Syeda Hajrah Rehman 1 , Zahid Akhter Rao 2 , Syed Samiuddin 3 , Muhsan Sultan Abbasi 4 , Muhammad Khalid 5
Authors affiliations:
Background & objective: Effective management of postoperative pain is challenging, as numerous patients encounter severe pain or face adverse events linked to analgesic medication. The study aims to compare the effect of intravenous low-dose ketamine versus lignocaine infusion on post-operative pain in patients undergoing laparoscopic cholecystectomy under general anesthesia in a tertiary care hospital.
Methods: This randomized controlled trial was performed in the Department of Anesthesia at Fazaia Ruth Pfau Medical College, PAF Hospital, Karachi, from January to November 2024. Group K received ketamine at a 0.2 mg/kg bolus intravenously and the same dose per hour throughout the surgery, whereas Group L received lignocaine at a rate of 1 mg/kg bolus and the same dose per hour throughout the surgery. Pain status was monitored postoperatively at 0, 2, 4, 6, 12, and 24 hours using the Numerical Rating Score (NRS).
Results: Fifty patients were studied in each arm. Postoperative pain score was similar in Group K and Group L at 0 hour (1.5 ± 0.8 versus 1.4 ± 0.5, P = 0.395), 2nd hour (2.3 ± 0.9 versus 2.4 ± 1.2; P = 0.847), 4th hour (2.8 ± 1.3 versus 2.9 ± 1.1, P = 0.872), 6th hour (2.8 ± 1.3 versus 2.8 ± 1.1, P = 1.000) and 12th hour (2.2 ± 1.2 versus 2.1 ± 0.8, P = 0.714). Mean pain score was found to be significantly different between Group K and Group L at 24th hour (1.3 ± 0.5 versus 2.2 ± 0.4, P < 0.001). The time to first rescue analgesia and total analgesic consumption did not differ significantly between the two groups.
Conclusions: The findings suggested that both low-dose ketamine and lignocaine had similar pain relief effect in early postoperative period. However, ketamine was found to be superior to lignocaine at 24 hours in terms of lower pain score.
Abbreviations: LC: laparoscopic cholecystectomy, NRS: Numerical Rating Score, NMDA: N-methyl-D-aspartate
Keywords: Laparoscopic Cholecystectomy; Lignocaine; Low-dose Ketamine; Pain; Post-operative analgesia
Citation: Rehman SH, Rao ZA, Sammiuddin S, Abbasi MS, Khalid M. Comparison of low-dose infusions of ketamine with lignocaine for post-operative pain in patients undergoing laparoscopic cholecystectomy under general anesthesia. Anaesth. pain intensive care 2025;29(8):861-867. DOI: 10.35975/apic.v29i8.3008.
Received: June 30, 2025. Revised: August 17, 2025. Accepted: August 22, 2025
Effective management of postoperative pain is challenging, as numerous patients encounter severe pain or face adverse events linked to analgesic medication.1 Approximately 86% of individuals undergoing surgery indicate the presence of postoperative pain, frequently characterized as moderate to severe.2 Inadequate pain control can lead to various unfavorable outcomes for the patient, such as delayed hospital discharge, hindered functional recovery, and an elevated likelihood of developing chronic pain.3,4
Various methods are employed to alleviate postoperative pain, such as intravenous or intramuscular administration of NSAIDs and opioids.5,6 Regrettably, the effectiveness of these approaches in pain relief comes at the cost of a range of undesirable short-term and long-term side effects, including respiratory depression, hypotension, sedation, nausea and vomiting, urinary retention, and ileus.7,8
In the current age of opioid-sparing practices, a multimodal analgesia approach is employed to achieve effective postoperative pain relief while minimizing the occurrence of opioid-related adverse events. This technique encompasses various methods such as epidural blocks, peripheral nerve blocks, local wound infiltration, and the use of systemic analgesic adjuncts.9 Recently, there has been a growing interest in exploring different analgesic adjuncts, including intravenous infusion of local anesthetics (such as lignocaine), N-methyl-D-aspartate (NMDA) receptor antagonists (like ketamine), alpha agonists (for example, dexmedetomidine), and anticonvulsants (such as gabapentin).10,11
Lignocaine, also known as lidocaine, is a local anesthetic primarily acting as a voltage-gated sodium channel blocker, and possesses analgesic, anti-hyperalgesic, and anti-inflammatory properties. A recent study demonstrated that intravenous lidocaine usage reduces opioid consumption, enhances postoperative analgesia, and improves bowel function without adverse effects following abdominal surgery.10
Pain stands out as the most crucial independent predictor of recovery time. Inadequately managed pain can result in adverse physiological effects and give rise to psychological, economic, and social consequences. Following surgery, a key concern is identifying a medication that offers prolonged pain relief and relaxation for the patient while minimizing side effects. Previous studies have explored the effects of ketamine and lignocaine, but many of these investigations did not involve low-dose ketamine. Additionally, these studies primarily focused on major surgeries like open abdominal surgeries,12 open nephrectomy,13 inguinal hernia repair,14 or included various elective surgical procedures.15 There is a scarcity of studies specifically examining the use of low-dose ketamine and lignocaine in patients undergoing laparoscopic cholecystectomy (LC). Thus, we planned the present study to compare the effect of low-dose ketamine versus lignocaine on post-operative pain in patients undergoing LC under general anesthesia in a tertiary care hospital.
