Taufan Pramadika 1 , Mochamat 2 , Widya Istanto Nurcahyo 3 , Satrio Adi Wicaksono 4 , Taufik Saputra 5 , Chandra Hermawan Manapa 6 , Alesandro Ksatriaputra 7
Authors affiliations:
Introduction: Spinal anesthesia (SA) is widely used for lower abdominal and limb surgeries due to its rapid onset and favorable safety profile. However, because of sympathetic blockade, it frequently results in bradycardia and hypotension, increasing the risk of perioperative complications and postoperative nausea and vomiting (PONV). Intrathecal atropine has shown potential as an adjuvant to counteract these effects and improve anesthesia outcomes. This review evaluates current evidence on the safety and efficacy of intrathecal atropine in SA.
Methods: MEDLINE, EMBASE, CENTRAL, Scopus, ProQuest, and Google Scholar were searched for randomized controlled trials published in English up to April 2025 on intrathecal atropine as an adjuvant in SA. The primary outcome was PONV; secondary outcomes included sensory block onset, postoperative pain, and adverse effects. Risk of bias was assessed using the Cochrane RoB 2.0 tool.
Results: Five RCTs involving 584 patients were included. Intrathecal atropine significantly reduced the incidence and severity of PONV in most studies (P < 0.05). However, it did not show significant effects on postoperative pain scores, sensory block onset, or hemodynamic parameters compared to control or other adjuvants. Adverse effects were generally mild and comparable across groups, though one study reported a higher incidence of pruritus with atropine (P < 0.001). No serious complications were observed. Nonetheless, study limitations included small sample sizes, non-standardized outcomes, and single-center designs with limited follow-up.
Conclusions: Intrathecal atropine may reduce PONV after SA without serious adverse effects. However, its impact on pain, sensory block, and hemodynamics remains uncertain, warranting further large-scale, standardized trials.
Abbreviations: GA: general anesthesia, RCT: randomized controlled trial, SA: Spinal anesthesia, PONV: postoperative nausea and vomiting
Keywords: Adjuvant; Atropine; Complications; Intrathecal; Outcome; PONV; Postoperative Nausea and Vomiting; Spinal Anesthesia
Citation: Pramadika T, Mochamat, Nurcahyo WI, Wicaksono SA, Saputra T, Manapa CH, Ksatriaputra A. Intrathecal atropine as an adjuvant in spinal anesthesia: a systematic review of efficacy and safety. Anaesth. pain intensive care 2025;29(7):796-807. DOI: 10.35975/apic.v29i7.2947
Received: July 01, 2025; Revised: August 03, 2024; Accepted: August 07, 2025
Spinal anesthesia (SA), also known as a subarachnoid block, remains one of the most commonly used techniques in regional anesthesia, especially for surgeries involving the lower abdomen, pelvis, and lower limbs.1 Its frequent use is supported by several well-known advantages, including rapid onset, reliable sensory and motor blockade, and better postoperative outcomes such as a lower incidence of nausea, vomiting, and respiratory depression when compared to general anesthesia (GA).2 However, it is also linked to hemodynamic adverse effects such as hypotension and bradycardia. These effects are caused by a sympathovagal imbalance that occurs from the blockade of the sympathetic nervous system and a relative increase in parasympathetic tone, which can be particularly concerning in patients with high block levels or limited cardiovascular reserve, potentially increasing perioperative risk.3,4 In addition to cardiovascular effects, this sympathetic blockade-induced hypotension has also been linked to an increased risk of postoperative nausea and vomiting (PONV), which can further affect patient comfort and recovery.5
Pharmacologic adjuvants administered intrathecally alongside SA have garnered increasing interest as a solution to these concerns. These agents aim to enhance the quality of analgesia during surgery, prolong the duration of anesthesia, and minimize the unwanted physiological effects.6 Among these options, atropine has gained particular attention. While it’s traditionally been used intravenously to manage bradyarrhythmias, atropine is a well-understood antimuscarinic agent. When administered intrathecally, it is thought to work by blocking muscarinic acetylcholine receptors in the spinal cord, which may reduce excessive vagal activity without causing significant systemic adverse effects.7,8
Although early findings have shown promise, the current research on intrathecal atropine varies greatly in terms of study design, patient characteristics, and outcome measures. Because of this variability, its true clinical value remains uncertain. Therefore, this review aims to evaluate the available evidence on the use of intrathecal atropine as an adjuvant to SA, focusing on both its safety and efficacy. By bringing together the findings from different clinical studies, this review endeavors to provide useful insights for clinical practice and highlight areas where further research is needed.
This systematic review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines to evaluate the efficacy and safety of intrathecal atropine as an adjuvant in SA. There was no need for formal ethical approval because the review was based only on previously published research and did not directly involve any patient participation.9
2.1. Study Identification and Eligibility Criteria
A comprehensive literature search was conducted to find studies that evaluate the use of intrathecal atropine as an adjuvant in SA. This search covered all publications available up to April 2025 and included multiple electronic databases such as MEDLINE ([PubMed]), Embase (OVID), Cochrane Central Register of Controlled Trials (CENTRAL), Scopus, and ProQuest. To make sure no important studies were missed, we also manually searched Google Scholar alongside the database searches. The literature search was carefully structured using the PICO framework to maintain clarity and precision. We combined keywords “atropine,” “atropine sulfate,” “intrathecal,” and “spinal anesthesia” using Boolean operators to get the most relevant results. The search focused on:
2.2. Data Collection
All records identified through the database searches were compiled and organized using Rayyan software. After removing duplicate entries, reviewers carefully reviewed the titles and abstracts to see which studies were relevant to the review. Full-text articles were then retrieved for studies that appeared to meet the eligibility criteria or required further evaluation. These full texts were reassessed in detail based on the predefined inclusion and exclusion criteria. If there were any differences in interpretation during the selection process, they were resolved through thoughtful discussion until agreement was reached. In cases where essential data were missing or unclear, study authors were contacted directly to request further information. For all studies that met the final inclusion criteria, data were extracted using a standardized form. This form captured key information such as the study’s publication year, author(s), country, number of participants, mean age, type of surgery, characteristics of the intervention, duration of follow-up, and reported clinical outcomes.
2.3. Risk of Bias Assessment
All studies included in this systematic review were evaluated for potential risk of bias according to their study design. Each study was independently reviewed by all members of the research team. To assess the quality and reliability of the evidence, the updated Cochrane Risk of Bias Tool for Randomized Trials (RoB 2.0) was used.10 In cases where there were differences in scoring between reviewers, any disparities were settled by discussion.
3.1. Study Selection
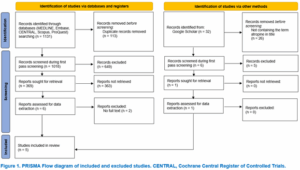
After a comprehensive search of five major databases (MEDLINE, Embase, CENTRAL, Scopus, and ProQuest), 1,113 records were identified. After removing 113 duplicates, 1,018 records were screened. Based on title and abstract review, 649 were excluded, and 363 full-text articles could not be retrieved. Six articles were assessed in detail, with two excluded due to lack of full text, resulting in four studies meeting the inclusion criteria. An additional search through Google Scholar identified 32 records; after title screening and eligibility assessment, one further study was included. In total, five randomized controlled trials were included in this review.11–15 The study selection process is illustrated in Figure 1.
3.2. Study characteristics
The five included studies were all RCTs published in three different countries between 2011 and 2025: Egypt (three studies),13–15 India (one study),12 and Italy (one study).11 The study included 584 patients who underwent elective surgery under SA. Of these, 242 patients received intrathecal atropine, 160 received other adjuvants such as dexamethasone, ondansetron, or metoclopramide, 67 received intravenous atropine, and 115 received normal saline. The surgical procedures included cesarean section (three studies),11,13,14 perineal surgery (one study),15 and lower abdominal surgery (one study).12 The postoperative follow-up duration ranged from 12 to 24 hours. Detailed characteristics of the included studies are summarized in Table 1.
Risk of bias assessment using the Cochrane RoB 2.0 tool showed that most studies had a low risk of bias (Figure 2).

Figure 2: Cochrane risk of bias summary in included studies.
