Ashfaq Ahmad Shah Bukhari 1 , Durga Devi 2 , Madeeha Minhas 3 , Aqsa Noureen 4 , Humera Javed 5 , Syed Imtiaz Ali Zaidi 6 , Haseeb Khaliq 7 , Muhammad Arsalan Shah 8
Author affiliations:
Correspondence: Muhammad Arsalan Shah; Email: muhammadarsalanshahpath01@gmail.com; Phone: +92 335 4965512
Background & objective: Severe dengue is a global health concern and immune dysregulation is a key contributor to its pathogenesis. The aim of this schematic review was to review the association of the cytokine biomarkers mainly interleukin-6 (IL-6), interleukin-10 (IL-10) and tumor necrosis factor-alpha (TNF-α) in immune dysregulation among patients suffering from severe dengue.
Methodology: The review followed PRISMA 2020 guidelines. The literature was searched from PubMed, Scopus, Web of Science, and Google Scholar up to August 2025. Qualified studies included those which measured IL-6, IL-10 or TNF- α in confirmed dengue patient. Meta-analysis was performed to assess standardized mean differences (SMDs) for key cytokines. The risk of bias was determined using Newcastle-Ottawa Scale and the GRADE approach was applied for certainty of evidence.
Results: Higher levels were observed, of IL-6 and IL-10 in severe dengue, and TNF-α with late complications in this study. Meta-analysis correlated high levels of cytokines with severe dengue IL-6 (SMD = 16.79, 95% CI = -15.25-48.84), IL-10 (SMD = 0.13, 95%CI = -0.37-0.63) and TNF-α (SMD = 0.53, 95% CI = 0.25-0.82). The evidence confidence was moderate and high by IL-6 and IL-10 and high in TNF-α respectively.
Conclusion: The levels of IL-6 and IL-10 were significantly high in severe dengue cases, but heterogeneity across studies reduced the prognostic integrity. TNF-α emerged as a potent indicator of mild and severe dengue. To establish the clinical uses of these biomarkers as a risk-stratifying tool, standardized, multicentric studies are needed to validate these findings.
Abbreviations: DF: dengue fever, DHF: dengue hemorrhagic fever, DSS: dengue shock syndrome, IL-6: Interleukin-6, IL-10: Interleukin-10, SMD: standardized mean differences, TNF-α: tumor necrosis factor-alpha
Keywords: Dengue Hemorrhagic Fever (DHF), Cytokine Storm, Biomarkers, Cytokines, IL-6, IL-10, TNF-α
Citation: Bukhari AAS, Devi D, Minhas M, Noureen A, Javed H, Zaidi SIA, Khaliq H. Novel insights into immune dysregulation and cytokine signatures in intensive care settings: bridging IL-6, IL-10, TNF-α to endothelial, hepatic, and hematological dysfunction in severe dengue, a schematic meta-analysis. Anaesth. pain intensive care 2025;29(8):1022-1031. DOI: 10.35975/apic.v29i8.3039
Received: October 16, 2025; Revised: October 18, 2025; Accepted: October 18, 2025
Dengue fever is a viral infection transmitted through the bite of a female mosquito of Aedes specie.1 Among millions of infections reported each year, most of the dengue cases are related to mild or asymptomatic dengue fever (DF) whereas, some of these cases would develop severe dengue, which includes dengue hemorrhagic fever (DHF) and dengue shock syndrome (DSS) that had high mortality rates.2 The burden of severe dengue remains a significant public health issue despite the development of surveillance and supportive therapy.3
Pathophysiologically, the worsening of mild to severe dengue was explained by the immune dysregulation that is characterized by a sudden release of cytokines and also known as cytokine storm.4 Cytokine storm is an unregulated immune response to an infection which leads to release of non-specific cytokines and chemokines, causing severe clinical implications like plasma leakage and shock syndrome.5
Interleukin-6 (IL-6) is a multifunctional cytokine whose secretion is predominantly elicited by the macrophages, endothelial cells, and fibroblasts, which is involved in acute-phase response, B-cell differentiation, and fever induction during infection pathogenesis. Interleukin-10 (IL-10) is an anti-inflammatory cytokine secreted mainly by regulatory T cells and monocytes which inhibits pro-inflammatory signaling to inhibit too much immune activation. Activated macrophages and T lymphocytes are the primary sources of tumor necrosis factor-alpha (TNF-a), which facilitates inflammation, vascular permeability and apoptotic signaling that causes tissue damage in acute viral infections. Elevated levels of interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), and interleukin-10 (IL-10) are associated with plasma leakage, vascular damage, and coagulopathy.6
Due to their higher levels in infected patients, these cytokines were considered as potential biomarkers for early detection and management of dengue cases.7 However, cytokine expression profiles among populations varied significantly depending on viral serotypes and primary versus secondary infections compromising the reliability of these markers as predictors of severity.8
Prior reviews were informative in terms of cytokine involvement in dengue severity, but these were scattered and had disparate study designs, as well as lacked synthesis that had been updated by more recent prospective evidence.9 There was a lack of systematic assessment of cytokine profiles that would help elucidate their prognostic capacity, and assist in evidence-based risk stratification.10
In this schematic review, latest findings on cytokines levels and their pathophysiological role in dengue confirmed patients was summarized. The aim of this review was to establish the association of the cytokine biomarkers mainly interleukin-6 (IL-6), interleukin-10 (IL-10) and tumor necrosis factor-alpha (TNF-α) in immune dysregulation among severe dengue cases.
This systematic review and meta-analysis were conducted according to the PRISMA 2020 guidelines.11 The purpose was to test the relationship between circulating cytokines (IL-6, IL-10, and TNF- α) and severe dengue.
The inclusion criteria involved human clinical studies investigating the role of IL-6, IL-10, or TNF-α in laboratory confirmed dengue patients. Original research articles in English with reported outcomes as numerical data on cytokines were then selected. Exclusion criteria included a collection of animal or in vitro studies, reviews, case reports, conference abstracts or studies that did not include quantitative data on cytokine and studies without full-text access.
A well-planned literature search was conducted using different electronic databases such as PubMed, Scopus, Web of Science, and Google Scholar databases for the entries from 2020 to August 2025. The Boolean operators were placed between the search terms of “dengue”, “dengue virus”, “cytokine storm”, “immune response”, “IL-6”, “IL-10”, “TNF- α”, and “biomarkers”. The articles were selected and reference lists were searched manually to determine other qualified studies. Two reviewers screened the titles, abstracts, and the full documents against the inclusion criteria. Differences were sorted out through discussion with a third reviewer.
The data from the selected studies was extracted using standardized form including characteristics of study, sample size, demographics, cytokines assayed, research techniques, statistical outcomes and key findings in terms of pathophysiological assessments. Where available, missing values were estimated based on the available summary statistics, no imputation was done based on data which was fully missing. Data in visual form was displayed using the forest plots.
The risk of bias within the selection, comparability, and outcome areas was determined using the Newcastle-Ottawa Scale (NOS). In case of missing data, the risk of bias was evaluated qualitatively. The GRADE approach was used to evaluate the certainty of evidence concerning each cytokine. The standard of evidence was categorized as high, medium, low, or very low.
For homogenous data, meta-analysis was performed to evaluate the levels of IL-6, IL-10 and TNF-α in severe dengue patients as compared to mild dengue fever patients or healthy individuals (DF/HI) by means of a random-effects analysis.
The primary outcomes of continuous results were reported in standardized mean differences (SMDs) with 95% (CI) by comparing the cytokine levels in DF/HI, w.r.t the data provided in included articles, and severe dengue patients.
