Putri Amelia 1 , Najib Advani 2 , Mulyadi M. Djer 3 , Bastian Lubis 4*
Authors affiliations:
Background & objective: The Blalock-Taussig (BT) shunt maintains pulmonary blood flow in patients with congenital heart defects. It is used before corrective surgery to prevent cyanosis and is associated with morbidity and declining survival. This study evaluated survival rates and identified predictors of morbidity and mortality in children who underwent BT shunt surgery at Dr. Cipto Mangunkusumo Hospital, Indonesia, retrospectively.
Methods: This retrospective study included 197 pediatric patients who underwent BT shunt surgery between 2016 and 2020. The patients' ages at the time of surgery ranged from 20 months to 17 years, and their weights varied from 2.7–42 kg. Patients with incomplete medical records were excluded from the study. The primary goal of this study was to evaluate the 5-year survival rate and identify predictors of mortality. The Cox regression test was applied, including factors with P < 0.25 from the bivariate analysis.
Results: Postoperative complications included prolonged mechanical ventilation (> 24 h) in 52% of cases. The survival rate declined from 68% immediately after surgery to 50% at 60 months postoperatively. The significant mortality risk factors included age < 263 days [Hazards Ratio (HR), 3.097], single-ventricle defects (HR, 1.834), low body weight (< 4.25 kg) (HR, 3.802), preoperative prostaglandin use (HR, 2.818), hematocrit levels < 45% (HR, 1.946), and postoperative saturation < 70% (HR, 3.562). Younger age and low saturation levels were associated with higher mortality risk.
Conclusion: The BT shunt, which is critical for children with heart defects, is associated with significant morbidity and decreased survival rates. Key mortality factors include young age, low body weight, prostaglandin use, poor postoperative oxygen saturation, and single ventricle defects. These findings emphasize the need for careful patient selection, surgical planning, and care to optimize survival.
Abbreviations: BT shunt: Blalock-Taussig shunt, CHD: Congenital heart disease, CICU: Cardiac Intensive Care, UnitHR: Hazards Ratio, SDGs: Sustainable Development Goals, TOF: tetralogy of Fallot,
Keywords: Blalock-Taussig shunt; Cardiac surgery; Congenital heart disease; Morbidity; Mortality; Postoperative outcomes
Citation: Amelia P, Advani N, Djer MM, Lubis B. Survival of Blalock-Taussig shunt surgery in children with congenital heart disease in the postoperative cardiac ICU of a national referral hospital in Jakarta. Anaesth. pain intensive care 2025;29(6):483-91 DOI: 10.35975/apic.v29i6.2891
Received: June 18, 2025; Revised: August 04, 2025; Accepted: August 04, 2025
Congenital heart disease (CHD) is a common congenital anomaly that occurs in approximately 0.8% of live births worldwide. Neonates with CHD up to 50% do not exhibit anomalies at birth; however, 25% manifest severe anomalies and necessitate prompt surgical treatment.1 The advancement of CHD protocols has been swift since the introduction of heart-lung bypass. Authoritative rectification through surgery has substantially reduced the morbidity and mortality rates.2 This aligns with the Sustainable Development Goals (SDGs), a worldwide strategy accepted by global leaders, including those in Indonesia. The third aim of the SDG is to guarantee robust lives and enhance the well-being of all age groups. One of the objectives anticipated by 2030 is to stop avoidable fatalities in newborns and young children. Each nation aims to decrease neonatal mortality to less than 12 per 1000 births and young child fatalities to as low as 25 per 1000 births.3 Furthermore, another anticipated objective is to reduce premature fatalities from noncommunicable illnesses by one-third through prevention, therapy, and enhancing psychological health and well-being. Non-communicable illnesses involve four fundamental conditions: cardiovascular disease, diabetes mellitus, cancer, and chronic respiratory disease. In babies with critical CHD, immediate surgical steps, such as palliative correction, are performed before definitive actions are taken.4
Approximately 30% of children with CHD undergo cardiac intervention within the first year of birth. Forty-five percent of children with cyanotic CHD, such as tetralogy of Fallot (TOF), require palliative procedures before conclusive rectification.5,6 Patients requiring urgent intervention are infants with severe heart disease who rely on the ductus arteriosus to sustain blood flow to the lungs (e.g., pulmonary atresia), sustain blood flow to the body (e.g., hypoplastic left heart syndrome), or sustain blood flow to both the body and lungs (e.g., transposition of the great arteries).7 In this scenario, the linkage between the pulmonary artery and systemic circulation is established through a surgical procedure referred to as the Blalock-Taussig shunt (BT shunt).8 BT shunt techniques have multiple objectives, specifically enhancing blood circulation to the lungs, resolving cyanosis, promoting pulmonary artery development, increasing the size of the left ventricle (LV), sustaining sufficient preload and averting hemodilution, preserving afterload via systemic vascular resistance, and regulating diastolic pressure to sustain coronary perfusion pressure.9 The risk of surgery may manifest intraoperatively, in the cardiac intensive care unit (CICU), or during the time spent in an outpatient area. The postoperative consequences of the BT shunt exhibit a broad range of outcomes. Several markers were evaluated, including the duration of stay in the standard room or CICU, the necessity for anticoagulant and antithrombotic treatment, the interval before definitive surgery, morbidity, and mortality.10
Morbidities associated with BT shunt operations may include postoperative hemorrhage, reoperation resulting from acute shunt thrombosis, decreased heart function syndrome due to excessive shunting, sepsis, paralysis of the diaphragm, paralysis of the vagus nerve, infection of the surgical incision, lung bleeding, collapsed lung, and treatment in the Cardiac Intensive Care Unit (CICU) lasting more than 48 hours.11 Based on data from 2016-2017 in the National Cardiovascular Center Harapan Kita (NCCHK), the overall mortality was 13.5% with 1.5%, 41%, and 57.5% of the population proportion undergoing BTS being neonates, infants, and older children or adults, respectively.11 In developed countries, BT shunt palliative operation is seldom conducted because of the ample resources, proficiency, and skill of the pediatric cardiac surgery group and the satisfactory count of personnel, enabling definitive interventions to be executed promptly. Nonetheless, in emerging nations, this operation is frequently undertaken because of restricted personnel count, proficiency and skill, and facilities.12,13 To date, assessments of the postoperative outer surface of the BT shunt have not been consistently conducted; therefore, information to assess the effectiveness of BT shunt procedures is not extensively recognized. Likewise, in Indonesia, external evaluations of morbidity and mortality following BT shunt surgery are limited because the resources for treating congenital heart disease are insufficient and unevenly distributed.13
The evaluation of the standards of pediatric cardiac surgery facilities is essential to enhance services in an institution.14 This evaluation may manifest as morbidity and mortality. In the past, mortality figures were the primary method for evaluating treatment efficacy. Nonetheless, advancements in surgical procedures, enhancements in the skills of cardiac surgical teams, and postoperative management have resulted in relatively low mortality rates, thereby rendering sole reliance on mortality assessment as inadequate.5,15 Thus, this study aimed to evaluate the risk factors that influence BT shunt postoperative morbidity and mortality in Indonesia. Consequently, the determinants can be addressed prior to, during, and after BT shunt surgery to enhance the standard of heart surgery provisions.
The current study aimed to evaluate the survival rates and identify the predictive factors of morbidity and mortality in children undergoing BT shunt surgery at a national referral hospital in Indonesia.
