Perisetty Brahmani 1 , Prabhu Thilaak 2 , Senthil Marappan 3 , Brindha Rathnasabapathy 4 , Raja Ganapathy 5*, Arbind Kumar Choudhary 6 , Panneerselvam Periasamy 7
Authors affiliations:
Background & objective: Managing preoperative anxiety and ensuring cooperation in children undergoing surgery remains a key concern in pediatric anesthesia. Nebulized premedication offers a non-invasive method that may enhance comfort and compliance. This study aimed to compare the effects of nebulized dexmedetomidine and midazolam on sedation, anxiety reduction, cooperation, and recovery in pediatric patients scheduled for elective surgical procedures.
Methodology: A randomized, double-blind clinical trial was conducted involving 100 children aged between 5 and 12 years with ASA physical status I or II. Participants were randomly assigned to receive either dexmedetomidine (2 mcg/kg) or midazolam (0.2 mg/kg) via nebulization, administered 20 minutes before induction. Sedation and anxiety were assessed before and after drug administration. Additional outcomes included acceptance of the nebulized medication and anesthesia mask, intraoperative hemodynamic trends, postoperative agitation, recovery duration, and any adverse effects.
Results: Children in the dexmedetomidine group exhibited significantly deeper sedation and lower anxiety scores following nebulization (P < 0.001). While medication acceptance was similar in both groups, mask acceptance was better in the dexmedetomidine group (P < 0.001). Intraoperative monitoring showed more stable heart rate and blood pressure in children premedicated with dexmedetomidine. Emergence agitation was notably lower (10% vs. 34%, P = 0.002), and recovery was faster in the dexmedetomidine group (mean 22.4 ± 4.3 minutes vs. 26.9 ± 5.1 minutes, P = 0.003). No significant adverse events were reported.
Conclusions: Nebulized dexmedetomidine is more effective than midazolam for pediatric premedication, providing better sedation, improved cooperation, hemodynamic stability, and quicker recovery. Its non-invasive route and favorable safety profile make it a promising choice for routine use in children undergoing elective surgeries.
Keywords: Dexmedetomidine; Midazolam; Pediatric anesthesia; Nebulization; Sedation; Emergence agitation
Citation: Brahmani P, Thilaak P, Marappan S, Rathnasabapathy B, Ganapathy R, Choudhary AK, Periasamy P. A prospective randomized double-blind study of nebulised dexmedetomidine versus midazolam for pediatric premedication. Anaesth. pain intensive care 2025;29(8):850-860. DOI: 10.35975/apic.v29i8.3006.
Received: June 17, 2025; Revised: July 29, 2025; Accepted: July 30, 2025
Preoperative anxiety is a prevalent concern in pediatric patients undergoing surgery and can lead to significant physiological and psychological disturbances if not adequately addressed. Heightened anxiety in children often triggers autonomic responses such as tachycardia, hypertension, increased catecholamine release, and elevated anesthetic requirements.1,2 Beyond the immediate intraoperative challenges, untreated anxiety may contribute to postoperative maladaptive behaviors including sleep disturbances, separation anxiety, and emergence delirium.3 Therefore, optimizing preoperative anxiolysis is essential to ensure smoother induction, hemodynamic stability, and better overall perioperative outcomes.
Midazolam, a short-acting benzodiazepine, is widely used in pediatric anesthesia for its sedative, anxiolytic, and amnestic properties.4 Despite its widespread use, midazolam poses several challenges when administered orally or intravenously. Oral midazolam often has a bitter taste, variable bioavailability, and a relatively delayed onset of action.5 Intravenous administration, while more predictable, is invasive and can provoke anxiety in children. Furthermore, paradoxical reactions such as agitation and restlessness have been reported, particularly in younger age groups.6
To overcome these limitations, alternative agents and non-invasive delivery methods are being increasingly explored. Dexmedetomidine, a highly selective α2-adrenergic agonist, has emerged as a promising alternative due to its ability to provide cooperative sedation, anxiolysis, and mild analgesia with minimal respiratory depression.7,8 Unlike benzodiazepines, dexmedetomidine exerts a sympatholytic effect, promoting hemodynamic stability during the perioperative period. It has demonstrated efficacy when administered via intravenous, intranasal, and buccal routes, and more recently through inhalational and nebulized methods.9
The nebulized route offers several advantages in the pediatric population. It is non-invasive, well-tolerated, and can facilitate faster drug absorption through the rich vascular network of the pulmonary mucosa, bypassing first-pass metabolism.10 This method may be particularly beneficial in uncooperative or needle-phobic children. Although the use of nebulized dexmedetomidine is supported by several clinical studies, it remains underutilized in routine practice.11 Additionally, direct comparisons between nebulized dexmedetomidine and midazolam are limited, especially within the Indian context.
This prospective, randomized, double-blind clinical study was conducted to compare the efficacy of nebulized dexmedetomidine and midazolam in children aged 5–12 years undergoing elective surgeries under general anesthesia. The primary objectives were to assess preoperative sedation levels, anxiety at parental separation, and acceptance of medication and anesthesia mask. Secondary outcomes included intraoperative hemodynamic parameters, emergence agitation, recovery time, and adverse effects. Through this investigation, we aim to provide evidence on the clinical utility of nebulized premedication in pediatric anesthesia and identify the agent that offers the best balance of efficacy, safety, and patient acceptability.12
This was a prospective, randomized, double-blind, comparative clinical trial conducted over 12 months in the Department of Anaesthesiology at Vinayaka Mission's Kirupananda Variyar Medical College and Hospitals, Vinayaka Missions Research Foundation (VMRF), Salem, Tamil Nadu, India. The study received ethical clearance from the Institutional Ethics Committee (Approval No: VMKVMC&H IEC/22/113), and written informed consent was obtained from all parents or legal guardians. Assent was also obtained from children aged 7 years and older, in accordance with institutional policy.
Sample size was calculated based on a pilot study and previous literature comparing sedation scores between dexmedetomidine and midazolam. Assuming a power of 80%, a Type I error (α) of 0.05, and a minimum detectable difference of 0.5 in sedation score, it was estimated that 45 participants per group would be sufficient. To account for potential dropouts, 50 patients were included in each group, a total of 100 children.
Children aged 5–12 years with American Society of Anesthesiologists (ASA) physical status I or II, scheduled for elective surgery under general anesthesia, were eligible. Exclusion criteria included known allergy to study drugs, upper respiratory tract infections, cardiovascular or neurological comorbidities, congenital airway anomalies, or emergency surgeries.
Participants were scheduled for a range of minor to intermediate elective procedures, including herniotomy, circumcision, and adenotonsillectomy. The average duration of surgery across both groups ranged from 30 to 45 minutes. Types and durations were evenly distributed between groups to minimize procedural bias influencing recovery outcomes.
Randomization was performed using computer-generated sequences and sealed opaque envelopes to ensure allocation concealment. Children were assigned to one of two groups: Group A received nebulized dexmedetomidine at 2 mcg/kg, and Group B received nebulized midazolam at 0.2 mg/kg. Both drugs were diluted with normal saline to a final volume of 3 mL and administered via a pediatric jet nebulizer with a facemask approximately 20 minutes before anesthesia induction. The study maintained a double-blind design, with drug preparation carried out by an independent anesthesiologist not involved in patient assessment, while both the administering clinician and the evaluator remained unaware of group allocation. Dexmedetomidine (Dexem, Neon Laboratories Ltd., India) and midazolam (Mezolam, Abbott Healthcare Pvt. Ltd., India) were used for the study.
Primary outcomes included the sedation level and anxiety score at the time of separation from parents. Sedation was assessed using the Modified Ramsay Sedation Scale1, while anxiety was evaluated using a validated 4-point behavioral scale2. Secondary outcomes included acceptance of nebulized medication and mask application using a 4-point cooperation scale, intraoperative hemodynamic parameters (heart rate, systolic and diastolic blood pressures), emergence agitation assessed with the Pediatric Anesthesia Emergence Delirium (PAED) Scale3, recovery time defined as the duration from cessation of anesthesia to achieving a Modified Aldrete Score ≥ 9, and any adverse events such as bradycardia, hypotension, nausea, vomiting, or respiratory difficulty. Sedation and anxiety scores were measured at two time points: immediately before nebulization and approximately 20 minutes after drug administration, just prior to anesthesia induction. Following premedication, anesthesia was induced using sevoflurane in 100% oxygen via facemask, and intravenous access was obtained after loss of consciousness. Standard intraoperative monitoring included non-invasive blood pressure, pulse oximetry, ECG, and capnography, with hemodynamic parameters recorded at baseline, post-nebulization, and every 5 minutes for the first 20 minutes of surgery.
