Marwa Eloraby 1*, Hend Ghoneem 2 , Marwa Elgohary 3 , Nashwa Ebrahim 4
Authors affiliations:
Objectives: To assess the efficacy of peritonsillar infiltration of ketamine and dexmedetomidine on controlling post-tonsillectomy pain in children undergoing tonsillectomy.
Methodology: This randomized, controlled, double-blind study involved 60 patients aged 5-12 years, both genders, and scheduled for elective tonsillectomy. General anesthesia (GA) was induced in all of the patients. The patients were randomized and divided into three groups. All three groups received a peritonsillar normal saline injection (1 mL for each tonsil). Group I received a saline injection without any additive; Group II with 1 μg/kg of dexmedetomidine, and Group III with 0.5 mg/kg of ketamine. After the anesthesia was over, the time to the first rescue analgesia was noted. The total amount of pethidine consumed, the time to start oral intake, and the Children's Hospital of Eastern Ontario Pain Scale (CHEOPS) scores were noted at 30 min and 1, 2, and 4 hours postoperatively.
Results: The time of 1st rescue analgesia was significantly prolonged in group II than group I and III (P1 < 0.001, P3 < 0.001, respectively) and in Group III than Group I (P2 = 0.011). The total amount of pethidine consumed by Group II and the time for starting oral intake were significantly lower in Group II (P < 0.001) and were insignificantly different between Groups I and III. Compared to Groups I and III, Group II had significantly lower Children's Hospital of Eastern Ontario Pain Scale (CHEOPS) scores at 30 min and 1, 2, and 4 hours postoperatively (P < 0.05). In terms of postoperative complications, none of the three groups differed significantly (P > 0.05). There was no significant variation in the dosage of metoclopramide among the three groups.
Conclusion: Compared to placebo, dexmedetomidine and ketamine infiltrated into the tonsils reduced postoperative pain more effectively. Dexmedetomidine also improved pain scores according to the CHEOPS scale at different postoperative time points and reduced the need for additional analgesics more effectively than ketamine.
Abbreviations: CHEOPS: Children's Hospital of Eastern Ontario Pain Scale, GA: General anesthesia, NMDA: N-methyl-D-aspartate, PACU: post-anesthesia care unit
Keywords: Anesthesia; Anaesthesia, General; Dexmedetomidine; Preemptive Analgesia; Peritonsillar Infiltration; Ketamine; Postoperative Pain; Tonsillectomy
Citation: Eloraby M, Ghoneem H, ElgoharyM, Ebrahim N.Comparative effect of preemptive peritonsillar infiltration of ketamine versus dexmedetomidine on postoperative pain in children: a randomized controlled trial, Anaesth. pain intensive care 2025;29(6):497-504; DOI: 10.35975/apic.v29i6.2897
Received: February 04, 2025; Revised: July 09, 2025; Accepted: July 09, 2025
Tonsillectomy remains the procedure of choice for pediatric otolaryngologists.1 Most patients undergoing tonsillectomy experience some pain in the days immediately following the procedure.2 Delays in oral intake, longer hospital stays, higher expenses, and an increased risk of subsequent bleeding are among the several negative outcomes that can result from inadequate pain management following tonsillectomy.3
Traditional methods of pain relief have relied on opioid and non-steroidal anti-inflammatory drugs; however, these medications are not without their drawbacks, including nausea, vomiting, constipation, postoperative hemorrhage, sedation, and respiratory depression.4
The tonsillar fossae and oropharynx are very delicate structures. Trigeminal and glossopharyngeal nerve branches provide good local innervation, and the somatic cerebral cortex has a good representation of these areas.5 One promising approach to alleviating pain after tonsillectomy is the use of a local anesthetic that can be injected into the peritonsillar region. This medication would give pain relief with few side effects.6
Dexmedetomidine is a dual-action alpha 2-adrenergic receptor agonist as it inhibits nociceptive neurotransmission via the posterior horn of the spinal cord and has a dose-dependent inhibitory effect on C and alpha fibers, which in turn inhibits pain signals to the brain. It can also inhibit the release of norepinephrine, which in turn induces hyperpolarization, so it has a centrally mediated analgesic effect.7,8 In addition to modulating analgesia, it enhances acetylcholine release from spinal interneurons and may have a role in enhanced nitric oxide release and synthesis involved in the regulation of analgesia.9,10
Ketamine regulates central sensitization and opiate resistance by exerting antagonistic effects on N-methyl-D-aspartate (NMDA) receptors.11 Another potential benefit is that it can reduce pain by binding to mu receptors in the central nervous system.12
Research has shown that peritonsillar infiltration of dexmedetomidine and ketamine is an effective pain reliever following tonsillectomy, but no studies have compared the two drugs.13-15 Therefore, this study aimed to compare the efficacy and safety of peritonsillar infiltration of dexmedetomidine and ketamine for managing postoperative pain in children who underwent tonsillectomy surgery.
This randomized, double-blind controlled trial involved 60 patients aged 5-12 years, both sexes, American Society of Anesthesiologists physical status I or II, and scheduled for elective tonsillectomy. The study was done from September 2024 to January 2025 after approval from the Ethical Committee Tanta University Hospitals, Egypt, and registration at the Pan African Trial Registry (ID: PACTR202409660479092). The patient's parents provided their signed informed consent.
Exclusion criteria were children who had a history of peritonsillar abscess, problems perceiving or assessing pain, impairments in cardiac, renal, liver, neurological, psychiatric, or coagulation functions, or who were suspected of or demonstrated hypersensitivity to the medications used.
2.1. Randomization and blindness:
A random list was generated using an online randomization application (http://www.randomizer.org), and the codes for each patient were stored in an opaque sealed envelope. Using a 1:1:1 allocation ratio, each patient was randomly assigned to one of three groups. Group I received 1 mL of normal saline for each tonsil; Group II received 1 mL of normal saline with 1 μg/kg of dexmedetomidine, and Group III received 1 mL of normal saline with 0.5 mg/kg of ketamine for each nostril. A pharmacist who had no affiliation with the research prepared the interventional medications. Parents of children and outcome assessors were unaware of which group they were in.
All patients had to undergo a thorough history, physical examination, and standard laboratory tests, such as a complete blood count and coagulation assessment. The children were fasted for 2 hours from clear fluid, 4 hours from unclear fluid, and 6 hours from solid food before surgery.