This investigation was carried out retrospectively. Pediatric patients aged 20 months to 17 years with CHD who underwent BT shunt operations at Jakarta Dr. Cipto Mangunkusumo Hospital, from January 2016 to December 2020, were enrolled. The hospital functions as a national referral center for government hospitals across Indonesia, and began service in the field of pediatric cardiac care in 2002. The hospital has an 8-bed CICU that provides postoperative care for both pediatric and adult cardiac patients, and is managed by pediatric and adult intensivists.
In this study, the convenient sampling method was used with the following inclusion criteria: pediatric patients with CHD, who underwent BT shunt surgery between January 2016 to December 2020. Pediatric patients with incomplete medical records and incomplete observation data were excluded, resulting in a total of 197 eligible samples out of 226 samples. To address missing data, we conducted a sensitivity analysis by comparing the baseline characteristics (age, sex, and body weight) between the included and excluded patients to ensure that no significant bias was introduced. No substantial differences were found, suggesting that the excluded cases did not skew the results.
Every child with CHD who underwent BT shunt surgery at Dr. Cipto Mangunkusumo Hospital was evaluated for short-term outcome data until hospital discharge. The data collected were as follows: 1) demographic data, including age at the time of CHD diagnosis, age at BT shunt surgery, sex, and body weight before surgery; 2) BT shunt postoperative morbidity (duration of mechanical ventilation and length of CICU stay); prolonged mechanical ventilation was defined as ventilation exceeding 72 h, based on established pediatric cardiac surgery literature;15 3) BT shunt postoperative mortality during hospitalization; and 4) predictors of postoperative morbidity.
To assess long-term outcomes, patients were contacted by phone calls or WhatsApp to determine their condition (alive or deceased) after discharge from the hospital. If the patient died, the exact time of death was determined to determine how long the patient survived after surgery.
The independent variables comprised age, prognosis, body weight, nutritional status, cardiopulmonary bypass (CPB), prostaglandin use, pre-and postoperative oxygen saturation, pre-surgical hemoglobin concentrations, manner of junction, manner of intervention, surgical technique, transplant dimensions, postoperative hemoglobin (Hb) concentrations, activated partial thromboplastin time (aPTT), and postoperative blood transfusions. The dependent variable in this study was postoperative mortality after BT shunt placement, and morbidity was assessed based on the duration of mechanical ventilation and length of the CICU stay. The numeric data obtained from the samples were then categorized based on the previous literature11 and the cut-off obtained from either the median or mean of the samples of this study for the age variable. Nutritional status was assessed using the WHO chart, with those in the range of +2 SD to -2 SD categorized as having a normal nutritional status, and those below were considered malnourished.16
Hospital medical records were analyzed to evaluate morbidity and mortality. To explore enduring results in the shape of subsequent phase operation and tube openness, examination of the medical record was conducted either through telephone or WhatsApp. An interview via WhatsApp was conducted to assess the mortality of the patient, and the patient’s medical records were reviewed to assess factors contributing to the patient’s outcome.
2.1. Statistical analysis
Data analysis was performed using IBM SPSS Statistics for Windows version 25 (IBM Corp., Armonk, N.Y., USA). The survival rates of the samples are shown in the Kaplan-Meier survival curve. The Cox regression test was used to determine the factors linked with the 5-year survival rate of the sample. All factors with P < 0.25 in the bivariate analysis were included in the Cox proportional hazards model.17 The outcomes of the multivariate analysis are presented as hazard ratios (HR) with 95% confidence intervals. To address missing data, we performed a sensitivity analysis using multiple imputations for key variables (e.g., body weight and oxygen saturation) to assess the robustness of our findings. The results were consistent with the primary analysis, confirming the minimal impact of missing data.
Through the 60-month follow-up, the participants’ survival post-BT shunt operation is depicted in Figure 1 and Table 1. Table 1 shows that at the 0th month of postoperative BT shunt, 68% of individuals survived. At the 12th, 24th, and 36th months, 62%, 55%, and 50% of the individuals remained alive, respectively. At the culmination of the observation in the 60th month, only 50% of patients survived. From this information, it is evident that the percentage of fatality in the initial 1-year postoperative period was 32%. At 12, 24, and 36 months following BT shunt surgery, the fatality rates were 38%, 45%, and 50%, respectively. These numbers were obtained by deducting a survival rate of 100%.
From the Kaplan-Meier plot, it is evident that the midpoint survival (i.e., at a point when half of the research participants were still alive) is 60 months after the BT shunt procedure (Figure 1).
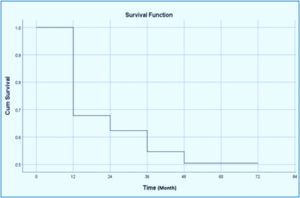
Figure 1: Kaplan-Meier estimation of survival at 5 years following postoperative BT shunt.
Bivariate examination was performed to evaluate the variables influencing the survival of individuals who underwent BT shunt postoperative death using Cox regression analysis. The magnitude of the relationship was expressed as the hazard ratio (HR).
Patients younger than 263 days at the time of BT surgery had a higher risk of death within 5 years (95% CI: 1.918–5.001) than those older than 263 days, indicating that age had a statistically significant survival outcome (P < 0.001). Similarly, patients diagnosed with a single-ventricle defect faced a 1.8-fold higher risk of mortality within five years after undergoing BT shunt surgery (95% CI: 1.105–3.044, P = 0.019) compared to those with a dual-ventricle defect.
Body weight emerged as a crucial factor, with patients weighing less than 4.25 kg at the time of surgery experiencing a dramatic 3.8-fold increase in mortality risk within five years (95% CI: 2.267–6.374, p < 0.001) compared to those above this threshold, underscoring the vulnerability of low-weight patients and the need for tailored perioperative strategies.
Preoperative prostaglandin administration significantly elevated the risk of mortality, with a 2.8-fold increase observed within five years after surgery. This association (P = 0.016) highlights the potential risks of this intervention and its influence on the long-term outcomes. Additionally, preoperative hematocrit levels < 45% were associated with a 1.9-fold higher risk of death within five years post-surgery (P = 0.013).
Surgical techniques and procedural factors further influenced these outcomes. Patients who underwent innominate artery-to-pulmonary artery (PA) anastomosis had a 1.9-fold higher likelihood of mortality within five years (P = 0.015). Similarly, median sternotomy was associated with a 2.1-fold greater risk of mortality within five years (95% CI: 1.338–3.597) than thoracotomy, emphasizing the need for careful consideration of the surgical approach. Graft size was another critical factor, with patients receiving grafts smaller than 3.5 mm facing a 2.1-fold higher risk of death within five years, underscoring the importance of selecting appropriate graft sizes for improved outcomes.
Postoperative oxygen saturation (SpO2) < 70% emerged as a key predictor of mortality, with affected patients experiencing a striking 3.5-fold higher risk of death within five years. This highlights the vital role of maintaining optimal postoperative oxygenation. Elevated postoperative activated partial thromboplastin time (aPTT) levels above 60 s were associated with a 0.3-fold increase in mortality risk within five years.
The multiple regression analysis in this study utilized the Cox Proportional Hazard Model. Factors included as contenders for multiple regression variables were variables with P < 0.25, specifically age, diagnosis, mass, CPB utilization, preoperative prostaglandin utilization, preoperative Hb concentration, Hb concentration before surgery, Ht concentration before surgery, anastomosis, type of procedure, surgical approach, graft magnitude, postoperative SpO2, postoperative aPTT concentrations, and postoperative pO2 concentrations.