[Legend: Green Circle, Low Risk of Bias; Yellow Circle, Unclear Risk of Bias; Red Circle, High Risk of Bias]
3.3. Incidence of PONV
The effect of intrathecal atropine on PONV was assessed in five studies,11–15 which included 584 patients in total (242 in the intrathecal atropine group, 227 in the comparator group, and 115 in the control group), evaluated the effect of intrathecal atropine on PONV. Baciarello et al. (2011) showed significantly lower PONV incidence with intrathecal atropine (15%) compared to intravenous atropine (37%) and normal saline (49%) (P < 0.001, respectively).11 Abdalla et al. (2019), using a 4-point scale (0: no nausea/vomiting; 1: mild, not requesting rescue; 2: moderate, requesting pharmacologic rescue; 3: severe, resistant to pharmacologic treatment), found that both nausea and vomiting scores were significantly reduced in the dexamethasone–atropine group (P = 0.025 and 0.013 for nausea vs dexamethasone and atropine monotherapy, respectively; P = 0.021 and 0.041 for vomiting).13
Turai et al. (2019), who applied a simplified grading system (Grade 1: >2 episodes/hour; Grade 2: >3 episodes/15 minutes), reported significant reductions in PONV incidence at 0–6 h and 6–12 h postoperatively in the intrathecal atropine group (P = 0.001).12 Farahat et al. (2019) found no significant differences in early nausea scores using a 3-point scale (0: none; 1: nausea; 2: nausea and vomiting); however, atropine significantly reduced late nausea scores (P = 0.005).14
Using the same 4-point rating system as Abdalla et al., Soliman et al. (2023) found that the atropine group had lower nausea and vomiting scores (P = 0.049 and 0.027, respectively).15 Overall, intrathecal atropine significantly reduced the incidence and severity of PONV, as shown in Table 2.
3.4. Postoperative Pain
Three studies used visual analogue scale (VAS) scores to assess the analgesic efficacy of different interventions. Using area under the curve (AUC) to measure pain intensity over a period of 0–12 hours, Baciarello et al. (2011) found no significant difference between intrathecal atropine, intravenous atropine, and normal saline groups (P = 0.935).11 Abdalla et al. (2019) reported no significant differences between atropine, dexamethasone, and atropine + dexamethasone groups pairwise (all P > 0.05).13
Farahat et al. (2019) assessed VAS scores on a categorical scale and found no significant difference between atropine and metoclopramide groups (P = 1.00).14 As shown in Table 3, no study found a statistically significant difference in the outcomes of early postoperative pain among the interventions that were evaluated.
3.5. Hemodynamic Parameters
In one study, Abdalla et al. (2019) evaluated the effect of intrathecal atropine on hemodynamic parameters over 24 hours postoperatively.13 The results showed no significant differences in systolic blood pressure (SBP), diastolic blood pressure (DBP), mean blood pressure (MBP), or heart rate (HR) between the atropine, dexamethasone, and atropine + dexamethasone groups pairwise (all P > 0.05), as shown in Table 4. Additionally, Soliman et al. (2023) compared intrathecal atropine with intravenous ondansetron in terms of MBP and HR based on graphical data; no significant difference was reported.15 Due to the absence of extractable numerical values, Soliman’s data on hemodynamic parameters were not included in the quantitative synthesis.
3.6. Onset of Sensory Block
Two studies evaluated the time to sensory block following spinal anesthesia, with or without intrathecal atropine. Abdalla et al. reported no statistically significant differences in sensory block onset time between the atropine, dexamethasone, and atropine + dexamethasone groups, with all pairwise comparisons showing P > 0.05.13 Similarly, Turai et al. found no significant difference between the intrathecal atropine and the normal saline group (P = 0.314). As shown in Table 5, the addition of intrathecal atropine did not significantly alter the onset of sensory block in either study.12
3.7. Adverse Effects
Table 6 summarizes five studies that reported the adverse effects of intrathecal atropine. According to Baciarello et al. (2011), there was no statistically significant difference in the incidence of intraoperative hypotension across groups (P = 1.00).11 Similarly, there were no significant differences between the groups in other adverse effects, including unexplained anxiety, visual disturbances, and xerostomia (P = 0.926, 0.128, and 0.532, respectively). Abdalla et al. observed that atropine group experienced 80% more postoperative itching than the dexamethasone (12.5%) and the atropine + dexamethasone group (15%).13 The difference was statistically significant when comparing atropine to both groups (P < 0.001), but there was no significant difference between the combination and dexamethasone groups (P = 0.75). No significant differences were observed in intraoperative hypotension among the three groups (P = 0.56 for all pairwise comparisons). Farahat et al. observed similar rates of intraoperative hypotension between the atropine and metoclopramide groups (P = 1.00).14 Turai et al. likewise reported no significant differences in the incidence of pruritus (P = 0.677), urinary retention (P = 0.749), intraoperative hypotension (P = 0.766), or unexplained anxiety (P = 1.00) between the atropine and normal saline groups.12 With all P-values > 0.05, Soliman et al. reported no significant differences in adverse effects between the atropine and ondansetron groups, including mouth dryness, initial bradycardia, headache, drowsiness, and abdominal distension.15 Except for a higher incidence of pruritus reported in one study, intrathecal atropine was not generally linked to an increased risk of common perioperative adverse effects when compared to other agents or a placebo.
This systematic review evaluated randomized controlled trials examining intrathecal atropine as an adjuvant in SA. After an extensive literature review across multiple databases, five studies with a total of 584 patients were ultimately included. According to the results, intrathecal atropine may enhance postoperative recovery by significantly lowering the incidence and severity of postoperative nausea and vomiting. Unlike other interventions or a placebo, it showed no significant effect on managing postoperative pain or the onset of sensory block. All groups showed mostly stable hemodynamic parameters, and no significant cardiovascular events were reported. Most studies did not show a significant increase in adverse effects, although one study indicated an increased incidence of pruritus in the atropine group.
The prototypical tertiary amine antimuscarinic agent, atropine, acts as a reversible and nonspecific antagonist of muscarinic acetylcholine receptors. Through competitive binding to all five muscarinic receptor subtypes (M1–M5), atropine effectively blocks parasympathetic signalling in central and peripheral tissues. It works by interfering with the muscarinic effects of acetylcholine on smooth muscles and glands that are responsive to endogenous acetylcholine but lacking direct innervation, as well as effector organs innervated by postganglionic cholinergic fibers.16–18 Atropine serves as the first-line therapy for symptomatic bradycardia, while also working as an anti-sialagogue and organophosphate poisoning antidote in clinical practice.19 In anesthesia practice, it is routinely administered intravenously to prevent or manage vagally mediated bradyarrhythmias, especially during procedures like laparoscopy, where vagal stimulation can occur.20 However, its ability to cross the blood–brain barrier because of the structural characteristic and exert central anticholinergic effects has spurred interest in alternative routes of administration, including intrathecal delivery. Intrathecal administration of atropine enables its central muscarinic receptor antagonism to potentially modulate autonomic tone at the spinal level.21,22 This route of administration may affect the visceral reflex pathways and reduce common postoperative complications such as nausea and vomiting, particularly in patients receiving intrathecal opioids. Furthermore, by delivering the drug directly to the central nervous system, intrathecal administration may reduce peripheral side effects that are commonly linked to systemic atropine use.11 Because of pharmacologic versatility and selective central action, atropine is an intriguing option for further study as an SA adjuvant to improve perioperative outcomes.
4.1. PONV
Intrathecal atropine has consistently reported significant efficacy in reducing both the incidence and severity of PONV compared to commonly used agents, including ondansetron and metoclopramide, during the early postoperative period. In all five studies reviewed,11–15 its antiemetic effect was most effective during the first 12 hours postoperatively since it aligned with the period when emetogenic stimuli peaked, which included elevated vagal activity, intra-abdominal manipulation, spinal-induced hypotension, and residual sedative or opioid effects.23–25 Although intrathecal atropine is not inherently time-dependent, its observed efficacy appears to align with the temporal pattern of these emetogenic stimuli. The duration of its central antiemetic effect has not been clearly defined, as no studies have characterized its overall pharmacologic profile via the intrathecal route. However, Turai et al. found that the antiemetic effects of intrathecal atropine lasted for up to 12 hours and hypothesized that the reduced efficacy after this period might be due to the drug clearance. This suggests a possible duration of action within this timeframe, although such inference is drawn from clinical outcomes rather than direct pharmacokinetic evidence.12
The antiemetic effect from atropine is thought to come from its central anticholinergic activity, particularly through inhibition of muscarinic receptors in the chemoreceptor trigger zone (CTZ) and vomiting center of the area postrema in the medulla oblongata, which is believed to underlie its antiemetic effect.26 The enhanced antiemetic efficacy also observed with the combination of intrathecal atropine and intravenous dexamethasone, as shown in the study by Abdalla et al., may be explained by their complementary mechanisms of action. Atropine exerts central anticholinergic effects, while dexamethasone provides potent anti-inflammatory effects that suppress the release of proinflammatory cytokines involved in nausea and vomiting.27 It may also reduce emetogenic signaling by inhibiting serotonin (5-HT) release from enterochromaffin cells and by dampening the activity of 5-HT₃ receptors.28 In addition, it can act centrally through glucocorticoid receptors found in areas including the nucleus of the solitary tract, the area postrema, and the raphe nuclei.13 A significant decrease in late nausea scores was found at 2–6 hours, but no significant difference was reported during the first two hours, according to Farahat et al., who measured nausea at discrete time intervals.14 This delayed onset may be attributed to the time required for atropine to exert sufficient central muscarinic blockade, possibly due to redistribution dynamics within the cerebrospinal fluid (CSF) or delayed interaction with its central targets. However, these interpretations remain speculative, and more study is necessary to determine the precise onset and duration of the central action of intrathecal atropine.