A 22-G intravenous cannula was inserted to establish intravenous access upon arrival at the operating theatre, and the patient was premedicated with 0.15 mg/kg IV dexamethasone. The patient's vitals were tracked with the use of non-invasive devices that measured blood pressure, electrocardiogram, capnography, temperature, and pulse oximetry. Propofol 2-3 mg/kg and 1 μg/kg fentanyl were used to induce anesthesia. Intubation of the trachea is facilitated three min following the IV administration of 0.5 mg/kg atracurium.
Following this, the patient was placed on the mechanical ventilator after a cuffed endotracheal tube of the appropriate size had been inserted. Normocapnia (EtCO2 35-40 mmHg) was maintained by adjusting the settings. The anesthetic was maintained with a combination of 1.2% isoflurane and a 1:1 ratio of oxygen to air.
After intubation and the application of a mouth gag, an ENT surgeon performed pre-incisional submucosal infiltration of the study drugs in the peritonsillar area 3–5 min before the commencement of the surgery. One milliliter of the solution was inserted into each tonsil in a fan-shaped fashion from the top and bottom of the fossa as part of the process.
The same surgical team operated on all children using the same surgical technique (bipolar electrocautery). All children were given an IV injection of Ringer's solution at a rate of 5 mL/kg, along with 15 mg/kg paracetamol and 0.15 mg/kg of ondansetron, twenty min before the completion of the procedures.
Stopping the inhalational anesthesia was done at the end of the procedure. To reverse muscular relaxation, the children were given neostigmine at a dosage of 0.04 mg/kg and atropine at a dosage of 0.01 mg/kg. They were then transferred to the post-anesthesia care unit (PACU) for further observation.
The Children's Hospital of Eastern Ontario Pain Scale (CHEOPS) score was used to assess the pain at 30 min, 1, 2, 4, 6, and 12 hours after surgery.16 The patient was given IV pethidine at a dose of 0.5 mg/kg as a rescue analgesic if the pain score rose to five. The total amount of pethidine was calculated and recorded in the first postoperative 12 h. Hypotension (below 20% of baseline) and IV atropine 0.01-0.02 mg/kg were administered to patients with bradycardia ( heart rate < 60 beats/min).17 Using the Apfel score,18 we measured postoperative nausea and vomiting (PONV), where 0 indicates no nausea or vomiting, 1 indicates nausea without vomiting, 2 indicates one attack of vomiting, and 3 indicates more than one attack of vomiting. If the score was equal to or more than 1, an intravenous dose of metoclopramide (100-150 µg/kg, with a maximum of 10 mg) was given. Time for starting oral intake and other complications like hallucinations, negative behaviors, or secondary hemorrhage were assessed and recorded.
The primary outcome was time to the 1st rescue analgesia. The secondary outcome was CHEOPS score, postoperative dose of pethidine, time to start oral intake, hemodynamics, and postoperative complications.
Sample size calculation:
We estimated the sample size employing G*Power 3.1.9.2 (Universitat Kiel, Germany). Based on our pilot investigation, which comprised five cases in each group, we found that the mean (± SD) time to the 1st rescue analgesia was 1.40 ± 0.82 with placebo, 3± 1.22 with dexmedetomidine, and 2.20 ± 1.30 with ketamine. Each group comprised twenty patients based on a 95% confidence limit, 95% power, an allocation ratio of 1:1:1, and an effect size of 0.583 in the study, and three cases were supplemented to each group to cater for the drop-outs.
Statistical analysis:
SPSS v27 (IBM©, Chicago, IL, USA) was employed to analyze the data. The Shapiro-Wilks test and histograms were employed to verify the normality of the data distribution. To analyze quantitative parametric data, which were represented by mean ± SD, an ANOVA (F) test was employed in conjunction with a post hoc test (Tukey). We analyzed quantitative non-parametric data, which was represented as the median and IQR, using the Kruskal-Wallis’s test and the Mann-Whitney U test. We used the Chi-square test to analyze qualitative variables, which were given as percentages and reported as frequencies. A two-tailed P < 0.05 was deemed statistically significant.
Seventy-three patients were considered for inclusion in this randomized trial. Nine individuals were deemed ineligible for inclusion in the trial, while four patients chose not to participate. The remaining 60 patients were divided into three groups of 20 each, and all patients' data were successfully followed up on and analyzed (Figure 1).
Figure 1: CONSORT flowchart of the enrolled patients.
Patients' ages, sex, body weights, and ASA classes did not differ significantly (P > 0.05) across the three groups. Also, the three groups did not differ in the duration of surgery (P = 0.726) as shown in Table 1.
There was no discernible variation across the three groups concerning HR and MAP. Group II had a significantly lower HR after 30 min and one hour after the operation compared to Groups I and III (P < 0.05), while Group III had a significantly lower HR than Group I (P < 0.05). Compared to Groups I and III, Group II had a considerably lower HR in the two and four hours after the procedure (P < 0.05), but there was no discernible distinction between Groups I and III (Figure 2).
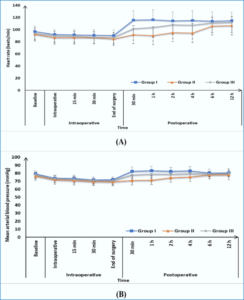
Figure 2: (A) Heart rate and (B) mean arterial blood pressure changes of the three studied groups.
CHEOPS scoring measurements at 30 min postoperative were significantly lower in Group II and Group III than in Group I (P < 0.05) and were insignificantly different between Group II and Group III (P < 0.05). CHEOPS scoring at 1h, 2h, and 4h postoperative was significantly lower in Group II than (Group I and Group III) (P < 0.05) and was insignificantly different between Group I and Group III. The CHEOPS score at 6 h and 12 h postoperative was insignificantly different among the three groups (Table 2).
The time to first rescue analgesic was seen to be significantly longer in Group II compared to Groups I and III (P < 0.05) and in Group III than in Group I (P < 0.05). In addition, Group II had a significantly lower postoperative dose of pethidine and a shorter time to start oral intake compared to Groups I and III (P < 0.05); however, Groups I and III were not significantly different. There was no discernible variation in the dosage of metoclopramide among the three groups (Table 3).
In terms of postoperative complications, none of the three groups differed significantly (P > 0.05). Only one case of hallucination and negative behavior was recorded in Group III, and none of the three groups reported postoperative hoarseness of voice, local anesthetic toxicity, or delayed hospital discharge (Table 4).