After formulating the preliminary framework of the 14 factors from the outset to the final stage, the remaining two factors were derived by deducting all factors, leading to the identification of a noteworthy factor (P < 0.05), specifically, age and post-surgery SpO2.
From Table 2, it can be inferred that among all the variables examined, the variables that predominantly influenced the survival of BT shunt postoperative individuals were life ≤ 263 days and postoperative SpO2 < 70%. Individuals with a lifespan of ≤ 263 days had a mortality risk three times compared with those with an age > 263 days. Patients with SpO2 < 70% had a mortality risk of 3.2 times compared with those with postoperative SpO2 ≥70%. The other factors did not influence the precision of the research participants in the multivariate analysis.
The findings of this study show that the mortality proportion of patients undergoing BT shunt increases each year, with the highest mortality rate of 50% from the third to fifth years of follow-up. The postoperative death rate of BT shunt in children is declining each year due to advancements in surgical procedures, enhanced capabilities of cardiac surgery teams, and postoperative management, leading to exceedingly low fatality rates.18 Previous research has documented fatality percentages for postoperative BT shunt individuals varying from 3.7-14%. Investigations carried out over a span of five years indicated the most minimal fatality rate; however, the age of the individuals examined was below 3 months, and half of those individuals received a diagnosis of Tetralogy of Fallot (TOF), which is a component of the biventricular diagnosis with a minor fatality rate of 5%.18
The findings indicate that young age, type of defect, body weight less than 4.25 kg, application of prostaglandins prior to operation, higher preoperative hematocrit levels, location of anastomosis, types of surgery approach, graft size, postoperative SpO2, postoperative aPTT, and postoperative pO2 play a role in the survival rate of patients who have undergone BT shunt procedure. In contrast, numerous studies have demonstrated that variables influencing BT shunt postoperative death rate include body mass under 3 kg, hematocrit concentration exceeding 45%, and aPTT <60 seconds.11,19
Patients with a single ventricular defect were likely to have a higher risk of mortality following BT shunt procedures (HR = 1.834 (1.105–3.044)). Syaikh et al. (2021) also stated that, except for one case, all patients who died within 30 days of the BT shunt procedure had single-ventricle defects.20 Patients with single-ventricle defects are more prone to adverse outcomes due to several factors, including the higher prevalence of associated anomalies, genetic syndromes, the complexity of their cardiac anatomy, and the presence of coronary pathologies. Additionally, volume overload caused by systemic-to-pulmonary artery shunts, such as modified Blalock-Taussig (mBT) shunts, can further increase susceptibility to complications, even in the long term.21
In this study, most BT shunt procedures were performed using sternotomy. Rasool et al. (2024) stated that the thoracostomy approach was used more often because it was considered more accessible, quicker, and safer. However, one life-threatening complication associated with thoracotomy is phrenic nerve paralysis.21
Ventilatory support is necessary for postoperative patients for a minimum of 12–24 h. Nevertheless, the process of transitioning to the removal of the endotracheal tube is advised to be conducted on the second postoperative day if the patient is stable.21 Oxygen saturation was maintained at 70–85%. The optimal goal of the anticipated saturation was 75–80%. Oxygen therapy is gradually tapered when saturation reaches 80% to prevent excessive shunting.21 Arterial blood gas analysis was performed every 6 h if the patient was in a stable state.22 As outlined in a clinical directive put forth by the Australian government, the utilization of a ventilator is considered prolonged if it surpasses 24 hours.22
In this investigation, we discovered that the central value of respirator utilization in the study participants was not significantly associated with mortality. Extended utilization of respiratory support is frequently associated with increased fatality rates. One of the most frequently mentioned reasons was pneumonia associated with respiratory support. The frequency of pneumonia associated with respiratory support in post-surgery patients with CHD at the Integrated Heart Centre Dr. Cipto Mangunkusumo Hospital Jakarta was documented as 6%.23
The duration of hospital stays in the CICU in this investigation had a median of 2 days, with the briefest 0 hours and the longest 5 months and 18 days. The duration of post-surgery CICU care from certain investigations fluctuated, ranging from 18 h to 4 days. This number fluctuates and is believed to be connected to the age bracket that was the focus of the research.24,25
Currently, the choice of conclusive amendment in TOF individuals within the first year of life surpasses the option of correction in two stages (palliative shunt surgery and successive surgical rectification of TOF). A study from Germany revealed that the fatality rate in individuals receiving definitive correction under 12 months of age was merely 3.2%, in contrast to 12.9% in individuals surpassing that age.26
Some of the benefits of conducting palliative operations initially include more straightforward interventions, leading to a reduced length of hospital stay, enhanced development of the pulmonary artery, and diminished utilization of transannular patches. Conversely, the benefits of directly performing definitive surgery are to avoid deformation of the pulmonary arteries, lower the necessity for hospitalization for follow-up procedures, and lessen early cyanosis.27
In this study, age and postoperative SpO2 were significant variables for predicting mortality after the BT shunt procedure. This result is different from the study conducted at the National Cardiovascular Center Harapan Kita by Riyandi et al. in 2019, which stated that age does not affect the mortality rate. Nonetheless, in their study, the highest mortality rate was found in the group of neonates (≤ 28 days) with a rate of 32.1%, which indicates that young age might contribute to mortality after the BT shunt procedure.11 A study by Tamisier et al. (1990) also found that younger age at surgery and lower weight were significantly associated with shunt thrombosis, which is one of the most lethal complications of the BT shunt procedure, contributing to mortality post-BT shunt procedure.28,29 A systematic review by Awori et al. in 2023 stated that most patients undergoing the BT shunt procedure had a maximum increase in oxygen saturation of 20%.30 According to a study by Singh et al. (2014), young age and an increase in PaO2 below 25% are predictors of mortality in patients.31 The post-shunt rise in PaO2 (P Diff) plays a crucial role in postoperative weaning and recovery, particularly in children undergoing emergency procedures.
This study provides valuable insights into the outcomes of BT shunt procedures in Indonesia, addressing the previously limited availability of such data in the literature. The large sample size strengthens the reliability of these findings. To account for the significant differences in expertise and resources across centers in Indonesia, adopting a multicenter approach in future studies will be essential.
The study limitations include it being a single-center study, which resulted in less variable results, because it only came from one hospital. Future studies that incorporate several referral hospitals across the country could help show the bigger picture of the factors affecting the mortality of children with congenital heart disease in Indonesia. This study also relied on data from the families of patients via phone calls, which could have lead to a risk of loss to follow-up or a high risk of developing bias.
This study underscores the BT shunt as a critical palliative procedure for children with congenital heart defects, although it is associated with significant morbidity and declining survival rates. Key predictors of mortality include younger age, low body weight, preoperative and postoperative physiological challenges, and specific surgical factors, highlighting the need for meticulous patient selection, individualized surgical planning, and enhanced perioperative care to improve outcomes. These findings emphasize the importance of early intervention, improved risk stratification, and targeted management strategies to optimize outcomes in this vulnerable population group.
7. Ethics approval
The Ethics Committee of the Faculty of Medicine, Universitas Indonesia, Cipto Mangunkusumo Hospital approved this study (KET-1301/UN2.F1/ETIK/PPM.00.02/2020). The consent of the parents of the research participants was obtained when possible.
8. Data availability
The numerical data generated during this research are available from the authors.
9. Conflict of interest
All authors declare that there was no conflict of interest.
10. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
11. Acknowledgments
The authors thank Universitas Indonesia, Dr. Cipto Mangunkusumo Hospital, Jakarta, Indonesia.
12. Authors’ contribution
All authors are responsible for the conceptualization, preparation, editing, and assurance of the manuscript. All authors have approved the final draft.
Authors affiliations:
- Putri Amelia, Department of Pediatrics, Faculty of Medicine, Universitas Sumatera Utara, Medan, Indonesia; Email: putri.amelia@usu.ac.id; {ORCID:0000-0001-9941-2555}
- Najib Advani, Department of Child Health, Faculty of Medicine, Universitas Indonesia, Jakarta, Indonesia; Email: najib2023july@gmail.com
- Mulyadi M. Djer, Department of Child Health, Faculty of Medicine, Universitas Indonesia, Jakarta, Indonesia; Email: mulyadiqwert2023@gmail.com
- Bastian Lubis, Department of Anesthesiology and Intensive Care, Faculty of Medicine, Universitas Sumatera Utara, Indonesia; Email: bastian.lubis@usu.ac.id; {ORCID:0000-0002-1839-4146}
ABSTRACT
Background & objective: The Blalock-Taussig (BT) shunt maintains pulmonary blood flow in patients with congenital heart defects. It is used before corrective surgery to prevent cyanosis and is associated with morbidity and declining survival. This study evaluated survival rates and identified predictors of morbidity and mortality in children who underwent BT shunt surgery at Dr. Cipto Mangunkusumo Hospital, Indonesia, retrospectively.
Methods: This retrospective study included 197 pediatric patients who underwent BT shunt surgery between 2016 and 2020. The patients' ages at the time of surgery ranged from 20 months to 17 years, and their weights varied from 2.7–42 kg. Patients with incomplete medical records were excluded from the study. The primary goal of this study was to evaluate the 5-year survival rate and identify predictors of mortality. The Cox regression test was applied, including factors with P < 0.25 from the bivariate analysis.
Results: Postoperative complications included prolonged mechanical ventilation (> 24 h) in 52% of cases. The survival rate declined from 68% immediately after surgery to 50% at 60 months postoperatively. The significant mortality risk factors included age < 263 days [Hazards Ratio (HR), 3.097], single-ventricle defects (HR, 1.834), low body weight (< 4.25 kg) (HR, 3.802), preoperative prostaglandin use (HR, 2.818), hematocrit levels < 45% (HR, 1.946), and postoperative saturation < 70% (HR, 3.562). Younger age and low saturation levels were associated with higher mortality risk.
Conclusion: The BT shunt, which is critical for children with heart defects, is associated with significant morbidity and decreased survival rates. Key mortality factors include young age, low body weight, prostaglandin use, poor postoperative oxygen saturation, and single ventricle defects. These findings emphasize the need for careful patient selection, surgical planning, and care to optimize survival.
Abbreviations: BT shunt: Blalock-Taussig shunt, CHD: Congenital heart disease, CICU: Cardiac Intensive Care, UnitHR: Hazards Ratio, SDGs: Sustainable Development Goals, TOF: tetralogy of Fallot,
Keywords: Blalock-Taussig shunt; Cardiac surgery; Congenital heart disease; Morbidity; Mortality; Postoperative outcomes
Citation: Amelia P, Advani N, Djer MM, Lubis B. Survival of Blalock-Taussig shunt surgery in children with congenital heart disease in the postoperative cardiac ICU of a national referral hospital in Jakarta. Anaesth. pain intensive care 2025;29(6):483-91 DOI: 10.35975/apic.v29i6.2891
Received: June 18, 2025; Revised: August 04, 2025; Accepted: August 04, 2025
1. INTRODUCTION
Congenital heart disease (CHD) is a common congenital anomaly that occurs in approximately 0.8% of live births worldwide. Neonates with CHD up to 50% do not exhibit anomalies at birth; however, 25% manifest severe anomalies and necessitate prompt surgical treatment.1 The advancement of CHD protocols has been swift since the introduction of heart-lung bypass. Authoritative rectification through surgery has substantially reduced the morbidity and mortality rates.2 This aligns with the Sustainable Development Goals (SDGs), a worldwide strategy accepted by global leaders, including those in Indonesia. The third aim of the SDG is to guarantee robust lives and enhance the well-being of all age groups. One of the objectives anticipated by 2030 is to stop avoidable fatalities in newborns and young children. Each nation aims to decrease neonatal mortality to less than 12 per 1000 births and young child fatalities to as low as 25 per 1000 births.3 Furthermore, another anticipated objective is to reduce premature fatalities from noncommunicable illnesses by one-third through prevention, therapy, and enhancing psychological health and well-being. Non-communicable illnesses involve four fundamental conditions: cardiovascular disease, diabetes mellitus, cancer, and chronic respiratory disease. In babies with critical CHD, immediate surgical steps, such as palliative correction, are performed before definitive actions are taken.4
Approximately 30% of children with CHD undergo cardiac intervention within the first year of birth. Forty-five percent of children with cyanotic CHD, such as tetralogy of Fallot (TOF), require palliative procedures before conclusive rectification.5,6 Patients requiring urgent intervention are infants with severe heart disease who rely on the ductus arteriosus to sustain blood flow to the lungs (e.g., pulmonary atresia), sustain blood flow to the body (e.g., hypoplastic left heart syndrome), or sustain blood flow to both the body and lungs (e.g., transposition of the great arteries).7 In this scenario, the linkage between the pulmonary artery and systemic circulation is established through a surgical procedure referred to as the Blalock-Taussig shunt (BT shunt).8 BT shunt techniques have multiple objectives, specifically enhancing blood circulation to the lungs, resolving cyanosis, promoting pulmonary artery development, increasing the size of the left ventricle (LV), sustaining sufficient preload and averting hemodilution, preserving afterload via systemic vascular resistance, and regulating diastolic pressure to sustain coronary perfusion pressure.9 The risk of surgery may manifest intraoperatively, in the cardiac intensive care unit (CICU), or during the time spent in an outpatient area. The postoperative consequences of the BT shunt exhibit a broad range of outcomes. Several markers were evaluated, including the duration of stay in the standard room or CICU, the necessity for anticoagulant and antithrombotic treatment, the interval before definitive surgery, morbidity, and mortality.10
Morbidities associated with BT shunt operations may include postoperative hemorrhage, reoperation resulting from acute shunt thrombosis, decreased heart function syndrome due to excessive shunting, sepsis, paralysis of the diaphragm, paralysis of the vagus nerve, infection of the surgical incision, lung bleeding, collapsed lung, and treatment in the Cardiac Intensive Care Unit (CICU) lasting more than 48 hours.11 Based on data from 2016-2017 in the National Cardiovascular Center Harapan Kita (NCCHK), the overall mortality was 13.5% with 1.5%, 41%, and 57.5% of the population proportion undergoing BTS being neonates, infants, and older children or adults, respectively.11 In developed countries, BT shunt palliative operation is seldom conducted because of the ample resources, proficiency, and skill of the pediatric cardiac surgery group and the satisfactory count of personnel, enabling definitive interventions to be executed promptly. Nonetheless, in emerging nations, this operation is frequently undertaken because of restricted personnel count, proficiency and skill, and facilities.12,13 To date, assessments of the postoperative outer surface of the BT shunt have not been consistently conducted; therefore, information to assess the effectiveness of BT shunt procedures is not extensively recognized. Likewise, in Indonesia, external evaluations of morbidity and mortality following BT shunt surgery are limited because the resources for treating congenital heart disease are insufficient and unevenly distributed.13
The evaluation of the standards of pediatric cardiac surgery facilities is essential to enhance services in an institution.14 This evaluation may manifest as morbidity and mortality. In the past, mortality figures were the primary method for evaluating treatment efficacy. Nonetheless, advancements in surgical procedures, enhancements in the skills of cardiac surgical teams, and postoperative management have resulted in relatively low mortality rates, thereby rendering sole reliance on mortality assessment as inadequate.5,15 Thus, this study aimed to evaluate the risk factors that influence BT shunt postoperative morbidity and mortality in Indonesia. Consequently, the determinants can be addressed prior to, during, and after BT shunt surgery to enhance the standard of heart surgery provisions.