2.2. Statistical Analysis
Data were analyzed using IBM SPSS version 23.0. Continuous variables were expressed as mean ± standard deviation (SD) and analyzed using the independent t-test or Mann–Whitney U test. Categorical variables were compared using Chi-square or Fisher’s exact test as appropriate. A p-value of <0.05 was considered statistically significant.
A total of 100 children aged 5–12 years were enrolled and randomized equally into two groups. The demographic and baseline clinical characteristics of both groups were statistically comparable, as shown in Table 1, ensuring that observed outcome differences were likely due to the interventions rather than pre-existing variability.
There were no significant differences between the two groups regarding age, sex distribution, anthropometric measurements, or ASA classification (P > 0.05 for all). This confirms that the study groups were well balanced at baseline, minimizing confounding in outcome assessments.
Sedation and anxiety were assessed using validated clinical scales both before and after nebulized drug administration. Sedation was evaluated using the Modified Ramsay Sedation Scale, while anxiety at parental separation was scored using a 4-point behavioural scale. Table 2 presents a comparative analysis of these scores between the two study groups.
There was no significant difference in sedation or anxiety scores between the groups at baseline (P > 0.05). Post-nebulization, Group A (dexmedetomidine) demonstrated significantly deeper sedation and greater anxiety reduction than Group B (P < 0.001), indicating superior preoperative calming effects. These findings highlight the enhanced efficacy of dexmedetomidine in managing pre-induction stress in pediatric patients.
Cooperation during drug administration and induction of anesthesia is critical in pediatric practice. In this study, children's behavioral responses to nebulized drug delivery and mask placement were evaluated using a 4-point Likert scale, where lower scores indicated better cooperation. The results are detailed in Table 3.
Both groups demonstrated similar levels of cooperation during administration of the nebulized premedication. In Group A (dexmedetomidine), 40% of children accepted the medication calmly (Grade 1 – Excellent), compared to an identical 40% in Group B (midazolam). The distribution of acceptance scores was comparable between the groups, with no statistically significant difference (P = 0.734). The median acceptance score in both groups was 2 [IQR: 1–2], indicating generally good cooperation.
Mask acceptance showed a significant difference between groups. In Group A, 72% of children were rated as “Excellent” (Grade 1) in mask acceptance following dexmedetomidine, compared to only 30% in the midazolam group. Group A also had a significantly lower median mask acceptance score (1 [1–2]) and lower mean score (1.42 ± 0.67), indicating better behavioral compliance. This difference was statistically significant (P < 0.001), suggesting that dexmedetomidine produced more effective pre-induction sedation and reduced anxiety, leading to smoother transitions.
While both drugs were similarly accepted in nebulized form, dexmedetomidine significantly improved cooperation during mask application. This reflects superior sedation depth and reduced anxiety at separation, making dexmedetomidine a more effective agent for achieving a calm anesthetic induction in pediatric patients.
Hemodynamic parameters were recorded at five-time intervals: baseline (pre-nebulization), and at 5, 10, 15, and 20 minutes post-nebulization. The mean heart rate (HR) and systolic blood pressure (SBP) values for each group across these time points are illustrated in Figure 1. In Group A (dexmedetomidine), HR showed a gradual and consistent decline from a baseline of 98 bpm to 88 bpm at 20 minutes. In contrast, Group B (midazolam) exhibited only a slight decrease in HR from 97 bpm to 94 bpm, with relatively higher values throughout. Similarly, SBP in Group A steadily decreased from 110 mmHg at baseline to 104 mmHg by 20 minutes, whereas Group B showed a more modest reduction from 111 mmHg to 107 mmHg. These hemodynamic trends were more pronounced and stable in the dexmedetomidine group, indicating a more potent sympatholytic effect and improved cardiovascular modulation. Despite the reductions, all values remained within normal pediatric physiological limits, and no adverse cardiovascular events were recorded in either group.
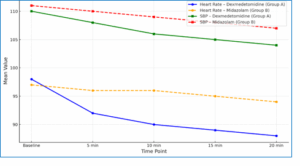
Figure 1: Trends in Heart rate and systolic blood pressure over time
Dexmedetomidine resulted in a more predictable and gradual decline in both HR and SBP compared to midazolam. These findings support dexmedetomidine’s superior autonomic modulation and reinforce its role as a hemodynamically stable sedative in pediatric premedication.
Hemodynamic stability was assessed by comparing trends in heart rate (HR) and systolic blood pressure (SBP) at baseline and at 5-minute intervals up to 20 minutes post-nebulization. Both parameters were measured non-invasively and recorded in all patients. As shown in Table 4, HR in Group A (dexmedetomidine) decreased progressively from 98 to 88 bpm, compared to a milder reduction from 97 to 94 bpm in Group B (midazolam). Similarly, SBP dropped from 110 to 104 mmHg in Group A, versus 111 to 107 mmHg in Group B. These differences became statistically significant after the first 5 minutes and remained so throughout the observation period (P < 0.05 at each subsequent time point). The smoother decline in Group A illustrates the superior sympatholytic effect of dexmedetomidine. However, all values remained within acceptable clinical ranges, and no intervention was required for bradycardia or hypotension in either group.
Dexmedetomidine produced a significantly greater and steadier reduction in both HR and SBP compared to midazolam beginning from the 5-minute mark. These findings suggest a more predictable and beneficial cardiovascular response, reinforcing dexmedetomidine’s utility in pediatric premedication protocols.
Sedation and anxiety levels were assessed before and after nebulization using standardized behavioral scales. The Modified Ramsay Sedation Scale and a 4-point Behavioral Anxiety Scale were employed to quantify the depth of sedation and the degree of separation-related anxiety, respectively. Results are illustrated in Figure 2.
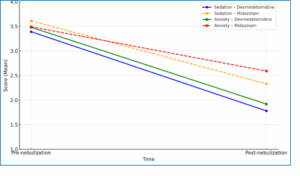
Figure 2: Sedation and anxiety score comparison before and after nebulization
At baseline, sedation and anxiety scores were comparable between the two groups (p > 0.05). Post-nebulization, Group A (dexmedetomidine) exhibited a significantly greater reduction in both sedation and anxiety scores compared to Group B (midazolam). The mean post-nebulization sedation score in Group A was 1.78 ± 0.40 versus 2.33 ± 0.35 in Group B (P < 0.001). Similarly, anxiety scores dropped to 1.92 ± 0.38 in Group A and 2.59 ± 0.49 in Group B (P < 0.001).
These findings suggest that nebulized dexmedetomidine provides more effective preoperative sedation and anxiolysis in pediatric patients undergoing elective surgery. Dexmedetomidine resulted in significantly deeper sedation and greater anxiety reduction post- nebulization compared to midazolam, affirming its superior efficacy for pediatric premedication.
Postoperative recovery was assessed using two primary parameters: recovery time (defined as the time from cessation of anesthesia to meeting discharge criteria) and emergence delirium, evaluated using the Pediatric Anesthesia Emergence Delirium (PAED) scale. Additionally, adverse events such as nausea, vomiting, bradycardia, and hypotension were documented and compared between groups. As shown in Table 5, Group A (dexmedetomidine) had a significantly shorter mean recovery time (22.4 ± 4.8 minutes) compared to Group B (midazolam) (26.9 ± 5.3 minutes; P = 0.002). Emergence delirium, defined as a PAED score ≥10, was observed in only 2 patients (4%) in Group A versus 7 patients (14%) in Group B, which was statistically significant (p = 0.048). Minor adverse effects were reported in both groups, but the difference was not statistically significant. Nausea and vomiting were slightly more common in Group B (6%) than in Group A (2%). Bradycardia occurred in 4% of patients in Group A versus 2% in Group B, while hypotension was rare in both groups. No life-threatening or serious complications were recorded.