4.2. Postoperative Pain
Three studies consistently reported no statistically significant differences in postoperative pain between intrathecal atropine with comparator or control groups.11,13,14 This lack of observed difference may be explained by the residual analgesic effect of spinal anesthesia, which, in these studies, included the use of intrathecal opioids as adjuvants. The combined use of local anesthetics and opioids likely contributed to a prolonged and potent sensory blockade during the early postoperative period across all groups.6 As a result, any potential difference in analgesic effect between groups may have been masked. Additionally, atropine does not interact with key excitatory neurotransmitters involved in pain transmission, such as glutamate or substance P, nor does it affect N-methyl-D-aspartate (NMDA) receptors,29 resulting in minimal influence on pain intensity. Both VAS scores and AUC analyses were used in these studies to assess pain. Whereas VAS reflects pain intensity at isolated time points, AUC offers a more sensitive measure of cumulative pain over time and is therefore more sensitive to subtle differences in analgesic effect.30 The consistent findings across all three studies support the conclusion that intrathecal atropine does not affect postoperative analgesia.
4.3. Hemodynamic Parameter
Based on the evidence that is currently available, intrathecal atropine has no significant impact on postoperative hemodynamic parameters. Abdalla et al. found no significant differences in SBP, DBP, MBP, or HR among the groups receiving intrathecal atropine, dexamethasone, or their combination.13 These results demonstrate that intrathecal atropine is well tolerated and does not affect cardiovascular stability, whether used alone or in combination. Although the exact mechanism remains unclear, the stability may be related to its limited systemic absorption and predominantly localized spinal action,31 which reduces the risk of systemic antimuscarinic effects. Interestingly, the antimuscarinic properties of atropine, such as inhibition of vagal tone, could theoretically attenuate bradycardia and hypotension often associated with SA,18 but no significant evidence from current studies to support this. Similarly, Soliman et al. found no significant difference in MAP and HR between intrathecal atropine and intravenous ondansetron, although their data were excluded in our quantitative synthesis because there were no extractable numerical values.15
4.4. Onset of Sensory Block
The administration of atropine via the intrathecal route does not appear to affect or have a minimal effect on the onset time of sensory block during SA, as supported by consistent findings in multiple studies. Both Abdalla et al.13 and Turai et al.12 found no significant differences in the onset of sensory block, indicating that atropine does not affect the initiation phase of SA. This finding concurs with its pharmacological role as an antimuscarinic agent18, which does not act on the voltage-gated sodium channels responsible for sensory blockade. The onset of sensory block in SA is predominantly determined by the diffusion and binding of local anesthetic agents to sodium channels in the nerve roots within the subarachnoid space, resulting in a reversible inhibition of action potential propagation.32 Since atropine does not interfere with this mechanism, its negligible effect on sensory block onset is pharmacologically consistent and reflects its limited involvement in spinal nociceptive modulation.
4.5. Adverse Effects
The safety profile of intrathecal atropine reported in the included studies is generally favorable, showing no significant increase in common perioperative adverse effects compared to other agents or placebo. This favorable tolerability is probably due to the relatively low and consistent dose used in these studies, namely 100 μg, which allows direct delivery to the central nervous system while minimizing exposure to peripheral tissues, which reduces the risk of systemic adverse effects.14,33 Systemic muscarinic blockade effects commonly seen with higher doses include tachycardia, which is mediated by M2 receptors in the heart, xerostomia through M3 receptors in the salivary glands, or urinary retention involving M3 receptors in the detrusor muscle of the bladder.8 Interestingly, one study by Abdalla et al.13 reported an increased incidence of postoperative pruritus in the group who received intrathecal atropine alone. This finding suggests that, although systemic adverse effects may be limited, intrathecal atropine can still induce central nervous system-mediated adverse effects, possibly by the activation of muscarinic receptors, particularly the M3 subtype, which has been involved in sensory pathways related to itch perception.34 Notably, none of the included studies reported adverse effects such as blurry vision, which could be related to M3 receptors in the ciliary body,8,35 or headache and drowsiness, which are associated with M1 receptor blockade.36 The absence of these effects may suggest that low-dose intrathecal atropine primarily acts at the spinal level, with minimal cephalad spread and limited supraspinal involvement.
This review has several limitations, primarily related to methodological weaknesses and clinical heterogeneity among the included studies. Although all trials used a uniform intrathecal atropine dose of 100 μg, outcome definitions, assessment intervals, and comparator interventions varied considerably, limiting data pooling. Visual Analog Scale scoring and PONV grading systems were inconsistently applied; for pain assessment, some studies used VAS while others used AUC methods. Hemodynamic outcomes in one study were reported only graphically, preventing numerical data extraction and restricting direct comparison. Moreover, all studies were single-center trials with limited sample sizes and minimal demographic stratification, which may reduce external validity. The geographic distribution was limited to only three countries, and postoperative follow-up duration ranged from 12 to 24 hours, limiting assessment of longer-term outcomes. The lack of standardized endpoints and inconsistent reporting intervals also hindered quantitative synthesis. Potential publication bias and language bias are also concerns, as only English-language studies were included, and negative results may have been underreported. Future trials should adopt standardized outcome measures, ensure rigorous trial design and reporting, and consider multicenter collaboration to improve the quality of evidence on intrathecal atropine.
This systematic review indicates that intrathecal atropine at a dose of 100 µg may help reduce both the incidence and severity of PONV in patients undergoing surgery under SA. However, no significant benefits were observed in levels of postoperative pain, onset of sensory block, or hemodynamic parameters when compared with other agents or placebo. Overall, adverse effects were mild and similar between groups, though one study noted a higher occurrence of postoperative pruritus. Given the limited number of trials, small sample sizes, and methodological variability, further well-designed, multicenter studies with standardized outcome measures are needed to confirm these findings and strengthen the evidence base.
7. Data availability
The numerical data generated during this research are available from the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
TP: Conceptualization; Protocol design; Supervision; Database search strategy; Manuscript writing; Final approval.
M: Methodology; Literature search; Screening of studies; Data extraction; Manuscript review.
WIN: Risk of bias assessment; Data curation; Results synthesis; Visualizations (tables, figures).
SAW: Software and tools; Data validation; Critical revision. TS: Full-text eligibility assessment; Statistical support; Data interpretation; Triage of discrepancies; Formatting and PRISMA checklist; Proofreading.
CHM: Reference management; Submission handling; Final review.
AK: Project administration; Overall quality control
Authors affiliations:
- Taufan Pramadika, Sp. An-TI, Department of Anesthesiology and Intensive Therapy, Faculty of Medicine, Universitas Diponegoro, Semarang, Indonesia; Email: taufanpramadikamd@lecturer.undip.ac.id, ORCID ID: 0000-0001-5571-3671
- Mochamat, M.Si.Med, SpAn-TI, FIP, Department of Anesthesiology and Intensive Therapy, Faculty of Medicine, Universitas Diponegoro, Semarang, Indonesia; Email: mochamat@uni-bonn.de
- Widya Istanto Nurcahyo, Sp.An-TI, Subsp.An.KV.(K), An.R.(K), Department of Anesthesiology and Intensive Therapy, Dr. Kariadi General Hospital, Semarang, Indonesia; Email: widya_istanto2@yahoo.com
- Satrio Adi Wicaksono, SpAn-TI, Subsp.An.O(K), Department of Anesthesiology and Intensive Therapy, Faculty of Medicine, Universitas Diponegoro, Semarang, Indonesia; Email: drsaw11@yahoo.com
- Taufik Saputra, Department of Anesthesiology and Intensive Therapy, Faculty of Medicine, Universitas Diponegoro, Semarang, Indonesia; Email: taufiksaputra99@gmail.com; {ORCID:0009-0009-0346-8227}
- Chandra Hermawan Manapa, Department of Anesthesiology and Intensive Therapy, Faculty of Medicine, Universitas Diponegoro, Semarang, Indonesia; Email: chmanapa95@gmail.com
- Alesandro Ksatriaputra, Faculty of Medicine, Universitas Diponegoro, Semarang, Indonesia; Email: androksatria@gmail.com; {ORCID:0000-0002-6089-8743}
ABSTRACT
Introduction: Spinal anesthesia (SA) is widely used for lower abdominal and limb surgeries due to its rapid onset and favorable safety profile. However, because of sympathetic blockade, it frequently results in bradycardia and hypotension, increasing the risk of perioperative complications and postoperative nausea and vomiting (PONV). Intrathecal atropine has shown potential as an adjuvant to counteract these effects and improve anesthesia outcomes. This review evaluates current evidence on the safety and efficacy of intrathecal atropine in SA.