The statistical analysis and forest plots were generated in MetaAnalysisOnline Tool.12 The mean and standard deviation were calculated using standard protocols where researchers had reported data in the form of medians and interquartile ranges. As a measure of heterogeneity, the I2 value was used and considered as low (> 50), moderate (50-75) and high (>75) heterogeneity. In study design, populations, or measurement methods that could not be statistically pooled because of heterogeneity, a narrative synthesis was offered in a more structured format. Cytokines were classified into pro-inflammatory (IL-6, TNF-α); and anti-inflammatory (IL-10), with expression of cytokines correlated with clinical outcome. The sensitivity analysis and subgroup analysis were performed for eligible studies. The articles included 7 prospective, 4 cross-sectionals and 1 retrospective study.13-24
Among the four searched electronic databases and other sources, 222 research articles were initially selected. The number was reduced to 92 records after removing the duplicates. Title and abstract screening further eliminated 30 studies. From the remaining 62 articles, 22 were removed due to unavailability of access to the full-texts. Further articles (28) were eliminated due to lack of stratified cytokine data and studies including animals, in vitro findings, reviews, case reports, or languages other than English. Ultimately, twelve studies that passed the inclusion criteria were included in this schematic review. The PRISMA flow diagram presented in Figure 1 illustrates the selection process.
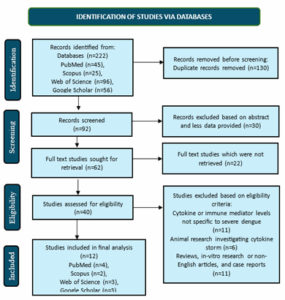
Figure 1: PRISMA Flow Diagram for Study Selection.
The systematic review included 12 human studies, reported in the last five years, containing data from 1934 individuals (including HI, DF and severe dengue patients) from eight countries including cross-sectional, prospective and retrospective cohort studies. All studies used laboratory-confirmed dengue cases and measured levels of cytokines (IL-6, IL-10, and TNF-α) primarily by enzyme-linked immunosorbent assay (ELISA) with majority of the samples assessed at the acute or critically ill stages. Table 1 presents the nature and the outcomes of the chosen studies in evaluating the profile of cytokines during severe dengue.
In all twelve studies, IL-6 levels were significantly higher in patients with severe dengue as compared to mild dengue fever (DF). Multiple studies revealed substantially greater levels of IL-6 in severe dengue, frequently accompanied by vascular leakage, hypovolemic shock, or liver complications. Temporal analyses proved that the IL-6 levels were higher at an earlier stage and persists in advance stages of infection, implying that IL-6 was a probable early predictive biomarker. Nevertheless, absolute values varied across inter-studies as evidence of methodological heterogeneity, especially regarding assay platforms and measurements timings. The pattern of IL-10 varied more across studies. Although a number of studies reported relatively greater levels of IL-10 in case of severe dengue, the rest reported comparably less and some reported insignificant difference, when contrasting dengue severe cases with DF/HI groups.
Kinetic models showed high levels of IL-10 in severe dengue, and its positive correlation with viraemia and prolonged fever. These results pointed to a dual action of IL-10 as both an anti-inflammatory regulator as well as a deregulator of the immune system. In the case of TNF-α, low heterogeneity was observed. It was found to be linked with severe dengue especially vascular leakage and liver damage, with limited predictive power because of its delayed expression. TNF-α used as a biomarker, pooled prevalence, seemed to be more useful as an indicator of disease progression, and not as a predictor of early disease severity.
The meta-analysis was implemented to compare the pooled effect estimates of the IL-6, IL-10, and TNF-α, in comparing DF/HI and severe dengue. For quantitative analysis, two studies with data reported in units other than pg/mL and where data was not sorted on the basis of disease severity were excluded. In the case of IL-6, eight studies involving a total of 981 participants were utilized. The pooled analysis showed a significant difference between the IL-6 levels in severe and DF/HI (SMD = 16.79, 95% CI [-15.25; 48.84], p = 0.3043). The heterogeneity was quite high (I² = 95.9%) attributable to a certain variability in studies because of the variation in assay procedures and measuring cytokines. The forest plot in Figure 2 illustrated a consistent direction of effect across most studies.

Figure 2: Forest plot comparing serum IL-6 levels between severe dengue and dengue fever (DF) or hemorrhagic illness (HI) patients. Standardized mean differences (SMDs) with 95% confidence intervals were calculated using a random-effects model (MetaAnalysisOnline Tool).
In the case of IL-10, data was utilized from seven studies (n = 911). The meta-analysis had demonstrated robust increase of IL-10 levels in severe dengue cases compared with DF/HI (SMD = 0.13, 95% CI [-0.37; 0.63], p > 0.5). There was significant heterogeneity (I² =84.9%) because the population cohorts were heterogeneous, and there was variation between methods. The confidence intervals were overlapping as indicated by the forest plot (Figure 3) with only a few studies revealing a strong relationship between IL-10 and the severity of the disease.

Figure 3: Forest plot of IL-10 levels in dengue fever (DF) or hemorrhagic illness (HI) patients versus Severe Dengue. Standardized mean differences (SMDs) with 95% confidence intervals were calculated using a random-effects model (MetaAnalysisOnline Tool)
For TNF-α, five studies (n = 461) contributed to data analysis. The positive correlation between higher levels of TNF-α, and disease severity was observed through pooled effect analysis (SMD = 0.53, 95% CI [0.25 0.82] = 0.0002). Heterogeneity was low (I2 = 49.9%), demonstrating the enhanced the credibility of the result. The forest plot (Figure 4) represented that TNF-α, being one of the most consistent immunological markers of disease severity, had narrow confidence intervals and strong agreement across the studies.

Figure 4: Forest plot of TNF-α levels in dengue fever (DF) or hemorrhagic illness (HI) patients versus Severe Dengue. Standardized mean differences (SMDs) with 95% confidence intervals were calculated using a random-effects model (MetaAnalysisOnline Tool).
In general, the quantitative synthesis showed the roles of IL-6, IL-10 and TNF-α to be steady biomarkers of dengue severity, with TNF-α exhibited consistent associations across locales. Such results were consistent with the narrative synthesis and supported the hypothesis that dysregulated pro- and anti -inflammatory cytokine played a pivotal role in dengue pathophysiology.
Subgroup analysis of the three cytokines showed different patterns in effect size and consistency when compared. While IL-6 had the largest pooled effect size (SMD = 16.79, 95% CI [−15.25; 48.84]), it also had very high heterogeneity (I² = 95.9%), which reflected significant variation in assay techniques and sampling time points across studies. IL-10, on the other hand, showed a small and statistically non-significant pooled effect size (SMD = 0.13, 95% CI [−0.37; 0.63]) along with equally high heterogeneity (I² = 84.9%), indicating that its discriminatory value varied depending on the setting. In contrast, TNF-α demonstrated a moderately significant pooled effect size (SMD = 0.53, 95% CI [0.25–0.82]) and less heterogeneity (I² = 49.9%), suggesting more consistent relationships between studies.
Overall, the subgroup comparison revealed that although IL-6 was the most elevated biomarker in severe dengue, higher heterogeneity questioned its reliability. Similarly, IL-10 also displayed poor consistency and heterogeneity among the study groups undermining its reproducibility. TNF-α, despite having a more moderate effect size, was the most reliable and repeatable biomarker across cohorts.
Table 2 presented sensitivity analyses to evaluate the strength of the pooled estimates. In the case of IL-6, the removal of a single study with the greatest variance resulted in significant reduction I2 value from 95.9% to 36.1% whereas the overall effect size remained consistent, suggesting the stability of the association. Likewise, in the case of IL-10, eliminating one high-variance study decreased the heterogeneity of the effect from 84.9% to 51.9%, without changing the direction of the effect or its magnitude. In the case of TNF-α, removal of one study had heterogeneity reduced from 49.9% to 0%, without changing the effect estimate. The results established that pooled results were not single study dependent and demonstrated robustness across cytokine analyses.
The Newcastle- Ottawa Scale had been used to determine the risk of bias and majority of the studies had been rated as moderate risk to low risk. The included studies had methodological diversity, which led to the heterogeneity of involving variables in syntheses. Moreover, the missing or selective disclosure of results were also observed which could influence the overall confidence of findings. The outcomes of risk of bias of included studies were listed in Table 3. The certainty of evidence was moderate for IL-6, IL-10 but high for TNF-α across the studies, as rated by GRADE assessment. Overall, the findings supported the potential roles of IL-6, and IL-10 as predictive biomarkers of severe dengue whereas, TNF-α was found as a predictor of disease progression.