Children in the dexmedetomidine group experienced faster recovery, less emergence delirium, and similar or fewer minor side effects compared to those in the midazolam group. These outcomes affirm dexmedetomidine’s efficacy and safety as a pediatric premedication agent for elective surgeries.
Figure 3 presents a side-by-side comparison of behavioral cooperation during preoperative care and postoperative recovery outcomes across the two groups. The panel on the left illustrates mean scores for medication and mask acceptance, while the right panel shows differences in emergence agitation and recovery time.
In the postoperative phase, Group A demonstrated a lower emergence agitation score (median: 1.5) and shorter recovery time (22.4 minutes) compared to Group B (agitation: 2.5; recovery time: 26.9 minutes). These findings emphasize the superior recovery profile associated with dexmedetomidine, including smoother emergence and more efficient return to baseline consciousness.
The combined behavioral and recovery outcomes strongly favor dexmedetomidine as a more effective and child-compliant premedication agent in pediatric anesthesia. Its advantages in both preoperative cooperation and postoperative stability make it a valuable option for enhancing patient experience and clinical workflow. These data reinforce the role of dexmedetomidine in settings where anxiolysis, cooperation, and smooth emergence are paramount.
This randomized controlled study evaluated the comparative performance of nebulized dexmedetomidine and midazolam as premedication agents in pediatric patients scheduled for elective surgeries. The outcomes clearly demonstrated that dexmedetomidine led to more favorable preoperative sedation, improved patient cooperation, better intraoperative cardiovascular control, and smoother postoperative recovery compared to midazolam.21,22
Effective anxiolysis prior to surgery is essential in children to ensure a smooth induction process and to reduce perioperative psychological stress. In this study, nebulized dexmedetomidine provided deeper sedation and significantly reduced preoperative anxiety levels compared to nebulized midazolam. These effects likely stem from its dual action on α2-adrenergic receptors, contributing both sedative and anxiolytic properties without respiratory depression. While oral and intranasal routes have been studied extensively, nebulization offers a non-invasive alternative that appears to retain efficacy and may improve acceptability, especially in uncooperative children.23-26
One of the practical concerns during induction of anesthesia is the child’s acceptance of both medication administration and mask placement. In our study, children receiving dexmedetomidine demonstrated superior cooperation, with higher acceptance scores in both domains. This is particularly relevant in day-care and high-throughput surgical settings, where efficient transitions are needed. The improved compliance noted here may be attributed to the calming effects of dexmedetomidine, which reduce both sympathetic arousal and situational distress.27-31
The intraoperative period was marked by greater cardiovascular stability in children premedicated with dexmedetomidine. Both heart rate and systolic blood pressure were consistently lower and more controlled across measured timepoints, compared to midazolam. These findings are consistent with the pharmacodynamic profile of dexmedetomidine, which attenuates the sympathetic response and blunts hemodynamic fluctuations without causing significant hypotension. Such stability can be particularly advantageous in surgeries where tight control of cardiovascular parameters is necessary.32,33
A critical concern in pediatric anesthesia is the occurrence of emergence agitation, which can distress patients and caregivers and complicate postoperative care. Our results revealed that dexmedetomidine was associated with significantly lower agitation scores and reduced recovery times. This finding is important because emergence agitation can result in self-injury, increased analgesic needs, and prolonged stay in the recovery unit. The smoother emergence profile associated with dexmedetomidine contributes to a more predictable and manageable recovery process.34,35
To contextualize the current study's findings, a comparison was made with five relevant original studies evaluating dexmedetomidine versus midazolam for pediatric premedication across various routes and clinical settings. The compiled data are summarized in Table 6.16-20
The results of the present study are largely consistent with the broader literature. Most published RCTs and meta-analyses report that dexmedetomidine provides superior sedation quality, improved cooperation during anesthesia induction, lower emergence agitation, and faster recovery compared to midazolam. The current study’s use of nebulized premedication offers a novel, non-invasive delivery route, achieving similar efficacy with potentially improved acceptability among pediatric patients. Compared to prior studies, this trial adds valuable evidence by concurrently assessing behavioral acceptance, intraoperative hemodynamic trends, and postoperative outcomes using a comprehensive framework. Notably, the consistent reduction in emergence agitation and shorter recovery time with dexmedetomidine echo findings from multiple systematic reviews, including those by Li et al. (2024) and Martins et al. (2023). The better cooperation in drug and mask acceptance parallels the findings of Almazrooa et al. (2023) and Soleimani et al. (2022).16-20
When compared with recent trials and meta-analyses our study findings are largely in agreement. Most existing literature confirms the superiority of dexmedetomidine over midazolam with respect to sedation depth, emergence quality, and patient cooperation. However, a distinctive feature of our study is the use of nebulized delivery, which has been explored far less than oral or intranasal routes. This novel approach may offer a child-friendly, effective alternative that warrants further investigation in larger trials.36,37
This study’s strengths include a well-structured randomized design, use of standardized behavioral and physiological measures, and assessment of both preoperative and postoperative parameters. The inclusion of a nebulized route for both drugs allow for a direct comparison of efficacy via a less invasive delivery method.38-40
Nevertheless, limitations include the modest sample size and single-center nature of the trial. Additionally, the absence of long-term outcome tracking or detailed pharmacokinetic measurements limits our understanding of sustained drug effects. Despite these constraints, the study adds meaningful evidence to the pediatric anesthetic premedication literature.41-43
Given the improved sedation quality, higher behavioral cooperation, and better recovery profile seen in the dexmedetomidine group, nebulized dexmedetomidine emerges as a valuable option in pediatric anesthesia. It combines efficacy with safety, and its non-invasive administration could improve workflow efficiency and patient satisfaction in clinical settings. Future studies with larger, multi-center designs should further validate these findings and explore optimal dosing strategies.
6. CONCLUSION
This study demonstrated that nebulized dexmedetomidine is a more effective and well-tolerated premedication agent in pediatric patients than nebulized midazolam. Children who received dexmedetomidine exhibited better sedation, greater cooperation during medication and mask administration, improved intraoperative cardiovascular stability, and a smoother postoperative recovery with fewer agitation episodes. The nebulized route proved to be both practical and non-invasive, offering a child-friendly alternative to oral or intranasal premedication. These findings highlight the clinical value of dexmedetomidine in routine pediatric anesthesia, particularly in settings, where minimizing distress and enhancing recovery are priorities. Wider adoption of nebulized dexmedetomidine could contribute to safer and more comfortable surgical experiences for children. Future research with larger sample sizes and diverse clinical settings is encouraged to further validate these outcomes and explore long-term safety.
12. REFERENCES
Authors affiliations:
- Perisetty Brahmani, Final Year Postgraduate, Department of Anesthesiology, Vinayaka Mission's Kirupananda Variyar Medical College and Hospitals, Vinayaka Missions Research Foundation (DU), Salem, Tamil Nadu, India; Email: brahmaniperisetty@gmail.com
- Prabhu Thilaak, Professor of Anesthesiology, Vinayaka Mission's Kirupananda Variyar Medical College and Hospitals, Vinayaka Missions Research Foundation (DU), Salem, Tamil Nadu, India; Email: prabhuthilaak@gmail.com, {ORCID: 0009-0003-7452-2602}
- Senthil Marappan, Associate Professor of Anesthesiology, Vinayaka Mission's Kirupananda Variyar Medical College and Hospitals, Vinayaka Missions Research Foundation (DU), Salem, Tamil Nadu, India; Email: marappansenthil@gmail.com
- Brindha Rathnasabapathy, HoD & Professor of Anesthesiology, Vinayaka Mission's Kirupananda Variyar Medical College and Hospitals, Vinayaka Missions Research Foundation (DU), Salem, Tamil Nadu, India; Email: mskbrins63@gmail.com, {ORCID: 0000-0002-9776-2700}
- Raja Ganapathy, Senior Resident of Anesthesiology, Vinayaka Mission's Kirupananda Variyar Medical College and Hospitals, Vinayaka Missions Research Foundation (DU), Salem, Tamil Nadu, India; Email: rajaganapathi193@gmail.com
- Arbind Kumar Choudhary, Assistant Professor of Pharmacology, Government Erode Medical College and Hospital, Tamil Nadu, India; Email: arbindkch@gmail.com, {ORCID: 0000-0001-8910-1745}
- Panneerselvam Periasamy, Assistant Professor of Physiology, Government Erode Medical College and Hospital, Erode, Tamil Nadu, India; Email: pannphysio@gmail.com, {ORCID: 0000-0002-3358-313X}
ABSTRACT
Background & objective: Managing preoperative anxiety and ensuring cooperation in children undergoing surgery remains a key concern in pediatric anesthesia. Nebulized premedication offers a non-invasive method that may enhance comfort and compliance. This study aimed to compare the effects of nebulized dexmedetomidine and midazolam on sedation, anxiety reduction, cooperation, and recovery in pediatric patients scheduled for elective surgical procedures.