Methods: MEDLINE, EMBASE, CENTRAL, Scopus, ProQuest, and Google Scholar were searched for randomized controlled trials published in English up to April 2025 on intrathecal atropine as an adjuvant in SA. The primary outcome was PONV; secondary outcomes included sensory block onset, postoperative pain, and adverse effects. Risk of bias was assessed using the Cochrane RoB 2.0 tool.
Results: Five RCTs involving 584 patients were included. Intrathecal atropine significantly reduced the incidence and severity of PONV in most studies (P < 0.05). However, it did not show significant effects on postoperative pain scores, sensory block onset, or hemodynamic parameters compared to control or other adjuvants. Adverse effects were generally mild and comparable across groups, though one study reported a higher incidence of pruritus with atropine (P < 0.001). No serious complications were observed. Nonetheless, study limitations included small sample sizes, non-standardized outcomes, and single-center designs with limited follow-up.
Conclusions: Intrathecal atropine may reduce PONV after SA without serious adverse effects. However, its impact on pain, sensory block, and hemodynamics remains uncertain, warranting further large-scale, standardized trials.
Abbreviations: GA: general anesthesia, RCT: randomized controlled trial, SA: Spinal anesthesia, PONV: postoperative nausea and vomiting
Keywords: Adjuvant; Atropine; Complications; Intrathecal; Outcome; PONV; Postoperative Nausea and Vomiting; Spinal Anesthesia
Citation: Pramadika T, Mochamat, Nurcahyo WI, Wicaksono SA, Saputra T, Manapa CH, Ksatriaputra A. Intrathecal atropine as an adjuvant in spinal anesthesia: a systematic review of efficacy and safety. Anaesth. pain intensive care 2025;29(7):796-807. DOI: 10.35975/apic.v29i7.2947
Received: July 01, 2025; Revised: August 03, 2024; Accepted: August 07, 2025
1. INTRODUCTION
Spinal anesthesia (SA), also known as a subarachnoid block, remains one of the most commonly used techniques in regional anesthesia, especially for surgeries involving the lower abdomen, pelvis, and lower limbs.1 Its frequent use is supported by several well-known advantages, including rapid onset, reliable sensory and motor blockade, and better postoperative outcomes such as a lower incidence of nausea, vomiting, and respiratory depression when compared to general anesthesia (GA).2 However, it is also linked to hemodynamic adverse effects such as hypotension and bradycardia. These effects are caused by a sympathovagal imbalance that occurs from the blockade of the sympathetic nervous system and a relative increase in parasympathetic tone, which can be particularly concerning in patients with high block levels or limited cardiovascular reserve, potentially increasing perioperative risk.3,4 In addition to cardiovascular effects, this sympathetic blockade-induced hypotension has also been linked to an increased risk of postoperative nausea and vomiting (PONV), which can further affect patient comfort and recovery.5
Pharmacologic adjuvants administered intrathecally alongside SA have garnered increasing interest as a solution to these concerns. These agents aim to enhance the quality of analgesia during surgery, prolong the duration of anesthesia, and minimize the unwanted physiological effects.6 Among these options, atropine has gained particular attention. While it’s traditionally been used intravenously to manage bradyarrhythmias, atropine is a well-understood antimuscarinic agent. When administered intrathecally, it is thought to work by blocking muscarinic acetylcholine receptors in the spinal cord, which may reduce excessive vagal activity without causing significant systemic adverse effects.7,8
Although early findings have shown promise, the current research on intrathecal atropine varies greatly in terms of study design, patient characteristics, and outcome measures. Because of this variability, its true clinical value remains uncertain. Therefore, this review aims to evaluate the available evidence on the use of intrathecal atropine as an adjuvant to SA, focusing on both its safety and efficacy. By bringing together the findings from different clinical studies, this review endeavors to provide useful insights for clinical practice and highlight areas where further research is needed.
2. METHODOLOGY
This systematic review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines to evaluate the efficacy and safety of intrathecal atropine as an adjuvant in SA. There was no need for formal ethical approval because the review was based only on previously published research and did not directly involve any patient participation.9
2.1. Study Identification and Eligibility Criteria
A comprehensive literature search was conducted to find studies that evaluate the use of intrathecal atropine as an adjuvant in SA. This search covered all publications available up to April 2025 and included multiple electronic databases such as MEDLINE ([PubMed]), Embase (OVID), Cochrane Central Register of Controlled Trials (CENTRAL), Scopus, and ProQuest. To make sure no important studies were missed, we also manually searched Google Scholar alongside the database searches. The literature search was carefully structured using the PICO framework to maintain clarity and precision. We combined keywords “atropine,” “atropine sulfate,” “intrathecal,” and “spinal anesthesia” using Boolean operators to get the most relevant results. The search focused on:
- Population (P): Adults aged 18 and older undergoing SA for any type of surgery
- Intervention (I): Use of intrathecal atropine as an adjuvant
- Comparison (C): Other adjuvant agents
- Outcomes (O):
- Primary: Incidence of PONV
- Secondary: Levels of postoperative pain, hemodynamic parameters, onset time to sensory block, and any adverse effects reported
2.2. Data Collection
All records identified through the database searches were compiled and organized using Rayyan software. After removing duplicate entries, reviewers carefully reviewed the titles and abstracts to see which studies were relevant to the review. Full-text articles were then retrieved for studies that appeared to meet the eligibility criteria or required further evaluation. These full texts were reassessed in detail based on the predefined inclusion and exclusion criteria. If there were any differences in interpretation during the selection process, they were resolved through thoughtful discussion until agreement was reached. In cases where essential data were missing or unclear, study authors were contacted directly to request further information. For all studies that met the final inclusion criteria, data were extracted using a standardized form. This form captured key information such as the study’s publication year, author(s), country, number of participants, mean age, type of surgery, characteristics of the intervention, duration of follow-up, and reported clinical outcomes.
2.3. Risk of Bias Assessment
All studies included in this systematic review were evaluated for potential risk of bias according to their study design. Each study was independently reviewed by all members of the research team. To assess the quality and reliability of the evidence, the updated Cochrane Risk of Bias Tool for Randomized Trials (RoB 2.0) was used.10 In cases where there were differences in scoring between reviewers, any disparities were settled by discussion.
3. RESULTS
3.1. Study Selection
After a comprehensive search of five major databases (MEDLINE, Embase, CENTRAL, Scopus, and ProQuest), 1,113 records were identified. After removing 113 duplicates, 1,018 records were screened. Based on title and abstract review, 649 were excluded, and 363 full-text articles could not be retrieved. Six articles were assessed in detail, with two excluded due to lack of full text, resulting in four studies meeting the inclusion criteria. An additional search through Google Scholar identified 32 records; after title screening and eligibility assessment, one further study was included. In total, five randomized controlled trials were included in this review.11–15 The study selection process is illustrated in Figure 1.

3.2. Study characteristics
The five included studies were all RCTs published in three different countries between 2011 and 2025: Egypt (three studies),13–15 India (one study),12 and Italy (one study).11 The study included 584 patients who underwent elective surgery under SA. Of these, 242 patients received intrathecal atropine, 160 received other adjuvants such as dexamethasone, ondansetron, or metoclopramide, 67 received intravenous atropine, and 115 received normal saline. The surgical procedures included cesarean section (three studies),11,13,14 perineal surgery (one study),15 and lower abdominal surgery (one study).12 The postoperative follow-up duration ranged from 12 to 24 hours. Detailed characteristics of the included studies are summarized in Table 1.