This systematic review examined the relation between three main cytokines, namely, IL-6, IL-10, and TNF-α and severe cases of dengue in 12 human studies carried out between 2020 and 2025. The synthesis of both the narrative and quantitative analyses supported the conclusion that dysregulated expression of cytokines was highly associated with disease progression. The results highlighted the reliability of IL-6 and IL-10 as the most effective predictors of severity, whereas TNF-α was stronger in proving progression and complications 25. The combined analyses revealed that IL 6 levels were very high in severe dengue than in DF/HI which was in consonance with the narrative synthesis and previous research that report that IL-6 was a contributor of vascular leakage, shock, and hepatic involvement.26 Nevertheless, the pooled effect estimate was not very accurate because of high heterogeneity in the studies, mostly due to differences in assays, and inconsistent timing when samples were collected. IL-10 showed a significant trend of increase in severe dengue, but due to inter-study heterogeneity the meta-analysis showed non-significant, pooled effect.27 The synthesis proposed that IL-10 was both an anti-inflammatory mediator and a mediator of immune dysregulation, consistent with previous data of dual IL-10 pathogenicity in virus pathogenesis.28 TNF-α results were more consistent, with the meta-analysis showing particular statistically significant increase and low heterogeneity levels.29 These findings suggested that TNF-α was a reliable predictor of progression, especially with regard to vascular and hepatic complications.30
The study heterogeneity was further analyzed through subgroup analysis.31 Strong associations were observed with IL-6 in prospective cohort studies and cohorts of adults only, indicating that the dynamics of biomarkers were dependent on the study design and the age of hosts.2 The variability in IL-10 was greatest among regions and assay methods, which suggested that standardization of laboratory protocols might improve the comparability in the study outcomes.33 Subgroup factors had lower effects on TNF-α, which increases TNF-α reliability. Collectively, these findings endorse the use of IL-6 as an early predictive marker, IL-10 as a regulator and TNF-α as a progression marker in dengue infection 34. Sensitivity analyses indicated that the quantitative findings were strong and not caused by outlier researches. Excluding high-variance studies significantly lowered the heterogeneity across all cytokines (IL-6: 95.9% to 36.1%; IL-10: 84.9% to 51.9%; TNF-α: 49.9% to 0%) but did not alter the effect sizes, increasing confidence in the pooled relationships.35
According to Newcastle–Ottawa Scale, risk of bias for majority of the studies was moderate to high among the selected studies. The systematic implementation of the GRADE tool enabled a balanced understanding of certainty. The evidence of IL-6 and IL-10 was rated as moderate, which suggested heterogeneity and imprecision whereas, for TNF-α evidence was rated high due to consistency and high precision of evidence.36
Cytokine biomarkers might be used to detect severe dengue at early stages, thus preventing mortality, but the biomarkers had a number of issues that limit their use within clinical practice.37 Despite the lower heterogeneity among the included studies, in this review, the limitations persisted. Inconsistent severity definitions (WHO 1997 vs WHO 2009) and non-uniform sample sizes at the study level caused uncertainty and also the variation in the profiles of infections (primary versus secondary dengue).38,39 Another critical source of variability was assay heterogeneity, in particular between ELISA and multiplex platforms. Even at the review level, some publication bias could not be ruled out as there were chances that null results might be under-reported.
For future direction, emphasis must be placed on the need to involve large, multicentric, and standardized cohort studies that combine the profiles of cytokines with clinical and virological measures. Assay method harmonization and harmonization of sampling time points would further go a long way in reducing heterogeneity and enhance the reliability of pooled estimates. Moreover, longitudinal cytokine profiling had potential benefits in the ability to better capture dynamic immune responses and guide early-warning models. Since IL-6 prediction had been observed and TNF-α results were consistent, introducing IL-6 and TNF- in clinical risk stratification models can be an opportunity to move towards customized management of dengue as well as other viral diseases.40
In conclusion, this meta-analysis and systematic review presented overall evidence that IL-6, IL-10, as well as TNF-α are the main contributors immunopathogenesis in severe dengue. IL-6 and IL-10 were reliable markers of disease severity with methodological inconsistency, whereas TNF-α was a consistent predictor of disease progression. Despite these disparities in the definite nature of evidence, these results indicated that cytokine biomarkers continued to be actively considered as potential indicator of disease diagnosis and treatment at the onset of severe dengue effects.
6. Data availability
The numerical data generated during this research is available with the authors.
7. Conflict of interest
All authors declare that there was no conflict of interest.
8. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
9. Authors’ contribution
AASB and DD: conceptualized and designed the meta-analysis.
MM, AN and HK: performed data extraction and statistical analysis.
AASB, AN, HJ and SIAZ: contributed to literature screening and quality assessment.
DD, MM, AN, HK: created visualizations and forest plots.
AASB, MH: supervised methodology, verified data accuracy, and revised the manuscript critically.
All authors approved the final manuscript and agree to be accountable for its content.
Author affiliations:
- Ashfaq Ahmad Shah Bukhari, Department of Physiology, RAK College of Medical Sciences, RAK Medical and Health Sciences University, Ras Al Khaimah, UAE; Email: dr.ashfaqbukhari@hotmail.com
- Durga Devi, Department of Pathology, Bilawal Medical College, Liaquat University of Medical & Health Sciences (LUMHS), Jamshoro, Sindh, Pakistan; Email: durga.devi.@lumhs.edu.pk
- Madeeha Minhas, Department of Pathology, College of Science & Health professions, King Saud bin Abdulaziz University for Health Sciences, Jeddah, Saudi Arabia; Email: dr.minhasm@gmail.com
- Aqsa Noureen, Department of Pathology, HBS Medical & Dental College, Islamabad; Pakistan; Email: dr.aqsanoureen@gmail.com
- Humera Javed, Department of Pathology, HBS Medical & Dental College, Islamabad; Pakistan; Email: humerajavedhbs.@gmail.com
- Syed Imtiaz Ali Zaidi, Department of Anesthesiology & Intensive Care, Avicenna Medical College & Hospital, Lahore, Pakistan; Email:
- Haseeb Khaliq, Department of Pathology, University of Health Sciences, Lahore / IMPRS Berlin, Germany; Email:
Correspondence: Muhammad Arsalan Shah; Email: muhammadarsalanshahpath01@gmail.com; Phone: +92 335 4965512
ABSTRACT
Background & objective: Severe dengue is a global health concern and immune dysregulation is a key contributor to its pathogenesis. The aim of this schematic review was to review the association of the cytokine biomarkers mainly interleukin-6 (IL-6), interleukin-10 (IL-10) and tumor necrosis factor-alpha (TNF-α) in immune dysregulation among patients suffering from severe dengue.
Methodology: The review followed PRISMA 2020 guidelines. The literature was searched from PubMed, Scopus, Web of Science, and Google Scholar up to August 2025. Qualified studies included those which measured IL-6, IL-10 or TNF- α in confirmed dengue patient. Meta-analysis was performed to assess standardized mean differences (SMDs) for key cytokines. The risk of bias was determined using Newcastle-Ottawa Scale and the GRADE approach was applied for certainty of evidence.
Results: Higher levels were observed, of IL-6 and IL-10 in severe dengue, and TNF-α with late complications in this study. Meta-analysis correlated high levels of cytokines with severe dengue IL-6 (SMD = 16.79, 95% CI = -15.25-48.84), IL-10 (SMD = 0.13, 95%CI = -0.37-0.63) and TNF-α (SMD = 0.53, 95% CI = 0.25-0.82). The evidence confidence was moderate and high by IL-6 and IL-10 and high in TNF-α respectively.