The current study aimed to evaluate the survival rates and identify the predictive factors of morbidity and mortality in children undergoing BT shunt surgery at a national referral hospital in Indonesia.
2. METHODOLOGY
This investigation was carried out retrospectively. Pediatric patients aged 20 months to 17 years with CHD who underwent BT shunt operations at Jakarta Dr. Cipto Mangunkusumo Hospital, from January 2016 to December 2020, were enrolled. The hospital functions as a national referral center for government hospitals across Indonesia, and began service in the field of pediatric cardiac care in 2002. The hospital has an 8-bed CICU that provides postoperative care for both pediatric and adult cardiac patients, and is managed by pediatric and adult intensivists.
In this study, the convenient sampling method was used with the following inclusion criteria: pediatric patients with CHD, who underwent BT shunt surgery between January 2016 to December 2020. Pediatric patients with incomplete medical records and incomplete observation data were excluded, resulting in a total of 197 eligible samples out of 226 samples. To address missing data, we conducted a sensitivity analysis by comparing the baseline characteristics (age, sex, and body weight) between the included and excluded patients to ensure that no significant bias was introduced. No substantial differences were found, suggesting that the excluded cases did not skew the results.
Every child with CHD who underwent BT shunt surgery at Dr. Cipto Mangunkusumo Hospital was evaluated for short-term outcome data until hospital discharge. The data collected were as follows: 1) demographic data, including age at the time of CHD diagnosis, age at BT shunt surgery, sex, and body weight before surgery; 2) BT shunt postoperative morbidity (duration of mechanical ventilation and length of CICU stay); prolonged mechanical ventilation was defined as ventilation exceeding 72 h, based on established pediatric cardiac surgery literature;15 3) BT shunt postoperative mortality during hospitalization; and 4) predictors of postoperative morbidity.
To assess long-term outcomes, patients were contacted by phone calls or WhatsApp to determine their condition (alive or deceased) after discharge from the hospital. If the patient died, the exact time of death was determined to determine how long the patient survived after surgery.
The independent variables comprised age, prognosis, body weight, nutritional status, cardiopulmonary bypass (CPB), prostaglandin use, pre-and postoperative oxygen saturation, pre-surgical hemoglobin concentrations, manner of junction, manner of intervention, surgical technique, transplant dimensions, postoperative hemoglobin (Hb) concentrations, activated partial thromboplastin time (aPTT), and postoperative blood transfusions. The dependent variable in this study was postoperative mortality after BT shunt placement, and morbidity was assessed based on the duration of mechanical ventilation and length of the CICU stay. The numeric data obtained from the samples were then categorized based on the previous literature11 and the cut-off obtained from either the median or mean of the samples of this study for the age variable. Nutritional status was assessed using the WHO chart, with those in the range of +2 SD to -2 SD categorized as having a normal nutritional status, and those below were considered malnourished.16
Hospital medical records were analyzed to evaluate morbidity and mortality. To explore enduring results in the shape of subsequent phase operation and tube openness, examination of the medical record was conducted either through telephone or WhatsApp. An interview via WhatsApp was conducted to assess the mortality of the patient, and the patient’s medical records were reviewed to assess factors contributing to the patient’s outcome.
2.1. Statistical analysis
Data analysis was performed using IBM SPSS Statistics for Windows version 25 (IBM Corp., Armonk, N.Y., USA). The survival rates of the samples are shown in the Kaplan-Meier survival curve. The Cox regression test was used to determine the factors linked with the 5-year survival rate of the sample. All factors with P < 0.25 in the bivariate analysis were included in the Cox proportional hazards model.17 The outcomes of the multivariate analysis are presented as hazard ratios (HR) with 95% confidence intervals. To address missing data, we performed a sensitivity analysis using multiple imputations for key variables (e.g., body weight and oxygen saturation) to assess the robustness of our findings. The results were consistent with the primary analysis, confirming the minimal impact of missing data.
3. RESULTS
Through the 60-month follow-up, the participants’ survival post-BT shunt operation is depicted in Figure 1 and Table 1. Table 1 shows that at the 0th month of postoperative BT shunt, 68% of individuals survived. At the 12th, 24th, and 36th months, 62%, 55%, and 50% of the individuals remained alive, respectively. At the culmination of the observation in the 60th month, only 50% of patients survived. From this information, it is evident that the percentage of fatality in the initial 1-year postoperative period was 32%. At 12, 24, and 36 months following BT shunt surgery, the fatality rates were 38%, 45%, and 50%, respectively. These numbers were obtained by deducting a survival rate of 100%.
| Table 1: Postoperative survival outcomes following the BT shunt procedure | |||
| Period (Months) | Survival | Deaths | Cumulative survival |
| 0 | 197 | 57 | 0.68 |
| 12 | 100 | 6 | 0.62 |
| 24 | 44 | 4 | 0.55 |
| 36 | 17 | 1 | 0.50 |
| 48 | 8 | 5 | 0.50 |
| 60 | 3 | 3 | 0.50 |
From the Kaplan-Meier plot, it is evident that the midpoint survival (i.e., at a point when half of the research participants were still alive) is 60 months after the BT shunt procedure (Figure 1).

Figure 1: Kaplan-Meier estimation of survival at 5 years following postoperative BT shunt.
Bivariate examination was performed to evaluate the variables influencing the survival of individuals who underwent BT shunt postoperative death using Cox regression analysis. The magnitude of the relationship was expressed as the hazard ratio (HR).
Patients younger than 263 days at the time of BT surgery had a higher risk of death within 5 years (95% CI: 1.918–5.001) than those older than 263 days, indicating that age had a statistically significant survival outcome (P < 0.001). Similarly, patients diagnosed with a single-ventricle defect faced a 1.8-fold higher risk of mortality within five years after undergoing BT shunt surgery (95% CI: 1.105–3.044, P = 0.019) compared to those with a dual-ventricle defect.
Body weight emerged as a crucial factor, with patients weighing less than 4.25 kg at the time of surgery experiencing a dramatic 3.8-fold increase in mortality risk within five years (95% CI: 2.267–6.374, p < 0.001) compared to those above this threshold, underscoring the vulnerability of low-weight patients and the need for tailored perioperative strategies.
Preoperative prostaglandin administration significantly elevated the risk of mortality, with a 2.8-fold increase observed within five years after surgery. This association (P = 0.016) highlights the potential risks of this intervention and its influence on the long-term outcomes. Additionally, preoperative hematocrit levels < 45% were associated with a 1.9-fold higher risk of death within five years post-surgery (P = 0.013).
