Bhavini Shah 1 , Brinda Badam 2 , Reshma Salim 3 , Pragya Pramanik 4
Authors affiliations:
Background & objective: Effective pain management during laparoscopic surgery under general anesthesia remains a challenge due to the lack of objective tools to assess nociception. The Perfusion Index (PI), derived from pulse oximetry, reflects peripheral perfusion and may serve as a non-invasive marker for analgesia adequacy by detecting sympathetic activation caused by nociceptive stimuli. We evaluated PI as a tool for assessing analgesia during laparoscopic surgeries by comparing its changes in response to painful stimuli and correlating it with hemodynamic parameters.
Methodology: This prospective observational study was conducted in the Department of Anesthesiology at Dr. D. Y. Patil Medical College, Hospital and Research Centre, Pimpri, Pune, Maharashtra, India, after the approval by Institutional Ethics Committee. In this prospective observational study, 90 ASA I/II patients, aged 18–60 years, undergoing elective laparoscopic surgery, were enrolled. PI, heart rate (HR), systolic blood pressure (SBP), diastolic blood pressure (DBP), and mean arterial pressure (MAP) were recorded at baseline, after induction, during pneumoperitoneum creation (P0), and after port insertions (P1–P3), every 1 min for first 10 min followed by every 5 min for 30 min. Fentanyl 1 µg/kg was administered after P1, and changes in PI and hemodynamic were analysed.
Results: PI decreased significantly during P1 (5.49 ± 0.05) compared to P0 (6.33 ± 0.06, P < 0.001), reflecting nociceptive response. After fentanyl administration, PI increased at P2 (5.72 ± 0.13) and P3 (6.01 ± 0.06, P < 0.001), indicating significant analgesia efficacy. HR spiked at P1 (88.18 ± 17.70 bpm) but stabilized post-fentanyl. No significant correlations were found between PI and SBP, DBP, or MAP.
Conclusion: PI effectively tracks nociceptive stimuli and analgesia efficacy during laparoscopic surgery, offering a real-time, non-invasive monitoring tool. Its dynamic changes align with painful events, supporting its utility in optimizing intraoperative pain management.
Abbreviations: DBP: diastolic blood pressure, HR: heart rate, MAP: mean arterial pressure, PI: Perfusion Index, SBP: systolic blood pressure,
Keywords: Analgesia; Fentanyl; General Anesthesia; Hemodynamic; Laparoscopic Surgery; Nociception; Perfusion Index; Peripheral Perfusion
Citation: Shah B, Badam B, Salim R, Pramanik P. Perfusion index as a valuable tool to assess analgesia during laparoscopic surgeries under general anesthesia. Anaesth. pain intensive care 2025;29(5):360-366. DOI: 10.35975/apic.v29i5.2871
Received: May 08, 2024; Revised: June 07, 2024; Accepted: June 09, 2025
The perioperative management of pain is an essential component of anesthetic practice, as it not only ensures that the patient is comfortable during the surgery but also serves to optimize surgical outcomes. Uncontrolled pain during surgery can lead to unpleasant physiological responses.1 These responses, collectively known as the ‘stress response to surgery’, can have harmful impact on organ function, leading to delayed recovery, and make patients susceptible to chronic pain syndromes. Issues of pain management associated with laparoscopic surgery are amplified through physiological changes induced by the surgeon for the use of minimally invasive techniques.2
Laparoscopic surgeries are preferred for their numerous benefits, such as smaller incisions, less postoperative pain, quicker recovery, and shorter hospital stays. However, these procedures also pose unique challenges. Although this technique is indispensable, it significantly affects the patient's physiology.3 The pneumoperitoneum increases intra-abdominal pressure, reduces venous return, compromises cardiac output, and stimulates nociceptive pathways that lead to an autonomic response characterized by vasoconstriction, tachycardia, and elevated blood pressure.4 Steep Trendelenburg or reverse Trendelenburg positions further complicate these effects, as they influence venous return, haemodynamic stability, and respiratory mechanics. Therefore, intraoperative adequate analgesia must be implemented to dampen nociceptive and physiological effects induced by pneumoperitoneum and surgical manipulation.
Pain, especially during a surgical process, cannot be accurately timed and evaluated.5 Pain in the operating room used to be estimated based on other indirect signs and symptoms, which include HR and blood pressure, among other effects on hemodynamics. These parameters are helpful but not specific as they can be influenced by several factors that are independent of nociception, such as depth of anesthesia, fluid shifts, and pharmacological interventions.6 The limitations of conventional methods underscore the necessity for a more reliable, objective, and non-invasive tool to assess analgesia during surgery.7 It is in this scenario that the perfusion index (PI), a parameter derived from pulse oximetry, comes to the fore. The PI reflects the ratio of pulsatile to non-pulsatile blood flow in the peripheral circulation, thereby serving as an indirect measure of peripheral perfusion.8
The autonomic nervous system actively controls peripheral perfusion and responds dynamically to any nociceptive stimulus. If there is pain or insufficient analgesia, sympathetic activation leads to peripheral vasoconstriction that reduces the blood flow of the periphery and PI.9 Conversely, when adequate analgesia is established, the nociceptive input is decreased, thus bringing about better peripheral perfusion and stabilization or elevation of PI. Different clinical situations have been used to test the use of the PI as a marker of nociception and adequacy of analgesia.10 In neonatal care, where traditional pain assessment techniques cannot be employed, promising findings have emerged regarding pain detection and guidance for analgesic interventions. In the case of regional anesthesia, the PI has been used to predict the success of nerve blocks through changes in peripheral perfusion. Additionally, research has shown that the PI can be used in predicting hypotension due to spinal anesthesia, indicating that it is sensitive to changes in autonomic and hemodynamic functions.11,12
Despite these observations, the use of the PI as a monitor for intraoperative analgesia during general anesthesia (GA) in laparoscopic surgery is an underexplored area. The autonomic and physiological changes that occur during laparoscopic surgery make it an ideal setting to evaluate the role of the PI in assessing nociception.13 Integration of PI monitoring into routine anesthetic practice may revolutionize intraoperative pain management. This could potentially allow anesthesiologists to tailor analgesic interventions more precisely by providing a continuous, non-invasive, and real-time measure of nociceptive states. This may reduce the overuse of opioids and their side effects, including respiratory depression, nausea, and postoperative ileus. This might further lead to improved intraoperative hemodynamic stability, diminished stress responses, and improved recovery in the postoperative period, thereby enhancing patient satisfaction and outcomes.14,15
The integration of PI into the anesthetic monitoring could help to shift away from the use of a "one-size-fits-all" approach to analgesia toward strategies that are dynamically adapted according to real-time patient needs. It may especially be helpful in patients at increased risk, like the elderly, patients with cardiovascular comorbidities, or patients undergoing lengthy and complicated surgeries.16
Hence, the current study was conducted to evaluate the changes in the PI in response to painful stimuli during laparoscopic surgery under GA.