It is crucial to ensure that tonsillectomy patients receive reliable and efficient analgesia after the procedure. Failure to comply with this requirement may result in complications, including delayed oral intake, extended hospital admissions, an elevated risk of secondary hemorrhage, and a delayed return to routine activities.19 When it comes to alleviating pain after tonsillectomy, a local anesthetic that can be injected into the peritonsillar region and has few side effects would be ideal.5
Our result demonstrated that dexmedetomidine greatly reduced the amount of pethidine needed, the time to first oral intake, the CHEOPS score at 30 min, 1, 2, and 4 hours postoperatively, and the time it took to request pethidine as a rescue analgesic. Also, ketamine was more effective than the placebo group but had a short duration of analgesia of 120 min. The incidence of postoperative complications (PONV, hallucination/ negative behaviors, and secondary hemorrhage) was insignificantly different among the three groups; only one case in the ketamine group had a hallucination or negative behaviors.
Lahane et al. and Abdel-Ghaffar HS et al. revealed that peritonsillar dexmedetomidine had several advantages over intravenous dexmedetomidine.9,20 Reductions in postoperative analgesic demands, recovery time, sedation, bradycardia, total oral intake on day one following surgery, time to first request, and family satisfaction were all positive outcomes.
Hatami et al. demonstrated that peritonsillar dexmedetomidine significantly reduced pain scores compared to placebo.21 Additionally, dexmedetomidine lengthened the time it took to request analgesics. Moreover, dexmedetomidine had considerably reduced hemodynamic parameters.
Modir et al. compared dexmedetomidine‑ropivacaine and placebo‑ropivacaine only and concluded that on the CHEOPS scale, the dexmedetomidine group had the fewest cases of pain and the fewest individuals requiring acetaminophen.15 Moreover, Singh et al. proved that ropivacaine with dexmedetomidine added provided longer analgesia, reduced the need for further analgesics, and increased patient satisfaction.22 Weheba et al. showed that dexmedetomidine decreases pain following tonsillectomy.5
Khademi et al. revealed that ketamine, when administered intravenously or injected into the tonsils before surgery, significantly lessens the likelihood and intensity of pain experienced by patients after adenotonsillectomy.23 Peritonsillar ketamine, on the other hand, significantly reduced postoperative pain, analgesic need, and antiemetics.
Concordant with our findings, Ayatollahi et al. demonstrated the pain control after surgery was comparable in the ketamine and placebo groups.24 This could be due to the fact that ketamine does not effectively control postoperative pain or that its effects wear off quickly. Furthermore, the ketamine group had a greater incidence of hallucinations and emerging reactions (11.9%), which may have impacted the assessment of pain in the recovery room. Opioid painkillers may amplify the placebo group's perception of pain by hiding the antagonistic impact of NMDA receptors, which is another probable reason.
Tong et al. (2014) conducted a meta-analysis that studied the effects of ketamine and a placebo on PONV and pain.25 The ketamine group showed a substantial decrease in PONV compared to the placebo group at 30 min, but there was no change at 120 min (likely due to the short-acting duration). This suggests that fewer people in the ketamine group experienced PONV, while those given the placebo did not.
Also, Pirzadeh et al. demonstrated that the ketamine group reported less pain on average compared to the control group using CHEOPS. There was no discernible difference in the number of analgesics required between the ketamine group and the control group. Only 1.333% of the ketamine group experienced delirium in the recovery room.26
In their meta-analysis, Dahmani et al. examined the effects of ketamine in the PACU during the initial two hours, as well as early pain scores and the necessity of analgesics. From 6 to 24 hours after administration, ketamine decreased pain scores without influencing the requirement for immediate analgesics.27
Khademi et al. demonstrated that compared to IV ketamine, peritonsillar infiltration of ketamine resulted in a greater reduction in pain, analgesic and antiemetic needs, and VAS scores.23 Contrary to our findings, Honarmand et al. (2008) demonstrated that children undergoing adenotonsillectomy experience good pain relief for 24 hours following surgery after a ketamine injection into the peritonsillar space about 3 min before the procedure.28 This treatment reduces postoperative pain and delays the initial request for analgesics, resulting in a substantial pethidine-sparing effect during the first 24 hours in comparison to peritonsillar saline infusion. Also, Marzban and Hadadi revealed that ketamine's benefits lingered for a few days following the surgery.29 Since ketamine inhibits the transmission of cerebral sensitivity to peripheral pain and/or secondary hyperalgesia, it has long-lasting effects.
Reducing postoperative pain is a major goal of early and frequent oral intake following tonsillectomy. The dexmedetomidine group had an earlier time for first oral intake (3.05 ± 0.79) hr, while the ketamine group (4.5 ± 1.1) hr and the placebo group (4.95 ± 1.5) hr.
In consistency with our study, Meena et al. investigated the impact of various agents infiltrating the peritonsillar space before an incision was made on pain levels and when patients could take oral medications after tonsillectomy.30 They found that 80% of patients in the group given bupivacaine and ketamine began oral intake four hours after surgery. In comparison, 50% of patients in the ketamine group started at the same time.
We observed no sedation and reduced nausea due to the slower rate at which the amount and dose of ketamine and dexmedetomidine entered the systemic circulation in our trial. Consequently, the adverse effects of the ketamine and dexmedetomidine groups were not substantially distinct. Understanding how these analgesics work when injected into the peritonsillar region is challenging because of the lack of clarity surrounding their mechanism of action or systemic absorption. Although we did not measure plasma levels, our study did find that one participant in the ketamine group experienced hallucinations and negative behavior.
This study has a few limitations, such as a limited sample size, and it was conducted at only one center. We suggest conducting more prospective, multicenter studies with a larger sample size to compare different doses and different combinations with other drugs.
Placing dexmedetomidine or ketamine peritonsillar reduced postoperative pain better than a placebo. Dexmedetomidine also improved pain scores according to the CHEOPS scale at different postoperative time points and reduced the need for supplemental analgesics more effectively than ketamine.
Acknowledgment: There is none to be declared.
7. Financial support and sponsorship:
Nil
8. Conflict of interests:
None to be declared.