This double-blinded randomized control trial was performed in the Department of Anesthesia at Fazaia Ruth Pfau Medical College, PAF Hospital Faisal Base, Karachi, with approval of the institutional Review Board (FRPMC-IRB-2024-41). The trial was registered with the number NCT06964555. The study was performed from January to November 2024. Patients were enlisted with their written informed consent. Both male and female patients were included in this study. Inclusion criteria included patients aged 18 years or older, scheduled for elective laparoscopic cholecystectomy (LC), and classified as ASA grade I–II. Exclusion criteria comprised obese patients (BMI ≥25 kg/m²), those with a history of alcohol or drug abuse, uncontrolled hypertension or diabetes, chronic pain, allergy to study drugs, neurological disorders, inability to understand the pain scoring system, or procedures converted to open cholecystectomy.
A sample size of 38 patients per group was estimated, taking pain scores of 2.47 ± 1.5 and 1.40 ± 1.8 in the lignocaine and the ketamine group, respectively16 at a power of 80% and 95% confidence interval. Sample size calculation was performed using an online calculator, Open-epi. For better results, 50 patients were enrolled per arm. The consecutive sampling technique was used to enroll participants into the study, whereas group allocation was done in a random fashion using sequentially numbered opaque sealed envelopes.14 Patients receiving ketamine were labeled as Group K, whereas patients receiving lignocaine were labeled as Group L.
Patients scheduled for LC were admitted a day before surgery for pre-anesthesia assessment, during which informed consent was obtained and patients were educated about the pain scale. After completing the assessment checklist, they were moved to the holding area and randomly assigned to either Group K or Group L. Patients were randomly assigned to study groups using a sequentially numbered opaque sealed envelope (SNOSE) protocol.17 Both patients and study investigators were blinded to the study groups. Only the anesthetist who prepared the medication and was not part of this study was aware of group identification.
After transferring the patient to the operating room, standard monitors were attached, and vital signs, including blood pressure, heart rate, and mean arterial pressure, were continuously monitored. Electrocardiography (ECG) was also performed as part of the standard anesthesia protocol. Anesthesia induction included premedication with nalbuphine 0.1 mg/kg, propofol 2 mg/kg, atracurium 0.5 mg/kg, and ondansetron 0.15 mg/kg. Just before the skin incision, Group K received ketamine 0.2 mg/kg as an intravenous bolus, followed by an infusion at 0.2 mg/kg/hr throughout the surgery, while Group L received lignocaine 1 mg/kg as an intravenous bolus, followed by an infusion at 1 mg/kg/hr. Anesthesia was maintained with 100% oxygen at 3 L/min without any carrier gas, along with isoflurane (1–1.5%), atracurium 0.1 mg/kg, and paracetamol 10 mg/kg intravenously. After procedure completion, all patients were shifted to post-operative recovery room. They were observed in the recovery unit with continuous monitoring of vital signs, including heart rate, blood pressure, oxygen saturation, and respiratory rate. Routine resuscitation equipment and emergency medications were available at the bedside, including provisions for treatment of potential adverse effects such as bradycardia, hypotension, arrhythmias, and emergence reactions. The anesthetic team was available throughout the recovery phase for immediate intervention should it be required. Patients stayed there for the next 2 hours and then were shifted to the ward and discharged after 24 hours if found stable as per hospital protocol.
Post-operatively, pain status was continuously monitored using the Numeric Rating Scale (NRS) at 0 hours, 2 hours, 4 hours, 6 hours, 12 hours, and then finally at 24 hours. As part of multi-model analgesia, paracetamol 10 mg/kg was administered post-operatively at 8-hour intervals as per routine protocol. Additional analgesia was administered to patients upon demand or if their pain score was 5 or higher. For rescue analgesia, an injection of tramadol 30 mg was administered. The assigned residents collected the data from time to time and recorded it in the study proforma.
The NRS is a ten-point scoring scale used for pain assessment. It ranges from 0 to 10 for ranking pain, where 0 means no pain and 10 means the worst pain.18 Time to rescue analgesia was defined as the time point at which the patient would be given the first rescue analgesia for a pain score of 5 or above. The time between the incision and the wound's closure was used to define the length of the surgery.
The collected data were put into the statistical package SPSS version 26 to perform data analysis. Frequencies and percentages were computed for categorical variables. Numerical variables were expressed as mean ± standard deviation or median with inter-quartile range (IQR) based on the assumption of normality that was tested using the Shapiro-Wilk test. Categorical variables were compared using the Chi-square or Fisher’s exact test, while numerical variables were analyzed using the independent t-test or Mann-Whitney U test, as appropriate. A P-value ≤ 0.05 was considered statistically significant.
A total of 100 patients were studied with a 1:1 ratio allocation in study groups. Table 1 displays patients’ sociodemographic and baseline features among the two groups. The two groups did not differ based on age (P = 0.398), gender (P = 0.542), hypertension (P = 0.137), diabetes (P = 0.473), ASA grade (P = 0.537), or surgery duration (P = 0.334). At baseline, none of the patients’ vitals, including systolic blood pressure (P = 0.303), diastolic blood pressure (P = 0.342), heart rate (P = 0.173), and mean arterial pressure (P = 0.157), were significantly different between the two groups. At the end of surgery, the two groups did not differ based on systolic blood pressure (P = 0.679), diastolic blood pressure (P = 0.261), heart rate (P = 0.261), and mean arterial pressure (P = 0.340).
Figure 1 compares patients’ pain status from 0-24 hours post-operatively. Mean score was not significantly different in Group K and Group L at 0 hour ( P = 0.395), 2nd hour (P = 0.847), 4th hour (P = 0.872), 6th hour (P = 1.000), and 12th hour (P = 0.714). Mean pain score was found to be significantly different between Group K and Group L at 24th hour (P < 0.001).