| Table 1: Comparative results of the studies with the main outcome as nausea/vomiting | ||||||
| Study | Outcome | Results (n (%)) | Effect (95% CI) | P-value | ||
| I | C1 | C2/CG | ||||
| Baciarello et al.[11] | PONV | 10 (15) | 24 (37) | 33 (49) | I: 0.28 (0.15-0.53) C1: 0.75 (0.50-1.12) |
PIC1: < 0.001* PICG: < 0.001* |
| Abdalla et al.[13] | Nausea | Grade 1: 8 (20) | Grade 1: 1 (2.5) | Grade 2: 7 (17.5) | N/A | PIC1: 0.013*; PIC2: 0.778; PC1C2: 0.025* |
| Vomiting | Grade 1: 4 (10) | 0 (0) | Grade 1: 5 (12.5) | N/A | PIC1: 0.041*; PIC2: 0.728; PC1C2: 0.021* |
|
| Farahat et al.[14] | Early Nausea Score | 0: 11 (36.7); 1: 14 (46.7); 2: 5 (16.6) |
0: 10 (33.3); 1: 1 (46.7); 2: 6 (20) |
N/A | N/A | 0.933 |
| Late Nausea Score | 0: 13 (43.3); 1: 15 (50); 2: 2 (6.7) |
0: 3 (10); 1: 19 (63.3); 2: 8 (26.7) |
N/A | N/A | 0.005* | |
| Turai et al.[12] | PONV | 0-6 h: 14 (28); 6-12 h: 14 (28); 12-18 h: 10 (20) 18-24 h: 8 (16) |
N/A | 0-6h: 33 (66); 6-12h: 32 (64); 12-18h: 18 (36) 18-24h: 10 (20) |
N/A | 0–6 h: 0.001*; 6–12 h: 0.001*; 12–18 h: 0.074; 18–24 h: 0.271 |
| Soliman et al.[15] | Nausea | Grade 1: 1 (2); Grade 2: 1 (2) |
Grade 1:6 (12); Grade 2: 3 (6) |
N/A | N/A | 0.049* |
| Vomiting | 0 (0) | Grade 1: 5 (10); Grade 2: 1 (2) |
N/A | N/A | 0.027* | |
| I, Intervention; C1: Comparator 1; C2: Comparator 2; CG, Control Group; IT, Intrathecal; IV, Intravenous; N/A, Not Available; PONV, Postoperative Nausea and Vomiting; VAS, Visual Analog Scale; P < 0.05 considered as significant; PIC1= P-value between I and C1; PIC2 = P-value between I and C2; PC1C2 = P-value between C1 and C2 | ||||||
Risk of bias assessment using the Cochrane RoB 2.0 tool showed that most studies had a low risk of bias (Figure 2).

Figure 2: Cochrane risk of bias summary in included studies.
[Legend: Green Circle, Low Risk of Bias; Yellow Circle, Unclear Risk of Bias; Red Circle, High Risk of Bias]
3.3. Incidence of PONV
The effect of intrathecal atropine on PONV was assessed in five studies,11–15 which included 584 patients in total (242 in the intrathecal atropine group, 227 in the comparator group, and 115 in the control group), evaluated the effect of intrathecal atropine on PONV. Baciarello et al. (2011) showed significantly lower PONV incidence with intrathecal atropine (15%) compared to intravenous atropine (37%) and normal saline (49%) (P < 0.001, respectively).11 Abdalla et al. (2019), using a 4-point scale (0: no nausea/vomiting; 1: mild, not requesting rescue; 2: moderate, requesting pharmacologic rescue; 3: severe, resistant to pharmacologic treatment), found that both nausea and vomiting scores were significantly reduced in the dexamethasone–atropine group (P = 0.025 and 0.013 for nausea vs dexamethasone and atropine monotherapy, respectively; P = 0.021 and 0.041 for vomiting).13
Turai et al. (2019), who applied a simplified grading system (Grade 1: >2 episodes/hour; Grade 2: >3 episodes/15 minutes), reported significant reductions in PONV incidence at 0–6 h and 6–12 h postoperatively in the intrathecal atropine group (P = 0.001).12 Farahat et al. (2019) found no significant differences in early nausea scores using a 3-point scale (0: none; 1: nausea; 2: nausea and vomiting); however, atropine significantly reduced late nausea scores (P = 0.005).14
Using the same 4-point rating system as Abdalla et al., Soliman et al. (2023) found that the atropine group had lower nausea and vomiting scores (P = 0.049 and 0.027, respectively).15 Overall, intrathecal atropine significantly reduced the incidence and severity of PONV, as shown in Table 2.
| Table 2: Comparative results of the studies using bupivacaine + morphine or fentanyl, with the main outcome as PONV, VAS etc. | ||||||||
| Author (Country, Year) | Number of Participants | Type of Surgery | Intervention | Comparator | Comparator 2/Control | Outcome | ||
| I | C1 | C2/CG | ||||||
| Baciarello et al.[11] (Italy, 2011) | 72 | 67 | 65 | Cesarean section | 12.5 mg 0.5% hyperbaric bupivacaine + 200 μg morphine + 100 μg IT atropine | 12.5 mg 0.5% hyperbaric bupivacaine + 200 μg morphine + 100 μg IV atropine | 12.5 mg 0.5% hyperbaric bupivacaine + 200 μg morphine + Normal Saline | PONV, Hemodynamic Parameter, Adverse Effect |
| Abdalla et al.[13] (Egypt, 2019) | 40 | 40 | 40 | Cesarean section | 10 mg 0.5% hyperbaric bupivacaine + 200 μg morphine + 100 μg IT atropine | 10 mg 0.5% hyperbaric bupivacaine + 200 μg morphine + 100 μg IT atropine + 8 mg IV Dexamethasone | 10 mg 0.5% hyperbaric bupivacaine + 200 μg morphine + 8 mg IV Dexamethasone | PONV, VAS Score, Hemodynamic Parameter, Onset of Sensory Block, Adverse Effect |
| Farahat et al.[14] (Egypt, 2019) | 30 | 30 | N/A | Cesarean section | 12.5 mg 0.5% hyperbaric bupivacaine + 25 μg fentanyl + 100 μg IT atropine | 12.5 mg 0.5% hyperbaric bupivacaine + 25 μg fentanyl + 10 mg IV Metoclopramide | N/A | PONV, VAS Score, Hemodynamic Parameter, Adverse Effect |
| Turai et al.[12] (India, 2019) | 50 | N/A | 50 | Lower abdominal surgery | 15 mg 0.5% hyperbaric bupivacaine + 200 μg morphine + 100 μg IT atropine | N/A | 15 mg 0.5% hyperbaric bupivacaine + 200 μg morphine + Normal Saline | PONV, Onset of Sensory Block, Adverse Effect |
| Soliman et al.[15] (Egypt, 2023) | 50 | 50 | N/A | Perineal surgery | 10 mg 0.5% hyperbaric bupivacaine + 250 μg morphine + 100 μg IT atropine | 10 mg 0.5% hyperbaric bupivacaine + 250 μg morphine + 4 mg IV Ondansetron | N/A | PONV, Hemodynamic Parameter, Adverse Effect |
3.4. Postoperative Pain
Three studies used visual analogue scale (VAS) scores to assess the analgesic efficacy of different interventions. Using area under the curve (AUC) to measure pain intensity over a period of 0–12 hours, Baciarello et al. (2011) found no significant difference between intrathecal atropine, intravenous atropine, and normal saline groups (P = 0.935).11 Abdalla et al. (2019) reported no significant differences between atropine, dexamethasone, and atropine + dexamethasone groups pairwise (all P > 0.05).13
Farahat et al. (2019) assessed VAS scores on a categorical scale and found no significant difference between atropine and metoclopramide groups (P = 1.00).14 As shown in Table 3, no study found a statistically significant difference in the outcomes of early postoperative pain among the interventions that were evaluated.
| Table 3: Comparative results of the studies with the main outcome as pain | ||||||
| Study | Outcome | Results | Effect (95% CI) | P-value | ||
| I | C1 | C2/CG | ||||
| Baciarello et al.[11] (Median (IQR)) | AUC (0-12h) | 20 (9-22) | 19 (15-24) | 20 (14-26) | N/A | 0.935 |
| Abdalla et al.[13]
(Mean + SD) |
VAS | 1.3 ± 1.12 | 1.1 ± 1.1 | 1.2 ± 1.1 | N/A | PIC1: 0.47; PIC2: 0.69; PC1C2: 0.69 |
| Farahat et al.[14]
(n (%)) |
VAS | 3: 17 (56.7); 4: 13 (43.3) |
3: 18 (60); 4: 12 (40) |
N/A | N/A | 1.00 |
| I, Intervention; C1: Comparator 1; C2: Comparator 2; CG, Control Group; N/A, Not Available; VAS, Visual Analog Scale; PIC1 = P-value between I and C1; PIC2 = P-value between I and C2; PC1C2 = P-value between C1 and C2 | ||||||
3.5. Hemodynamic Parameters
In one study, Abdalla et al. (2019) evaluated the effect of intrathecal atropine on hemodynamic parameters over 24 hours postoperatively.13 The results showed no significant differences in systolic blood pressure (SBP), diastolic blood pressure (DBP), mean blood pressure (MBP), or heart rate (HR) between the atropine, dexamethasone, and atropine + dexamethasone groups pairwise (all P > 0.05), as shown in Table 4. Additionally, Soliman et al. (2023) compared intrathecal atropine with intravenous ondansetron in terms of MBP and HR based on graphical data; no significant difference was reported.15 Due to the absence of extractable numerical values, Soliman’s data on hemodynamic parameters were not included in the quantitative synthesis.