This prospective observational study was conducted in the Department of Anesthesiology at Dr. D. Y. Patil Medical College, Hospital and Research Centre, Pimpri, Pune, Maharashtra, India, after approval by the Institutional Ethics Committee. The study was carried out over 18 months, with an additional 6 months allocated for data analysis and manuscript preparation.
Based on prior research by Atef et al. (2013), which compared PI with non-invasive hemodynamic parameters during insertion of I-gel, classic laryngeal mask airway and endotracheal tube, the sample size was estimated using “WIN PEPI” software (11.3). Considering the SD of A as 14.39 (HR) and a SD of B as 10.06 (DBP) with mean difference of 10.2 and 95% confidence interval, the sample would be 84. But for effective results we chose a total sample size of 90.
Patients of either gender, aged between 18 and 60 years, classified as American Society of Anesthesiologists (ASA) physical status I or II, and scheduled for elective laparoscopic surgeries under general anesthesia (GA) were included. Only those who were hemodynamically stable with normal routine investigations and who provided written informed consent were enrolled in the study.
Patients were excluded with anticipated difficult intubation or obese individuals, those with psychiatric, neurological, neuromuscular, or cardiovascular disease, or those with impaired hepatic or renal function. Patients or their guardians who declined participation were also excluded from the study.
A detailed preoperative assessment was conducted one day before surgery, and clinical parameters were reconfirmed on the day of the procedure. After securing a 20G IV cannula under sterile precautions, a balanced crystalloid infusion was started. Standard ASA monitors—including ECG, NIBP, pulse oximetry, and capnography—were applied, and baseline vital signs were recorded.
Patients were preoxygenated with 100% oxygen for 3 min. Premedication included glycopyrrolate 0.004 mg/kg, midazolam 0.02 mg/kg, and fentanyl 2 µg/kg IV. Induction was achieved using propofol 2 mg/kg, followed by succinylcholine 1.5 mg/kg IV for intubation. Anesthesia was maintained with oxygen, nitrous oxide, and sevoflurane. Pneumoperitoneum was created with CO₂ at 12–15 mmHg pressure, after which the first laparoscopic port was inserted. An additional dose of fentanyl 1 µg/kg was administered immediately after this. Two more ports were inserted under direct vision for optimal access.
Vital parameters including PI, HR, NIBP, and oxygen saturation were monitored at key points: pre-induction, post-induction, post-intubation, during pneumoperitoneum (P0), and after each port insertion (P1, P2, P3). Measurements were taken every min for the first 10 min and then at 5-min intervals up to 30 min to capture changes due to anesthetic and surgical stimuli.
2.1. Statistical Analysis
The analysis was performed using the data analysis tool in Microsoft Excel. Paired t-tests were used to compare the two specific time points. The null hypothesis was that the differences between paired values were 0, and a 95% CI was used to determine statistical significance, with P < 0.05 considered as significant. In addition, Pearson's correlation coefficients were calculated to ascertain the correlation between PI and other hemodynamic parameters. The latter included MAP and HR. This was a measure of the strength and direction of linear associations and was obtained using the correlation function in MS Excel. Coefficients were positive when they represented direct relationships and negative when representing inverse relationships. The more significant the coefficient values, the stronger the association. The combination of paired t-tests and correlation analysis enabled a detailed evaluation of the hemodynamic changes and their relationship to PI.
A total of 90 patients were enrolled for the study, out of which 45 were females and 45 were males; with a mean age of 35.9 ± 9.56 years. All participants enrolled in the study completed the study. All tests were conducted with a confidence level of 95%. PI values after administering fentanyl between P1 and P2 increased from 5.49 ± 0.05 to 5.72 ± 0.13 with a P < 0.001 (Table 1).
Similarly, these values saw a significant increase between P1 and P3, with P3 6.01 ± 0.06 (P < 0.001) (Table 1); whereas the changes in the HR, SBP, DBP, and MAP were not significant (Table 2).
A Pearson’s correlation (r) test was also run on the difference in PI, HR, and MAP values (Table 3, 4, 5, and 6). The increase in PI values between both these port insertions did not correlate with the changes in HR and MAP and was found to be statistically insignificant.