Surgical techniques and procedural factors further influenced these outcomes. Patients who underwent innominate artery-to-pulmonary artery (PA) anastomosis had a 1.9-fold higher likelihood of mortality within five years (P = 0.015). Similarly, median sternotomy was associated with a 2.1-fold greater risk of mortality within five years (95% CI: 1.338–3.597) than thoracotomy, emphasizing the need for careful consideration of the surgical approach. Graft size was another critical factor, with patients receiving grafts smaller than 3.5 mm facing a 2.1-fold higher risk of death within five years, underscoring the importance of selecting appropriate graft sizes for improved outcomes.
Postoperative oxygen saturation (SpO2) < 70% emerged as a key predictor of mortality, with affected patients experiencing a striking 3.5-fold higher risk of death within five years. This highlights the vital role of maintaining optimal postoperative oxygenation. Elevated postoperative activated partial thromboplastin time (aPTT) levels above 60 s were associated with a 0.3-fold increase in mortality risk within five years.
The multiple regression analysis in this study utilized the Cox Proportional Hazard Model. Factors included as contenders for multiple regression variables were variables with P < 0.25, specifically age, diagnosis, mass, CPB utilization, preoperative prostaglandin utilization, preoperative Hb concentration, Hb concentration before surgery, Ht concentration before surgery, anastomosis, type of procedure, surgical approach, graft magnitude, postoperative SpO2, postoperative aPTT concentrations, and postoperative pO2 concentrations.
After formulating the preliminary framework of the 14 factors from the outset to the final stage, the remaining two factors were derived by deducting all factors, leading to the identification of a noteworthy factor (P < 0.05), specifically, age and post-surgery SpO2.
From Table 2, it can be inferred that among all the variables examined, the variables that predominantly influenced the survival of BT shunt postoperative individuals were life ≤ 263 days and postoperative SpO2 < 70%. Individuals with a lifespan of ≤ 263 days had a mortality risk three times compared with those with an age > 263 days. Patients with SpO2 < 70% had a mortality risk of 3.2 times compared with those with postoperative SpO2 ≥70%. The other factors did not influence the precision of the research participants in the multivariate analysis.
| Table 2: A Bivariate analysis of factors associated with survival 5 years post-BT shunt | |||||||
| Variable | Mortality | Bivariate | Multivariate | ||||
| Yes | No | Hazard ratio (CI 95%) | P-value | HR
(CI 95%) |
P-value | ||
| Age |
≤ 263 Days > 263 Days |
25 (44.6) 16 (11.3) |
31 (55.4) 125 (88.7) |
.097 (1.918–5.001) Ref |
0.001* | 3.020 (1.790–5.095) | < 0.001 |
| Type of Defect |
Single Biventricular |
17 (35.4) 24 (16.1) |
31 (64.6) 125 (83.9) |
1.834 (1.105–3.044) Ref |
0.019* | ||
| Weight |
< 4.25 Kg ≥ 4.25 Kg |
21 (70.0) 20 (12.0) |
9 (30.0) 147 (88.0) |
3.802 (2.267–6.374) Ref |
< 0.001* | ||
| Nutritional State |
Malnutrition Good nutrition |
22 (21.2) 19 (20.4) |
82 (78.8) 74 (79.5) |
1.245 (0.769–2.014) Ref |
0.373 | ||
| CPB (Cardiopulmonary bypass) | Yes No |
11 (40.7) 30 (17.6) |
16 (59.3) 140 (82.4) |
1.631 (0.905-2.939) Ref |
0.103 | ||
| Prostaglandin |
Yes No |
6 (75.0) 35 (18.5) |
(25.0) 154 (81.5) |
2.818 (1.212–6.554) Ref |
0.016* | ||
| Preoperative SpO2
|
< 70 % ≥ 70 % |
11 (20.8) 30 (20.8) |
2 (79.2) 114 (79.2) |
1.171 (0.699-1.961) Ref |
0.549 | ||
| Preoperative Hb |
< 15 gr/dL ≥ 15 gr/dL |
17 (30.4) 24 (17.0) |
39 (69.6) 117 (83.0) |
1.626 (0.997–2.654) Ref |
0.052 | ||
| Preoperative Ht (hematocrit) | < 45 % ≥ 45 % |
16 (42.1) 25 (15.7) |
22 (57.9) 134 (84.3) |
1.946 (1.152–3.284) Ref |
0.013* | ||
| Location of Anastomosis Innominate Artery– pulmonary artery Aorta – pulmonary artery Carotid artery– pulmonary artery Subclavian – pulmonary artery |
20 (32.8) 10 (28.6) 1 (20.0) 10 (10.4) |
41 (67.2) 25 (71.4) 4 (80.0) 86 (89.6) |
1.989 (1.146–3.455) 1.823 (0.968–3.435) 0.714 (0.097–5.288) Ref |
0.015* 0.063 0.742 |
|||
| Category of procedure |
Urgent/Emergency Elective |
3 (35.1) 28 (17.5) |
24 (64.9) 132 (82.5) |
1.515 (0.873–2.628) Ref |
0.140 | ||
| Surgical approach |
Median Sternotomy Thoracotomy |
32 (35.6) 9 (8.4) |
58 (64.4) 98 (91.6) |
2.194 (1.338–3.597) Ref |
0.002* | ||
| Graft Size |
<3.5 mm ≥3.5 mm |
14 (42.4) 27 (16.5) |
19 (57.6) 137 (83.5) |
2.126 (1.250–3.618) Ref |
0.005* | ||
| Duration of mechanical ventilation | >24 Hours ≤24 Hours |
20 (19.6) 21 (22.1) |
82 (80.4) 74 (77.9) |
1.201 (0.744–1.939) Ref |
0.454 | ||
| Duration in CICU |
>48 Hours ≤48 Hours |
11 (16.4) 30 (23.1) |
56 (83.6) 100 (76.9) |
1.146 (0.696–1.888) Ref |
0.591 | ||
| Postoperative SpO2
|
<70 % ≥70 % |
3 (37.5) 35 (18.8) |
5 (62.5) 151 (81.2) |
3.562 (1.696–7.482) Ref |
0.001* | 3.262 (1.532–6.946) | 0.002 |
| Postoperative aPTT (activated partial thromboplastin time) | ≥60 Seconds <60 Seconds |
28 (17.9) 3 (10.3) |
128 (82.1) 26 (89.7) |
0.359 (0.137–0.940) Ref |
0.037* | ||
| Postoperative pO2
|
<30 mmHg >45 mmHg 30-45 mmHg (normal) |
1 (25.0) 23 (14.2) 9 (42.9) |
3 (75.0) 139 (85.8) 12 (57.1) |
0.683 (0.155–3.013) 0.354 (0.194–0.648) Ref |
0.615 0.001* |
||
| Postoperative Transfusion | Yes No |
19 (23.2) 22 (19.1) |
63 (76.9) 93 (80.9) |
1.277 (0.793–2.056) Ref |
0.314 | ||
| Data presented as n (%); P 0.05 considered as significant; CICU: Cardiac Intensive Care Unit, HR - Hazard Ratio | |||||||
4. DISCUSSION
The findings of this study show that the mortality proportion of patients undergoing BT shunt increases each year, with the highest mortality rate of 50% from the third to fifth years of follow-up. The postoperative death rate of BT shunt in children is declining each year due to advancements in surgical procedures, enhanced capabilities of cardiac surgery teams, and postoperative management, leading to exceedingly low fatality rates.18 Previous research has documented fatality percentages for postoperative BT shunt individuals varying from 3.7-14%. Investigations carried out over a span of five years indicated the most minimal fatality rate; however, the age of the individuals examined was below 3 months, and half of those individuals received a diagnosis of Tetralogy of Fallot (TOF), which is a component of the biventricular diagnosis with a minor fatality rate of 5%.18
The findings indicate that young age, type of defect, body weight less than 4.25 kg, application of prostaglandins prior to operation, higher preoperative hematocrit levels, location of anastomosis, types of surgery approach, graft size, postoperative SpO2, postoperative aPTT, and postoperative pO2 play a role in the survival rate of patients who have undergone BT shunt procedure. In contrast, numerous studies have demonstrated that variables influencing BT shunt postoperative death rate include body mass under 3 kg, hematocrit concentration exceeding 45%, and aPTT <60 seconds.11,19
Patients with a single ventricular defect were likely to have a higher risk of mortality following BT shunt procedures (HR = 1.834 (1.105–3.044)). Syaikh et al. (2021) also stated that, except for one case, all patients who died within 30 days of the BT shunt procedure had single-ventricle defects.20 Patients with single-ventricle defects are more prone to adverse outcomes due to several factors, including the higher prevalence of associated anomalies, genetic syndromes, the complexity of their cardiac anatomy, and the presence of coronary pathologies. Additionally, volume overload caused by systemic-to-pulmonary artery shunts, such as modified Blalock-Taussig (mBT) shunts, can further increase susceptibility to complications, even in the long term.21