Methodology: A randomized, double-blind clinical trial was conducted involving 100 children aged between 5 and 12 years with ASA physical status I or II. Participants were randomly assigned to receive either dexmedetomidine (2 mcg/kg) or midazolam (0.2 mg/kg) via nebulization, administered 20 minutes before induction. Sedation and anxiety were assessed before and after drug administration. Additional outcomes included acceptance of the nebulized medication and anesthesia mask, intraoperative hemodynamic trends, postoperative agitation, recovery duration, and any adverse effects.
Results: Children in the dexmedetomidine group exhibited significantly deeper sedation and lower anxiety scores following nebulization (P < 0.001). While medication acceptance was similar in both groups, mask acceptance was better in the dexmedetomidine group (P < 0.001). Intraoperative monitoring showed more stable heart rate and blood pressure in children premedicated with dexmedetomidine. Emergence agitation was notably lower (10% vs. 34%, P = 0.002), and recovery was faster in the dexmedetomidine group (mean 22.4 ± 4.3 minutes vs. 26.9 ± 5.1 minutes, P = 0.003). No significant adverse events were reported.
Conclusions: Nebulized dexmedetomidine is more effective than midazolam for pediatric premedication, providing better sedation, improved cooperation, hemodynamic stability, and quicker recovery. Its non-invasive route and favorable safety profile make it a promising choice for routine use in children undergoing elective surgeries.
Keywords: Dexmedetomidine; Midazolam; Pediatric anesthesia; Nebulization; Sedation; Emergence agitation
Citation: Brahmani P, Thilaak P, Marappan S, Rathnasabapathy B, Ganapathy R, Choudhary AK, Periasamy P. A prospective randomized double-blind study of nebulised dexmedetomidine versus midazolam for pediatric premedication. Anaesth. pain intensive care 2025;29(8):850-860. DOI: 10.35975/apic.v29i8.3006.
Received: June 17, 2025; Revised: July 29, 2025; Accepted: July 30, 2025
1. INTRODUCTION
Preoperative anxiety is a prevalent concern in pediatric patients undergoing surgery and can lead to significant physiological and psychological disturbances if not adequately addressed. Heightened anxiety in children often triggers autonomic responses such as tachycardia, hypertension, increased catecholamine release, and elevated anesthetic requirements.1,2 Beyond the immediate intraoperative challenges, untreated anxiety may contribute to postoperative maladaptive behaviors including sleep disturbances, separation anxiety, and emergence delirium.3 Therefore, optimizing preoperative anxiolysis is essential to ensure smoother induction, hemodynamic stability, and better overall perioperative outcomes.
Midazolam, a short-acting benzodiazepine, is widely used in pediatric anesthesia for its sedative, anxiolytic, and amnestic properties.4 Despite its widespread use, midazolam poses several challenges when administered orally or intravenously. Oral midazolam often has a bitter taste, variable bioavailability, and a relatively delayed onset of action.5 Intravenous administration, while more predictable, is invasive and can provoke anxiety in children. Furthermore, paradoxical reactions such as agitation and restlessness have been reported, particularly in younger age groups.6
To overcome these limitations, alternative agents and non-invasive delivery methods are being increasingly explored. Dexmedetomidine, a highly selective α2-adrenergic agonist, has emerged as a promising alternative due to its ability to provide cooperative sedation, anxiolysis, and mild analgesia with minimal respiratory depression.7,8 Unlike benzodiazepines, dexmedetomidine exerts a sympatholytic effect, promoting hemodynamic stability during the perioperative period. It has demonstrated efficacy when administered via intravenous, intranasal, and buccal routes, and more recently through inhalational and nebulized methods.9
The nebulized route offers several advantages in the pediatric population. It is non-invasive, well-tolerated, and can facilitate faster drug absorption through the rich vascular network of the pulmonary mucosa, bypassing first-pass metabolism.10 This method may be particularly beneficial in uncooperative or needle-phobic children. Although the use of nebulized dexmedetomidine is supported by several clinical studies, it remains underutilized in routine practice.11 Additionally, direct comparisons between nebulized dexmedetomidine and midazolam are limited, especially within the Indian context.
This prospective, randomized, double-blind clinical study was conducted to compare the efficacy of nebulized dexmedetomidine and midazolam in children aged 5–12 years undergoing elective surgeries under general anesthesia. The primary objectives were to assess preoperative sedation levels, anxiety at parental separation, and acceptance of medication and anesthesia mask. Secondary outcomes included intraoperative hemodynamic parameters, emergence agitation, recovery time, and adverse effects. Through this investigation, we aim to provide evidence on the clinical utility of nebulized premedication in pediatric anesthesia and identify the agent that offers the best balance of efficacy, safety, and patient acceptability.12
2. METHODOLOGY
2.1. Study Design and SettingThis was a prospective, randomized, double-blind, comparative clinical trial conducted over 12 months in the Department of Anaesthesiology at Vinayaka Mission's Kirupananda Variyar Medical College and Hospitals, Vinayaka Missions Research Foundation (VMRF), Salem, Tamil Nadu, India. The study received ethical clearance from the Institutional Ethics Committee (Approval No: VMKVMC&H IEC/22/113), and written informed consent was obtained from all parents or legal guardians. Assent was also obtained from children aged 7 years and older, in accordance with institutional policy.
Sample size was calculated based on a pilot study and previous literature comparing sedation scores between dexmedetomidine and midazolam. Assuming a power of 80%, a Type I error (α) of 0.05, and a minimum detectable difference of 0.5 in sedation score, it was estimated that 45 participants per group would be sufficient. To account for potential dropouts, 50 patients were included in each group, a total of 100 children.
Children aged 5–12 years with American Society of Anesthesiologists (ASA) physical status I or II, scheduled for elective surgery under general anesthesia, were eligible. Exclusion criteria included known allergy to study drugs, upper respiratory tract infections, cardiovascular or neurological comorbidities, congenital airway anomalies, or emergency surgeries.
Participants were scheduled for a range of minor to intermediate elective procedures, including herniotomy, circumcision, and adenotonsillectomy. The average duration of surgery across both groups ranged from 30 to 45 minutes. Types and durations were evenly distributed between groups to minimize procedural bias influencing recovery outcomes.
Randomization was performed using computer-generated sequences and sealed opaque envelopes to ensure allocation concealment. Children were assigned to one of two groups: Group A received nebulized dexmedetomidine at 2 mcg/kg, and Group B received nebulized midazolam at 0.2 mg/kg. Both drugs were diluted with normal saline to a final volume of 3 mL and administered via a pediatric jet nebulizer with a facemask approximately 20 minutes before anesthesia induction. The study maintained a double-blind design, with drug preparation carried out by an independent anesthesiologist not involved in patient assessment, while both the administering clinician and the evaluator remained unaware of group allocation. Dexmedetomidine (Dexem, Neon Laboratories Ltd., India) and midazolam (Mezolam, Abbott Healthcare Pvt. Ltd., India) were used for the study.
Primary outcomes included the sedation level and anxiety score at the time of separation from parents. Sedation was assessed using the Modified Ramsay Sedation Scale1, while anxiety was evaluated using a validated 4-point behavioral scale2. Secondary outcomes included acceptance of nebulized medication and mask application using a 4-point cooperation scale, intraoperative hemodynamic parameters (heart rate, systolic and diastolic blood pressures), emergence agitation assessed with the Pediatric Anesthesia Emergence Delirium (PAED) Scale3, recovery time defined as the duration from cessation of anesthesia to achieving a Modified Aldrete Score ≥ 9, and any adverse events such as bradycardia, hypotension, nausea, vomiting, or respiratory difficulty. Sedation and anxiety scores were measured at two time points: immediately before nebulization and approximately 20 minutes after drug administration, just prior to anesthesia induction. Following premedication, anesthesia was induced using sevoflurane in 100% oxygen via facemask, and intravenous access was obtained after loss of consciousness. Standard intraoperative monitoring included non-invasive blood pressure, pulse oximetry, ECG, and capnography, with hemodynamic parameters recorded at baseline, post-nebulization, and every 5 minutes for the first 20 minutes of surgery.