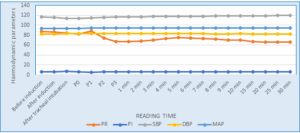
Figure 2: Comparison of hemodynamic parameters in PI, PR, SBP, DBP and MAP
Intraoperative pain management plays a vital role in modern anesthesia, directly impacting patient safety, comfort, and surgical outcomes. Effective pain control helps maintain physiological stability, as pain can activate the autonomic nervous system, leading to undesirable responses such as elevated HR and blood pressure.17 To assess intraoperative pain, anesthesiologists typically rely on a combination of physiological markers, autonomic indicators, and advanced monitoring tools. Each method has its limitations and advantages. Among the newer techniques, the PI has emerged as a promising, yet relatively underexplored, parameter for assessing intraoperative analgesia. It stands out for being non-invasive, simple to use, reliable, and cost-effective.
PI is influenced by sympathetic nervous system activity. When a patient experiences pain, sympathetic stimulation causes peripheral vasoconstriction, leading to a reduction in PI.18,19 In contrast, adequate analgesia leads to vasodilation and a subsequent increase in PI. Building on prior research by Surekha et al. (2022) this study was undertaken.18
In our study PI and other hemodynamic parameters were studied and compared from the baseline, after induction, during a nociceptive stimulus from the laparoscopic port insertion and after administration of analgesic dose of fentanyl. The data were systematically compiled, compared, and interpreted.
The PI was the highest during pneumoperitoneum creation [P0 (6.33 ± 0.06)] and decreased to 5.49 ± 0.05 at first port insertion (P1). After the administration of fentanyl 1µg IV, PI increased at P2 (5.72 ± 0.13) and P3 (6.01 ± 0.06). Hence, there is a positive association between PI and the adequacy of analgesia. These findings are consistent with previous research highlighting the PI (PI) as a valuable tool for monitoring analgesia during surgery. Surekha et al. (2022) demonstrated a significant rise in PI following fentanyl administration, accompanied by a reduction in HR—paralleling the trends observed in our study.18 Similarly, Kupeli et al. (2018) found that PI increased with the onset of epidural analgesia during labor and decreased as its effects diminished, which aligns with our observation of increased PI after fentanyl administration.19 Abdelnasser A, et al (2017) and Hasanin A, et al. (2017) also predicted successful supraclavicular brachial plexus block using pulse oximeter perfusion index.
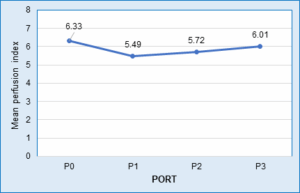
Figure 1 Mean perfusion index at port insertions.
In our study, pulse rate showed minor variations throughout the procedure, with a peak at first port insertion followed by a significant drop post-fentanyl administration, and weak correlation with perfusion index except for a moderate, statistically significant association at P2. These findings align with those of Saleh et al., who observed reduced HR and increased PI after analgesia, though without significant correlation.22 Similarly, Surekha et al. reported increased PI and decreased HR after fentanyl, supporting PI’s role in nociception monitoring.18 Vaghela et al. also found increased PI and reduced HR and MAP post-analgesia, consistent with our results, although correlations remained statistically weak.23
In our study, systolic, diastolic, and mean arterial pressures remained relatively stable throughout the laparoscopic procedure, with only minor fluctuations. Correlation analysis showed consistently weak and statistically non-significant relationships between perfusion index and SBP, DBP and MAP, at all measured time points. These results align with studies by Mehandale et al. (2016) and Saito et al. (2017), which also reported minimal or independent associations between PI and blood pressure variables.24 25 However, our findings contrast with Duggappa et al. (2017), who found a strong correlation between higher baseline PI and hypotension incidence post-spinal anesthesia, suggesting differing implications of PI across clinical settings and patient populations.26
From the results of our conducted study, it was found that PI is a reliable, non-invasive tool for assessing intraoperative nociception and analgesic adequacy during laparoscopic surgery. Its real-time responsiveness supports tailored analgesic dosing, potentially improving postoperative outcomes.
6. Data availability
The numerical data generated during this research is available with the authors, and can be available on a reasonable request.
7. Conflict of interest
All authors declare that there was no conflict of interest.
8. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
9. Acknowledgements
The authors express their sincere gratitude to the staff of the Departments of Anesthesiology as well as the surgical team, for their full cooperation.
10. Authors’ contribution
BS: Concept, conduct of the study; manuscript editing.
BB: Concept, conduct study, collecting data and manuscript editing.
RS, PP: contributed in collecting data, managing statistics
Authors affiliations:
- Bhavini Shah, Professor, Department of Anesthesia, Dr D Y Patil Medical College Hospital & Research Centre, Pune, Maharashtra, India; Email: drbhavinishah71@gmail.com
- Brinda Badam, Resident, Department of Anesthesia, Dr D Y Patil Medical College, Hospital & Research Centre, Pune, Maharashtra, India; Email: drbrindabadam@gmail.com; {ORCID:0009-0002-1341-0407}
- Reshma Salim, Resident, Department of Anesthesia, Dr D Y Patil Medical College, Hospital & Research Centre, Pune, Maharashtra, India; Email: reshmasalim1996@gmail.com
- Pragya Pramanik, Resident, Department of Anesthesia, Dr D Y Patil Medical College, Hospital & Research Centre, Pune, Maharashtra, India; Email: pramanik.pragya124@gmail.com
ABSTRACT
Background & objective: Effective pain management during laparoscopic surgery under general anesthesia remains a challenge due to the lack of objective tools to assess nociception. The Perfusion Index (PI), derived from pulse oximetry, reflects peripheral perfusion and may serve as a non-invasive marker for analgesia adequacy by detecting sympathetic activation caused by nociceptive stimuli. We evaluated PI as a tool for assessing analgesia during laparoscopic surgeries by comparing its changes in response to painful stimuli and correlating it with hemodynamic parameters.