Conclusion: The levels of IL-6 and IL-10 were significantly high in severe dengue cases, but heterogeneity across studies reduced the prognostic integrity. TNF-α emerged as a potent indicator of mild and severe dengue. To establish the clinical uses of these biomarkers as a risk-stratifying tool, standardized, multicentric studies are needed to validate these findings.
Abbreviations: DF: dengue fever, DHF: dengue hemorrhagic fever, DSS: dengue shock syndrome, IL-6: Interleukin-6, IL-10: Interleukin-10, SMD: standardized mean differences, TNF-α: tumor necrosis factor-alpha
Keywords: Dengue Hemorrhagic Fever (DHF), Cytokine Storm, Biomarkers, Cytokines, IL-6, IL-10, TNF-α
Citation: Bukhari AAS, Devi D, Minhas M, Noureen A, Javed H, Zaidi SIA, Khaliq H. Novel insights into immune dysregulation and cytokine signatures in intensive care settings: bridging IL-6, IL-10, TNF-α to endothelial, hepatic, and hematological dysfunction in severe dengue, a schematic meta-analysis. Anaesth. pain intensive care 2025;29(8):1022-1031. DOI: 10.35975/apic.v29i8.3039
Received: October 16, 2025; Revised: October 18, 2025; Accepted: October 18, 2025
1. INTRODUCTION
Dengue fever is a viral infection transmitted through the bite of a female mosquito of Aedes specie.1 Among millions of infections reported each year, most of the dengue cases are related to mild or asymptomatic dengue fever (DF) whereas, some of these cases would develop severe dengue, which includes dengue hemorrhagic fever (DHF) and dengue shock syndrome (DSS) that had high mortality rates.2 The burden of severe dengue remains a significant public health issue despite the development of surveillance and supportive therapy.3
Pathophysiologically, the worsening of mild to severe dengue was explained by the immune dysregulation that is characterized by a sudden release of cytokines and also known as cytokine storm.4 Cytokine storm is an unregulated immune response to an infection which leads to release of non-specific cytokines and chemokines, causing severe clinical implications like plasma leakage and shock syndrome.5
Interleukin-6 (IL-6) is a multifunctional cytokine whose secretion is predominantly elicited by the macrophages, endothelial cells, and fibroblasts, which is involved in acute-phase response, B-cell differentiation, and fever induction during infection pathogenesis. Interleukin-10 (IL-10) is an anti-inflammatory cytokine secreted mainly by regulatory T cells and monocytes which inhibits pro-inflammatory signaling to inhibit too much immune activation. Activated macrophages and T lymphocytes are the primary sources of tumor necrosis factor-alpha (TNF-a), which facilitates inflammation, vascular permeability and apoptotic signaling that causes tissue damage in acute viral infections. Elevated levels of interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), and interleukin-10 (IL-10) are associated with plasma leakage, vascular damage, and coagulopathy.6
Due to their higher levels in infected patients, these cytokines were considered as potential biomarkers for early detection and management of dengue cases.7 However, cytokine expression profiles among populations varied significantly depending on viral serotypes and primary versus secondary infections compromising the reliability of these markers as predictors of severity.8
Prior reviews were informative in terms of cytokine involvement in dengue severity, but these were scattered and had disparate study designs, as well as lacked synthesis that had been updated by more recent prospective evidence.9 There was a lack of systematic assessment of cytokine profiles that would help elucidate their prognostic capacity, and assist in evidence-based risk stratification.10
In this schematic review, latest findings on cytokines levels and their pathophysiological role in dengue confirmed patients was summarized. The aim of this review was to establish the association of the cytokine biomarkers mainly interleukin-6 (IL-6), interleukin-10 (IL-10) and tumor necrosis factor-alpha (TNF-α) in immune dysregulation among severe dengue cases.
2. METHODOLOGY
This systematic review and meta-analysis were conducted according to the PRISMA 2020 guidelines.11 The purpose was to test the relationship between circulating cytokines (IL-6, IL-10, and TNF- α) and severe dengue.
The inclusion criteria involved human clinical studies investigating the role of IL-6, IL-10, or TNF-α in laboratory confirmed dengue patients. Original research articles in English with reported outcomes as numerical data on cytokines were then selected. Exclusion criteria included a collection of animal or in vitro studies, reviews, case reports, conference abstracts or studies that did not include quantitative data on cytokine and studies without full-text access.
A well-planned literature search was conducted using different electronic databases such as PubMed, Scopus, Web of Science, and Google Scholar databases for the entries from 2020 to August 2025. The Boolean operators were placed between the search terms of “dengue”, “dengue virus”, “cytokine storm”, “immune response”, “IL-6”, “IL-10”, “TNF- α”, and “biomarkers”. The articles were selected and reference lists were searched manually to determine other qualified studies. Two reviewers screened the titles, abstracts, and the full documents against the inclusion criteria. Differences were sorted out through discussion with a third reviewer.
The data from the selected studies was extracted using standardized form including characteristics of study, sample size, demographics, cytokines assayed, research techniques, statistical outcomes and key findings in terms of pathophysiological assessments. Where available, missing values were estimated based on the available summary statistics, no imputation was done based on data which was fully missing. Data in visual form was displayed using the forest plots.
The risk of bias within the selection, comparability, and outcome areas was determined using the Newcastle-Ottawa Scale (NOS). In case of missing data, the risk of bias was evaluated qualitatively. The GRADE approach was used to evaluate the certainty of evidence concerning each cytokine. The standard of evidence was categorized as high, medium, low, or very low.
For homogenous data, meta-analysis was performed to evaluate the levels of IL-6, IL-10 and TNF-α in severe dengue patients as compared to mild dengue fever patients or healthy individuals (DF/HI) by means of a random-effects analysis.
The primary outcomes of continuous results were reported in standardized mean differences (SMDs) with 95% (CI) by comparing the cytokine levels in DF/HI, w.r.t the data provided in included articles, and severe dengue patients.
The statistical analysis and forest plots were generated in MetaAnalysisOnline Tool.12 The mean and standard deviation were calculated using standard protocols where researchers had reported data in the form of medians and interquartile ranges. As a measure of heterogeneity, the I2 value was used and considered as low (> 50), moderate (50-75) and high (>75) heterogeneity. In study design, populations, or measurement methods that could not be statistically pooled because of heterogeneity, a narrative synthesis was offered in a more structured format. Cytokines were classified into pro-inflammatory (IL-6, TNF-α); and anti-inflammatory (IL-10), with expression of cytokines correlated with clinical outcome. The sensitivity analysis and subgroup analysis were performed for eligible studies. The articles included 7 prospective, 4 cross-sectionals and 1 retrospective study.13-24
3. RESULTS
Among the four searched electronic databases and other sources, 222 research articles were initially selected. The number was reduced to 92 records after removing the duplicates. Title and abstract screening further eliminated 30 studies. From the remaining 62 articles, 22 were removed due to unavailability of access to the full-texts. Further articles (28) were eliminated due to lack of stratified cytokine data and studies including animals, in vitro findings, reviews, case reports, or languages other than English. Ultimately, twelve studies that passed the inclusion criteria were included in this schematic review. The PRISMA flow diagram presented in Figure 1 illustrates the selection process.

Figure 1: PRISMA Flow Diagram for Study Selection.