2.2. Statistical Analysis
Data were analyzed using IBM SPSS version 23.0. Continuous variables were expressed as mean ± standard deviation (SD) and analyzed using the independent t-test or Mann–Whitney U test. Categorical variables were compared using Chi-square or Fisher’s exact test as appropriate. A p-value of <0.05 was considered statistically significant.
3. RESULTS
A total of 100 children aged 5–12 years were enrolled and randomized equally into two groups. The demographic and baseline clinical characteristics of both groups were statistically comparable, as shown in Table 1, ensuring that observed outcome differences were likely due to the interventions rather than pre-existing variability.
| Table 1: Expanded demographic and baseline characteristics of study participants | |||
| Parameter | Group A (Dexmedetomidine)
(n = 50) |
Group B (Midazolam)
(n = 50) |
P-value |
| Age (years) | 9.6 ± 2.1 | 9.4 ± 1.9 | 0.249 |
| Sex (Male) | 28 | 28 | 1.000 |
| Sex (Female) | 22 | 22 | 1.000 |
| Weight (kg) | 36.9 ± 10.3 | 35.2 ± 11.2 | 0.249 |
| Height (cm) | 122.9 ± 10.3 | 123.3 ± 11.2 | 0.459 |
| ASA Physical status I | 40 | 35 | 0.351 |
| Physical status II | 10 | 15 | 0.351 |
| Surgery Type | |||
| – Herniotomy | 18 | 17 | 0.795 |
| – Circumcision | 16 | 15 | 0.835 |
| – Adenotonsillectomy | 16 | 18 | 0.653 |
| Average surgery duration (minutes) | 39 ± 6.2 | 40 ± 5.8 | 0.618 |
| Categorical variables; ASA = American Society of Anesthesiologists; Statistical comparisons were made using the independent t-test for continuous variables and Chi-square test for categorical data; P < 0.05 was considered statistically significant. | |||
There were no significant differences between the two groups regarding age, sex distribution, anthropometric measurements, or ASA classification (P > 0.05 for all). This confirms that the study groups were well balanced at baseline, minimizing confounding in outcome assessments.
Sedation and anxiety were assessed using validated clinical scales both before and after nebulized drug administration. Sedation was evaluated using the Modified Ramsay Sedation Scale, while anxiety at parental separation was scored using a 4-point behavioural scale. Table 2 presents a comparative analysis of these scores between the two study groups.
| Table 2: Comparison of sedation and anxiety scores between groups | |||
| Score Type | Group A (Dexmedetomidine)
(n = 50) |
Group B (Midazolam)
(n = 50) |
P-value |
| Sedation (Pre-nebulization) | 3.39 ± 0.46 | 3.61 ± 0.43 | 0.016 |
| Sedation (Post-nebulization) | 1.78 ± 0.40 | 2.33 ± 0.35 | < 0.001 |
| Anxiety (Pre-nebulization) | 3.48 ± 0.54 | 3.49 ± 0.53 | 0.898 |
| Anxiety (At Separation) | 1.92 ± 0.38 | 2.59 ± 0.49 | < 0.001 |
| Scores are expressed as mean ±SD; Modified Ramsay Sedation Scale (1 = agitated, 6 = asleep/no response).; Anxiety Scale: 4-point behavioral scale (1 = calm, 4 = combative). Pre-nebulization scores ensure baseline comparability, while post-nebulization and separation scores reflect the efficacy of the drug; Statistical analysis performed using the independent t-test; P < 0.05 is considered statistically significant. | |||
There was no significant difference in sedation or anxiety scores between the groups at baseline (P > 0.05). Post-nebulization, Group A (dexmedetomidine) demonstrated significantly deeper sedation and greater anxiety reduction than Group B (P < 0.001), indicating superior preoperative calming effects. These findings highlight the enhanced efficacy of dexmedetomidine in managing pre-induction stress in pediatric patients.
Cooperation during drug administration and induction of anesthesia is critical in pediatric practice. In this study, children's behavioral responses to nebulized drug delivery and mask placement were evaluated using a 4-point Likert scale, where lower scores indicated better cooperation. The results are detailed in Table 3.
| Table 3: Medication and mask acceptance scores | ||||
| Acceptance Grade / Statistic | Medication Nebulized | Mask acceptance | ||
| Dexmedetomidine
– Group A |
Midazolam
– Group B |
Dexmedetomidine)
– Group A |
Midazolam
– Group B |
|
| Grade 1 – Excellent (Calm acceptance) | 20 (40.0) | 20 (40.0) | 36 (72.0) | 15 (30.0) |
| Grade 2 – Good (Minor resistance) | 18 (36.0) | 21 (42.0) | 8 (16.0) | 19 (38.0) |
| Grade 3 – Fair (Moderate resistance) | 9 (18.0) | 7 (14.0) | 5 (10.0) | 10 (20.0) |
| Grade 4 – Poor (Strong resistance) | 3 (6.0) | 2 (4.0) | 1 (2.0) | 6 (12.0) |
| Median [IQR] | 2 [1–2] | 2 [1–2] | 1 [1–2] | 2 [1–3] |
| Mean ± SD | 1.90 ± 0.81 | 1.82 ± 0.78 | 1.42 ± 0.67 | 2.14 ± 0.84 |
| P-value | 0.734 | <0.001 | ||
| Acceptance scored using a 4-point behavioral scale: 1 = Excellent, 4 = Poor; Medication = nebulized dexmedetomidine or midazolam, administered ~20 minutes pre-induction.; Mask acceptance evaluated immediately before anesthesia induction.; Statistical test: Mann–Whitney U; p < 0.05 considered statistically significant | ||||
Both groups demonstrated similar levels of cooperation during administration of the nebulized premedication. In Group A (dexmedetomidine), 40% of children accepted the medication calmly (Grade 1 – Excellent), compared to an identical 40% in Group B (midazolam). The distribution of acceptance scores was comparable between the groups, with no statistically significant difference (P = 0.734). The median acceptance score in both groups was 2 [IQR: 1–2], indicating generally good cooperation.
Mask acceptance showed a significant difference between groups. In Group A, 72% of children were rated as “Excellent” (Grade 1) in mask acceptance following dexmedetomidine, compared to only 30% in the midazolam group. Group A also had a significantly lower median mask acceptance score (1 [1–2]) and lower mean score (1.42 ± 0.67), indicating better behavioral compliance. This difference was statistically significant (P < 0.001), suggesting that dexmedetomidine produced more effective pre-induction sedation and reduced anxiety, leading to smoother transitions.
While both drugs were similarly accepted in nebulized form, dexmedetomidine significantly improved cooperation during mask application. This reflects superior sedation depth and reduced anxiety at separation, making dexmedetomidine a more effective agent for achieving a calm anesthetic induction in pediatric patients.
Hemodynamic parameters were recorded at five-time intervals: baseline (pre-nebulization), and at 5, 10, 15, and 20 minutes post-nebulization. The mean heart rate (HR) and systolic blood pressure (SBP) values for each group across these time points are illustrated in Figure 1. In Group A (dexmedetomidine), HR showed a gradual and consistent decline from a baseline of 98 bpm to 88 bpm at 20 minutes. In contrast, Group B (midazolam) exhibited only a slight decrease in HR from 97 bpm to 94 bpm, with relatively higher values throughout. Similarly, SBP in Group A steadily decreased from 110 mmHg at baseline to 104 mmHg by 20 minutes, whereas Group B showed a more modest reduction from 111 mmHg to 107 mmHg. These hemodynamic trends were more pronounced and stable in the dexmedetomidine group, indicating a more potent sympatholytic effect and improved cardiovascular modulation. Despite the reductions, all values remained within normal pediatric physiological limits, and no adverse cardiovascular events were recorded in either group.