Methodology: This prospective observational study was conducted in the Department of Anesthesiology at Dr. D. Y. Patil Medical College, Hospital and Research Centre, Pimpri, Pune, Maharashtra, India, after the approval by Institutional Ethics Committee. In this prospective observational study, 90 ASA I/II patients, aged 18–60 years, undergoing elective laparoscopic surgery, were enrolled. PI, heart rate (HR), systolic blood pressure (SBP), diastolic blood pressure (DBP), and mean arterial pressure (MAP) were recorded at baseline, after induction, during pneumoperitoneum creation (P0), and after port insertions (P1–P3), every 1 min for first 10 min followed by every 5 min for 30 min. Fentanyl 1 µg/kg was administered after P1, and changes in PI and hemodynamic were analysed.
Results: PI decreased significantly during P1 (5.49 ± 0.05) compared to P0 (6.33 ± 0.06, P < 0.001), reflecting nociceptive response. After fentanyl administration, PI increased at P2 (5.72 ± 0.13) and P3 (6.01 ± 0.06, P < 0.001), indicating significant analgesia efficacy. HR spiked at P1 (88.18 ± 17.70 bpm) but stabilized post-fentanyl. No significant correlations were found between PI and SBP, DBP, or MAP.
Conclusion: PI effectively tracks nociceptive stimuli and analgesia efficacy during laparoscopic surgery, offering a real-time, non-invasive monitoring tool. Its dynamic changes align with painful events, supporting its utility in optimizing intraoperative pain management.
Abbreviations: DBP: diastolic blood pressure, HR: heart rate, MAP: mean arterial pressure, PI: Perfusion Index, SBP: systolic blood pressure,
Keywords: Analgesia; Fentanyl; General Anesthesia; Hemodynamic; Laparoscopic Surgery; Nociception; Perfusion Index; Peripheral Perfusion
Citation: Shah B, Badam B, Salim R, Pramanik P. Perfusion index as a valuable tool to assess analgesia during laparoscopic surgeries under general anesthesia. Anaesth. pain intensive care 2025;29(5):360-366. DOI: 10.35975/apic.v29i5.2871
Received: May 08, 2024; Revised: June 07, 2024; Accepted: June 09, 2025
1. INTRODUCTION
The perioperative management of pain is an essential component of anesthetic practice, as it not only ensures that the patient is comfortable during the surgery but also serves to optimize surgical outcomes. Uncontrolled pain during surgery can lead to unpleasant physiological responses.1 These responses, collectively known as the ‘stress response to surgery’, can have harmful impact on organ function, leading to delayed recovery, and make patients susceptible to chronic pain syndromes. Issues of pain management associated with laparoscopic surgery are amplified through physiological changes induced by the surgeon for the use of minimally invasive techniques.2
Laparoscopic surgeries are preferred for their numerous benefits, such as smaller incisions, less postoperative pain, quicker recovery, and shorter hospital stays. However, these procedures also pose unique challenges. Although this technique is indispensable, it significantly affects the patient's physiology.3 The pneumoperitoneum increases intra-abdominal pressure, reduces venous return, compromises cardiac output, and stimulates nociceptive pathways that lead to an autonomic response characterized by vasoconstriction, tachycardia, and elevated blood pressure.4 Steep Trendelenburg or reverse Trendelenburg positions further complicate these effects, as they influence venous return, haemodynamic stability, and respiratory mechanics. Therefore, intraoperative adequate analgesia must be implemented to dampen nociceptive and physiological effects induced by pneumoperitoneum and surgical manipulation.
Pain, especially during a surgical process, cannot be accurately timed and evaluated.5 Pain in the operating room used to be estimated based on other indirect signs and symptoms, which include HR and blood pressure, among other effects on hemodynamics. These parameters are helpful but not specific as they can be influenced by several factors that are independent of nociception, such as depth of anesthesia, fluid shifts, and pharmacological interventions.6 The limitations of conventional methods underscore the necessity for a more reliable, objective, and non-invasive tool to assess analgesia during surgery.7 It is in this scenario that the perfusion index (PI), a parameter derived from pulse oximetry, comes to the fore. The PI reflects the ratio of pulsatile to non-pulsatile blood flow in the peripheral circulation, thereby serving as an indirect measure of peripheral perfusion.8
The autonomic nervous system actively controls peripheral perfusion and responds dynamically to any nociceptive stimulus. If there is pain or insufficient analgesia, sympathetic activation leads to peripheral vasoconstriction that reduces the blood flow of the periphery and PI.9 Conversely, when adequate analgesia is established, the nociceptive input is decreased, thus bringing about better peripheral perfusion and stabilization or elevation of PI. Different clinical situations have been used to test the use of the PI as a marker of nociception and adequacy of analgesia.10 In neonatal care, where traditional pain assessment techniques cannot be employed, promising findings have emerged regarding pain detection and guidance for analgesic interventions. In the case of regional anesthesia, the PI has been used to predict the success of nerve blocks through changes in peripheral perfusion. Additionally, research has shown that the PI can be used in predicting hypotension due to spinal anesthesia, indicating that it is sensitive to changes in autonomic and hemodynamic functions.11,12
Despite these observations, the use of the PI as a monitor for intraoperative analgesia during general anesthesia (GA) in laparoscopic surgery is an underexplored area. The autonomic and physiological changes that occur during laparoscopic surgery make it an ideal setting to evaluate the role of the PI in assessing nociception.13 Integration of PI monitoring into routine anesthetic practice may revolutionize intraoperative pain management. This could potentially allow anesthesiologists to tailor analgesic interventions more precisely by providing a continuous, non-invasive, and real-time measure of nociceptive states. This may reduce the overuse of opioids and their side effects, including respiratory depression, nausea, and postoperative ileus. This might further lead to improved intraoperative hemodynamic stability, diminished stress responses, and improved recovery in the postoperative period, thereby enhancing patient satisfaction and outcomes.14,15
The integration of PI into the anesthetic monitoring could help to shift away from the use of a "one-size-fits-all" approach to analgesia toward strategies that are dynamically adapted according to real-time patient needs. It may especially be helpful in patients at increased risk, like the elderly, patients with cardiovascular comorbidities, or patients undergoing lengthy and complicated surgeries.16
Hence, the current study was conducted to evaluate the changes in the PI in response to painful stimuli during laparoscopic surgery under GA.