Authors affiliations:
- Marwa Eloraby, Lecturer, Anesthesiology, Department of Surgical Intensive Care & Pain Medicine, Faculty of Medicine, Tanta University, Tanta, Egypt; Email: dr.marwaoraby5@gmail.com; {ORCID:0000-0003-4032-9639}
- Hend Ghoneem, Lecturer, Anesthesiology, Department of Surgical Intensive Care & Pain Medicine, Faculty of Medicine, Tanta University, Tanta, Egypt; Email: hannod20389@gmail.com; {ORCID:0000-0001-7177-8184}
- Marwa Elgohary, Lecturer, Anesthesiology, Department of Surgical Intensive Care & Pain Medicine, Faculty of Medicine, Tanta University, Tanta, Egypt; Email: Marwaelgohary95@gmail.com
- Nashwa Ebrahim, Lecturer, Anesthesiology, Department of Surgical Intensive Care & Pain Medicine, Faculty of Medicine, Tanta University, Tanta, Egypt; Email: nashwa741987@gmail.com
ABSTRACT
Objectives: To assess the efficacy of peritonsillar infiltration of ketamine and dexmedetomidine on controlling post-tonsillectomy pain in children undergoing tonsillectomy.
Methodology: This randomized, controlled, double-blind study involved 60 patients aged 5-12 years, both genders, and scheduled for elective tonsillectomy. General anesthesia (GA) was induced in all of the patients. The patients were randomized and divided into three groups. All three groups received a peritonsillar normal saline injection (1 mL for each tonsil). Group I received a saline injection without any additive; Group II with 1 μg/kg of dexmedetomidine, and Group III with 0.5 mg/kg of ketamine. After the anesthesia was over, the time to the first rescue analgesia was noted. The total amount of pethidine consumed, the time to start oral intake, and the Children's Hospital of Eastern Ontario Pain Scale (CHEOPS) scores were noted at 30 min and 1, 2, and 4 hours postoperatively.
Results: The time of 1st rescue analgesia was significantly prolonged in group II than group I and III (P1 < 0.001, P3 < 0.001, respectively) and in Group III than Group I (P2 = 0.011). The total amount of pethidine consumed by Group II and the time for starting oral intake were significantly lower in Group II (P < 0.001) and were insignificantly different between Groups I and III. Compared to Groups I and III, Group II had significantly lower Children's Hospital of Eastern Ontario Pain Scale (CHEOPS) scores at 30 min and 1, 2, and 4 hours postoperatively (P < 0.05). In terms of postoperative complications, none of the three groups differed significantly (P > 0.05). There was no significant variation in the dosage of metoclopramide among the three groups.
Conclusion: Compared to placebo, dexmedetomidine and ketamine infiltrated into the tonsils reduced postoperative pain more effectively. Dexmedetomidine also improved pain scores according to the CHEOPS scale at different postoperative time points and reduced the need for additional analgesics more effectively than ketamine.
Abbreviations: CHEOPS: Children's Hospital of Eastern Ontario Pain Scale, GA: General anesthesia, NMDA: N-methyl-D-aspartate, PACU: post-anesthesia care unit
Keywords: Anesthesia; Anaesthesia, General; Dexmedetomidine; Preemptive Analgesia; Peritonsillar Infiltration; Ketamine; Postoperative Pain; Tonsillectomy
Citation: Eloraby M, Ghoneem H, ElgoharyM, Ebrahim N.Comparative effect of preemptive peritonsillar infiltration of ketamine versus dexmedetomidine on postoperative pain in children: a randomized controlled trial, Anaesth. pain intensive care 2025;29(6):497-504; DOI: 10.35975/apic.v29i6.2897
Received: February 04, 2025; Revised: July 09, 2025; Accepted: July 09, 2025
1. INTRODUCTION
Tonsillectomy remains the procedure of choice for pediatric otolaryngologists.1 Most patients undergoing tonsillectomy experience some pain in the days immediately following the procedure.2 Delays in oral intake, longer hospital stays, higher expenses, and an increased risk of subsequent bleeding are among the several negative outcomes that can result from inadequate pain management following tonsillectomy.3
Traditional methods of pain relief have relied on opioid and non-steroidal anti-inflammatory drugs; however, these medications are not without their drawbacks, including nausea, vomiting, constipation, postoperative hemorrhage, sedation, and respiratory depression.4
The tonsillar fossae and oropharynx are very delicate structures. Trigeminal and glossopharyngeal nerve branches provide good local innervation, and the somatic cerebral cortex has a good representation of these areas.5 One promising approach to alleviating pain after tonsillectomy is the use of a local anesthetic that can be injected into the peritonsillar region. This medication would give pain relief with few side effects.6
Dexmedetomidine is a dual-action alpha 2-adrenergic receptor agonist as it inhibits nociceptive neurotransmission via the posterior horn of the spinal cord and has a dose-dependent inhibitory effect on C and alpha fibers, which in turn inhibits pain signals to the brain. It can also inhibit the release of norepinephrine, which in turn induces hyperpolarization, so it has a centrally mediated analgesic effect.7,8 In addition to modulating analgesia, it enhances acetylcholine release from spinal interneurons and may have a role in enhanced nitric oxide release and synthesis involved in the regulation of analgesia.9,10
Ketamine regulates central sensitization and opiate resistance by exerting antagonistic effects on N-methyl-D-aspartate (NMDA) receptors.11 Another potential benefit is that it can reduce pain by binding to mu receptors in the central nervous system.12
Research has shown that peritonsillar infiltration of dexmedetomidine and ketamine is an effective pain reliever following tonsillectomy, but no studies have compared the two drugs.13-15 Therefore, this study aimed to compare the efficacy and safety of peritonsillar infiltration of dexmedetomidine and ketamine for managing postoperative pain in children who underwent tonsillectomy surgery.
2. METHODOLOGY
This randomized, double-blind controlled trial involved 60 patients aged 5-12 years, both sexes, American Society of Anesthesiologists physical status I or II, and scheduled for elective tonsillectomy. The study was done from September 2024 to January 2025 after approval from the Ethical Committee Tanta University Hospitals, Egypt, and registration at the Pan African Trial Registry (ID: PACTR202409660479092). The patient's parents provided their signed informed consent.
Exclusion criteria were children who had a history of peritonsillar abscess, problems perceiving or assessing pain, impairments in cardiac, renal, liver, neurological, psychiatric, or coagulation functions, or who were suspected of or demonstrated hypersensitivity to the medications used.
2.1. Randomization and blindness:
A random list was generated using an online randomization application (http://www.randomizer.org), and the codes for each patient were stored in an opaque sealed envelope. Using a 1:1:1 allocation ratio, each patient was randomly assigned to one of three groups. Group I received 1 mL of normal saline for each tonsil; Group II received 1 mL of normal saline with 1 μg/kg of dexmedetomidine, and Group III received 1 mL of normal saline with 0.5 mg/kg of ketamine for each nostril. A pharmacist who had no affiliation with the research prepared the interventional medications. Parents of children and outcome assessors were unaware of which group they were in.