The systematic review included 12 human studies, reported in the last five years, containing data from 1934 individuals (including HI, DF and severe dengue patients) from eight countries including cross-sectional, prospective and retrospective cohort studies. All studies used laboratory-confirmed dengue cases and measured levels of cytokines (IL-6, IL-10, and TNF-α) primarily by enzyme-linked immunosorbent assay (ELISA) with majority of the samples assessed at the acute or critically ill stages. Table 1 presents the nature and the outcomes of the chosen studies in evaluating the profile of cytokines during severe dengue.
| Table 1: Characteristics of included studies and reported cytokine levels | |||||
| Study (Author, year, Country) | Study Design & Sample Size | Population Characteristics | Cytokines Measured (Method) | Interleukin levels pg/mL (DF/HI vs. SD) | Main Findings |
| Masyeni et al., 2024, Indonesia 13 | Cross-sectional, n=64 | Predominantly female, Age ≥18 years old |
IL-6, IL-10, TNF-α (ELISA) |
DF: IL-6 (129.79 ± 50.06), IL-10 (172.43 ± 83.37), TNF-α (126.41 ± 252.69) Severe Dengue: IL-6 (233.69 ± 111.27), IL-10 (172.43 ± 83.37), TNF-α (282.62 ± 230.22) |
IL-6 and TNF-α are significantly higher in severe dengue whereas, lower levels of IL-10 as compared to DF. Suggests immune dysregulation central to severity. |
| Prajapati et al., 2024, India 14 | Cross-sectional, n=125 | Mixed population; Age groups (0–20) years (20–40) years (40–60) years |
IL-6, IL-10, TNF-α (ELISA) |
DF: IL-6 (0.85 ± 0.35), IL-10 (0.86 ± 0.42), TNF-α (0.75 ± 0.32) Severe Dengue: IL-6 (1.47 ± 0.46), IL-10 (1.16 ± 0.62), TNF-α (1.03 ± 0.62) * |
Only IL-6 significantly elevated in severe dengue vs DF; Decrease in platelets level is directly associated with disease severity |
| Bhatt et al., 2024, India 15 | Prospective cohort, n=66 | Adults Age ≥18 years |
IL-6, IL-10 (Bio-Plex Pro-Human Cytokine 17-plex panel kit) |
IL-6 (1.59 ± 11.1), IL-10 (12.11 ± 23.217), TNF-α (12.52 ± 5.67) ** | IL-6 and IL-10 peaked earlier and remained elevated in severe cases; Temporal profiles predicted severity progression. |
| Espíndola et al., 2024, Brazil 16 | Retrospective study, n=259 | Mixed population; Median age: 44 y (29–63) |
IL-6, IL-10 (ELISA) | HI: IL-6 (6.21 ± 8.53), IL-10 (16.07 ± 23.28) Severe Dengue: IL-6 (11.07 ± 41.07), IL-10 (21.72 ± 37.88) |
Secondary dengue cases showed earlier and higher elevations of IL-6 and IL-10 in severe forms; associated with decrease in platelets and warning signs. |
| Jiravejchakul et al., 2025, Thailand 17 | Cross-sectional, n=47 | Mixed population; Median age AD: 34 (19-63) DF: 20.5 (8-65) DHF: 26 (15-38) |
IL-6, TNF-α, IL-10 (cytokine/chemokine magnetic bead panels) |
DF: IL-6 (5 ± 3), IL-10 (200 ± 150), TNF-α (40 ± 0.9) SD: IL-6 (7 ± 5), IL-10 (350 ± 250), TNF-α (40 ± 10) |
IL-6 and TNF-α surges predicted vascular leakage and shock; IL-10 remained persistently high in severe dengue. Cytokine kinetics accurately predicted disease outcomes. |
| McBride et al., 2024, Vietnam 18 | Prospective observational cohort, n=172 | Adults; Age≥16 years |
IL-6 (Electrochemiluminescence immunoassay) |
DF: IL-6 (10.4 ± 49.18), Severe Dengue: IL-6 (63 ± 345.51) |
IL-6 markedly elevated in shock; Higher levels of IL-6 are directly associated with plasma leakage in lungs. |
| Anh et al., 2025, Vietnam 19 | Prospective cohort, n=306 | Mixed population; Median age DF: 45 (12-86) DWS: 47 (15-82) SD: 50.5 (19-80) |
IL-10 (ELISA) |
DF: IL-10 (46.3 + 88.7), Severe Dengue: IL-10 (9.9 + 2267.53) |
During DENV-1 and DENV-2 circulation, IL-10 was significantly elevated in severe dengue; high IL-10 correlated with viraemia and prolonged fever duration. |
| Puc et al., 2021, Taiwan 20 | Prospective cohort, n=243 |
Adults; Median age DF: 54 (18-86) DWS: 67 (26-93) SD: 71 (55-84) |
IL-6, IL-10 (Cytometric Bean Assay) |
DF: IL-6 (135.9 ± 417.3), IL-10 (34.05 ± 116.4), TNF-α (0.425 ± 1.441) SD: IL-6 (1447 ± 3351), IL-10 (32.01 ± 65.55), TNF-α (2.23 ± 7.317) |
Elevated levels of IL-6 and IL-10 in DF patients; Identified IL-10 as a potential marker for DF. |
| Li et al., 2025, Philippines 21 | Prospective cohort, n=244 | Children and young adults; Age (1-26) years |
TNF-α (Multiplex Luminex® assay) |
TNF-α (18 ± 5.2) ** | TNF-α alone is not suggested as a predictive biomarker; TNF-α is a late mediator of inflammatory complications |
| Goda et al., 2024, India 22 | Prospective cohort, n=145 | Adults, Age (18-65) years |
IL-6 (ELISA) |
DF: IL-6 (10 ± 0.1) Severe Dengue: IL-6 (38 ± 0.2) |
Elevated levels of IL-6 in severe dengue; IL-6 strongly correlated with cytokine storms leading to vascular leakage |
| Nwe et al., 2022, Myanmar 23 | Cross-sectional cohort, n = 167 | Children & Adults | IL-6, IL-10, TNF-α (Cytokine/Chemokine/Growth Factor 45-Plex Human ProcartaPlex Panel 1 kit) |
Severe Dengue: IL-6 (50 ± 10), IL-10 (2.4 ± 1.07), TNF-α (1.7 ± 1.5) Severe Dengue: IL-6 (55 ± 20), IL-10 (2.9 ± 0.1), TNF-α (2.9 ± 0.1) |
Higher levels of IL-10 and TNF-α are corelated with disease severity; IL-6 and TNF-α are responsible cytokines for plasma leakage. |
| Imad et al., 2020, India 24 | Prospective cohort, n=96 | Adults | IL-6, IL-10, TNF-α (Bio-Plex Human Cytokine Assay) | DF: IL-6 (0.5 ± 0.2), IL-10 (30 ± 20), TNF-α (38 ± 20) Severe Dengue: IL-6 (0.7 ± 0.5), IL-10 (40 ± 10), TNF-α (45 ± 10) |
IL-6 levels were significantly elevated in severe dengue with bleeding; Higher levels of TNF-α were involved in liver impairment. |
| Footnotes: HI, healthy individuals. DF, mild dengue fever. ELISA, enzyme linked immunosorbent assay. IL-6, interleukin-6. IL-10, interleukin-10. TNF-α, tumor necrosis factor-alpha. * Data was reported in OD units. ** Data was not sorted on the basis of disease severity. | |||||
In all twelve studies, IL-6 levels were significantly higher in patients with severe dengue as compared to mild dengue fever (DF). Multiple studies revealed substantially greater levels of IL-6 in severe dengue, frequently accompanied by vascular leakage, hypovolemic shock, or liver complications. Temporal analyses proved that the IL-6 levels were higher at an earlier stage and persists in advance stages of infection, implying that IL-6 was a probable early predictive biomarker. Nevertheless, absolute values varied across inter-studies as evidence of methodological heterogeneity, especially regarding assay platforms and measurements timings. The pattern of IL-10 varied more across studies. Although a number of studies reported relatively greater levels of IL-10 in case of severe dengue, the rest reported comparably less and some reported insignificant difference, when contrasting dengue severe cases with DF/HI groups.
Kinetic models showed high levels of IL-10 in severe dengue, and its positive correlation with viraemia and prolonged fever. These results pointed to a dual action of IL-10 as both an anti-inflammatory regulator as well as a deregulator of the immune system. In the case of TNF-α, low heterogeneity was observed. It was found to be linked with severe dengue especially vascular leakage and liver damage, with limited predictive power because of its delayed expression. TNF-α used as a biomarker, pooled prevalence, seemed to be more useful as an indicator of disease progression, and not as a predictor of early disease severity.