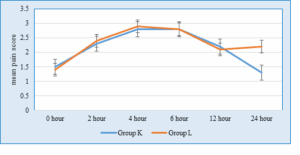
Figure 1: Comparison of mean pain score between the two groups at different time intervals post-operatively
Table 2 compares analgesic status among the two study groups. Time to first rescue analgesia and analgesic consumption did not significantly differ between the two study groups.
Postoperative pain influences complications, patients’ satisfaction, and overall recovery, so managing pain following surgical procedures is a challenging aspect for an anesthetist. In this study, both lignocaine and ketamine were evaluated for their analgesic properties and comparative effects. Ketamine, a well-known NMDA receptor antagonist, is recognized for its analgesic and opioid-sparing effects.19 Contrarily, lignocaine is a local anesthetic drug that exerts analgesic properties on intravenous administration and lowers pain via blocking sodium channels and has an anti-inflammatory impact.20
In the current study, low-dose ketamine and lignocaine are compared for their pain control effects among patients who underwent LC. The findings demonstrated that pain status was similar among both the study groups during 0-12 hours post-operatively. However, the lower pain scores were seen in the ketamine group at 24 hours post-operatively in comparison to lignocaine, suggesting a prolonged analgesic effect of ketamine in modulating pain associated with surgical trauma. The significantly lower pain score for ketamine at 24 hours also highlights that NMDA receptor blockade plays a role in diminishing long-term nociceptive responses. This finding is favorable for the hypothesis that low-dose ketamine lowers long-term pain and enhances pain control beyond the immediate post-operative duration.21 Moreover, the anti-inflammatory properties of ketamine also contribute to prolonged analgesic effects while reducing cytokine-mediated nociceptive responses.22
Previous research has demonstrated that low-dose ketamine alone provides effective analgesia and reduces opioid consumption postoperatively.23-25 Some studies have also reported that ketamine’s analgesic effects are particularly beneficial in the late postoperative period with different routes due to its ability to prevent central sensitization and hyperalgesia.9,26 However, when comparison of ketamine was made with lignocaine, results were variable. Singariya G, et al. (2023) analyzed that pain scores for ketamine and lignocaine were the same at 0 hours and 24 hours.9 Between 0-24 hours, pain scores were found to be lower in the ketamine group during the early post-operative period, 1-6 hours, which gradually increased and matched the pain levels in the lignocaine group. However, our findings are in agreement with the study of Toleska et al, (2022), who also reported effective pain control in the ketamine group at 24 hours than the lignocaine group (1.40 ± 1.8 versus 2.47 ± 1.5).16 Gesso AS et al. (2022) compared pain incidences between low-dose ketamine and lignocaine groups among all elective surgical procedures and reported that low-dose ketamine was superior to lignocaine in lowering pain, with low pain incidences in patients receiving low-dose ketamine.15
Although our study did not find long-term impacts of lignocaine in pain control, the literature documents its analgesic properties in different surgical procedures. Some studies report that lignocaine infusion diminishes opioid need and hastens recovery, mainly in abdominal procedures.27,28 The primary mechanism of lignocaine is based on modulating acute inflammatory responses and inhibiting ectopic discharges in injured nerves.29 We believe that because of this mechanism, which diminishes systemic effects with time, its analgesic advantages were not seen beyond the immediate post-operative period.
The clinical importance of this study is based on the potential function of ketamine in improving postoperative recovery and reducing opioid use. In recent years, the hunt for alternative analgesic techniques has been intensified due to opioid-associated side effects, including nausea, vomiting, and respiratory depression. Because of the well-documented opioid-sparing effects of ketamine, it has been increasingly recommended as part of multimodal analgesia protocols.30,31 Since patients receiving laparoscopic cholecystectomy frequently endure moderate post-operative pain, adding ketamine to pain management protocols may enhance patient comfort and decrease dependence on opioid analgesics.
Moreover, we found that total analgesic consumption and time to first rescue analgesia were not considerably different in Group K and Group L. This finding indicates that ketamine played a greater role in controlling late postoperative pain at 24 hours compared to lignocaine; however, it did not affect the overall need for additional analgesics, as no significant difference was observed between the groups during the immediate postoperative period (before 24 hours).
Despite the positive results, this study has some important limitations. First, pain assessment was performed at specific time intervals, and continuous monitoring could have provided a more comprehensive evaluation of the analgesic effects of the study drugs. Second, the side effects of ketamine, such as hallucinations or dysphoria, were not assessed. Third, hemodynamic stability was not compared between the two groups. Although low-dose ketamine is generally well-tolerated, future studies should investigate its safety profile during the postoperative period for further validation.
The findings suggested that both low-dose ketamine and lignocaine had similar pain-relieving effects in the early postoperative period. However, ketamine was found to be superior to lignocaine at 24 hours in terms of lower pain score.
Authors affiliations:
- Syeda Hajrah Rehman, Fazaia Ruth Pfau Medical College, PAF Base Faisal Karachi, Pakistan; Email: hajrahrehman@ymail.com
- Zahid Akhter Rao, Fazaia Ruth Pfau Medical College, PAF Base Faisal Karachi, Pakistan; Email: zahidrao57@hotmail.com
- Syed Samiuddin, Fazaia Ruth Pfau Medical College, PAF Base Faisal Karachi, Pakistan; Email: same_samee@hotmail.com
- Muhsan Sultan Abbasi, Fazaia Ruth Pfau Medical College, PAF Base Faisal Karachi, Pakistan; Email: abbasimuhsan@gmail.com
- Muhammad Khalid, Fazaia Ruth Pfau Medical College, PAF Base Faisal Karachi, Pakistan; Email: asrozs@yahoo.com
ABSTRACT
Background & objective: Effective management of postoperative pain is challenging, as numerous patients encounter severe pain or face adverse events linked to analgesic medication. The study aims to compare the effect of intravenous low-dose ketamine versus lignocaine infusion on post-operative pain in patients undergoing laparoscopic cholecystectomy under general anesthesia in a tertiary care hospital.