All patients had to undergo a thorough history, physical examination, and standard laboratory tests, such as a complete blood count and coagulation assessment. The children were fasted for 2 hours from clear fluid, 4 hours from unclear fluid, and 6 hours from solid food before surgery.
A 22-G intravenous cannula was inserted to establish intravenous access upon arrival at the operating theatre, and the patient was premedicated with 0.15 mg/kg IV dexamethasone. The patient's vitals were tracked with the use of non-invasive devices that measured blood pressure, electrocardiogram, capnography, temperature, and pulse oximetry. Propofol 2-3 mg/kg and 1 μg/kg fentanyl were used to induce anesthesia. Intubation of the trachea is facilitated three min following the IV administration of 0.5 mg/kg atracurium.
Following this, the patient was placed on the mechanical ventilator after a cuffed endotracheal tube of the appropriate size had been inserted. Normocapnia (EtCO2 35-40 mmHg) was maintained by adjusting the settings. The anesthetic was maintained with a combination of 1.2% isoflurane and a 1:1 ratio of oxygen to air.
After intubation and the application of a mouth gag, an ENT surgeon performed pre-incisional submucosal infiltration of the study drugs in the peritonsillar area 3–5 min before the commencement of the surgery. One milliliter of the solution was inserted into each tonsil in a fan-shaped fashion from the top and bottom of the fossa as part of the process.
The same surgical team operated on all children using the same surgical technique (bipolar electrocautery). All children were given an IV injection of Ringer's solution at a rate of 5 mL/kg, along with 15 mg/kg paracetamol and 0.15 mg/kg of ondansetron, twenty min before the completion of the procedures.
Stopping the inhalational anesthesia was done at the end of the procedure. To reverse muscular relaxation, the children were given neostigmine at a dosage of 0.04 mg/kg and atropine at a dosage of 0.01 mg/kg. They were then transferred to the post-anesthesia care unit (PACU) for further observation.
The Children's Hospital of Eastern Ontario Pain Scale (CHEOPS) score was used to assess the pain at 30 min, 1, 2, 4, 6, and 12 hours after surgery.16 The patient was given IV pethidine at a dose of 0.5 mg/kg as a rescue analgesic if the pain score rose to five. The total amount of pethidine was calculated and recorded in the first postoperative 12 h. Hypotension (below 20% of baseline) and IV atropine 0.01-0.02 mg/kg were administered to patients with bradycardia ( heart rate < 60 beats/min).17 Using the Apfel score,18 we measured postoperative nausea and vomiting (PONV), where 0 indicates no nausea or vomiting, 1 indicates nausea without vomiting, 2 indicates one attack of vomiting, and 3 indicates more than one attack of vomiting. If the score was equal to or more than 1, an intravenous dose of metoclopramide (100-150 µg/kg, with a maximum of 10 mg) was given. Time for starting oral intake and other complications like hallucinations, negative behaviors, or secondary hemorrhage were assessed and recorded.
The primary outcome was time to the 1st rescue analgesia. The secondary outcome was CHEOPS score, postoperative dose of pethidine, time to start oral intake, hemodynamics, and postoperative complications.
Sample size calculation:
We estimated the sample size employing G*Power 3.1.9.2 (Universitat Kiel, Germany). Based on our pilot investigation, which comprised five cases in each group, we found that the mean (± SD) time to the 1st rescue analgesia was 1.40 ± 0.82 with placebo, 3± 1.22 with dexmedetomidine, and 2.20 ± 1.30 with ketamine. Each group comprised twenty patients based on a 95% confidence limit, 95% power, an allocation ratio of 1:1:1, and an effect size of 0.583 in the study, and three cases were supplemented to each group to cater for the drop-outs.
Statistical analysis:
SPSS v27 (IBM©, Chicago, IL, USA) was employed to analyze the data. The Shapiro-Wilks test and histograms were employed to verify the normality of the data distribution. To analyze quantitative parametric data, which were represented by mean ± SD, an ANOVA (F) test was employed in conjunction with a post hoc test (Tukey). We analyzed quantitative non-parametric data, which was represented as the median and IQR, using the Kruskal-Wallis’s test and the Mann-Whitney U test. We used the Chi-square test to analyze qualitative variables, which were given as percentages and reported as frequencies. A two-tailed P < 0.05 was deemed statistically significant.
3. RESULTS
Seventy-three patients were considered for inclusion in this randomized trial. Nine individuals were deemed ineligible for inclusion in the trial, while four patients chose not to participate. The remaining 60 patients were divided into three groups of 20 each, and all patients' data were successfully followed up on and analyzed (Figure 1).

Figure 1: CONSORT flowchart of the enrolled patients.
Patients' ages, sex, body weights, and ASA classes did not differ significantly (P > 0.05) across the three groups. Also, the three groups did not differ in the duration of surgery (P = 0.726) as shown in Table 1.
| Table 1: Demographic data and duration of surgery of the studied groups | |||||
| Variables | Group I (n = 20) |
Group II (n = 20) |
Group III (n = 20) |
P value | |
| Age (years) | 8.9 ± 2.02 | 7.6 ± 2.06 | 8.4 ± 2.37 | 0.145 | |
| Sex | Male | 9 (45%) | 11 (55%) | 8 (40%) | 0.626 |
| Female | 11 (55%) | 9 (45%) | 12 (60%) | ||
| Weight (kg) | 37.8 ± 9.01 | 32.8 ± 8.15 | 34.6 ± 9.48 | 0.201 | |
| ASA
physical status |
I | 16 (80%) | 14 (70%) | 15 (75%) | 0.766 |
| II | 4 (20%) | 6 (30%) | 5 (25%) | ||
| Duration of surgery (min) | 46.8 ± 11.73 | 44.5 ± 9.02 | 44.8 ± 8.03 | 0.726 | |
| Data are presented as mean ± SD or frequency (%). ASA: American society of anesthesiologists. | |||||
There was no discernible variation across the three groups concerning HR and MAP. Group II had a significantly lower HR after 30 min and one hour after the operation compared to Groups I and III (P < 0.05), while Group III had a significantly lower HR than Group I (P < 0.05). Compared to Groups I and III, Group II had a considerably lower HR in the two and four hours after the procedure (P < 0.05), but there was no discernible distinction between Groups I and III (Figure 2).