2. METHODOLOGY
This prospective observational study was conducted in the Department of Anesthesiology at Dr. D. Y. Patil Medical College, Hospital and Research Centre, Pimpri, Pune, Maharashtra, India, after approval by the Institutional Ethics Committee. The study was carried out over 18 months, with an additional 6 months allocated for data analysis and manuscript preparation.
Based on prior research by Atef et al. (2013), which compared PI with non-invasive hemodynamic parameters during insertion of I-gel, classic laryngeal mask airway and endotracheal tube, the sample size was estimated using “WIN PEPI” software (11.3). Considering the SD of A as 14.39 (HR) and a SD of B as 10.06 (DBP) with mean difference of 10.2 and 95% confidence interval, the sample would be 84. But for effective results we chose a total sample size of 90.
Patients of either gender, aged between 18 and 60 years, classified as American Society of Anesthesiologists (ASA) physical status I or II, and scheduled for elective laparoscopic surgeries under general anesthesia (GA) were included. Only those who were hemodynamically stable with normal routine investigations and who provided written informed consent were enrolled in the study.
Patients were excluded with anticipated difficult intubation or obese individuals, those with psychiatric, neurological, neuromuscular, or cardiovascular disease, or those with impaired hepatic or renal function. Patients or their guardians who declined participation were also excluded from the study.
A detailed preoperative assessment was conducted one day before surgery, and clinical parameters were reconfirmed on the day of the procedure. After securing a 20G IV cannula under sterile precautions, a balanced crystalloid infusion was started. Standard ASA monitors—including ECG, NIBP, pulse oximetry, and capnography—were applied, and baseline vital signs were recorded.
Patients were preoxygenated with 100% oxygen for 3 min. Premedication included glycopyrrolate 0.004 mg/kg, midazolam 0.02 mg/kg, and fentanyl 2 µg/kg IV. Induction was achieved using propofol 2 mg/kg, followed by succinylcholine 1.5 mg/kg IV for intubation. Anesthesia was maintained with oxygen, nitrous oxide, and sevoflurane. Pneumoperitoneum was created with CO₂ at 12–15 mmHg pressure, after which the first laparoscopic port was inserted. An additional dose of fentanyl 1 µg/kg was administered immediately after this. Two more ports were inserted under direct vision for optimal access.
Vital parameters including PI, HR, NIBP, and oxygen saturation were monitored at key points: pre-induction, post-induction, post-intubation, during pneumoperitoneum (P0), and after each port insertion (P1, P2, P3). Measurements were taken every min for the first 10 min and then at 5-min intervals up to 30 min to capture changes due to anesthetic and surgical stimuli.
2.1. Statistical Analysis
The analysis was performed using the data analysis tool in Microsoft Excel. Paired t-tests were used to compare the two specific time points. The null hypothesis was that the differences between paired values were 0, and a 95% CI was used to determine statistical significance, with P < 0.05 considered as significant. In addition, Pearson's correlation coefficients were calculated to ascertain the correlation between PI and other hemodynamic parameters. The latter included MAP and HR. This was a measure of the strength and direction of linear associations and was obtained using the correlation function in MS Excel. Coefficients were positive when they represented direct relationships and negative when representing inverse relationships. The more significant the coefficient values, the stronger the association. The combination of paired t-tests and correlation analysis enabled a detailed evaluation of the hemodynamic changes and their relationship to PI.
3. RESULTS
A total of 90 patients were enrolled for the study, out of which 45 were females and 45 were males; with a mean age of 35.9 ± 9.56 years. All participants enrolled in the study completed the study. All tests were conducted with a confidence level of 95%. PI values after administering fentanyl between P1 and P2 increased from 5.49 ± 0.05 to 5.72 ± 0.13 with a P < 0.001 (Table 1).
| Table 1: Mean perfusion index at port insertions | ||
| Port | Perfusion index | p |
| P0 | 6.33 ± 0.06 | < 0.001 |
| P1 | 5.49 ± 0.05 | |
| P2 | 5.72 ± 0.13 | |
| P3 | 6.01 ± 0.06 | |
| P0: During Pneumoperitoneum Creation; P1: After Insertion of The First Laparoscopic Port; P2: After Insertion of The Second Port; P3: After Insertion of The Third Port, P – probability value. | ||
Similarly, these values saw a significant increase between P1 and P3, with P3 6.01 ± 0.06 (P < 0.001) (Table 1); whereas the changes in the HR, SBP, DBP, and MAP were not significant (Table 2).