Methods: This randomized controlled trial was performed in the Department of Anesthesia at Fazaia Ruth Pfau Medical College, PAF Hospital, Karachi, from January to November 2024. Group K received ketamine at a 0.2 mg/kg bolus intravenously and the same dose per hour throughout the surgery, whereas Group L received lignocaine at a rate of 1 mg/kg bolus and the same dose per hour throughout the surgery. Pain status was monitored postoperatively at 0, 2, 4, 6, 12, and 24 hours using the Numerical Rating Score (NRS).
Results: Fifty patients were studied in each arm. Postoperative pain score was similar in Group K and Group L at 0 hour (1.5 ± 0.8 versus 1.4 ± 0.5, P = 0.395), 2nd hour (2.3 ± 0.9 versus 2.4 ± 1.2; P = 0.847), 4th hour (2.8 ± 1.3 versus 2.9 ± 1.1, P = 0.872), 6th hour (2.8 ± 1.3 versus 2.8 ± 1.1, P = 1.000) and 12th hour (2.2 ± 1.2 versus 2.1 ± 0.8, P = 0.714). Mean pain score was found to be significantly different between Group K and Group L at 24th hour (1.3 ± 0.5 versus 2.2 ± 0.4, P < 0.001). The time to first rescue analgesia and total analgesic consumption did not differ significantly between the two groups.
Conclusions: The findings suggested that both low-dose ketamine and lignocaine had similar pain relief effect in early postoperative period. However, ketamine was found to be superior to lignocaine at 24 hours in terms of lower pain score.
Abbreviations: LC: laparoscopic cholecystectomy, NRS: Numerical Rating Score, NMDA: N-methyl-D-aspartate
Keywords: Laparoscopic Cholecystectomy; Lignocaine; Low-dose Ketamine; Pain; Post-operative analgesia
Citation: Rehman SH, Rao ZA, Sammiuddin S, Abbasi MS, Khalid M. Comparison of low-dose infusions of ketamine with lignocaine for post-operative pain in patients undergoing laparoscopic cholecystectomy under general anesthesia. Anaesth. pain intensive care 2025;29(8):861-867. DOI: 10.35975/apic.v29i8.3008.
Received: June 30, 2025. Revised: August 17, 2025. Accepted: August 22, 2025
1. INTRODUCTION
Effective management of postoperative pain is challenging, as numerous patients encounter severe pain or face adverse events linked to analgesic medication.1 Approximately 86% of individuals undergoing surgery indicate the presence of postoperative pain, frequently characterized as moderate to severe.2 Inadequate pain control can lead to various unfavorable outcomes for the patient, such as delayed hospital discharge, hindered functional recovery, and an elevated likelihood of developing chronic pain.3,4
Various methods are employed to alleviate postoperative pain, such as intravenous or intramuscular administration of NSAIDs and opioids.5,6 Regrettably, the effectiveness of these approaches in pain relief comes at the cost of a range of undesirable short-term and long-term side effects, including respiratory depression, hypotension, sedation, nausea and vomiting, urinary retention, and ileus.7,8
In the current age of opioid-sparing practices, a multimodal analgesia approach is employed to achieve effective postoperative pain relief while minimizing the occurrence of opioid-related adverse events. This technique encompasses various methods such as epidural blocks, peripheral nerve blocks, local wound infiltration, and the use of systemic analgesic adjuncts.9 Recently, there has been a growing interest in exploring different analgesic adjuncts, including intravenous infusion of local anesthetics (such as lignocaine), N-methyl-D-aspartate (NMDA) receptor antagonists (like ketamine), alpha agonists (for example, dexmedetomidine), and anticonvulsants (such as gabapentin).10,11
Lignocaine, also known as lidocaine, is a local anesthetic primarily acting as a voltage-gated sodium channel blocker, and possesses analgesic, anti-hyperalgesic, and anti-inflammatory properties. A recent study demonstrated that intravenous lidocaine usage reduces opioid consumption, enhances postoperative analgesia, and improves bowel function without adverse effects following abdominal surgery.10
Pain stands out as the most crucial independent predictor of recovery time. Inadequately managed pain can result in adverse physiological effects and give rise to psychological, economic, and social consequences. Following surgery, a key concern is identifying a medication that offers prolonged pain relief and relaxation for the patient while minimizing side effects. Previous studies have explored the effects of ketamine and lignocaine, but many of these investigations did not involve low-dose ketamine. Additionally, these studies primarily focused on major surgeries like open abdominal surgeries,12 open nephrectomy,13 inguinal hernia repair,14 or included various elective surgical procedures.15 There is a scarcity of studies specifically examining the use of low-dose ketamine and lignocaine in patients undergoing laparoscopic cholecystectomy (LC). Thus, we planned the present study to compare the effect of low-dose ketamine versus lignocaine on post-operative pain in patients undergoing LC under general anesthesia in a tertiary care hospital.
2. METHODOLOGY
This double-blinded randomized control trial was performed in the Department of Anesthesia at Fazaia Ruth Pfau Medical College, PAF Hospital Faisal Base, Karachi, with approval of the institutional Review Board (FRPMC-IRB-2024-41). The trial was registered with the number NCT06964555. The study was performed from January to November 2024. Patients were enlisted with their written informed consent. Both male and female patients were included in this study. Inclusion criteria included patients aged 18 years or older, scheduled for elective laparoscopic cholecystectomy (LC), and classified as ASA grade I–II. Exclusion criteria comprised obese patients (BMI ≥25 kg/m²), those with a history of alcohol or drug abuse, uncontrolled hypertension or diabetes, chronic pain, allergy to study drugs, neurological disorders, inability to understand the pain scoring system, or procedures converted to open cholecystectomy.