Figure 2: (A) Heart rate and (B) mean arterial blood pressure changes of the three studied groups.
CHEOPS scoring measurements at 30 min postoperative were significantly lower in Group II and Group III than in Group I (P < 0.05) and were insignificantly different between Group II and Group III (P < 0.05). CHEOPS scoring at 1h, 2h, and 4h postoperative was significantly lower in Group II than (Group I and Group III) (P < 0.05) and was insignificantly different between Group I and Group III. The CHEOPS score at 6 h and 12 h postoperative was insignificantly different among the three groups (Table 2).
| Table 2: CHOPES pain scale of the studied groups | |||||
| Time | Group I (n = 20) |
Group II (n = 20) |
Group III (n = 20) |
P value | Post hoc |
| 30 min | 4(4 - 4.25) | 4(4 - 4) | 4(4 - 4) | 0.005* | P1 = 0.014*
P2 = 0.014* P3 = 0.999 |
| 1 h | 4(4 - 11) | 4(4 - 4) | 4(4 - 5) | 0.004* | P1 = 0.005*
P2 = 0.999 P3 = 0.046* |
| 2 h | 7.5(4 - 9.25) | 4(4 - 4) | 4.5(4 - 8) | < 0.001* | P1 < 0.001*
P2 = 0.999 P3 = 0.005* |
| 4 h | 9.5(4 - 12) | 4(4 - 4) | 9(4 - 10.25) | 0.007* | P1 = 0.013*
P2 = 0.999 P3 = 0.029* |
| 6 h | 6.5(4 - 10) | 4(4 - 5.25) | 5(4 - 8) | 0.235 | |
| 12 h | 7.5(4 - 10) | 5(4 - 6.25) | 6(5 - 9) | 0.205 | |
| Data is presented as median (IQR). *: Significant as p value < 0.05. (P1): P value between group I and group II, (P2): P value between group I and group III, and (P3): P value between group II and group III. | |||||
The time to first rescue analgesic was seen to be significantly longer in Group II compared to Groups I and III (P < 0.05) and in Group III than in Group I (P < 0.05). In addition, Group II had a significantly lower postoperative dose of pethidine and a shorter time to start oral intake compared to Groups I and III (P < 0.05); however, Groups I and III were not significantly different. There was no discernible variation in the dosage of metoclopramide among the three groups (Table 3).
| Table 3: Time of 1st rescue analgesia, pethidine dose, time for starting oral intake, and metoclopramide dose of the studied groups | |||||
| Analgesic variables | Group I
(n = 20) |
Group II
(n = 20) |
Group III
(n = 20) |
P value | Post hoc |
| Time of 1st rescue analgesia (hr) | 1.2 ± 0.62 | 5.1 ± 0.97 | 2.1 ± 1.12 | < 0.001* | P1 < 0.001*
P2 = 0.011* P3 < 0.001* |
| Pethidine dose (mg) | 57.3 ± 16.51 | 21.4 ± 9.39 | 49.3 ± 19.64 | < 0.001* | P1 < 0.001*
P2 = 0.173 P3 < 0.001* |
| Time for starting oral intake (h) | 4.95 ± 1.5 | 3.05 ± 0.79 | 4.5 ± 1.1 | < 0.001* | P1 < 0.001*
P2 = 0.443 P3 < 0.001* |
| Metoclopramide dose (mg) | 3.3 ± 0.61 | 3.7 ± 0.65 | 3.6 ± 1.05 | 0.763 | |
| Data are presented as mean (SD) *: Significant as P < 0.05; (P1): P value between group I and group II, (P2): P value between group I and group III, and (P3): P value between group II and group III. | |||||
In terms of postoperative complications, none of the three groups differed significantly (P > 0.05). Only one case of hallucination and negative behavior was recorded in Group III, and none of the three groups reported postoperative hoarseness of voice, local anesthetic toxicity, or delayed hospital discharge (Table 4).
| Table 4: Complications of the studied groups | ||||
| Complications | Group I (n = 20) |
Group II (n = 20) |
Group III (n = 20) |
P value |
| PONV | 3 (15%) | 5 (25%) | 4 (20%) | 0.732 |
| Hallucination/ Negative behaviors | 0 (0%) | 0 (0%) | 1 (5%) | 0.362 |
| Secondary hemorrhage | 0 (0%) | 0 (0%) | 0 (0%) | --- |
| Data is presented as frequency (%). PONV: Postoperative nausea and vomiting. | ||||
4. DISCUSSION
It is crucial to ensure that tonsillectomy patients receive reliable and efficient analgesia after the procedure. Failure to comply with this requirement may result in complications, including delayed oral intake, extended hospital admissions, an elevated risk of secondary hemorrhage, and a delayed return to routine activities.19 When it comes to alleviating pain after tonsillectomy, a local anesthetic that can be injected into the peritonsillar region and has few side effects would be ideal.5
Our result demonstrated that dexmedetomidine greatly reduced the amount of pethidine needed, the time to first oral intake, the CHEOPS score at 30 min, 1, 2, and 4 hours postoperatively, and the time it took to request pethidine as a rescue analgesic. Also, ketamine was more effective than the placebo group but had a short duration of analgesia of 120 min. The incidence of postoperative complications (PONV, hallucination/ negative behaviors, and secondary hemorrhage) was insignificantly different among the three groups; only one case in the ketamine group had a hallucination or negative behaviors.
Lahane et al. and Abdel-Ghaffar HS et al. revealed that peritonsillar dexmedetomidine had several advantages over intravenous dexmedetomidine.9,20 Reductions in postoperative analgesic demands, recovery time, sedation, bradycardia, total oral intake on day one following surgery, time to first request, and family satisfaction were all positive outcomes.
Hatami et al. demonstrated that peritonsillar dexmedetomidine significantly reduced pain scores compared to placebo.21 Additionally, dexmedetomidine lengthened the time it took to request analgesics. Moreover, dexmedetomidine had considerably reduced hemodynamic parameters.