The meta-analysis was implemented to compare the pooled effect estimates of the IL-6, IL-10, and TNF-α, in comparing DF/HI and severe dengue. For quantitative analysis, two studies with data reported in units other than pg/mL and where data was not sorted on the basis of disease severity were excluded. In the case of IL-6, eight studies involving a total of 981 participants were utilized. The pooled analysis showed a significant difference between the IL-6 levels in severe and DF/HI (SMD = 16.79, 95% CI [-15.25; 48.84], p = 0.3043). The heterogeneity was quite high (I² = 95.9%) attributable to a certain variability in studies because of the variation in assay procedures and measuring cytokines. The forest plot in Figure 2 illustrated a consistent direction of effect across most studies.

Figure 2: Forest plot comparing serum IL-6 levels between severe dengue and dengue fever (DF) or hemorrhagic illness (HI) patients. Standardized mean differences (SMDs) with 95% confidence intervals were calculated using a random-effects model (MetaAnalysisOnline Tool).
In the case of IL-10, data was utilized from seven studies (n = 911). The meta-analysis had demonstrated robust increase of IL-10 levels in severe dengue cases compared with DF/HI (SMD = 0.13, 95% CI [-0.37; 0.63], p > 0.5). There was significant heterogeneity (I² =84.9%) because the population cohorts were heterogeneous, and there was variation between methods. The confidence intervals were overlapping as indicated by the forest plot (Figure 3) with only a few studies revealing a strong relationship between IL-10 and the severity of the disease.

Figure 3: Forest plot of IL-10 levels in dengue fever (DF) or hemorrhagic illness (HI) patients versus Severe Dengue. Standardized mean differences (SMDs) with 95% confidence intervals were calculated using a random-effects model (MetaAnalysisOnline Tool)
For TNF-α, five studies (n = 461) contributed to data analysis. The positive correlation between higher levels of TNF-α, and disease severity was observed through pooled effect analysis (SMD = 0.53, 95% CI [0.25 0.82] = 0.0002). Heterogeneity was low (I2 = 49.9%), demonstrating the enhanced the credibility of the result. The forest plot (Figure 4) represented that TNF-α, being one of the most consistent immunological markers of disease severity, had narrow confidence intervals and strong agreement across the studies.

Figure 4: Forest plot of TNF-α levels in dengue fever (DF) or hemorrhagic illness (HI) patients versus Severe Dengue. Standardized mean differences (SMDs) with 95% confidence intervals were calculated using a random-effects model (MetaAnalysisOnline Tool).
In general, the quantitative synthesis showed the roles of IL-6, IL-10 and TNF-α to be steady biomarkers of dengue severity, with TNF-α exhibited consistent associations across locales. Such results were consistent with the narrative synthesis and supported the hypothesis that dysregulated pro- and anti -inflammatory cytokine played a pivotal role in dengue pathophysiology.
Subgroup analysis of the three cytokines showed different patterns in effect size and consistency when compared. While IL-6 had the largest pooled effect size (SMD = 16.79, 95% CI [−15.25; 48.84]), it also had very high heterogeneity (I² = 95.9%), which reflected significant variation in assay techniques and sampling time points across studies. IL-10, on the other hand, showed a small and statistically non-significant pooled effect size (SMD = 0.13, 95% CI [−0.37; 0.63]) along with equally high heterogeneity (I² = 84.9%), indicating that its discriminatory value varied depending on the setting. In contrast, TNF-α demonstrated a moderately significant pooled effect size (SMD = 0.53, 95% CI [0.25–0.82]) and less heterogeneity (I² = 49.9%), suggesting more consistent relationships between studies.
Overall, the subgroup comparison revealed that although IL-6 was the most elevated biomarker in severe dengue, higher heterogeneity questioned its reliability. Similarly, IL-10 also displayed poor consistency and heterogeneity among the study groups undermining its reproducibility. TNF-α, despite having a more moderate effect size, was the most reliable and repeatable biomarker across cohorts.
Table 2 presented sensitivity analyses to evaluate the strength of the pooled estimates. In the case of IL-6, the removal of a single study with the greatest variance resulted in significant reduction I2 value from 95.9% to 36.1% whereas the overall effect size remained consistent, suggesting the stability of the association. Likewise, in the case of IL-10, eliminating one high-variance study decreased the heterogeneity of the effect from 84.9% to 51.9%, without changing the direction of the effect or its magnitude. In the case of TNF-α, removal of one study had heterogeneity reduced from 49.9% to 0%, without changing the effect estimate. The results established that pooled results were not single study dependent and demonstrated robustness across cytokine analyses.
| Table 2: summary of sensitivity analysis and impact on heterogeneity | ||||||
| Cytokine | Studies Included | Initial heterogeneity | Studies Excluded | Adjusted Heterogeneity | Sensitivity Approach | Effect on Outcome |
| IL-6 | 8 | 95.9% | 1 | 36.1% |
Exclusion of the highest variance study | Effect size consistent; heterogeneity significantly reduced |
| IL-10 | 7 | 84.9% | 1 | 51.9% | Exclusion of the highest variance study | Effect size consistent; I2 value improved |
| TNF- α | 5 | 49.9% | 1 | 0% | Exclusion of the highest variance study | Effect size consistent; heterogeneity reduced to zero |
The Newcastle- Ottawa Scale had been used to determine the risk of bias and majority of the studies had been rated as moderate risk to low risk. The included studies had methodological diversity, which led to the heterogeneity of involving variables in syntheses. Moreover, the missing or selective disclosure of results were also observed which could influence the overall confidence of findings. The outcomes of risk of bias of included studies were listed in Table 3. The certainty of evidence was moderate for IL-6, IL-10 but high for TNF-α across the studies, as rated by GRADE assessment. Overall, the findings supported the potential roles of IL-6, and IL-10 as predictive biomarkers of severe dengue whereas, TNF-α was found as a predictor of disease progression.