| Table 4: Comparative hemodynamic parameters of a study | ||||||
| Study | Outcome | Results (Mean + SD) | Effect (95% CI) | P-value | ||
| I | C1 | C2/CG | ||||
| Abdalla et al.[13] | SBP 24h | 103.3 ± 8.4 | 106.5 ± 8.1 | 104.2 ± 6.2 | N/A | PIC1: 0.09; PIC2: 0.59; PC1C2: 0.16 |
| DBP 24h | 70.6 ± 4.5 | 71.6 + 5.4 | 69.3 ± 6.0 | N/A | PIC1: 0.37; PIC2: 0.28; PC1C2: 0.08 |
|
| MBP 24h | 81.9 ± 6.2 | 83.2 ± 8.2 | 81.1 ± 4.3 | N/A | PIC1: 0.16; PIC2: 0.50; PC1C2: 0.15 |
|
| HR 24h | 88.6 ± 12.9 | 86.6 + 12.3 | 86.3 ± 11.2 | N/A | PIC1: 0.48; PIC2: 0.39; PC1C2: 0.91 |
|
| I, Intervention; C1: Comparator 1; C2: Comparator 2; CG, Control Group; N/A, Not Available; SBP, Systolic Blood Pressure; DBP, Diastolic Blood Pressure; MBP, Mean Blood Pressure; HR, Heart Rate ; PIC1, P-value between I and C1; PIC2,P-value between I and C2; PC1C2, P-value between C1 and C2 | ||||||
3.6. Onset of Sensory Block
Two studies evaluated the time to sensory block following spinal anesthesia, with or without intrathecal atropine. Abdalla et al. reported no statistically significant differences in sensory block onset time between the atropine, dexamethasone, and atropine + dexamethasone groups, with all pairwise comparisons showing P > 0.05.13 Similarly, Turai et al. found no significant difference between the intrathecal atropine and the normal saline group (P = 0.314). As shown in Table 5, the addition of intrathecal atropine did not significantly alter the onset of sensory block in either study.12
| Table 5: Comparative complications / side-effects in the studies | ||||||
| Study | Outcome | Results (n (%)) | Effect (95% CI) | P-value | ||
| I | C1 | C2/CG | ||||
| Baciarello et al.[11] | Intraoperative Hypotension | 2 (3) | 2 (3) | 3 (5) | N/A | 1.00 |
| Unexplained Anxiety | 5 (7) | 4 (6) | 5 (8) | N/A | 0.926 | |
| Visual Disturbance | 4 (6) | 0 (0) | 4 (6) | N/A | 0.128 | |
| Xerostomia | 18 (25) | 15 (22) | 20 (31) | N/A | 0.532 | |
| Abdalla et al.[13] | Intraoperative Hypotension | 2 (5) | 1 (2.5) | 1 (2.5) | N/A | PIC1: 0.56; PIC2: 0.56; PC1C2: 1.00 |
| Postoperative Itching | 32 (80) | 6 (15) | 5 (12.5) | N/A | PIC1: <0.001* PIC2: <0.001* PC1C2: 0.75 |
|
| Farahat et al.[14] | Intraoperative Hypotension | 1: 14 (46.7) 2: 16 (53.3) |
1: 14 (46.7) 2: 16 (53.3) |
N/A | N/A | 1.00 |
| Turai et al.[12] | Pruritus | 17 (34) | N/A | 19 (38) | N/A | 0.677 |
| Urinary Retention | 6 (12) | N/A | 5 (10) | N/A | 0.749 | |
| Intraoperative Hypotension | 7 (14)_ | N/A | 6 (12) | N/A | 0.766 | |
| Unexplained Anxiety | 3 (6) | N/A | 2 (4) | N/A | 1.00 | |
| Soliman et al.[15] | Mouth Dryness | 2 (4) | 0 (0) | N/A | N/A | 0.495 |
| Initial Bradycardia | 1 (2) | 0 (0) | N/A | N/A | 1.00 | |
| Headache | 0 (0) | 3 (6) | N/A | N/A | 0.242 | |
| Drowsiness | 0 (0) | 1 (2) | N/A | N/A | 1.00 | |
| Distension | 0 (0) | 1 (2) | N/A | N/A | 1.00 | |
| I, Intervention; C1: Comparator 1; C2: Comparator 2; CG, Control Group; N/A, Not Available; PIC1, P-value between I and C1; PIC2,P-value between I and C2; PC1C2, P-value between C1 and C2 | ||||||
3.7. Adverse Effects
Table 6 summarizes five studies that reported the adverse effects of intrathecal atropine. According to Baciarello et al. (2011), there was no statistically significant difference in the incidence of intraoperative hypotension across groups (P = 1.00).11 Similarly, there were no significant differences between the groups in other adverse effects, including unexplained anxiety, visual disturbances, and xerostomia (P = 0.926, 0.128, and 0.532, respectively). Abdalla et al. observed that atropine group experienced 80% more postoperative itching than the dexamethasone (12.5%) and the atropine + dexamethasone group (15%).13 The difference was statistically significant when comparing atropine to both groups (P < 0.001), but there was no significant difference between the combination and dexamethasone groups (P = 0.75). No significant differences were observed in intraoperative hypotension among the three groups (P = 0.56 for all pairwise comparisons). Farahat et al. observed similar rates of intraoperative hypotension between the atropine and metoclopramide groups (P = 1.00).14 Turai et al. likewise reported no significant differences in the incidence of pruritus (P = 0.677), urinary retention (P = 0.749), intraoperative hypotension (P = 0.766), or unexplained anxiety (P = 1.00) between the atropine and normal saline groups.12 With all P-values > 0.05, Soliman et al. reported no significant differences in adverse effects between the atropine and ondansetron groups, including mouth dryness, initial bradycardia, headache, drowsiness, and abdominal distension.15 Except for a higher incidence of pruritus reported in one study, intrathecal atropine was not generally linked to an increased risk of common perioperative adverse effects when compared to other agents or a placebo.
4. DISCUSSION
This systematic review evaluated randomized controlled trials examining intrathecal atropine as an adjuvant in SA. After an extensive literature review across multiple databases, five studies with a total of 584 patients were ultimately included. According to the results, intrathecal atropine may enhance postoperative recovery by significantly lowering the incidence and severity of postoperative nausea and vomiting. Unlike other interventions or a placebo, it showed no significant effect on managing postoperative pain or the onset of sensory block. All groups showed mostly stable hemodynamic parameters, and no significant cardiovascular events were reported. Most studies did not show a significant increase in adverse effects, although one study indicated an increased incidence of pruritus in the atropine group.
The prototypical tertiary amine antimuscarinic agent, atropine, acts as a reversible and nonspecific antagonist of muscarinic acetylcholine receptors. Through competitive binding to all five muscarinic receptor subtypes (M1–M5), atropine effectively blocks parasympathetic signalling in central and peripheral tissues. It works by interfering with the muscarinic effects of acetylcholine on smooth muscles and glands that are responsive to endogenous acetylcholine but lacking direct innervation, as well as effector organs innervated by postganglionic cholinergic fibers.16–18 Atropine serves as the first-line therapy for symptomatic bradycardia, while also working as an anti-sialagogue and organophosphate poisoning antidote in clinical practice.19 In anesthesia practice, it is routinely administered intravenously to prevent or manage vagally mediated bradyarrhythmias, especially during procedures like laparoscopy, where vagal stimulation can occur.20 However, its ability to cross the blood–brain barrier because of the structural characteristic and exert central anticholinergic effects has spurred interest in alternative routes of administration, including intrathecal delivery. Intrathecal administration of atropine enables its central muscarinic receptor antagonism to potentially modulate autonomic tone at the spinal level.21,22 This route of administration may affect the visceral reflex pathways and reduce common postoperative complications such as nausea and vomiting, particularly in patients receiving intrathecal opioids. Furthermore, by delivering the drug directly to the central nervous system, intrathecal administration may reduce peripheral side effects that are commonly linked to systemic atropine use.11 Because of pharmacologic versatility and selective central action, atropine is an intriguing option for further study as an SA adjuvant to improve perioperative outcomes.