Modir et al. compared dexmedetomidine‑ropivacaine and placebo‑ropivacaine only and concluded that on the CHEOPS scale, the dexmedetomidine group had the fewest cases of pain and the fewest individuals requiring acetaminophen.15 Moreover, Singh et al. proved that ropivacaine with dexmedetomidine added provided longer analgesia, reduced the need for further analgesics, and increased patient satisfaction.22 Weheba et al. showed that dexmedetomidine decreases pain following tonsillectomy.5
Khademi et al. revealed that ketamine, when administered intravenously or injected into the tonsils before surgery, significantly lessens the likelihood and intensity of pain experienced by patients after adenotonsillectomy.23 Peritonsillar ketamine, on the other hand, significantly reduced postoperative pain, analgesic need, and antiemetics.
Concordant with our findings, Ayatollahi et al. demonstrated the pain control after surgery was comparable in the ketamine and placebo groups.24 This could be due to the fact that ketamine does not effectively control postoperative pain or that its effects wear off quickly. Furthermore, the ketamine group had a greater incidence of hallucinations and emerging reactions (11.9%), which may have impacted the assessment of pain in the recovery room. Opioid painkillers may amplify the placebo group's perception of pain by hiding the antagonistic impact of NMDA receptors, which is another probable reason.
Tong et al. (2014) conducted a meta-analysis that studied the effects of ketamine and a placebo on PONV and pain.25 The ketamine group showed a substantial decrease in PONV compared to the placebo group at 30 min, but there was no change at 120 min (likely due to the short-acting duration). This suggests that fewer people in the ketamine group experienced PONV, while those given the placebo did not.
Also, Pirzadeh et al. demonstrated that the ketamine group reported less pain on average compared to the control group using CHEOPS. There was no discernible difference in the number of analgesics required between the ketamine group and the control group. Only 1.333% of the ketamine group experienced delirium in the recovery room.26
In their meta-analysis, Dahmani et al. examined the effects of ketamine in the PACU during the initial two hours, as well as early pain scores and the necessity of analgesics. From 6 to 24 hours after administration, ketamine decreased pain scores without influencing the requirement for immediate analgesics.27
Khademi et al. demonstrated that compared to IV ketamine, peritonsillar infiltration of ketamine resulted in a greater reduction in pain, analgesic and antiemetic needs, and VAS scores.23 Contrary to our findings, Honarmand et al. (2008) demonstrated that children undergoing adenotonsillectomy experience good pain relief for 24 hours following surgery after a ketamine injection into the peritonsillar space about 3 min before the procedure.28 This treatment reduces postoperative pain and delays the initial request for analgesics, resulting in a substantial pethidine-sparing effect during the first 24 hours in comparison to peritonsillar saline infusion. Also, Marzban and Hadadi revealed that ketamine's benefits lingered for a few days following the surgery.29 Since ketamine inhibits the transmission of cerebral sensitivity to peripheral pain and/or secondary hyperalgesia, it has long-lasting effects.
Reducing postoperative pain is a major goal of early and frequent oral intake following tonsillectomy. The dexmedetomidine group had an earlier time for first oral intake (3.05 ± 0.79) hr, while the ketamine group (4.5 ± 1.1) hr and the placebo group (4.95 ± 1.5) hr.
In consistency with our study, Meena et al. investigated the impact of various agents infiltrating the peritonsillar space before an incision was made on pain levels and when patients could take oral medications after tonsillectomy.30 They found that 80% of patients in the group given bupivacaine and ketamine began oral intake four hours after surgery. In comparison, 50% of patients in the ketamine group started at the same time.
We observed no sedation and reduced nausea due to the slower rate at which the amount and dose of ketamine and dexmedetomidine entered the systemic circulation in our trial. Consequently, the adverse effects of the ketamine and dexmedetomidine groups were not substantially distinct. Understanding how these analgesics work when injected into the peritonsillar region is challenging because of the lack of clarity surrounding their mechanism of action or systemic absorption. Although we did not measure plasma levels, our study did find that one participant in the ketamine group experienced hallucinations and negative behavior.
5. LIMITATIONS
This study has a few limitations, such as a limited sample size, and it was conducted at only one center. We suggest conducting more prospective, multicenter studies with a larger sample size to compare different doses and different combinations with other drugs.
6. CONCLUSION
Placing dexmedetomidine or ketamine peritonsillar reduced postoperative pain better than a placebo. Dexmedetomidine also improved pain scores according to the CHEOPS scale at different postoperative time points and reduced the need for supplemental analgesics more effectively than ketamine.
Acknowledgment: There is none to be declared.
7. Financial support and sponsorship:
Nil
8. Conflict of interests:
None to be declared.
9. REFERENCES
- Mitchell RB, Archer SM, Ishman SL, Rosenfeld RM, Coles S, Finestone SA, et al. Clinical practice guideline: Tonsillectomy in children (update)-executive summary. Otolaryngol Head Neck Surg. 2019;160:187-205. [PubMed] DOI: 1177/0194599818807917
- Qian L, Li X, Chen H, Chi X. Recent advances in post-tonsillectomy analgesia. Dig Med Res. 2020;3:951-87. DOI: 10.21037/dmr-20-38
- Alkhars HF, Zakaria OM, Alkhars H. Parents' perception on post-tonsillectomy hemorrhage: A local survey. Cureus. 2024;16:629-707. [PubMed] DOI: 7759/cureus.62907
- Alorfi NM. Pharmacological methods of pain management: Narrative review of medication used. Int J Gen Med. 2023;16:3247-56. [PubMed] DOI: 2147/IJGM.S419239