A sample size of 38 patients per group was estimated, taking pain scores of 2.47 ± 1.5 and 1.40 ± 1.8 in the lignocaine and the ketamine group, respectively16 at a power of 80% and 95% confidence interval. Sample size calculation was performed using an online calculator, Open-epi. For better results, 50 patients were enrolled per arm. The consecutive sampling technique was used to enroll participants into the study, whereas group allocation was done in a random fashion using sequentially numbered opaque sealed envelopes.14 Patients receiving ketamine were labeled as Group K, whereas patients receiving lignocaine were labeled as Group L.
Patients scheduled for LC were admitted a day before surgery for pre-anesthesia assessment, during which informed consent was obtained and patients were educated about the pain scale. After completing the assessment checklist, they were moved to the holding area and randomly assigned to either Group K or Group L. Patients were randomly assigned to study groups using a sequentially numbered opaque sealed envelope (SNOSE) protocol.17 Both patients and study investigators were blinded to the study groups. Only the anesthetist who prepared the medication and was not part of this study was aware of group identification.
After transferring the patient to the operating room, standard monitors were attached, and vital signs, including blood pressure, heart rate, and mean arterial pressure, were continuously monitored. Electrocardiography (ECG) was also performed as part of the standard anesthesia protocol. Anesthesia induction included premedication with nalbuphine 0.1 mg/kg, propofol 2 mg/kg, atracurium 0.5 mg/kg, and ondansetron 0.15 mg/kg. Just before the skin incision, Group K received ketamine 0.2 mg/kg as an intravenous bolus, followed by an infusion at 0.2 mg/kg/hr throughout the surgery, while Group L received lignocaine 1 mg/kg as an intravenous bolus, followed by an infusion at 1 mg/kg/hr. Anesthesia was maintained with 100% oxygen at 3 L/min without any carrier gas, along with isoflurane (1–1.5%), atracurium 0.1 mg/kg, and paracetamol 10 mg/kg intravenously. After procedure completion, all patients were shifted to post-operative recovery room. They were observed in the recovery unit with continuous monitoring of vital signs, including heart rate, blood pressure, oxygen saturation, and respiratory rate. Routine resuscitation equipment and emergency medications were available at the bedside, including provisions for treatment of potential adverse effects such as bradycardia, hypotension, arrhythmias, and emergence reactions. The anesthetic team was available throughout the recovery phase for immediate intervention should it be required. Patients stayed there for the next 2 hours and then were shifted to the ward and discharged after 24 hours if found stable as per hospital protocol.
Post-operatively, pain status was continuously monitored using the Numeric Rating Scale (NRS) at 0 hours, 2 hours, 4 hours, 6 hours, 12 hours, and then finally at 24 hours. As part of multi-model analgesia, paracetamol 10 mg/kg was administered post-operatively at 8-hour intervals as per routine protocol. Additional analgesia was administered to patients upon demand or if their pain score was 5 or higher. For rescue analgesia, an injection of tramadol 30 mg was administered. The assigned residents collected the data from time to time and recorded it in the study proforma.
The NRS is a ten-point scoring scale used for pain assessment. It ranges from 0 to 10 for ranking pain, where 0 means no pain and 10 means the worst pain.18 Time to rescue analgesia was defined as the time point at which the patient would be given the first rescue analgesia for a pain score of 5 or above. The time between the incision and the wound's closure was used to define the length of the surgery.
The collected data were put into the statistical package SPSS version 26 to perform data analysis. Frequencies and percentages were computed for categorical variables. Numerical variables were expressed as mean ± standard deviation or median with inter-quartile range (IQR) based on the assumption of normality that was tested using the Shapiro-Wilk test. Categorical variables were compared using the Chi-square or Fisher’s exact test, while numerical variables were analyzed using the independent t-test or Mann-Whitney U test, as appropriate. A P-value ≤ 0.05 was considered statistically significant.
3. RESULTS
A total of 100 patients were studied with a 1:1 ratio allocation in study groups. Table 1 displays patients’ sociodemographic and baseline features among the two groups. The two groups did not differ based on age (P = 0.398), gender (P = 0.542), hypertension (P = 0.137), diabetes (P = 0.473), ASA grade (P = 0.537), or surgery duration (P = 0.334). At baseline, none of the patients’ vitals, including systolic blood pressure (P = 0.303), diastolic blood pressure (P = 0.342), heart rate (P = 0.173), and mean arterial pressure (P = 0.157), were significantly different between the two groups. At the end of surgery, the two groups did not differ based on systolic blood pressure (P = 0.679), diastolic blood pressure (P = 0.261), heart rate (P = 0.261), and mean arterial pressure (P = 0.340).