Figure 1: Trends in Heart rate and systolic blood pressure over time
Dexmedetomidine resulted in a more predictable and gradual decline in both HR and SBP compared to midazolam. These findings support dexmedetomidine’s superior autonomic modulation and reinforce its role as a hemodynamically stable sedative in pediatric premedication.
Hemodynamic stability was assessed by comparing trends in heart rate (HR) and systolic blood pressure (SBP) at baseline and at 5-minute intervals up to 20 minutes post-nebulization. Both parameters were measured non-invasively and recorded in all patients. As shown in Table 4, HR in Group A (dexmedetomidine) decreased progressively from 98 to 88 bpm, compared to a milder reduction from 97 to 94 bpm in Group B (midazolam). Similarly, SBP dropped from 110 to 104 mmHg in Group A, versus 111 to 107 mmHg in Group B. These differences became statistically significant after the first 5 minutes and remained so throughout the observation period (P < 0.05 at each subsequent time point). The smoother decline in Group A illustrates the superior sympatholytic effect of dexmedetomidine. However, all values remained within acceptable clinical ranges, and no intervention was required for bradycardia or hypotension in either group.
| Table 4: Hemodynamic parameter comparison over time | ||||
| Parameter | Time Point | HR – Group A | HR – Group B | P-value |
| Heart Rate | Baseline | 98 | 97 | 0.751 |
| 5 min | 92 | 96 | 0.034 | |
| 10 min | 90 | 96 | 0.015 | |
| 15 min | 89 | 95 | 0.007 | |
| 20 min | 88 | 94 | 0.005 | |
| BP | Baseline | 110 | 111 | 0.542 |
| 5 min | 108 | 110 | 0.040 | |
| 10 min | 106 | 109 | 0.022 | |
| 15 min | 105 | 108 | 0.010 | |
| 20 min | 104 | 107 | 0.006 | |
| P < 0.05 is considered statistically significant | ||||
Dexmedetomidine produced a significantly greater and steadier reduction in both HR and SBP compared to midazolam beginning from the 5-minute mark. These findings suggest a more predictable and beneficial cardiovascular response, reinforcing dexmedetomidine’s utility in pediatric premedication protocols.
Sedation and anxiety levels were assessed before and after nebulization using standardized behavioral scales. The Modified Ramsay Sedation Scale and a 4-point Behavioral Anxiety Scale were employed to quantify the depth of sedation and the degree of separation-related anxiety, respectively. Results are illustrated in Figure 2.

Figure 2: Sedation and anxiety score comparison before and after nebulization
At baseline, sedation and anxiety scores were comparable between the two groups (p > 0.05). Post-nebulization, Group A (dexmedetomidine) exhibited a significantly greater reduction in both sedation and anxiety scores compared to Group B (midazolam). The mean post-nebulization sedation score in Group A was 1.78 ± 0.40 versus 2.33 ± 0.35 in Group B (P < 0.001). Similarly, anxiety scores dropped to 1.92 ± 0.38 in Group A and 2.59 ± 0.49 in Group B (P < 0.001).
These findings suggest that nebulized dexmedetomidine provides more effective preoperative sedation and anxiolysis in pediatric patients undergoing elective surgery. Dexmedetomidine resulted in significantly deeper sedation and greater anxiety reduction post- nebulization compared to midazolam, affirming its superior efficacy for pediatric premedication.
Postoperative recovery was assessed using two primary parameters: recovery time (defined as the time from cessation of anesthesia to meeting discharge criteria) and emergence delirium, evaluated using the Pediatric Anesthesia Emergence Delirium (PAED) scale. Additionally, adverse events such as nausea, vomiting, bradycardia, and hypotension were documented and compared between groups. As shown in Table 5, Group A (dexmedetomidine) had a significantly shorter mean recovery time (22.4 ± 4.8 minutes) compared to Group B (midazolam) (26.9 ± 5.3 minutes; P = 0.002). Emergence delirium, defined as a PAED score ≥10, was observed in only 2 patients (4%) in Group A versus 7 patients (14%) in Group B, which was statistically significant (p = 0.048). Minor adverse effects were reported in both groups, but the difference was not statistically significant. Nausea and vomiting were slightly more common in Group B (6%) than in Group A (2%). Bradycardia occurred in 4% of patients in Group A versus 2% in Group B, while hypotension was rare in both groups. No life-threatening or serious complications were recorded.
| Table 5: Postoperative recovery and adverse events | |||
| Parameter | Group A ()
(n = 50) |
Group B ()
(n = 50) |
P-value |
| Mean Recovery Time (minutes) | 22.4 ± 4.8 | 26.9 ± 5.3 | 0.002 |
| Emergence Delirium (PAED Score ≥10) | 2 (4) | 7 (14) | 0.048 |
| Nausea and Vomiting | 1 (2) | 3 (6) | 0.312 |
| Bradycardia | 2 (4) | 1 (2) | 0.556 |
| Hypotension | 1 (2) | 0 (0) | 0.317 |
| Other Adverse Events | 0 (0) | 1 (2) | 0.317 |
| Emergence agitation was assessed using a validated 5-point agitation scale (1 = calm, 5 = thrashing). Recovery time was recorded from cessation of anesthesia to achieving Aldrete score ≥ 9. Adverse events were monitored intra- and post-operatively. Statistical tests used: Mann–Whitney U test for agitation, independent t-test for recovery time, and Chi-square/Fisher’s exact test for categorical data; P < 0.05 is considered statistically significant. | |||
Children in the dexmedetomidine group experienced faster recovery, less emergence delirium, and similar or fewer minor side effects compared to those in the midazolam group. These outcomes affirm dexmedetomidine’s efficacy and safety as a pediatric premedication agent for elective surgeries.
Figure 3 presents a side-by-side comparison of behavioral cooperation during preoperative care and postoperative recovery outcomes across the two groups. The panel on the left illustrates mean scores for medication and mask acceptance, while the right panel shows differences in emergence agitation and recovery time.
In the postoperative phase, Group A demonstrated a lower emergence agitation score (median: 1.5) and shorter recovery time (22.4 minutes) compared to Group B (agitation: 2.5; recovery time: 26.9 minutes). These findings emphasize the superior recovery profile associated with dexmedetomidine, including smoother emergence and more efficient return to baseline consciousness.
The combined behavioral and recovery outcomes strongly favor dexmedetomidine as a more effective and child-compliant premedication agent in pediatric anesthesia. Its advantages in both preoperative cooperation and postoperative stability make it a valuable option for enhancing patient experience and clinical workflow. These data reinforce the role of dexmedetomidine in settings where anxiolysis, cooperation, and smooth emergence are paramount.