| Table 2: Comparison of hemodynamic parameters in PI, PR,SBP, DBP and MAP | |||||
| Reading time | PI | PR | SBP | DBP | MAP |
| Before induction | 5.71 ± 0.06 | 87.28 ± 12.50 | 116.86 ± 11.18 | 82.24 ± 7.90 | 93.78 ± 6.95 |
| After induction | 6.47 ± 0.06 | 86.29 ± 12.18 | 115.34 ± 11.07 | 82.60 ± 7.67 | 93.51 ± 6.67 |
| After tracheal intubation | 6.62 ± 0.06 | 83.89 ± 12.11 | 113.94 ± 11.31 | 82.86 ± 7.51 | 93.22 ± 6.47 |
| P0 | 6.33 ± 0.06 | 82.22 ± 12.07 | 113.91 ± 11.96 | 83.32 ± 7.57 | 93.52 ± 6.66 |
| P1 | 5.49 ± 0.05 | 88.18 ± 17.70 | 114.99 ± 12.12 | 83.52 ± 7.25 | 94.01 ± 6.55 |
| P2 | 5.72 ± 0.13 | 74.04 ± 13.04 | 116.00 ± 12.18 | 83.61 ± 7.20 | 94.41 ± 6.52 |
| P3 | 6.01 ± 0.06 | 66.68 ± 10.87 | 117.10 ± 12.34 | 83.57 ± 7.07 | 94.74 ± 6.42 |
| 1 min | 6.01 ± 0.06 | 66.76 ± 9.88 | 117.02 ± 12.20 | 83.37 ± 6.95 | 94.59 ± 6.34 |
| 2 min | 5.92 ± 0.03 | 68.1 ± 10.82 | 117.02 ± 12.18 | 83.06 ± 6.91 | 94.38 ± 6.32 |
| 3 min | 5.95 ± 0.08 | 70.28 ±11.95 | 117.38 ± 12.08 | 82.76 ± 7.08 | 94.30 ± 6.37 |
| 4 min | 5.89 ± 0.23 | 72.9 ± 12.73 | 117.62 ± 12.41 | 82.71 ± 7.30 | 94.35 ± 6.58 |
| 5 min | 5.89 ± 0.23 | 75.40 ± 13.87 | 117.91 ± 12.37 | 82.90 ± 7.19 | 94.57 ± 6.59 |
| 6 min | 6.01 ± 0.04 | 73.98 ± 13.43 | 118.00 ± 12.05 | 82.89 ± 7.48 | 94.59 ± 6.60 |
| 7 min | 5.92 ± 0.04 | 72.65 ± 12.89 | 117.92 ± 12.01 | 82.77 ± 7.51 | 94.49 ± 6.55 |
| 8 min | 5.91 ± 0.23 | 71.6 ± 12.59 | 118.24 ± 11.98 | 82.62 ± 7.32 | 94.50 ± 6.38 |
| 9 min | 5.92 ± 0.23 | 70.3 ± 11.93 | 118.27 ± 12.03 | 82.56 ± 7.10 | 94.46 ± 6.30 |
| 10 min | 5.92 ± 0.23 | 69.73 ± 12.01 | 118.22 ± 12.03 | 82.67 ± 7.11 | 94.52 ± 6.38 |
| 15 min | 5.97 ± 0.04 | 67.21 ± 11.02 | 118.58 ± 11.82 | 82.31 ± 7.31 | 94.40 ± 6.30 |
| 20 min | 6.02 ± 0.05 | 66.12 ± 10.59 | 119.02 ± 12.00 | 82.21 ± 7.51 | 94.48 ± 6.41 |
| 25 min | 6.09 ± 0.05 | 66.03 ± 10. 68 | 119.42 ± 12.16 | 81.93 ± 7.53 | 94.43 ± 6.47 |
| 30 min | 6.20 ± 0.05 | 66.00 ± 10.72 | 119.71 ± 12.70 | 81.87 ± 7.53 | 94.48 ± 6.48 |
| p | < 0.001 | < 0.001 | < 0.001 | 0.992 | 0.931 |
| PR: Pulse rate; SBP: Systolic Blood Pressure; DBP: Diastolic Blood Pressure; MAP: Mean Arterial Pressure; P0: During Pneumoperitoneum Creation; P1: After Insertion of The First Laparoscopic Port; P2: After Insertion of The Second Port; P3: After Insertion of The Third Port, p – probability value. | |||||
A Pearson’s correlation (r) test was also run on the difference in PI, HR, and MAP values (Table 3, 4, 5, and 6). The increase in PI values between both these port insertions did not correlate with the changes in HR and MAP and was found to be statistically insignificant.
| Table 3: Correlation of hemodynamic parameters - perfusion index with PR | |||
| Parameters | r | P - value | |
| PR (/ min) vs PI | P0 | 0.095 | 0.375 |
| P1 | 0.116 | 0.276 | |
| P2 | 0.264 | 0.012* | |
| P3 | 0.140 | 0.188 | |
| SBP (mmHg) | P0 | -0.127 | 0.231 |
| P1 | -0.157 | 0.139 | |
| P2 | -0.131 | 0.219 | |
| P3 | -0.058 | 0.587 | |
| DBP (mmHg) | P0 | -0.006 | 0.954 |
| P1 | 0.061 | 0.568 | |
| P2 | -0.167 | 0.116 | |
| P3 | 0.007 | 0.951 | |
| MAP (mmHg) | P0 | -0.081 | 0.449 |
| P1 | -0.052 | 0.626 | |
| P2 | -0.204 | 0.053 | |
| P3 | -0.032 | 0.762 | |
| PR: Pulse rate; SBP: Systolic Blood Pressure; DBP: Diastolic Blood Pressure; MBP: mean Blood Pressure; P0: During Pneumoperitoneum Creation; P1: After Insertion of The First Laparoscopic Port; P2: After Insertion of The Second Port; P3: After Insertion of The Third Port; r: correlation coefficient, P < 0.05 considered as significant | |||

Figure 2: Comparison of hemodynamic parameters in PI, PR, SBP, DBP and MAP
4. DISCUSSION
Intraoperative pain management plays a vital role in modern anesthesia, directly impacting patient safety, comfort, and surgical outcomes. Effective pain control helps maintain physiological stability, as pain can activate the autonomic nervous system, leading to undesirable responses such as elevated HR and blood pressure.17 To assess intraoperative pain, anesthesiologists typically rely on a combination of physiological markers, autonomic indicators, and advanced monitoring tools. Each method has its limitations and advantages. Among the newer techniques, the PI has emerged as a promising, yet relatively underexplored, parameter for assessing intraoperative analgesia. It stands out for being non-invasive, simple to use, reliable, and cost-effective.