In this study, most BT shunt procedures were performed using sternotomy. Rasool et al. (2024) stated that the thoracostomy approach was used more often because it was considered more accessible, quicker, and safer. However, one life-threatening complication associated with thoracotomy is phrenic nerve paralysis.21
Ventilatory support is necessary for postoperative patients for a minimum of 12–24 h. Nevertheless, the process of transitioning to the removal of the endotracheal tube is advised to be conducted on the second postoperative day if the patient is stable.21 Oxygen saturation was maintained at 70–85%. The optimal goal of the anticipated saturation was 75–80%. Oxygen therapy is gradually tapered when saturation reaches 80% to prevent excessive shunting.21 Arterial blood gas analysis was performed every 6 h if the patient was in a stable state.22 As outlined in a clinical directive put forth by the Australian government, the utilization of a ventilator is considered prolonged if it surpasses 24 hours.22
In this investigation, we discovered that the central value of respirator utilization in the study participants was not significantly associated with mortality. Extended utilization of respiratory support is frequently associated with increased fatality rates. One of the most frequently mentioned reasons was pneumonia associated with respiratory support. The frequency of pneumonia associated with respiratory support in post-surgery patients with CHD at the Integrated Heart Centre Dr. Cipto Mangunkusumo Hospital Jakarta was documented as 6%.23
The duration of hospital stays in the CICU in this investigation had a median of 2 days, with the briefest 0 hours and the longest 5 months and 18 days. The duration of post-surgery CICU care from certain investigations fluctuated, ranging from 18 h to 4 days. This number fluctuates and is believed to be connected to the age bracket that was the focus of the research.24,25
Currently, the choice of conclusive amendment in TOF individuals within the first year of life surpasses the option of correction in two stages (palliative shunt surgery and successive surgical rectification of TOF). A study from Germany revealed that the fatality rate in individuals receiving definitive correction under 12 months of age was merely 3.2%, in contrast to 12.9% in individuals surpassing that age.26
Some of the benefits of conducting palliative operations initially include more straightforward interventions, leading to a reduced length of hospital stay, enhanced development of the pulmonary artery, and diminished utilization of transannular patches. Conversely, the benefits of directly performing definitive surgery are to avoid deformation of the pulmonary arteries, lower the necessity for hospitalization for follow-up procedures, and lessen early cyanosis.27
In this study, age and postoperative SpO2 were significant variables for predicting mortality after the BT shunt procedure. This result is different from the study conducted at the National Cardiovascular Center Harapan Kita by Riyandi et al. in 2019, which stated that age does not affect the mortality rate. Nonetheless, in their study, the highest mortality rate was found in the group of neonates (≤ 28 days) with a rate of 32.1%, which indicates that young age might contribute to mortality after the BT shunt procedure.11 A study by Tamisier et al. (1990) also found that younger age at surgery and lower weight were significantly associated with shunt thrombosis, which is one of the most lethal complications of the BT shunt procedure, contributing to mortality post-BT shunt procedure.28,29 A systematic review by Awori et al. in 2023 stated that most patients undergoing the BT shunt procedure had a maximum increase in oxygen saturation of 20%.30 According to a study by Singh et al. (2014), young age and an increase in PaO2 below 25% are predictors of mortality in patients.31 The post-shunt rise in PaO2 (P Diff) plays a crucial role in postoperative weaning and recovery, particularly in children undergoing emergency procedures.
This study provides valuable insights into the outcomes of BT shunt procedures in Indonesia, addressing the previously limited availability of such data in the literature. The large sample size strengthens the reliability of these findings. To account for the significant differences in expertise and resources across centers in Indonesia, adopting a multicenter approach in future studies will be essential.
5. LIMITATIONS
The study limitations include it being a single-center study, which resulted in less variable results, because it only came from one hospital. Future studies that incorporate several referral hospitals across the country could help show the bigger picture of the factors affecting the mortality of children with congenital heart disease in Indonesia. This study also relied on data from the families of patients via phone calls, which could have lead to a risk of loss to follow-up or a high risk of developing bias.
6. CONCLUSION
This study underscores the BT shunt as a critical palliative procedure for children with congenital heart defects, although it is associated with significant morbidity and declining survival rates. Key predictors of mortality include younger age, low body weight, preoperative and postoperative physiological challenges, and specific surgical factors, highlighting the need for meticulous patient selection, individualized surgical planning, and enhanced perioperative care to improve outcomes. These findings emphasize the importance of early intervention, improved risk stratification, and targeted management strategies to optimize outcomes in this vulnerable population group.
7. Ethics approval
The Ethics Committee of the Faculty of Medicine, Universitas Indonesia, Cipto Mangunkusumo Hospital approved this study (KET-1301/UN2.F1/ETIK/PPM.00.02/2020). The consent of the parents of the research participants was obtained when possible.
8. Data availability
The numerical data generated during this research are available from the authors.
9. Conflict of interest
All authors declare that there was no conflict of interest.
10. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
11. Acknowledgments
The authors thank Universitas Indonesia, Dr. Cipto Mangunkusumo Hospital, Jakarta, Indonesia.
12. Authors’ contribution
All authors are responsible for the conceptualization, preparation, editing, and assurance of the manuscript. All authors have approved the final draft.
13. REFERENCES
- Kantor PF, Lougheed J, Dancea A, McGillion M, Barbosa N, Wong K. Presentation, Diagnosis, and Medical Management of Heart Failure in Children: Canadian Cardiovascular Society Guidelines. Can J Cardiol. 2013;29(12):1535-52. [PubMed] DOI: 1016/j.cjca.2013.08.008
- Bouma BJ, Mulder BJM. Changing Landscape of Congenital Heart Disease. Circ Res. 2017;120(6):908-22. [PubMed] DOI: 1161/CIRCRESAHA.116.309302
- Sorooshian S. The sustainable development goals of the United Nations: A comparative midterm research review. J Clean Prod. 2024;:142272. DOI: 1016/j.jclepro.2024.142272
- Pemerintah Provinsi DKI Jakarta. Sustainable Development Goals Jakarta 2020. Jakarta: Bappeda DKI Jakarta; 2020.