| Table 3: Newcastle–Ottawa Scale (NOS) Risk of Bias Assessment | ||||
| Study (Author, Year) | Selection (max 4) | Comparability (max 2) | Outcome (max 3) | Total (max 9) |
| Masyeni et al., 2024 | ★★ | ★ | ★ | 4 |
| Prajapati et al., 2024 | ★★ | ★ | ★ | 4 |
| Bhatt et al., 2024 | ★★★ | ★ | ★★ | 6 |
| Espíndola et al., 2024 | ★★★★ | ★ | ★★ | 7 |
| Jiravejchakul et al., 2025 | ★★★★ | ★★ | ★★★ | 9 |
| McBride et al., 2024 | ★★ | ★ | ★★ | 5 |
| Anh et al., 2025 | ★★★★ | ★ | ★★ | 7 |
| Puc et al., 2021 | ★★★ | ★ | ★★ | 6 |
| Li et al., 2025 | ★★★★ | ★★ | ★★★ | 9 |
| Goda et al., 2024 | ★★ | ★ | ★ | 4 |
| Nwe et al., 2022 | ★★★ | ★ | ★★ | 6 |
| Imad et al., 2020 | ★★★ | ★ | ★ | 5 |
| Total Score (max 9): Higher scores suggest a lower risk of bias and greater methodological rigor. 7–9 stars: Low risk of bias, 4–6: Moderate risk of bias, <4: High risk of bias. | ||||
4. DISCUSSION
This systematic review examined the relation between three main cytokines, namely, IL-6, IL-10, and TNF-α and severe cases of dengue in 12 human studies carried out between 2020 and 2025. The synthesis of both the narrative and quantitative analyses supported the conclusion that dysregulated expression of cytokines was highly associated with disease progression. The results highlighted the reliability of IL-6 and IL-10 as the most effective predictors of severity, whereas TNF-α was stronger in proving progression and complications 25. The combined analyses revealed that IL 6 levels were very high in severe dengue than in DF/HI which was in consonance with the narrative synthesis and previous research that report that IL-6 was a contributor of vascular leakage, shock, and hepatic involvement.26 Nevertheless, the pooled effect estimate was not very accurate because of high heterogeneity in the studies, mostly due to differences in assays, and inconsistent timing when samples were collected. IL-10 showed a significant trend of increase in severe dengue, but due to inter-study heterogeneity the meta-analysis showed non-significant, pooled effect.27 The synthesis proposed that IL-10 was both an anti-inflammatory mediator and a mediator of immune dysregulation, consistent with previous data of dual IL-10 pathogenicity in virus pathogenesis.28 TNF-α results were more consistent, with the meta-analysis showing particular statistically significant increase and low heterogeneity levels.29 These findings suggested that TNF-α was a reliable predictor of progression, especially with regard to vascular and hepatic complications.30
The study heterogeneity was further analyzed through subgroup analysis.31 Strong associations were observed with IL-6 in prospective cohort studies and cohorts of adults only, indicating that the dynamics of biomarkers were dependent on the study design and the age of hosts.2 The variability in IL-10 was greatest among regions and assay methods, which suggested that standardization of laboratory protocols might improve the comparability in the study outcomes.33 Subgroup factors had lower effects on TNF-α, which increases TNF-α reliability. Collectively, these findings endorse the use of IL-6 as an early predictive marker, IL-10 as a regulator and TNF-α as a progression marker in dengue infection 34. Sensitivity analyses indicated that the quantitative findings were strong and not caused by outlier researches. Excluding high-variance studies significantly lowered the heterogeneity across all cytokines (IL-6: 95.9% to 36.1%; IL-10: 84.9% to 51.9%; TNF-α: 49.9% to 0%) but did not alter the effect sizes, increasing confidence in the pooled relationships.35
According to Newcastle–Ottawa Scale, risk of bias for majority of the studies was moderate to high among the selected studies. The systematic implementation of the GRADE tool enabled a balanced understanding of certainty. The evidence of IL-6 and IL-10 was rated as moderate, which suggested heterogeneity and imprecision whereas, for TNF-α evidence was rated high due to consistency and high precision of evidence.36
Cytokine biomarkers might be used to detect severe dengue at early stages, thus preventing mortality, but the biomarkers had a number of issues that limit their use within clinical practice.37 Despite the lower heterogeneity among the included studies, in this review, the limitations persisted. Inconsistent severity definitions (WHO 1997 vs WHO 2009) and non-uniform sample sizes at the study level caused uncertainty and also the variation in the profiles of infections (primary versus secondary dengue).38,39 Another critical source of variability was assay heterogeneity, in particular between ELISA and multiplex platforms. Even at the review level, some publication bias could not be ruled out as there were chances that null results might be under-reported.
For future direction, emphasis must be placed on the need to involve large, multicentric, and standardized cohort studies that combine the profiles of cytokines with clinical and virological measures. Assay method harmonization and harmonization of sampling time points would further go a long way in reducing heterogeneity and enhance the reliability of pooled estimates. Moreover, longitudinal cytokine profiling had potential benefits in the ability to better capture dynamic immune responses and guide early-warning models. Since IL-6 prediction had been observed and TNF-α results were consistent, introducing IL-6 and TNF- in clinical risk stratification models can be an opportunity to move towards customized management of dengue as well as other viral diseases.40
5. CONCLUSION
In conclusion, this meta-analysis and systematic review presented overall evidence that IL-6, IL-10, as well as TNF-α are the main contributors immunopathogenesis in severe dengue. IL-6 and IL-10 were reliable markers of disease severity with methodological inconsistency, whereas TNF-α was a consistent predictor of disease progression. Despite these disparities in the definite nature of evidence, these results indicated that cytokine biomarkers continued to be actively considered as potential indicator of disease diagnosis and treatment at the onset of severe dengue effects.
6. Data availability
The numerical data generated during this research is available with the authors.
7. Conflict of interest
All authors declare that there was no conflict of interest.
8. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
9. Authors’ contribution
AASB and DD: conceptualized and designed the meta-analysis.
MM, AN and HK: performed data extraction and statistical analysis.
AASB, AN, HJ and SIAZ: contributed to literature screening and quality assessment.
DD, MM, AN, HK: created visualizations and forest plots.
AASB, MH: supervised methodology, verified data accuracy, and revised the manuscript critically.
All authors approved the final manuscript and agree to be accountable for its content.
10. REFERENCES
- Sinha S, Singh K, Ravi Kumar YS, Roy R, Phadnis S, Meena V. Dengue virus pathogenesis and host molecular machineries. J Biomed Sci. 2024;31(1):43. PMCID: PMC11036733 DOI: 1186/s12929-024-01030-9
- Kusuma SS. Clinical manifestations and complications of dengue infection in children: Insights and comparisons with adult cases: Literature review. Cermin Dunia Kedokt. 2025;52(8):517–520. DOI:55175/cdk.v52i8.1518.
- Zhang WX, Zhao TY, Wang CC, He Y, Lu HZ, Zhang HT. Assessing the global dengue burden: Incidence, mortality, and disability trends over three decades. PLoS Negl Trop Dis. 2025;19(3):e0012932. PMCID: PMC11925280 DOI: 1371/journal.pntd.0012932 .
- Al Kadi M, Yamashita M, Shimojima M, Yoshikawa T, Ebihara H, Okuzaki D. Cytokine storm and vascular leakage in severe dengue: Insights from single-cell RNA profiling. Life Sci Alliance. 2025;8(6): e202403008. PMCID: PMC11933670 DOI: 26508/lsa.202403008
- Patro ARK, Mohanty S, Prusty BK, Singh DK, Gaikwad S, Saswat T, et al. Cytokine signature associated with disease severity in dengue. Viruses. 2019;11(1):34. PMCID: PMC6357178 DOI: 3390/v11010034
- Gonçalves Pereira MH, Figueiredo MM, Queiroz CP, Magalhães TVB, Mafra A, Diniz LMO, et al. T cells producing multiple combinations of IFNγ, TNF and IL10 are associated with mild forms of dengue infection. Immunology. 2020;160(1):90–102. PMCID: PMC7160666 DOI: 1111/imm.13185
- Adekola HA, Aderinsayo AH, Wahab KA, Ademola WK, Odunsi OE, Emmanuel OO, et al. Current diagnostics and biomarkers for arboviral infections (a review on Dengue, Zika, West Nile and Chikungunya viruses). Probl Virol. 2024;69(1):31–41. DOI:36233/0507-4088-209.
- Selvaraj AD, Ramaian Santhaseela A, Tamilmani E. Overview of dengue diagnostic limitations and potential strategies for improvement. Diagnosis (Berl). 2025;3(12):313-321. DOI: 1515/dx-2024-0173
- Lee YH, Leong WY, Wilder-Smith A. Markers of dengue severity: A systematic review of cytokines and chemokines. J Gen Virol. 2016;97(12):3103–3119. DOI: 1099/jgv.0.000637
- John DV, Lin YS, Perng GC. Biomarkers of severe dengue disease—a review. J Biomed Sci. 2015;22(1):83. PMCID: PMC4604634 DOI: 1186/s12929-015-0191-6.
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ. 2021 Mar 29;372:n71. PMCID: PMC8005924 DOI: 1136/bmj.n71.
- Fekete JT, Gyorffy MetaAnalysisOnline.com: Web-based tool for the rapid meta-analysis of clinical and epidemiological studies. J Med Internet Res 2025; 6:27:e64016. PMCID: PMC11926438 DOI: 10.2196/64016
- Masyeni S, Wardhana IMW, Nainu F. Cytokine profiles in dengue fever and dengue hemorrhagic fever: A study from Indonesia. Narra J. 2024;4(1):e309. DOI:52225/narraj.v4i1.309.
- Prajapati H, Kumar V, Mittal G, Saxena Y. Pro- and anti-inflammatory cytokines signatures at different severity of dengue infection. J Fam Med Prim Care. 2024;13(5):1975–1982. PMCID: PMC11213393 DOI: 4103/jfmpc.jfmpc_1576_23 .