PI is influenced by sympathetic nervous system activity. When a patient experiences pain, sympathetic stimulation causes peripheral vasoconstriction, leading to a reduction in PI.18,19 In contrast, adequate analgesia leads to vasodilation and a subsequent increase in PI. Building on prior research by Surekha et al. (2022) this study was undertaken.18
In our study PI and other hemodynamic parameters were studied and compared from the baseline, after induction, during a nociceptive stimulus from the laparoscopic port insertion and after administration of analgesic dose of fentanyl. The data were systematically compiled, compared, and interpreted.
The PI was the highest during pneumoperitoneum creation [P0 (6.33 ± 0.06)] and decreased to 5.49 ± 0.05 at first port insertion (P1). After the administration of fentanyl 1µg IV, PI increased at P2 (5.72 ± 0.13) and P3 (6.01 ± 0.06). Hence, there is a positive association between PI and the adequacy of analgesia. These findings are consistent with previous research highlighting the PI (PI) as a valuable tool for monitoring analgesia during surgery. Surekha et al. (2022) demonstrated a significant rise in PI following fentanyl administration, accompanied by a reduction in HR—paralleling the trends observed in our study.18 Similarly, Kupeli et al. (2018) found that PI increased with the onset of epidural analgesia during labor and decreased as its effects diminished, which aligns with our observation of increased PI after fentanyl administration.19 Abdelnasser A, et al (2017) and Hasanin A, et al. (2017) also predicted successful supraclavicular brachial plexus block using pulse oximeter perfusion index.

Figure 1 Mean perfusion index at port insertions.
In our study, pulse rate showed minor variations throughout the procedure, with a peak at first port insertion followed by a significant drop post-fentanyl administration, and weak correlation with perfusion index except for a moderate, statistically significant association at P2. These findings align with those of Saleh et al., who observed reduced HR and increased PI after analgesia, though without significant correlation.22 Similarly, Surekha et al. reported increased PI and decreased HR after fentanyl, supporting PI’s role in nociception monitoring.18 Vaghela et al. also found increased PI and reduced HR and MAP post-analgesia, consistent with our results, although correlations remained statistically weak.23
In our study, systolic, diastolic, and mean arterial pressures remained relatively stable throughout the laparoscopic procedure, with only minor fluctuations. Correlation analysis showed consistently weak and statistically non-significant relationships between perfusion index and SBP, DBP and MAP, at all measured time points. These results align with studies by Mehandale et al. (2016) and Saito et al. (2017), which also reported minimal or independent associations between PI and blood pressure variables.24 25 However, our findings contrast with Duggappa et al. (2017), who found a strong correlation between higher baseline PI and hypotension incidence post-spinal anesthesia, suggesting differing implications of PI across clinical settings and patient populations.26
5. CONCLUSION
From the results of our conducted study, it was found that PI is a reliable, non-invasive tool for assessing intraoperative nociception and analgesic adequacy during laparoscopic surgery. Its real-time responsiveness supports tailored analgesic dosing, potentially improving postoperative outcomes.
6. Data availability
The numerical data generated during this research is available with the authors, and can be available on a reasonable request.
7. Conflict of interest
All authors declare that there was no conflict of interest.
8. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
9. Acknowledgements
The authors express their sincere gratitude to the staff of the Departments of Anesthesiology as well as the surgical team, for their full cooperation.
10. Authors’ contribution
BS: Concept, conduct of the study; manuscript editing.
BB: Concept, conduct study, collecting data and manuscript editing.