- Sanders J, Keogh BE, Van der Meulen J, Browne JP, Treasure T, Montgomery HE. The development of a postoperative morbidity score to assess total morbidity burden after cardiac surgery. J Clin Epidemiol. 2012;65(4):423-33. [PubMed] DOI: 1016/j.jclinepi.2011.11.004
- Kanter KR, Kogon BE, Kirshbom PM, Carlock PR. Symptomatic Neonatal Tetralogy of Fallot: Repair or Shunt? Ann Thorac Surg. 2010;89(3):858-63. [PubMed] DOI: 1016/j.athoracsur.2009.12.060
- Yuan SM, Shinfeld A, Raanani E. The Blalock-Taussig Shunt. J Card Surg. 2009;24(2):101-8. [PubMed] DOI: 1111/j.1540-8191.2008.00758.x
- Li D, Wang Y, Lin K, An Q. Modified Blalock-Taussig Shunt: A Single-Center Experience and Follow-Up. Heart Surg Forum. 2020;23(1):E053-E057. [PubMed] DOI: 1532/hsf.2715
- McKenzie DE, Khan MS, Samayoa AX, Vener DS, Ishak YM, Fraser CD. The Blalock-Taussig Shunt Revisited: A Contemporary Experience. J Am Coll Surg. 2013;216(4):699-704. [PubMed] DOI: 1016/j.jamcollsurg.2012.12.027
- Sasikumar N, Hermuzi A, Fan CPS, Lee KJ, Chaturvedi R, Benson L. Outcomes of Blalock-Taussig shunts in current era: A single center experience. Congenit Heart Dis. 2017;12(6):808-14. [PubMed] DOI: 1111/chd.12516
- Riyandi M, Lilyasari O, Juzar DA, Rahmat B. Age Criteria As Operative Mortality Predictor After Modified Blalock-Taussig Shunt. Indones J Cardiol. 2019;40(1). DOI: 30701/ijc.v40i1.763
- Jonas RA. Congenital Heart Surgery in Developing Countries. Semin Thorac Cardiovasc Surg Pediatr Card Surg Annu. 2008;11(1):3-6. [PubMed] DOI: 1053/j.pcsu.2007.12.001
- Rana JS, Ahmad KA, Shamim AS, Hassan SB, Ahmed MA. Blalock-Taussig shunt: Experience from the developing world. Heart Lung Circ. 2002;11(3):152-6. [PubMed] DOI: 1046/j.1444-2892.2002.00145.x
- Eghtesady P, Brar AK, Hall M. Prioritizing quality improvement in pediatric cardiac surgery. J Thorac Cardiovasc Surg. 2013;145(3):631-40. [PubMed] DOI: 1016/j.jtcvs.2012.12.018
- Welke KF, Ungerleider RM. Mortality as an outcome parameter for pediatric heart surgery. ASAIO J. 2006;52(5):552-5. [PubMed] DOI: 1097/01.mat.0000235490.26754.09
- Unit Kerja Koordinasi Nutrisi dan Penyakit Metabolik Ikatan Dokter Anak Indonesia. Asuhan nutrisi pediatrik (Pediatric Nutrition Care). Jakarta: Ikatan Dokter Anak Indonesia; 2011.
- Dahlan MS. Statistik Untuk Kedokteran dan Kesehatan Edisi 6. Jakarta: Salemba Medika; 2014.
- Gold JP, Violaris K, Engle MA, Klein AA, Ehlers KH, Isom OW. A Five-Year Clinical Experience with 112 Blalock-Taussig Shunts. J Card Surg. 1993;8(1):9-17. [PubMed] DOI: 1111/j.1540-8191.1993.tb00571.x
- Küçük M, Özdemir R, Karaçelik M, Doksöz Ö, Karadeniz C, Sarıosmanoğlu N. Risk Factors for Thrombosis, Overshunting and Death in Infants after Modified Blalock-Taussig Shunt. Acta Cardiol Sin. 2016;32(3):337-42. [PubMed] DOI: 6515/acs20150731a
- Shaikh S, Al-Mukhaini KS, Al-Rawahi AH, Al-Dafie O. Outcomes of Infants Undergoing Modified Blalock-Taussig Shunt Procedures in Oman: A retrospective study. Sultan Qaboos Univ Med J. 2021;21(3):457-63. [PubMed] DOI: 18295/squmj.8.2021.125
- Rasool F, Qureshi AZ, Khan A, Kazmi T, Shah SA. Role of BT shunt in tetralogy of Fallot. Cardiol Young. 2024;34(11):2445-2448. [PubMed] DOI: 1017/S1047951124025836
- Government of Western Australia Child and Adolescent Health Service. Blalock-Taussig Shunt (BT Shunt) Management Following Insertion. Neonatal Guideline. Australia: CAHS; 2013.
- Murni IK, Djer MM, Yanuarso PB, Putra ST, Advani N, Sukardi R. Outcome of pediatric cardiac surgery and predictors of major complication in a developing country. Ann Pediatr Cardiol. 2019;12(1):38-44. [PubMed] DOI: 4103/apc.APC_146_17
- Oster ME, Watkins S, Hill KD, Knight JH, Meyer RE. Academic Outcomes in Children With Congenital Heart Defects: A Population-Based Cohort Study. Circ Cardiovasc Qual Outcomes. 2017;10(2). [PubMed] DOI: 1161/CIRCOUTCOMES.116.003074
- Yuan SM, Jing H. Palliative procedures for congenital heart defects. Arch Cardiovasc Dis. 2009;102(6-7):549-57. [PubMed] DOI: 1016/j.acvd.2009.04.011
- Gerling C, Rukosujew A, Kehl HG, Tjan TDT, Hoffmeier A, Krasemann T. Do the age of patients with tetralogy of fallot at the time of surgery and the applied surgical technique influence the reoperation rate? a single-center experience. Herz. 2009;34(2):155-60. [PubMed] DOI: 1007/s00059-009-3169-x
- Savla JJ, Faerber JA, Huang YS V, Zaoutis T, Goldmuntz E, Mercer-Rosa L. 2-Year Outcomes After Complete or Staged Procedure for Tetralogy of Fallot in Neonates. J Am Coll Cardiol. 2019;74(12):1570-9. [PubMed] DOI: 1016/j.jacc.2019.05.057
- Taksaudom N, Thanayodcharoen S, Tantraworasin A. Factors Related to Early Postoperative Modified Blalock-Taussig Shunt Thrombosis and In-Hospital Mortality in Infants. J Med Assoc Thai. 2024;107(10). Full Text
- Tamisier D, Vouhé PR, Vernant F, Leca F, Massot C, Neveux JY. Modified Blalock-Taussig shunts: results in infants less than 3 months of age. Ann Thorac Surg. 1990;49(5):797-801. [PubMed] DOI: 1016/0003-4975(90)90026-3
- Awori MN, Awori JA, Langat G, Kipkoech K. The magnitude of systemic arterial oxygen saturation improvement achievable by a Blalock-Taussig-Thomas shunt: A systematic review. Afr Ann Thorac Cardiovasc Surg. 2024;16(1):1-4. Full Text
- Singh SP, Chauhan S, Choudhury M, Malik V, Talwar S, Hote MP, et al. Modified Blalock Taussig shunt: comparison between neonates, infants and older children. Ann Card Anaesth. 2014;17(3):191-7. [PubMed] DOI: 4103/0971-9784.135847