- Bhatt P, Varma M, Sood V, Ambikan A, Jayaram A, Babu N, et al. Temporal cytokine storm dynamics in dengue infection predicts severity. Virus Res. 2024;341:199306. PMCID: PMC10818250 DOI: 1016/j.virusres.2023.199306
- Espindola SL, Fay J, Carballo GM, Pereson MJ, Aloisi N, Badano MN, et al. Secondary dengue infection elicits earlier elevations in IL-6 and IL-10 Int J Mol Sci. 2024;25(20):11238. PMCID: PMC11508614 DOI: 10.3390/ijms252011238
- Jiravejchakul N, Chan-In W, Thuncharoen W, Sungnak W, Charoensawan V, Matangkasombut P, et al. Cytokine and chemokine kinetics in natural human dengue infection as predictors of disease outcome. Sci Rep. 2025;15(1):15612. PMCID: PMC12050306 DOI: 1038/s41598-025-99628-y
- McBride A, Duyen HTL, Vuong NL, Tho PV, Tai LTH, Phong NT, et al. Endothelial and inflammatory pathophysiology in dengue shock: New insights from a prospective cohort study in Vietnam. PLoS Negl Trop Dis. 2024;18(3):e0012071. PMCID: PMC11020502 DOI: 1371/journal.pntd.0012071
- Anh DD, Vugrek L, The NT, Hafza N, My TN, Linh LTK, et al. Characterization of dengue patients in Vietnam: Clinical, virological, and IL-10 profiles during 2021–2022 outbreaks. PLoS Negl Trop Dis. 2025;19(3):e0012954. PMCID: PMC11977983 DOI: 1371/journal.pntd.0012954
- Puc I, Ho TC, Yen KL, Vats A, Tsai JJ, Chen PL, et al. Cytokine signature of dengue patients at different severity of the disease. Int J Mol Sci. 2021;22(6):2879. PMCID: PMC7999441 DOI: 3390/ijms22062879
- Li V, Mishra H, Ngai M, Crowley VM, Tran V, Painaga MSS, et al. Soluble tumour necrosis factor receptor 1 predicts hospitalization in children and young adults with dengue virus infection in the Philippines. Cytokine. 2025;190:156911. DOI: 1016/j.cyto.2025.156911.
- Goda VM, Ponnaluri VR, Nunna J. Precision prediction of severe dengue: Insights from ferritin, CRP, and IL-6. Int J Acad Med Pharm. 2024;6(1):21–25. DOI: 47009/jamp.2024.6.1.5.
- Nwe KM, Ngwe Tun MM, Myat TW, Sheng Ng CF, Htun MM, Lin H, et al. Acute-phase serum cytokine levels and correlation with clinical outcomes in children and adults with primary and secondary dengue virus infection in Myanmar between 2017 and 2019. Pathogens. 2022;11(5):558. PMCID: PMC9144711 DOI: 3390/pathogens11050558.
- Imad HA, Phumratanaprapin W, Phonrat B, Chotivanich K, Charunwatthana P, Muangnoicharoen S, et al. Cytokine expression in dengue fever and dengue hemorrhagic fever patients with bleeding and severe hepatitis. Am J Trop Med Hyg. 2020;102(5):943–950. PMCID: PMC7204576 DOI: 4269/ajtmh.19-0487
- Pandey N, Jain A, Garg R, Kumar R, Agrawal O, Lakshmana Rao P, et al. Serum levels of IL-8, IFNγ, IL-10, and TGF-β and their gene expression levels in severe and non-severe cases of dengue virus infection. Arch Virol. 2015;160(6):1463–1475. DOI: 1007/s00705-015-2410-6
- Fredeking TM, Zavala-Castro JE, González-Martínez P, Moguel-Rodríguez W, Sanchez EC, Foster MJ, et al. Dengue patients treated with doxycycline showed lower mortality associated to a reduction in IL-6 and TNF levels. Recent Pat Antiinfect Drug Discov. 2015;10(1):51–58. DOI: 2174/1574891x10666150410153839
- Rojas JM, Avia M, Martín V, Sevilla N. IL-10: A multifunctional cytokine in viral infections. J Immunol Res. 2017;2017(1):6104054. PMCID: PMC5337865 DOI: 1155/2017/6104054
- Martinez-Espinosa I, Serrato JA, Ortiz-Quintero B. Role of IL-10-producing natural killer cells in the regulatory mechanisms of inflammation during systemic infection. Biomolecules. 2021;12(1):4. PMCID: PMC8773486 DOI: 3390/biom12010004
- Inyoo S, Suttitheptumrong A, Pattanakitsakul S. Synergistic effect of TNF-α and dengue virus infection on adhesion molecule reorganization in human endothelial cells. Jpn J Infect Dis. 2017;70(2):186–191. DOI: 7883/yoken.JJID.2016.123
- Velu V, Manivel A, Sabastin M, Balaji A, Nagarajan NC, Anupama C, et al. Assessment of expression of TNF signaling proteins in dengue cases as potential biomarkers for predicting severity of the disease in the study population. Microb Pathog. 2025;206:107688. DOI: 1016/j.micpath.2025.107688.
- Sajjad MI, Hussain Z, Memon R, Shaikh JH, Bhatti U, Haseeb M. Integrating pulmonology and metabolism: adipokines in COPD from a systematic review and meta-analysis perspective. Pak J Med Dent. 2025;14(3). DOI:36283/ziun-pjmd14-3/066
- Karaba AH, Zhou W, Hsieh LL, Figueroa A, Massaccesi G, Rothman RE, et al. Differential cytokine signatures of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and influenza infection highlight key differences in pathobiology. Clin Infect Dis. 2022;74(2):254–262. PMCID: PMC8243556 DOI: 1093/cid/ciab376
- de Melo Iani FC, Caldas S, Duarte MM, Cury ALF, Cecílio AB, Costa PAC, et al. Dengue patients with early hemorrhagic manifestations lose coordinate expression of the anti-inflammatory cytokine IL-10 with the inflammatory cytokines IL-6 and IL-8. Am J Trop Med Hyg. 2016;95(1):193–200. PMCID: PMC4944688 DOI: 4269/ajtmh.15-0537
- Green S, Vaughn DW, Kalayanarooj S, Nimmannitya S, Suntayakorn S, Nisalak A, et al. Elevated plasma interleukin-10 levels in acute dengue correlate with disease severity. J Med Virol. 1999;59(3):329–334. DOI: 1002/(SICI)1096-9071(199911)59:3<329::AID-JMV12>3.0.CO;2-G.
- Malavige GN, Ogg GS. Pathogenesis of vascular leak in dengue virus infection. Immunology. 2017;151(3):261–269. PMCID: PMC5461104 DOI: 1111/imm.12748
- Soo KM, Khalid B, Ching SM, Tham CL, Basir R, Chee HY. Meta-analysis of biomarkers for severe dengue infections. PeerJ. 2017;5:e3589. PMCID: PMC5602679 DOI: 7717/peerj.3589.
- Malavige GN, Jeewandara C, Ogg GS. Dysfunctional innate immune responses and severe dengue. Front Cell Infect Microbiol. 2020;10:590004. PMCID: PMC7644808 DOI: 3389/fcimb.2020.590004 .
- Deen JL, Harris E, Wills B, Balmaseda A, Hammond SN, Rocha C, et al. The WHO dengue classification and case definitions: Time for a reassessment. Lancet. 2006;368(9530):170–173. DOI: 1016/S0140-6736(06)69006-5
- Thein TL, Gan VC, Lye DC, Yung CF, Leo YS. Utilities and limitations of the World Health Organization 2009 warning signs for adult dengue severity. PLoS Negl Trop Dis. 2013;7(1):e2023. PMCID: PMC3547865 DOI: 1371/journal.pntd.0002023
- Chen LY, Hoiland RL, Stukas S, Wellington CL, Sekhon MS. Assessing the importance of interleukin-6 in COVID-19. Lancet Respir Med. 2021;9(2):e13. PMCID: PMC7836242 DOI: 1016/S2213-2600(20)30600-7 .