RS, PP: contributed in collecting data, managing statistics
11. REFERENCES
- Chen Q, Chen E, Qian X. A narrative review on perioperative pain management strategies in enhanced recovery pathways—The past, present and future. J Clin Med. 2021;10(12):2568. [PubMed] DOI: 3390/jcm10122568
- Bain CR, Myles PS, Corcoran T, Dieleman JM. Postoperative systemic inflammatory dysregulation and corticosteroids: A narrative review. Anaesthesia. 2023;78(3):356–70. [PubMed] DOI: 1111/anae.15896
- Srivastava A, Niranjan A. Secrets of safe laparoscopic surgery: Anaesthetic and surgical considerations. J Min Access Surg. 2010;6(3):91–4. [PubMed] DOI: 4103/0972-9941.72593
- Ponduru S, Nanda A, Pakhare V, Khare R, Pandey N, Kumar A. The effect of different pressures of pneumoperitoneum on the dimensions of internal jugular vein – A prospective double-blind, randomised study. Indian J Anaesth. 2022;66(8):631–7. [PubMed] DOI: 4103/ija.ija_350_22
- He H, Gruartmoner G, Ince Y, Doganci S, Bissett IP, Eltzschig HK. Effect of pneumoperitoneum and steep reverse-Trendelenburg position on mean systemic filling pressure, venous return, and microcirculation during esophagectomy. J Thorac Dis. 2018;10(6):3399–408. [PubMed] DOI: 21037/jtd.2018.05.169
- Wang H, Wang Q, He Q, Liu L, Xie Y, Zhang Y. Current perioperative nociception monitoring and potential directions. Asian J Surg. 2024;47(3):2558–65. [PubMed] DOI: 1016/j.asjsur.2024.03.090
- Bohringer C, Liu H. Is it always necessary to reverse the neuromuscular blockade at the end of surgery? J Biomed Res. 2019;33(4):217–20. PMC DOI: 7555/JBR.33.20180123
- Sun X, He H, Xu M, Long Y. Peripheral perfusion index of pulse oximetry in adult patients: A narrative review. Eur J Med Res. 2024;29:457. [PubMed] DOI: 1186/s40001-024-02048-3
- Kyle BN, McNeil DW. Autonomic arousal and experimentally induced pain: A critical review of the literature. Pain Res Manag. 2014;19(3):159–67. [PubMed] DOI: 1155/2014/536859
- Kim D, Lee C, Bae H, Choi S, Park J, Kim H. Comparison of the perfusion index as an index of noxious stimulation in monitored anesthesia care. BMC Anesthesiol. 2023;23:183. [PubMed] DOI: 1186/s12871-023-02116-x
- Perry S, Tan Z, Chen J, Wallace MS, Khan S, Engelhardt T. Neonatal pain: Perceptions and current practice. Crit Care Nurs Clin North Am. 2018;30(4):549–61. [PubMed] DOI: 1016/j.cnc.2018.07.013
- Kinoshita M, Hawkes CP, Ryan CA, Dempsey EM. Perfusion index in the very preterm infant. Acta Paediatr. 2013;102:e398–401. [PubMed] DOI: 1111/apa.12322
- Kim MK, Choi GJ, Oh KS, Lee SP, Kang H. Pain assessment using the analgesia nociception index (ANI). J Pers Med. 2023;13(10):1461. [PubMed] DOI: 3390/jpm13101461
- Oh SK, Won YJ, Lim BG. Surgical pleth index monitoring in perioperative pain management: Usefulness and limitations. Korean J Anesthesiol. 2024;77(1):31–45. [PubMed] DOI: 4097/kja.23158
- Benyamin R, Trescot AM, Datta S, Buenaventura R, Adlaka R, Sehgal N, et al. Opioid complications and side effects. Pain Physician. 2008;11(Suppl 2):S105–20. [PubMed]
- González-González MA, Conde SV, Latorre R, Thébault SC, Pratelli M, Spitzer NC, et al. Bioelectronic medicine: A multidisciplinary roadmap. Front Integr Neurosci. 2024;18:1321872. [PubMed] DOI: 3389/fnint.2024.1321872
- Atef HM, Fattah SA, Abd Gaffer ME, Al Rahman AA. Perfusion index versus non-invasive hemodynamic parameters. Indian J Anaesth. 2013;57(2):156–62. [PubMed] DOI: 4103/0019-5049.111843
- Surekha C, Eadara VS, Satish Kumar MN. Evaluation of perfusion index as an objective tool to assess analgesia. Indian J Anaesth. 2022;66(3):260–5. [PubMed] DOI: 4103/ija.ija_658_21
- Kupeli I, Kulhan NG. Can perfusion index be used as an objective tool for pain assessment in labor analgesia? Pak J Med Sci. 2018;34(5):1262–6. [PubMed] DOI: 12669/pjms.345.15157
- Abdelnasser A, Abdelhamid B, Elsonbaty A, Hasanin A, Rady A. Predicting successful supraclavicular brachial plexus block using pulse oximeter perfusion index. Br J Anaesth. 2017;119(2):276–80. [PubMed] DOI: 1093/bja/aex166
- Hasanin A, Mohamed SAR, El-adawy A. Evaluation of perfusion index as a tool for pain assessment in critically ill patients. J Clin Monit Comput. 2017;31(5):961–5. [PubMed] DOI: 1007/s10877-016-9936-3
- Saleh AN, Mostafa RH, Hamdy AN, Hafez AF. Pulse-oximetry derived perfusion index as a predictor of the efficacy of rescue analgesia after major abdominal surgeries. Open J Anesthesiol. 2020;14:101–7. DOI: 2174/2589645802014010101
- Vaghela AP, Patel US, Saiyad JH. Role of perfusion index as a tool for acute post-operative pain assessment in laparoscopic surgery. Nat J Med Res. 2021;11(4):131–3. Full Text
- Mehandale SG, Rajasekhar P. Perfusion index as a predictor of hypotension following propofol induction – A prospective observational study. Indian J Anaesth. 2017;61(12):990–5. [PubMed] DOI: 4103/ija.IJA_352_17
- Saito J, Kitayama M, Amanai E, Yamaguchi K, Akiyama K, Tanaka H. Impact of acute changes in perfusion index and blood pressure on the accuracy of non-invasive.
- continuous hemoglobin concentration measurements during induction of anesthesia. J Anesth. 2017;31(2):193–7. [PubMed] DOI: 10.1007/s00540-017-2306-6
- Duggappa DR, Lokesh M, Dixit A, Paul R, Raghavendra Rao RS, Prabha P. Perfusion index as a predictor of hypotension following spinal anaesthesia in lower segment caesarean section. Indian J Anaesth. 2017;61(8):649–54. [PubMed] DOI: 4103/ija.IJA_429_16