4.1. PONV
Intrathecal atropine has consistently reported significant efficacy in reducing both the incidence and severity of PONV compared to commonly used agents, including ondansetron and metoclopramide, during the early postoperative period. In all five studies reviewed,11–15 its antiemetic effect was most effective during the first 12 hours postoperatively since it aligned with the period when emetogenic stimuli peaked, which included elevated vagal activity, intra-abdominal manipulation, spinal-induced hypotension, and residual sedative or opioid effects.23–25 Although intrathecal atropine is not inherently time-dependent, its observed efficacy appears to align with the temporal pattern of these emetogenic stimuli. The duration of its central antiemetic effect has not been clearly defined, as no studies have characterized its overall pharmacologic profile via the intrathecal route. However, Turai et al. found that the antiemetic effects of intrathecal atropine lasted for up to 12 hours and hypothesized that the reduced efficacy after this period might be due to the drug clearance. This suggests a possible duration of action within this timeframe, although such inference is drawn from clinical outcomes rather than direct pharmacokinetic evidence.12
The antiemetic effect from atropine is thought to come from its central anticholinergic activity, particularly through inhibition of muscarinic receptors in the chemoreceptor trigger zone (CTZ) and vomiting center of the area postrema in the medulla oblongata, which is believed to underlie its antiemetic effect.26 The enhanced antiemetic efficacy also observed with the combination of intrathecal atropine and intravenous dexamethasone, as shown in the study by Abdalla et al., may be explained by their complementary mechanisms of action. Atropine exerts central anticholinergic effects, while dexamethasone provides potent anti-inflammatory effects that suppress the release of proinflammatory cytokines involved in nausea and vomiting.27 It may also reduce emetogenic signaling by inhibiting serotonin (5-HT) release from enterochromaffin cells and by dampening the activity of 5-HT₃ receptors.28 In addition, it can act centrally through glucocorticoid receptors found in areas including the nucleus of the solitary tract, the area postrema, and the raphe nuclei.13 A significant decrease in late nausea scores was found at 2–6 hours, but no significant difference was reported during the first two hours, according to Farahat et al., who measured nausea at discrete time intervals.14 This delayed onset may be attributed to the time required for atropine to exert sufficient central muscarinic blockade, possibly due to redistribution dynamics within the cerebrospinal fluid (CSF) or delayed interaction with its central targets. However, these interpretations remain speculative, and more study is necessary to determine the precise onset and duration of the central action of intrathecal atropine.
4.2. Postoperative Pain
Three studies consistently reported no statistically significant differences in postoperative pain between intrathecal atropine with comparator or control groups.11,13,14 This lack of observed difference may be explained by the residual analgesic effect of spinal anesthesia, which, in these studies, included the use of intrathecal opioids as adjuvants. The combined use of local anesthetics and opioids likely contributed to a prolonged and potent sensory blockade during the early postoperative period across all groups.6 As a result, any potential difference in analgesic effect between groups may have been masked. Additionally, atropine does not interact with key excitatory neurotransmitters involved in pain transmission, such as glutamate or substance P, nor does it affect N-methyl-D-aspartate (NMDA) receptors,29 resulting in minimal influence on pain intensity. Both VAS scores and AUC analyses were used in these studies to assess pain. Whereas VAS reflects pain intensity at isolated time points, AUC offers a more sensitive measure of cumulative pain over time and is therefore more sensitive to subtle differences in analgesic effect.30 The consistent findings across all three studies support the conclusion that intrathecal atropine does not affect postoperative analgesia.
4.3. Hemodynamic Parameter
Based on the evidence that is currently available, intrathecal atropine has no significant impact on postoperative hemodynamic parameters. Abdalla et al. found no significant differences in SBP, DBP, MBP, or HR among the groups receiving intrathecal atropine, dexamethasone, or their combination.13 These results demonstrate that intrathecal atropine is well tolerated and does not affect cardiovascular stability, whether used alone or in combination. Although the exact mechanism remains unclear, the stability may be related to its limited systemic absorption and predominantly localized spinal action,31 which reduces the risk of systemic antimuscarinic effects. Interestingly, the antimuscarinic properties of atropine, such as inhibition of vagal tone, could theoretically attenuate bradycardia and hypotension often associated with SA,18 but no significant evidence from current studies to support this. Similarly, Soliman et al. found no significant difference in MAP and HR between intrathecal atropine and intravenous ondansetron, although their data were excluded in our quantitative synthesis because there were no extractable numerical values.15
4.4. Onset of Sensory Block
The administration of atropine via the intrathecal route does not appear to affect or have a minimal effect on the onset time of sensory block during SA, as supported by consistent findings in multiple studies. Both Abdalla et al.13 and Turai et al.12 found no significant differences in the onset of sensory block, indicating that atropine does not affect the initiation phase of SA. This finding concurs with its pharmacological role as an antimuscarinic agent18, which does not act on the voltage-gated sodium channels responsible for sensory blockade. The onset of sensory block in SA is predominantly determined by the diffusion and binding of local anesthetic agents to sodium channels in the nerve roots within the subarachnoid space, resulting in a reversible inhibition of action potential propagation.32 Since atropine does not interfere with this mechanism, its negligible effect on sensory block onset is pharmacologically consistent and reflects its limited involvement in spinal nociceptive modulation.
4.5. Adverse Effects
The safety profile of intrathecal atropine reported in the included studies is generally favorable, showing no significant increase in common perioperative adverse effects compared to other agents or placebo. This favorable tolerability is probably due to the relatively low and consistent dose used in these studies, namely 100 μg, which allows direct delivery to the central nervous system while minimizing exposure to peripheral tissues, which reduces the risk of systemic adverse effects.14,33 Systemic muscarinic blockade effects commonly seen with higher doses include tachycardia, which is mediated by M2 receptors in the heart, xerostomia through M3 receptors in the salivary glands, or urinary retention involving M3 receptors in the detrusor muscle of the bladder.8 Interestingly, one study by Abdalla et al.13 reported an increased incidence of postoperative pruritus in the group who received intrathecal atropine alone. This finding suggests that, although systemic adverse effects may be limited, intrathecal atropine can still induce central nervous system-mediated adverse effects, possibly by the activation of muscarinic receptors, particularly the M3 subtype, which has been involved in sensory pathways related to itch perception.34 Notably, none of the included studies reported adverse effects such as blurry vision, which could be related to M3 receptors in the ciliary body,8,35 or headache and drowsiness, which are associated with M1 receptor blockade.36 The absence of these effects may suggest that low-dose intrathecal atropine primarily acts at the spinal level, with minimal cephalad spread and limited supraspinal involvement.
5. LIMITATIONS
This review has several limitations, primarily related to methodological weaknesses and clinical heterogeneity among the included studies. Although all trials used a uniform intrathecal atropine dose of 100 μg, outcome definitions, assessment intervals, and comparator interventions varied considerably, limiting data pooling. Visual Analog Scale scoring and PONV grading systems were inconsistently applied; for pain assessment, some studies used VAS while others used AUC methods. Hemodynamic outcomes in one study were reported only graphically, preventing numerical data extraction and restricting direct comparison. Moreover, all studies were single-center trials with limited sample sizes and minimal demographic stratification, which may reduce external validity. The geographic distribution was limited to only three countries, and postoperative follow-up duration ranged from 12 to 24 hours, limiting assessment of longer-term outcomes. The lack of standardized endpoints and inconsistent reporting intervals also hindered quantitative synthesis. Potential publication bias and language bias are also concerns, as only English-language studies were included, and negative results may have been underreported. Future trials should adopt standardized outcome measures, ensure rigorous trial design and reporting, and consider multicenter collaboration to improve the quality of evidence on intrathecal atropine.
6. CONCLUSION
This systematic review indicates that intrathecal atropine at a dose of 100 µg may help reduce both the incidence and severity of PONV in patients undergoing surgery under SA. However, no significant benefits were observed in levels of postoperative pain, onset of sensory block, or hemodynamic parameters when compared with other agents or placebo. Overall, adverse effects were mild and similar between groups, though one study noted a higher occurrence of postoperative pruritus. Given the limited number of trials, small sample sizes, and methodological variability, further well-designed, multicenter studies with standardized outcome measures are needed to confirm these findings and strengthen the evidence base.
7. Data availability
The numerical data generated during this research are available from the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
TP: Conceptualization; Protocol design; Supervision; Database search strategy; Manuscript writing; Final approval.
M: Methodology; Literature search; Screening of studies; Data extraction; Manuscript review.
WIN: Risk of bias assessment; Data curation; Results synthesis; Visualizations (tables, figures).
SAW: Software and tools; Data validation; Critical revision. TS: Full-text eligibility assessment; Statistical support; Data interpretation; Triage of discrepancies; Formatting and PRISMA checklist; Proofreading.
CHM: Reference management; Submission handling; Final review.