| Table 1: Patients’ sociodemographic and baseline features among the two groups. | ||||
| Variables | Group A
(n = 50) |
Group B
(n = 50) |
P-value | |
| Demographics | Age (years)a | 37.9 ± 3.7 | 38.7 ± 5.5 | 0.398 |
| GenderbMale | 19 (38) | 22 (44) | 0.542 | |
| Female | 31 (62) | 28 (56) | ||
| Body mass indexa | 23.2 ± 2.1 | 23.3 ± 1.9 | 0.675 | |
| Comorbidityb | Controlled Hypertension | 4 (8) | 9 (18) | 0.137 |
| Diabetes | 2 (4) | 8 (16) | 0.473 | |
| ASA gradeb | ASA-I | 37 (74) | 29 (58) | 0.091 |
| ASA-II | 13 (26) | 21 (42) | ||
| Surgery duration (min)a | 72.4 ± 14.7 | 73.7 ± 13.7 | 0.841 | |
| Baseline hemodynamic features | Systolic blood pressure (mmHg)c | 131 (130-133) | 133 (128.5-140) | 0.303 |
| Diastolic blood pressure (mmHg)c | 86 (80-90) | 80 (79-87) | 0.342 | |
| Heart rate (BPM)c | 84 (74.8-90.3) | 79 (70-81.5) | 0.173 | |
| Mean arterial pressure (mmHg)c | 103 (96-106.3) | 98 (95-103) | 0.157 | |
| Hemodynamics at the end of surgery | Systolic blood pressure (mmHg)c | 133 (131-136) | 133 (131-135.3) | 0.679 |
| Diastolic blood pressure (mmHg)c | 83 (80.8-85) | 83 (81-85) | 0.488 | |
| Heart rate (BPM)c | 85 (81.8-86) | 84 (80-86) | 0.261 | |
| Mean arterial pressure (mmHg)c | 102 (96.8-106) | 101 (98-109) | 0.340 | |
| a: Normally distributed numerical data is expressed as mean ± SD, b: Categorical data is represented as n (%), c: Non-normal numerical data is presented as median (Q3 – Q1), *Significant at P < 0.05 | ||||
Figure 1 compares patients’ pain status from 0-24 hours post-operatively. Mean score was not significantly different in Group K and Group L at 0 hour ( P = 0.395), 2nd hour (P = 0.847), 4th hour (P = 0.872), 6th hour (P = 1.000), and 12th hour (P = 0.714). Mean pain score was found to be significantly different between Group K and Group L at 24th hour (P < 0.001).

Figure 1: Comparison of mean pain score between the two groups at different time intervals post-operatively
Table 2 compares analgesic status among the two study groups. Time to first rescue analgesia and analgesic consumption did not significantly differ between the two study groups.
| Table 2: Analgesic status and hospital stay among the two study groups. | |||
| Variables | Group A
(n = 50) |
Group B
(n = 50) |
P-value |
| Time for first rescue analgesia (in hours)a | 5.0 ± 3.1 | 4.7 ± 2.2 | 0.605 |
| Total number of analgesic doses | 1.1 ± 0.5 | 1.04 ± 0.2 | 0.060 |
| a: Normally distributed numerical data is expressed as mean ± SD, *Significant at P < 0.05 | |||
4. DISCUSSION
Postoperative pain influences complications, patients’ satisfaction, and overall recovery, so managing pain following surgical procedures is a challenging aspect for an anesthetist. In this study, both lignocaine and ketamine were evaluated for their analgesic properties and comparative effects. Ketamine, a well-known NMDA receptor antagonist, is recognized for its analgesic and opioid-sparing effects.19 Contrarily, lignocaine is a local anesthetic drug that exerts analgesic properties on intravenous administration and lowers pain via blocking sodium channels and has an anti-inflammatory impact.20
In the current study, low-dose ketamine and lignocaine are compared for their pain control effects among patients who underwent LC. The findings demonstrated that pain status was similar among both the study groups during 0-12 hours post-operatively. However, the lower pain scores were seen in the ketamine group at 24 hours post-operatively in comparison to lignocaine, suggesting a prolonged analgesic effect of ketamine in modulating pain associated with surgical trauma. The significantly lower pain score for ketamine at 24 hours also highlights that NMDA receptor blockade plays a role in diminishing long-term nociceptive responses. This finding is favorable for the hypothesis that low-dose ketamine lowers long-term pain and enhances pain control beyond the immediate post-operative duration.21 Moreover, the anti-inflammatory properties of ketamine also contribute to prolonged analgesic effects while reducing cytokine-mediated nociceptive responses.22
Previous research has demonstrated that low-dose ketamine alone provides effective analgesia and reduces opioid consumption postoperatively.23-25 Some studies have also reported that ketamine’s analgesic effects are particularly beneficial in the late postoperative period with different routes due to its ability to prevent central sensitization and hyperalgesia.9,26 However, when comparison of ketamine was made with lignocaine, results were variable. Singariya G, et al. (2023) analyzed that pain scores for ketamine and lignocaine were the same at 0 hours and 24 hours.9 Between 0-24 hours, pain scores were found to be lower in the ketamine group during the early post-operative period, 1-6 hours, which gradually increased and matched the pain levels in the lignocaine group. However, our findings are in agreement with the study of Toleska et al, (2022), who also reported effective pain control in the ketamine group at 24 hours than the lignocaine group (1.40 ± 1.8 versus 2.47 ± 1.5).16 Gesso AS et al. (2022) compared pain incidences between low-dose ketamine and lignocaine groups among all elective surgical procedures and reported that low-dose ketamine was superior to lignocaine in lowering pain, with low pain incidences in patients receiving low-dose ketamine.15
Although our study did not find long-term impacts of lignocaine in pain control, the literature documents its analgesic properties in different surgical procedures. Some studies report that lignocaine infusion diminishes opioid need and hastens recovery, mainly in abdominal procedures.27,28 The primary mechanism of lignocaine is based on modulating acute inflammatory responses and inhibiting ectopic discharges in injured nerves.29 We believe that because of this mechanism, which diminishes systemic effects with time, its analgesic advantages were not seen beyond the immediate post-operative period.