- El Madany MM, Eloraby MA. Comparison of post-operative analgesic effects of peritonsillar infiltration of dexmedetomidine, lidocaine or both in children following tonsillectomy. Egypt J Hosp Med. 2021;85:4308-13. [PubMed] DOI: 21608/ejhm.2021.209421
- Swain SK. Current treatment of post-tonsillectomy pain: a review. Int J Otorhinolaryngol Head Neck Surg. 2021;10:170-98. DOI: 18203/issn.2454-5929.ijohns20213911
- Zhao Y, He J, Yu N, Jia C, Wang S. Mechanisms of dexmedetomidine in neuropathic pain. Front Neurosci. 2020;14:330-98. [PubMed] DOI: 3389/fnins.2020.00330
- Liu Y, Ma H, Wang X, Yang I, Wang J. Immunomodulatory potential of dexmedetomidine in perioperative pain management for patients with cancer. Cancer Pathog Ther. 2025;78:654-987. [PubMed] DOI: 1016/j.cpt.2024.12.007
- Lahane PV, Nandanvankar NK, Pradeep MS, Memon NY, Yennawar SD. A comparative study of intraoperative peritonsillar infiltration versus intravenous dexemedetomidine for perioperative analgesia in tonsillectomy. Asian J Med Sci. 2020;11:89-95. Full Text DOI: 3126/ajms.v11i6.30639
- Edinoff AN, Houk GM, Patil S, Siddaiah HB, Kaye AJ, Iyengar PS, et al. Adjuvant drugs for peripheral nerve blocks: the role of alpha-2 agonists, dexamethasone, midazolam, and non-steroidal anti-inflammatory drugs. Anesth Pain Med. 2021;11:987-96. [PubMed] DOI: 5812/aapm.117197
- Subramanian S, Haroutounian S, Palanca BJA, Lenze EJ. Ketamine as a therapeutic agent for depression and pain: mechanisms and evidence. J Neurol Sci. 2022;434:120-52. [PubMed] DOI: 1016/j.jns.2022.120152
- Ma X, Yan J, Jiang H. Application of ketamine in pain management and the underlying mechanism. Pain Res Manag. 2023;2023:192-896. [PubMed] DOI: 1155/2023/1928969
- Wang L, Guo Y, Tian J. The comparison of ketamine with tramadol for postoperative pain relief on children following adenotonsillectomy or tonsillectomy: a meta-analysis of randomized controlled trials. Medicine. 2021;100:225-341. [PubMed] DOI: 1097/MD.0000000000022541
- Nuhu SI, Atteh DF, Embu HY, Isamade ES, Kokong DD, Ma'an ND, et al. Evaluation of the effects of peritonsillar infiltration of tramadol-ketamine combination versus tramadol alone on posttonsillectomy pain in children. J West Afr Coll Surg. 2023;13:84-90. [PubMed] DOI: 4103/jwas.jwas_31_23
- Modir H, Moshiri E, Naghavi F. Efficacy of peritonsillar infiltration with dexmedetomidine versus tramadol in comparison to placebo for pain control and sedation after tonsillectomy in pediatric patients: A randomized clinical trial. Natl J Maxillofac Surg. 2024;15:40-6. [PubMed] DOI: 4103/njms.njms_507_21
- Gerstman MD, Rolland LR, Tramèr MR, Habre W, Elia N. Researchers’ choice of pain scales in trials of children undergoing surgery: A cross‐sectional analysis of systematically searched randomized controlled trials and survey of authors. Paediatr Anaesth. 2021;31:1194-207. [PubMed] DOI: 1111/pan.14264
- Mason KP, Lönnqvist PA. Bradycardia in perspective-not all reductions in heart rate need immediate intervention. Paediatr Anaesth. 2015;25:44-51. [PubMed] DOI: 1111/pan.12584
- Jai D, Handscombe M, Brooke M, Karena S, Arune S, Leslie K. Interpretation of the four risk factors for postoperative nausea and vomiting in the Apfel simplified risk score: an analysis of published studies. Can J Anaesth. 2021;68:1057-63. [PubMed] DOI: 1007/s12630-021-01974-8
- Jensen DR. Pharmacologic management of post-tonsillectomy pain in children. World J Otorhinolaryngol Head Neck Surg. 2021;7:186-93. [PubMed] DOI: 1016/j.wjorl.2021.03.004
- Abdel-Ghaffar HS, Abdel-Haleem AK. Efficacy and safety of intraoperative dexmedetomidine in pediatric posttonsillectomy pain: Peritonsillar versus intravenous administration. Egypt J Anaesth. 2011;27:219-25. DOI; 1016/j.egja.2011.08.001
- Hatami M, Jalali M, Ayatollahi V, Baradaranfar M, Vaziribozorg S. Comparison of the effect of peritonsillar infiltration of tramadol vs dexmedetomidine on post-tonsillectomy pain. Eur Arch Otorhinolaryngol. 2022;279:2665-9. [PubMed] DOI: 1007/s00405-021-07102-y
- Singh R, Trivedi LH, Parmar RM. Comparison of the efficacy of ropivacaine and ropivacaine with dexmedetomidine in post-tonsilectomy pain relief after peritonsillar infiltration: a prospective randomized double blind study. Indian J Clin Anaesth. 2019;6:263-9. DOI: 18231/j.ijca.2019.049
- Khademi S, Ghaffarpasand F, Heiran HR, Yavari MJ, Motazedian S, Dehghankhalili M. Intravenous and peritonsillar infiltration of ketamine for postoperative pain after adenotonsillectomy: a randomized placebo-controlled clinical trial. Med Princ Pract. 2011;20:433-7. [PubMed] DOI: 1159/000327657
- Ayatollahi V, Behdad S, Hatami M, Moshtaghiun H, Baghianimoghadam B. Comparison of peritonsillar infiltration effects of ketamine and tramadol on post tonsillectomy pain: a double-blinded randomized placebo-controlled clinical trial. Croat Med J. 2012;53:155-61. [PubMed] DOI: 3325/cmj.2012.53.155
- Tong Y, Ding XB, Wang X, Ren H, Chen ZX, Li Q. Ketamine peritonsillar infiltration during tonsillectomy in pediatric patients: An updated meta-analysis. Int J Pediatr Otorhinolaryngol. 2014;78:1735-41. [PubMed] DOI: 1016/j.ijporl.2014.07.036
- Pirzadeh A, Mohammadi MA, Allaf-Akbari S, Entezariasl M. The effect of ketamine on posttonsillectomy pain in children: a clinical trial. Iran J Otorhinolaryngol. 2012;24:23-8. [PubMed]
- Dahmani S, Michelet D, Abback PS, Wood C, Brasher C, Nivoche Y, et al. Ketamine for perioperative pain management in children: a meta-analysis of published studies. Paediatr Anaesth. 2011;21:636-52. [PubMed] DOI: 1111/j.1460-9592.2011.03566.x
- Honarmand A, Safavi MR, Jamshidi M. The preventative analgesic effect of preincisional peritonsillar infiltration of two low doses of ketamine for postoperative pain relief in children following adenotonsillectomy. A randomized, double-blind, placebo-controlled study. Paediatr Anaesth. 2008;18:508-14. [PubMed] DOI: 1111/j.1460-9592.2008.02461.x
- Marzban S, Hadadi S. The effect of low dose ketamine on sore throat after tonsillectomy. Anesth Pain. 2011;1:9-16. Full Text
- Meena SK, Jain RK, Meena VK, Meena DR. Comparative study on Start oral intake after tonsillectomy by pretonsillectomy peritonsillar infiltration various agents. IJASHNB. 2016;3:65-9. Full Text