AK: Project administration; Overall quality control
11. REFERENCES
- Olawin AM, Das JM. Spinal Anesthesia [Internet]. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025 [cited 2025 Mar 21]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK537299/
- Sun S, Wang J, Bao N, Chen Y, Wang J. Comparison of dexmedetomidine and fentanyl as local anesthetic adjuvants in spinal anesthesia: a systematic review and meta-analysis of randomized controlled trials. Drug Des Devel Ther. 2017;11:3413–24. [PubMed] DOI: 2147/DDDT.S146092
- Hofhuizen C, Lemson J, Snoeck M, Scheffer GJ. Spinal anesthesia-induced hypotension is caused by a decrease in stroke volume in elderly patients. Local Reg Anesth. 2019;12:19–26. [PubMed] DOI: 2147/LRA.S193925
- Ferré F, Martin C, Bosch L, Kurrek M, Lairez O, Minville V. Control of Spinal Anesthesia-Induced Hypotension in Adults. Local Reg Anesth. 2020;13:39–46. [PubMed] DOI: 2147/LRA.S240753
- Koo BW. Achieving relief from nausea and vomiting: from intraoperative to postoperative management. Korean J Anesthesiol. 2024;77(6):573–4. [PubMed] DOI: 4097/kja.24779
- Swain A, Nag DS, Sahu S, Samaddar DP. Adjuvants to local anesthetics: Current understanding and future trends. World J Clin Cases. 2017;5(8):307–23. [PubMed] DOI: 12998/wjcc.v5.i8.307
- Aldossary SA. Review on Pharmacology of Atropine, Clinical Use and Toxicity. Biomed Pharmacol J. 2022;15(2):691–7. DOI: 13005/bpj/2408
- Naji A, Gatling JW. Muscarinic Antagonists [Internet]. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025 [cited 2025 Mar 21]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK557541/
- Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M, et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev. 2015;4(1):1. [PubMed] DOI: 1186/2046-4053-4-1
- Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. [PubMed] DOI: 1136/bmj.l4898
- Baciarello M, Cornini A, Zasa M, Pedrona P, Scrofani G, Venuti FS, et al. Intrathecal atropine to prevent postoperative nausea and vomiting after cesarean section: A randomized, controlled trial. Minerva Anestesiol. 2011;77(8):781–8. [PubMed]
- Turai A, Prabha P, Rachana. A Clinical Study to Evaluate the Effect of Intrathecal Atropine on Post Operative Nausea and Vomiting in Patients Receiving Intrathecal Morphine and Hyperbaric Bupivacaine for Spinal Anaesthesia: Prospective Randomized Trial. Karnataka Anaesth J. 2019;17(1-2):23–31. Full Text
- Abdalla E, Kamel EZ, Farrag WS. Intravenous dexamethasone combined with intrathecal atropine to prevent morphine-related nausea and vomiting after cesarean delivery: A randomized double-blinded study. Egypt J Anaesth. 2019;35(1):65–70. Full Text
- Farahat TEM, Abdelhafez MS. Intrathecal atropine versus intravenous metoclopramide for prevention of nausea and vomiting during cesarean section under spinal anesthesia. Res Opin Anesth Intensive Care. 2019;6(4):393. Full Text
- Soliman FI, Mousa EA, Ahmed SM, Hassan AH. Intrathecal atropine versus preoperative intravenous ondasetron for prevention of postoperative nausea and vomiting due to intrathecal morphine in perineal surgery. Res Opin Anesth Intensive Care. 2023;10(3):271. Full Text DOI: 4103/roaic.roaic_24_23
- Waseem R, Hussein MIH, Salih TSM, Ahmed SMG. Pharmacology of the Autonomic Nervous System [Internet]. In: Topics in Autonomic Nervous System. IntechOpen; 2023 [cited 2025 Mar 21]. Available from: https://www.intechopen.com/chapters/88194
- Buels KS, Fryer AD. Muscarinic receptor antagonists: effects on pulmonary function. Handb Exp Pharmacol. 2012;(208):317–41. [PubMed] DOI: 1007/978-3-642-23274-9_14
- Howland MA. Atropine. In: Nelson LS, Howland MA, Lewin NA, Smith SW, Goldfrank LR, Hoffman RS, editors. Goldfrank’s Toxicologic Emergencies. 11th ed. New York, NY: McGraw-Hill Education; 2019 [cited 2025 Mar 21]. Available from: mhmedical.com/content.aspx?aid=1163003488
- Eddleston M, Chowdhury FR. Pharmacological treatment of organophosphorus insecticide poisoning: the old and the (possible) new. Br J Clin Pharmacol. 2016;81(3):462–70. [PubMed] DOI: 1111/bcp.12784
- Heyba M, Khalil A, Elkenany Y. Severe Intraoperative Bradycardia during Laparoscopic Cholecystectomy due to Rapid Peritoneal Insufflation. Case Rep Anesthesiol. 2020;2020:8828914. [PubMed] DOI: 1155/2020/8828914
- Robinson D, Araklitis G. Anticholinergic therapy: A case-based approach. Case Rep Womens Health. 2020;25:e00164. [PubMed] DOI: 1016/j.crwh.2019.e00164
- Jordan LM, McVagh JR, Noga BR, Cabaj AM, Majczyński H, Sławińska U, et al. Cholinergic mechanisms in spinal locomotion—potential target for rehabilitation approaches [Internet]. Front Neural Circuits. 2014;8:132 [cited 2025 Mar 21]. [PubMed] DOI: 3389/fncir.2014.00132
- Maisiyiti A, Tian M, Chen JDZ. Acceleration of postoperative recovery with brief intraoperative vagal nerve stimulation mediated via the autonomic mechanism [Internet]. Front Neurosci. 2023;17:1188781. [PubMed] DOI: 3389/fnins.2023.1188781
- Shaikh SI, Nagarekha D, Hegade G, Marutheesh M. Postoperative nausea and vomiting: A simple yet complex problem. Anesth Essays Res. 2016;10(3):388–96. [PubMed] DOI: 4103/0259-1162.179310
- Chatterjee S, Rudra A, Sengupta S. Current Concepts in the Management of Postoperative Nausea and Vomiting. Anesthesiol Res Pract. 2011;2011:748031. [PubMed] DOI: 1155/2011/748031
- Zhong W, Shahbaz O, Teskey G, Beever A, Kachour N, Venketaraman V, et al. Mechanisms of Nausea and Vomiting: Current Knowledge and Recent Advances in Intracellular Emetic Signaling Systems. Int J Mol Sci. 2021;22(11):5797. [PubMed] DOI: 3390/ijms22115797
- Ottaiano A, Santorsola M, Capuozzo M, Scala S. Balancing immunotherapy and corticosteroids in cancer treatment: dilemma or paradox? Oncologist. 2025;30(3):oyaf045. [PubMed] DOI: 1093/oncolo/oyaf045
- Li BUK, Sunku BK. Vomiting and Nausea. In: Wyllie R, Hyams JS, editors. Pediatric Gastrointestinal and Liver Disease. 4th ed. Saint Louis: W.B. Saunders; 2011. p. 88-105.
- Deng M, Chen SR, Pan HL. Presynaptic NMDA receptors control nociceptive transmission at the spinal cord level in neuropathic pain. Cell Mol Life Sci. 2019;76(10):1889–99. [PubMed] DOI: 1007/s00018-019-03047-y
- De Andres J, Hayek S, Perruchoud C, Lawrence MM, Reina MA, De Andres-Serrano C, et al. Intrathecal Drug Delivery: Advances and Applications in the Management of Chronic Pain Patient. Front Pain Res. 2022;3:900566. [PubMed] DOI: 3389/fpain.2022.900566
- Hermanns H, Hollmann MW, Stevens MF, Lirk P, Brandenburger T, Piegeler T, et al. Molecular mechanisms of action of systemic lidocaine in acute and chronic pain: a narrative review. Br J Anaesth. 2019;123(3):335–49. [PubMed] DOI: 1016/j.bja.2019.06.014
- Lu CT, Zhao YZ, Wong HL, Cai J, Peng L, Tian XQ. Current approaches to enhance CNS delivery of drugs across the brain barriers. Int J Nanomedicine. 2014;9:2241–57. [PubMed] DOI: 2147/IJN.S61288
- Hassan I, Haji MLI. Understanding itch: An update on mediators and mechanisms of pruritus. Indian J Dermatol Venereol Leprol. 2014;80(2):106-14. [PubMed] DOI: 4103/0378-6323.129377
- Kudlak M, Tadi P. Physiology, Muscarinic Receptor [Internet]. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025 [cited 2025 Mar 21]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK555909/
- Scarr E. Muscarinic M1 receptor agonists: can they improve cognitive performance? Int J Neuropsychopharmacol. 2013;16(4):717–20. [PubMed] DOI: 1017/S1461145712001113