The clinical importance of this study is based on the potential function of ketamine in improving postoperative recovery and reducing opioid use. In recent years, the hunt for alternative analgesic techniques has been intensified due to opioid-associated side effects, including nausea, vomiting, and respiratory depression. Because of the well-documented opioid-sparing effects of ketamine, it has been increasingly recommended as part of multimodal analgesia protocols.30,31 Since patients receiving laparoscopic cholecystectomy frequently endure moderate post-operative pain, adding ketamine to pain management protocols may enhance patient comfort and decrease dependence on opioid analgesics.
Moreover, we found that total analgesic consumption and time to first rescue analgesia were not considerably different in Group K and Group L. This finding indicates that ketamine played a greater role in controlling late postoperative pain at 24 hours compared to lignocaine; however, it did not affect the overall need for additional analgesics, as no significant difference was observed between the groups during the immediate postoperative period (before 24 hours).
5. LIMITATIONS
Despite the positive results, this study has some important limitations. First, pain assessment was performed at specific time intervals, and continuous monitoring could have provided a more comprehensive evaluation of the analgesic effects of the study drugs. Second, the side effects of ketamine, such as hallucinations or dysphoria, were not assessed. Third, hemodynamic stability was not compared between the two groups. Although low-dose ketamine is generally well-tolerated, future studies should investigate its safety profile during the postoperative period for further validation.
6. CONCLUSION
The findings suggested that both low-dose ketamine and lignocaine had similar pain-relieving effects in the early postoperative period. However, ketamine was found to be superior to lignocaine at 24 hours in terms of lower pain score.
- Data availability
- Conflict of interest
- Authors’ contribution
11. REFERENCES
- Gan TJ, Epstein RS, Leone-Perkins ML, Salimi T, Iqbal SU, Whang PG. Practice Patterns and Treatment Challenges in Acute Postoperative Pain Management: A Survey of Practicing Physicians. Pain Ther. 2018;7(2):205-216. PMCID: PMC6251830 DOI: 10.1007/s40122-018-0106-9
- Gan TJ, Habib AS, Miller TE, White W, Apfelbaum JL. Incidence, patient satisfaction, and perceptions of post-surgical pain: results from a US national survey. Curr Med Res Opin. 2014;30:149–160. DOI: 10.1185/03007995.2013.860019
- Willingham M, Rangrass G, Curcuru C, Abdallah AB, Wildes TS, McKinnon S, Kronzer A, Sharma A, Helsten D, Hall B, Avidan MS. Association between postoperative complications and lingering post-surgical pain: an observational cohort study. Br J Anaesth. 2020;124(2):214-21. DOI: 10.1016/j.bja.2019.10.012
- Komann M, Baumbach P, Stamer UM, Weinmann C, Arnold C, Pogatzki-Zahn E, et al. Desire to Receive More Pain Treatment - A Relevant Patient-Reported Outcome Measure to Assess Quality of Post-Operative Pain Management? Results From 79,996 Patients Enrolled in the Pain Registry QUIPS from 2016 to 2019. J Pain. 2021;22(6):730-738. DOI: 10.1016/j.jpain.2021.01.002
- Gupta A, Bah M: NSAIDs in the treatment of postoperative pain. Curr Pain Headache Rep. 2016, 20:62. DOI: 10.1007/s11916-016-0591-7
- Yu JM, Tao QY, He Y, Liu D, Niu JY, Zhang Y. Opioid-Free Anesthesia for Pain Relief After Laparoscopic Cholecystectomy: A Prospective Randomized Controlled Trial. J Pain Res. 202330;16:3625-3632. PMCID: PMC10624184 DOI: 10.2147/JPR.S432601
- Imani F, Rahimzadeh P, Faiz H, Nowruzina S, Shakeri A, Ghahremani M. Comparison of the Post-Caesarean Analgesic Effect of Adding Dexmedetomidine to Paracetamol and Ketorolac: A Randomized Clinical Trial. Anesth Pain Med. 2018;8(5):e85311. PMCID: PMC6252045 DOI: 10.5812/aapm.85311
- White PF. The Changing Role of Non-Opioid Analgesic Techniques in the Management of Postoperative Pain. Anesth Analg. 2005;101(5S):S5–S22.
- Singariya G, Sharma J, Kumari K, Kamal M, Khatri C, Selvin CC. Comparison of the effects of intravenous lignocaine and ketamine on postoperative pain after lower abdominal surgeries under general anaesthesia. Indian J Anaesth. 2023 Feb;67(Suppl 1):S41-S47. PMCID: PMC10104105 DOI: 10.4103/ija.ija_691_22
- Kranke P, Jokinen J, Pace NL, Schnabel A, Hollmann MW, Hahnenkamp K, et al. Continuous intravenous perioperative lidocaine infusion for postoperative pain and recovery. Cochrane Database Syst Rev. 2015;7:CD009642.
- Radvansky BM, Shah K, Parikh A, Sifonios AN, Le V, Eloy JD. Role of ketamine in acute postoperative pain management: A narrative review. Biomed Res Int. 2015;2015:749837. PMCID: PMC4606413 DOI: 10.1155/2015/749837
- Imani F, Bagheri AR, Arvin E, Gatt SP, Sarveazad A. Effects of Ketamine and Lidocaine Infusion on Acute Pain after Elective Open Abdominal Surgery, a Randomized, Double-Blinded Study. Med J Islam Repub Iran. 2022;36:60. PMCID: PMC9448502 DOI: 10.47176/mjiri.36.60
- Jendoubi A, Naceur IB, Bouzouita A, Trifa M, Ghedira S, Chebil M, Houissa M. A comparison between intravenous lidocaine and ketamine on acute and chronic pain after open nephrectomy: A prospective, double-blind, randomized, placebo-controlled study. Saudi J Anaesth. 2017;11(2):177-184. PMCID: PMC5389236 DOI: 10.4103/1658-354X.203027
- Fahad Najam M, Jafri N. A Comparison Between the Effectiveness of Ketamine Bolus and Intradermal Lidocaine in Reducing Acute Postoperative Pain. Cureus. 2022;14(7):e26563. PMCID: PMC9349481 DOI: 10.7759/cureus.26563
- Gesso AS, Yuya SA, Fekede MS, Delile ST, Asrat EB. Effectiveness of low-dose ketamine versus lignocaine pre-treatment to prevent propofol injection pain and hemodynamic instability in adult elective surgical patients at Tikur Anbessa specialized hospital, 2022: A prospective observational cohort study. Int J Surg Open. 2022;47:100537.