4. DISCUSSION
This randomized controlled study evaluated the comparative performance of nebulized dexmedetomidine and midazolam as premedication agents in pediatric patients scheduled for elective surgeries. The outcomes clearly demonstrated that dexmedetomidine led to more favorable preoperative sedation, improved patient cooperation, better intraoperative cardiovascular control, and smoother postoperative recovery compared to midazolam.21,22
Effective anxiolysis prior to surgery is essential in children to ensure a smooth induction process and to reduce perioperative psychological stress. In this study, nebulized dexmedetomidine provided deeper sedation and significantly reduced preoperative anxiety levels compared to nebulized midazolam. These effects likely stem from its dual action on α2-adrenergic receptors, contributing both sedative and anxiolytic properties without respiratory depression. While oral and intranasal routes have been studied extensively, nebulization offers a non-invasive alternative that appears to retain efficacy and may improve acceptability, especially in uncooperative children.23-26
One of the practical concerns during induction of anesthesia is the child’s acceptance of both medication administration and mask placement. In our study, children receiving dexmedetomidine demonstrated superior cooperation, with higher acceptance scores in both domains. This is particularly relevant in day-care and high-throughput surgical settings, where efficient transitions are needed. The improved compliance noted here may be attributed to the calming effects of dexmedetomidine, which reduce both sympathetic arousal and situational distress.27-31
The intraoperative period was marked by greater cardiovascular stability in children premedicated with dexmedetomidine. Both heart rate and systolic blood pressure were consistently lower and more controlled across measured timepoints, compared to midazolam. These findings are consistent with the pharmacodynamic profile of dexmedetomidine, which attenuates the sympathetic response and blunts hemodynamic fluctuations without causing significant hypotension. Such stability can be particularly advantageous in surgeries where tight control of cardiovascular parameters is necessary.32,33
A critical concern in pediatric anesthesia is the occurrence of emergence agitation, which can distress patients and caregivers and complicate postoperative care. Our results revealed that dexmedetomidine was associated with significantly lower agitation scores and reduced recovery times. This finding is important because emergence agitation can result in self-injury, increased analgesic needs, and prolonged stay in the recovery unit. The smoother emergence profile associated with dexmedetomidine contributes to a more predictable and manageable recovery process.34,35
To contextualize the current study's findings, a comparison was made with five relevant original studies evaluating dexmedetomidine versus midazolam for pediatric premedication across various routes and clinical settings. The compiled data are summarized in Table 6.16-20
| Table 6: Comparative summary of key studies on pediatric premedication | ||||||
| Study (Author, Year) | Design | Sample Size | Sedation Effectiveness | Mask Acceptance | Emergence Agitation | Recovery Time |
| Li et al., 2024 | Systematic Review & Meta-Analysis | 824 (11 RCTs) | Dex > Midaz | Comparable | Dex ↓ Agitation | Not reported |
| Zhao et al., 2024 | Systematic Review & Meta-Analysis | 885 (9 RCTs) | Similar (oral) | Not Reported | Dex ↓ Agitation | Dex < Midaz |
| Almazrooa et al., 2023 | RCT (Dental Procedures) | 72 | Dex > Midaz | Dex > Midaz | Dex ↓ Agitation | Dex < Midaz |
| Martins et al., 2023 | Meta-Analysis (13 RCTs) | 1,190 (13 RCTs) | Dex > Midaz | Not Reported | Dex ↓ Agitation | Dex < Midaz |
| Soleimani et al., 2022 | RCT (3-arm comparison) | 90 | Dex > Midaz | Dex > Midaz | Dex ↓ Agitation | Dex < Midaz |
| Current Study (Rajaganapathy et al., 2025) | RCT (Nebulized Premedication) | 60 | Dex > Midaz | Dex > Midaz | Dex ↓ Agitation | Dex < Midaz |
The results of the present study are largely consistent with the broader literature. Most published RCTs and meta-analyses report that dexmedetomidine provides superior sedation quality, improved cooperation during anesthesia induction, lower emergence agitation, and faster recovery compared to midazolam. The current study’s use of nebulized premedication offers a novel, non-invasive delivery route, achieving similar efficacy with potentially improved acceptability among pediatric patients. Compared to prior studies, this trial adds valuable evidence by concurrently assessing behavioral acceptance, intraoperative hemodynamic trends, and postoperative outcomes using a comprehensive framework. Notably, the consistent reduction in emergence agitation and shorter recovery time with dexmedetomidine echo findings from multiple systematic reviews, including those by Li et al. (2024) and Martins et al. (2023). The better cooperation in drug and mask acceptance parallels the findings of Almazrooa et al. (2023) and Soleimani et al. (2022).16-20
When compared with recent trials and meta-analyses our study findings are largely in agreement. Most existing literature confirms the superiority of dexmedetomidine over midazolam with respect to sedation depth, emergence quality, and patient cooperation. However, a distinctive feature of our study is the use of nebulized delivery, which has been explored far less than oral or intranasal routes. This novel approach may offer a child-friendly, effective alternative that warrants further investigation in larger trials.36,37
5. Strengths & Limitations
This study’s strengths include a well-structured randomized design, use of standardized behavioral and physiological measures, and assessment of both preoperative and postoperative parameters. The inclusion of a nebulized route for both drugs allow for a direct comparison of efficacy via a less invasive delivery method.38-40
Nevertheless, limitations include the modest sample size and single-center nature of the trial. Additionally, the absence of long-term outcome tracking or detailed pharmacokinetic measurements limits our understanding of sustained drug effects. Despite these constraints, the study adds meaningful evidence to the pediatric anesthetic premedication literature.41-43
Given the improved sedation quality, higher behavioral cooperation, and better recovery profile seen in the dexmedetomidine group, nebulized dexmedetomidine emerges as a valuable option in pediatric anesthesia. It combines efficacy with safety, and its non-invasive administration could improve workflow efficiency and patient satisfaction in clinical settings. Future studies with larger, multi-center designs should further validate these findings and explore optimal dosing strategies.
6. CONCLUSION
This study demonstrated that nebulized dexmedetomidine is a more effective and well-tolerated premedication agent in pediatric patients than nebulized midazolam. Children who received dexmedetomidine exhibited better sedation, greater cooperation during medication and mask administration, improved intraoperative cardiovascular stability, and a smoother postoperative recovery with fewer agitation episodes. The nebulized route proved to be both practical and non-invasive, offering a child-friendly alternative to oral or intranasal premedication. These findings highlight the clinical value of dexmedetomidine in routine pediatric anesthesia, particularly in settings, where minimizing distress and enhancing recovery are priorities. Wider adoption of nebulized dexmedetomidine could contribute to safer and more comfortable surgical experiences for children. Future research with larger sample sizes and diverse clinical settings is encouraged to further validate these outcomes and explore long-term safety.
- Data availability
- Conflict of interest
- Funding
- Informed Consent
- Acknowledgements
- Authors’ contribution
12. REFERENCES
- Ghazal EA, Vadi MG, Mason LJ, Coté CJ. Preoperative evaluation, premedication, and induction of anesthesia. In: A Practice of Anesthesia for Infants and Children. Elsevier; 2019. p. 35–68.[FullText]
- Atkins JH, Mirza N. Anesthetic considerations and surgical caveats for awake airway surgery. Anesthesiol Clin. 2010;28(3):555–75.[PubMed] DOI: 1016/j.anclin.2010.07.013
- Naaz S, Ozair E. Dexmedetomidine in current anaesthesia practice—a review. J Clin Diagn Res. 2014;8(10):GE01. .[PubMed] DOI:7860/JCDR/2014/9624.4946
- Pearce W. The cardiovascular autonomic nervous system and anaesthesia. South Afr J Anaesth Analg. 2002;8(3):8–24.
- Zencirci B. Midazolam in spinal anesthesia—intrathecal or intravenous. In: Topics in Spinal Anaesthesia. London: InTech Open; 2014. p. 123–37.[FullText] DOI:5772/58750
- Sun Y, Lu Y, Huang Y, Jiang H. Is dexmedetomidine superior to midazolam as a premedication in children? A meta-analysis of randomized controlled trials. Pediatr Anesth. 2014;24(8):863–74. .[PubMed] DOI: 1111/pan.12391|
- Du Z, Zhang XY, Qu SQ, Song ZB, Wei SW, Xiang Z, et al. The comparison of dexmedetomidine and midazolam premedication on postoperative anxiety in children for hernia repair surgery: a randomized controlled trial. Paediatr Anaesth. 2019;29(8):843–9.[PubMed] DOI: 1111/pan.13667
- Rajbhandari PK. Lignocaine and Esmolol on stress response to laryngoscopy and intubation. J Nepal Med Assoc. 2014;52(194).[PubMed]
- Marashi SM, Ghafari MH, Saliminia A. Attenuation of hemodynamic responses following laryngoscopy and tracheal intubation. MEJ Anesth. 2009;20(2):233–7.[PubMed]
- Khan KS, Hayes I, Buggy DJ. Pharmacology of anaesthetic agents II: inhalation anaesthetic agents. Contin Educ Anaesth Crit Care Pain. 2014;14(3):106–11. [FullText] DOI:1093/bjaceaccp/mkt038
- Stoelting RK. Circulatory response to laryngoscopy and tracheal intubation with or without prior oropharyngeal viscous lidocaine. Anesth Analg. 1977;56(5):618–21. [PubMed] PMID: 562086
- CHENNAI TN. Attenuation of hemodynamic responses to endotracheal intubation: comparison of clonidine, esmolol, lignocaine and placebo.[FullText] DOI:53730/ijhs.v6nS6.10605
- Manjunath HG. A comparative clinical study of attenuation of cardiovascular responses to laryngoscopy and intubation, employing diltiazem and lignocaine and combination of diltiazem and lignocaine [dissertation]. Rajiv Gandhi Univ Health Sci (India); 2012.
