13(4):R117. [PubMed] DOI: 1186/cc7967
- Klompas M, Speck K, Howell MD, Greene LR, Berenholtz SM. Reappraisal of routine oral care with chlorhexidine gluconate for patients receiving mechanical ventilation: systematic review and meta-analysis. JAMA Intern Med. 2014;174(5):751-61. [PubMed] DOI: 1001/jamainternmed.2014.359
- Hua F, Xie H, Worthington HV, Furness S, Zhang Q, Li C, Shi L. Oral hygiene care for critically ill patients to prevent ventilator-associated pneumonia. Cochrane Database Syst Rev. 2016;2016(10):CD008367. [PubMed] DOI: 1002/14651858.CD008367.pub3
- Chandel S, Khan MA, Singh N, Agrawal A, Khare V. The effect of sodium bicarbonate oral rinse on salivary pH and oral microflora: A prospective cohort study. Natl J Maxillofac Surg. 2017;8(2):106-109. [PubMed] DOI: 4103/njms.NJMS_36_17
- Bescos R, Ashworth A, Cutler C, Brookes ZL, Belfield LA, Rodríguez-Lopez J, et al. Effects of chlorhexidine mouthwash on the oral microbiome. Sci Rep. 2020;10(1):5254. [PubMed] DOI: 1038/s41598-020-61912-4
- Shi Z, Xie H,ript Wang P, Zhang Q, Wu Y, Chen E, Ng L, Worthington HV, Needleman I, Furness S. Oral hygiene care for critically ill patients to prevent ventilator-associated pneumonia. Cochrane Database Syst Rev. 2013;(8):CD008367. [PubMed] DOI: 1002/14651858.CD008367.pub2
- Labeau SO, Van de Vyver K, Brusselaers N, Vogelaers D, Blot SI. Prevention of ventilator-associated pneumonia with oral antiseptics: a systematic review and meta-analysis. Lancet Infect Dis. 2011;11(11):845-54. [PubMed] DOI: 1016/S1473-3099(11)70127-X
- Pant A, Prasai A, Rauniyar AK, Adhikary L, Basnet K, Khadka T. Pneumonia in Patients with Chronic Kidney Disease Admitted to Nephrology Department of a Tertiary Care Center: A Descriptive Cross-sectional Study. JNMA J Nepal Med Assoc. 2021;59(242):1000-1003. [PubMed] DOI: 31729/jnma.7074
- Xu H, Gasparini A, Ishigami J, Matsushita K, et al. eGFR and the risk of community-acquired infections. Clin J Am Soc Nephrol. 2017;12(9):1399-1408. [PubMed] DOI: 2215/CJN.00250117
- McDonald HI, Thomas SL, Nitsch D. Chronic kidney disease as a risk factor for acute community-acquired infections in high-income countries: a systematic review. BMJ Open. 2014;4(4):e004100. [PubMed] DOI: 1136/bmjopen-2013-004100
- Calfee DP. Multidrug-resistant organisms within the dialysis population: a potentially preventable perfect storm. Am J Kidney Dis. 2015;65(1):3-5. [PubMed] DOI: 1053/j.ajkd.2014.10.003
- Safarabadi M, Ghaznavi-Rad E, Pakniyat A, Rezaie K, Jadidi A. Comparing the Effect of Echinacea and Chlorhexidine Mouthwash on the Microbial Flora of Intubated Patients Admitted to the Intensive Care Unit. Iran J Nurs Midwifery Res. 2017;22(6):481-485. [PubMed] DOI: 4103/ijnmr.IJNMR_92_16
- La Combe B, Mahérault AC, Messika J, Billard-Pomares T, Branger C, Landraud L, et al. Oropharyngeal Bacterial Colonization after Chlorhexidine Mouthwash in Mechanically Ventilated Critically Ill Patients. Anesthesiology. 2018;129(6):1140-1148. [PubMed] DOI:1097/ALN.0000000000002451
- Choi S, Kim H. Sodium Bicarbonate Solution versus Chlorhexidine Mouthwash in Oral Care of Acute Leukemia Patients Undergoing Induction Chemotherapy: A Randomized Controlled Trial. Asian Nurs Res (Korean Soc Nurs Sci). 2012;6(2):60-66. [PubMed] DOI: 1016/j.anr.2012.05.004
- Berry AM, Davidson PM, Masters J, Rolls K, Ollerton R. Effects of three approaches to standardized oral hygiene to reduce bacterial colonization and ventilator-associated pneumonia in mechanically ventilated patients: a randomized control trial. Int J Nurs Stud. 2011;48(6):681-8. [PubMed] DOI: 1016/j.ijnurstu.2010.11.004
- Munro CL, Grap MJ, Elswick RK, McKinney J, Sessler CN, Hummel R. Oral health status and development of ventilator-associated pneumonia: A descriptive study. Am J Crit Care. 2006;15(5):453-60. [PubMed]
- Tablan OC, Anderson LJ, Besser R, Bridges C, Hajjeh R; CDC; Healthcare Infection Control Practices Advisory Committee. Guidelines for preventing health-care--associated pneumonia, 2003: recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee. MMWR Recomm Rep. 2004 53(RR-3):1-36. [PubMed]
- Kollef MH, Chastre J, Fagon JY, et al. Ventilator-associated pneumonia. J Crit Care. 2008;23(3):404-410. [PubMed] DOI: 1164/ajrccm.165.7.2105078
- Klompas M, Sreedhara S. Ventilator-associated pneumonia: diagnosis, treatment, and prevention. Clin Chest Med. 2017;38(1):139-150. [PubMed] DOI: 1128/CMR.00051-05
- Labeau SO, Vereecke A, Vandijck DM, et al. Critical care nurses' knowledge, attitudes, and compliance with oral care guidelines in Belgium. Intensive Care Med. 2008;34(6):1140-6. [PubMed]
- Shi Z, Xie H, Wang P, Zhang Q, Wu Y, Chen E, Ng L, Worthington HV, Needleman I, Furness S. Oral hygiene care for critically ill patients to prevent ventilator-associated pneumonia. Cochrane Database Syst Rev. 2013;(8):CD008367. [PubMed] DOI: 1002/14651858.CD008367.pub2