- Toleska M, Dimitrovski A, Shosholcheva M, Kartalov A, Kuzmanovska B, Dimitrovska NT. Pain and multimodal analgesia in laparoscopic cholecystectomy. prilozi. 2022 Jul 13;43(2):41-9. DOI: 10.2478/prilozi-2022-0017
- Clark L, Dean A, Mitchell A, Torgerson DJ. Envelope use and reporting in randomised controlled trials: a guide for researchers. Research Methods in Medicine & Health Sciences. 2021 Jan;2(1):2-11.
- Breivik H, Borchgrevink PC, Allen SM, et al. Assessment of pain. Br J Anaesth 2008;101(1):17–24.
- Ma X, Yan J, Jiang H. Application of Ketamine in Pain Management and the Underlying Mechanism. Pain Res Manag. 2023 Aug 16;2023:1928969. PMCID: PMC10447145 DOI: 10.1155/2023/1928969
- Yang X, Wei X, Mu Y, Li Q, Liu J. A review of the mechanism of the central analgesic effect of lidocaine. Medicine (Baltimore). 2020 Apr;99(17):e19898. PMCID: PMC7440315 DOI: 10.1097/MD.0000000000019898
- Riccardi A, Guarino M, Serra S, Spampinato MD, Vanni S, Shiffer D, Voza A, Fabbri A, De Iaco F; Study and Research Center of the Italian Society of Emergency Medicine. Narrative Review: Low-Dose Ketamine for Pain Management. J Clin Med. 2023 May 2;12(9):3256. PMCID: PMC10179418 DOI: 10.3390/jcm12093256 .
- Richards ND, Howell SJ, Bellamy MC, Beck J. The diverse effects of ketamine, a jack-of-all-trades: a narrative review. Br J Anaesth. 2025:S0007-0912(24)00695-0. PMCID: PMC11867090 DOI: 10.1016/j.bja.2024.11.018
- Jain, S., Nazir, N. and Mustafi, S.M., 2022. Preemptive low-dose intravenous ketamine in the management of acute and chronic postoperative pain following laparoscopic cholecystectomy: a prospective randomized control study. Medical gas research, 12(4), pp.141-145.
- Zhao L, Li Z, Jin B, Hou N, Yang H. Safety and efficacy of low-dose esketamine in laparoscopic cholecystectomy: a prospective, double-blind randomized controlled trial. BMC anesthesiology. 2024 Feb 1;24(1):47. PMCID: PMC10832235 DOI: 10.1186/s12871-024-02429-5
- Sudersan S, Thangavelu R, Segaran S. Intraoperative low-dose ketamine infusion for postoperative pain relief in laparoscopic surgeries–A randomised, double blind controlled clinical trial. Journal of Current Research in Scientific Medicine. 2024 Jan 1;10(1):67-73. DOI: 10.4103/jcrsm.jcrsm_130_23
- Xu B, Wang Y, Zeng C, Wei J, Li J, Wu Z, et al. Analgesic efficacy and safety of ketamine after total knee or hip arthroplasty: a meta-analysis of randomised placebo-controlled studies. BMJ Open. 2019;9(9):e028337. PMCID: PMC6747636 DOI: 10.1136/bmjopen-2018-028337
- Weibel S, Jokinen J, Pace NL, Schnabel A, Hollmann MW, Hahnenkamp K et al. Efficacy and safetyof intravenous lidocaine for postoperative analgesia and
- Chu R, Umukoro N, Greer T, Roberts J, Adekoya P, Odonkor CA, Hagedorn JM, Olatoye D, Urits I, Orhurhu MS, Umukoro P, Viswanath O, Hasoon J, Kaye AD, Orhurhu V. Intravenous Lidocaine Infusion for the Management of Early Postoperative Pain: A Comprehensive Review of Controlled Trials. Psychopharmacol Bull. 2020 Oct 15;50(4 Suppl 1):216-259. PMCID: PMC7901134 DOI: 10.64719/pb.4391
- Hermanns H, Hollmann MW, Stevens MF, Lirk P, Brandenburger T, Piegeler T, et al. Molecular mechanisms of action of systemic lidocaine in acute and chronic pain: a narrative review. Br J Anaesth. 2019;123(3):335-349. DOI: 10.1016/j.bja.2019.06.014
- Chaouch MAMA, Daghmouri MA, Boutron MC, Ferraz JM, Usai S, Soubrane O, et al. Ketamine as a component of multimodal analgesia for pain management in bariatric surgery: A systematic review and meta-analysis of randomized controlled trials. Ann Med Surg (Lond). 2022 May 14;78:103783. PMCID: PMC9121244 DOI: 10.1016/j.amsu.2022.103783
- Stojanović M, Marinković M, Miličić B, Stojičić M, Jović M, Jovanović M, et al. The Role of Ketamine as a Component of Multimodal Analgesia in Burns: A Retrospective Observational Study. J Clin Med. 2024;13(3):764. PMCID: PMC10856234 DOI: 10.3390/jcm13030764