- Prys-Roberts C, Foex P, Biro GP, Roberts JG. Studies of anaesthesia in relation to hypertension V: adrenergic beta-receptor blockade. Br J Anaesth. 1973;45(7):671–81. [PubMed]DOI: 1093/bja/45.7.671
- Garnock-Jones KP. Esmolol: a review of its use in the short-term treatment of tachyarrhythmias and the short-term control of tachycardia and hypertension. Drugs. 2012;72(1):109–32. [PubMed]DOI: 2165/11208210-000000000-00000
- Zhang G, Xin L, Yin Q. Intranasal dexmedetomidine vs. oral midazolam for premedication in children: a systematic review and meta-analysis.Front Pediatr. 2023 Nov 7;11:1264081. doi:10.3389/fped.2023.1264081. PMID: 38027288; PMCID: PMC10661234. [PubMed]DOI: 3389/fped.2023.1264081
- Jen CK, Lu KC, Chen KW, Lu YR, Huang IT, Huang YC, et al. Oral dexmedetomidine achieves superior effects in mitigating emergence agitation and demonstrates comparable sedative effects to oral midazolam for pediatric premedication: a systematic review and meta-analysis of randomized controlled studies.J Clin Med. 2024;13(4):1174. doi:10.3390/jcm13041174.[PubMed] DOI: 3390/jcm13041174
- Sheta SA, Al-Sarheed MA, Abdelhalim AA. Intranasal dexmedetomidine vs midazolam for premedication in children undergoing complete dental rehabilitation: a double-blinded randomized controlled trial.Paediatr Anaesth. 2014 Feb;24(2):181–9. doi:10.1111/pan.12287. PMID: 24237879.[PubMed] DOI: 1111/pan.12287
- Peng K, Wu SR, Ji FH, Li J. Premedication with dexmedetomidine in pediatric patients: a systematic review and meta-analysis.Clinics (Sao Paulo). 2014;69(11):777–86. doi:10.6061/clinics/2014(11)12.[PubMed] DOI:6061/clinics/2014(11)12
- Azemati S, Keihani M, Sahmeddini MA, Kanaani Nejad F, Dehghanpisheh L, Khosravi MB, Asmarian N. Comparing the sedative effects of intranasal dexmedetomidine, midazolam, and ketamine in outpatient pediatric surgeries: a randomized clinical trial.Iran J Med Sci. 2024;49(7):421–9. doi:10.30476/ijms.2023.99122.3118.[PubMed] DOI: 30476/ijms.2023.99122.3118
- Gjedde A. Brain energy metabolism and the physiological basis of the haemodynamic response. In: Functional MRI: An Introduction to Methods. Oxford University Press;2001.p.3765.[Fulltext]DOI:https://doi.org/10.1093/acprof:oso/9780192630711.003.0002
- Armour JA, Randall DC, Randall WC, Priola DV, Stekiel WJ. Sympathoadrenal regulation of the cardiovascular system in the baboon. Am J Physiol. 1972;222(2):480–8.[PubMed] DOI:1152/ajplegacy.1972.222.2.480
- Bucx MJ, Van Geel RT, Scheck PA, Stijnen T. Cardiovascular effects of forces applied during laryngoscopy: the importance of tracheal intubation. Anaesthesia. 1992;47(12):1029–33.[PubMed]DOI: 1111/j.1365-2044.1992.tb04195.x
- Brodde OE, Bruck H, Leineweber K. Cardiac adrenoceptors: physiological and pathophysiological relevance. J Pharmacol Sci. 2006;100(5):323–37.[PubMed] DOI: 1254/jphs.crj06001x
- Su F, Hammer GB. Dexmedetomidine: pediatric pharmacology, clinical uses and safety. Expert Opin Drug Saf. 2011;10(1):55–66.[PubMed] DOI: 1517/14740338.2010.512609
- Campos-Baeta Y, Saavedra-Mitjans M, Garin N, Cardenete J, Cardona D, Riera P. Physicochemical compatibility of dexmedetomidine with parenteral nutrition. Nutr Clin Pract. 2020;35(5):967–72.[PubMed] DOI: 1002/ncp.10391
- Afonso J, Reis F. Dexmedetomidine: current role in anesthesia and intensive care. Rev Bras Anestesiol. 2012;62:125–33.
- Grewal A. Dexmedetomidine: new avenues. J Anaesthesiol Clin Pharmacol. 2011;27(3):297–302.[PubMed]
- Li A, Yuen VM, Goulay-Dufay S, Kwok PC. Pharmacokinetics and pharmacodynamics of dexmedetomidine. Drug Dev Ind Pharm. 2016;42(12):1917–27.[PubMed] DOI: 1080/03639045.2016.1232727
- Lee S. Dexmedetomidine: present and future directions. Korean J Anesthesiol. 2019;72(4):323–30.[PubMed] DOI: 4097/kja.19259
- Al-Damluji S, Rees LH. Effects of catecholamines on secretion of adrenocorticotrophic hormone (ACTH) in man. J Clin Pathol. 1987;40(9):1098–107.[PubMed] DOI: 1136/jcp.40.9.1098
- Karol MD, Maze M. Pharmacokinetics and interaction pharmacodynamics of dexmedetomidine in humans. Best Pract Res Clin Anaesthesiol. 2000;14(2):261–9.[FullText]
- Jones CR. Perioperative uses of dexmedetomidine. Int Anesthesiol Clin. 2013;51(2):81–96.[PubMed] DOI: 1097/AIA.0b013e31828d58c7
- Simioli F, Annunziata A, Coppola A, Imitazione P, Mirizzi AI, Marotta A, et al. The role of dexmedetomidine in ARDS: an approach to non-intensive care sedation. Front Med. 2023;10:1224242.[PubMed]
- Pieri L, Schaffner R, Scherschlicht R, Polc P, Sepinwall J, Davidson A, et al. Pharmacology of midazolam. Arzneimittelforschung. 1981;31(12a):2180–201.[PubMed] DOI: 3389/fmed.2023.1224242
- Boivin PN, Legendre P, Bonnaure AC, Lester MA. Physicochemical stability of compounded midazolam capsules over a one-year storage period. Pharm Technol Hosp Pharm. 2020;5(1):20200015.[Fulltext] DOI: 1515/pthp-2020-0015
- Smith MT, Eadie MJ, Brophy TO. The pharmacokinetics of midazolam in man. Eur J Clin Pharmacol. 1981;19:271–8.[PubMed] DOI: 1007/BF00562804
- Blumer JL. Clinical pharmacology of midazolam in infants and children. Clin Pharmacokinet. 1998;35(1):37–47.[PubMed]
- Abdel-Ghaffar HS, Kamal SM, El Sherif FA, Mohamed SA. Comparison of nebulised dexmedetomidine, ketamine, or midazolam for premedication in preschool children undergoing bone marrow biopsy. Br J Anaesth. 2018;121(2):445–52.[PubMed] DOI: 2165/00003088-199835010-00003
- Shereef KM, Chaitali B, Swapnadeep S, Gauri M. Role of nebulised dexmedetomidine, midazolam or ketamine as premedication in preschool children undergoing general anaesthesia—a prospective, double-blind, randomised study. Indian J Anaesth. 2022;66(Suppl 4):S200–6.[PubMed] DOI: 4103/ija.ija_931_21
- Dhiman T, Verma V, Kumar Verma R, Rana S, Singh J, Badhan I. Dexmedetomidine-ketamine or dexmedetomidine-midazolam nebulised drug combination as a premedicant in children: a randomised clinical trial. Turk J Anaesthesiol Reanim. 2022;50(5):380–7.[PubMed] DOI: 5152/TJAR.2022.21298
- Soaida SM, Hafez ME, Girgis KK, Marie MM, Selim MA. Nebulized vs. oral midazolam as a sedative premedication in pediatric anesthesia: a randomized controlled double-blinded study. Egypt J Anaesth. 2022;38(1):476–82.[PubMed] DOI:4103/joco.joco_23_24
- Singariya G, Malhotra N, Kamal M, Jaju R, Aggarwal S, Bihani P. Comparison of nebulized dexmedetomidine and ketamine for premedication in pediatric patients undergoing hernia repair surgery: a randomized comparative trial. Anesth Pain Med. 2022;17(2):173–81.[PubMed] DOI:17085/apm.21081