Karrar Talib Awad 1 , Hussein Nasser AL-Salman 2 , Mazin Abed Al-Ghani Najm 3
Authors affiliations:
Background & objective: Cyclin-dependent kinases (CDKs), particularly CDK2, are essential regulators of cell cycle progression and have recently been implicated in inflammation and immune modulation. Inhibitors like Rescovitine promote neutrophil apoptosis, reduce cytokine release, and show anti-inflammatory. However, its short half-life and off-target effects limit its clinical utility.
To synthesize and evaluate five novel thiourea derivatives of N-((4-cyano-1H-pyrazol-5-yl) carbamothioyl) benzamide that not only structurally resemble Rescovitine but also exhibit enhanced biological activity—particularly in inflammation control—through potent and selective inhibition of CDK2. This study aims to identify compounds with superior anti-inflammatory efficacy by targeting CDK2, a key regulator of neutrophil apoptosis and inflammatory resolution, while also offering improved pharmacokinetic properties.
Methodology: The derivatives VIa–VIe were synthesized through a multi-step reaction sequence, and their CDK2 inhibitory activity was evaluated in vitro using the MTT assay on human cell lines. The IC₅₀ values were determined for each compound.
Results: All compounds showed varying levels of CDK2 inhibition. VIa and VIe demonstrated the highest inhibitory activity with IC₅₀ values of 1.28 µM and 0.94 µM, respectively, comparable to Rescovitine (0.60 µM). VId and VId had moderate effects, while VIb was the least active. Fluorine—substitution in VIe enhanced activity, suggesting a key role of electronic substituents in CDK2 binding.
Conclusion: VIa and VIe are biologically promising anti-inflammatory candidates, and their ability to inhibit CDK2 could make them useful in treating inflammatory and proliferative diseases, where CDK2 plays a critical pathological role. Further preclinical studies could confirm their therapeutic value.
Abbreviations: CDK: Cyclin-dependent kinase, CDKI: CDK inhibitor, IL-10: Interleukin-10, MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide, TGF-β: Transforming growth factor beta, TLC: thin layer chromatography, C NMR: Computational nuclear magnetic resonance
Keywords: Anti-inflammatory agents; CDK2 inhibition; Inflammation; Kinase inhibitors; Rescovitine; Thiourea derivatives
Citation: Awad KT, AL-Salman HN, Najm MAA. Synthesis and CDK2 inhibition profiling of novel pyrazolyl-thiourea derivatives as potential anti-inflammatory agents. Anaesth. pain intensive care 2025;29(6):594-599; DOI: 10.35975/apic.v29i6.2855
Received: July 31, 2025; Revised: August 05, 2024; Accepted: August 05, 2025
Cyclin-dependent kinases (CDKs) are critical enzymes belonging to the serine/threonine kinase family, primarily known for their role in orchestrating the progression of the cell cycle through interactions with cyclin proteins. While their classical function involves cell proliferation, research has uncovered broader roles for certain CDKs—particularly CDK1 and CDK2—in transcription regulation, apoptosis, and immune cell function.1 Recent interest has emerged around the use of CDK inhibitors (CDKIs) as potential anti-inflammatory agents. Compounds such as Rrescovitine have demonstrated the capacity to accelerate neutrophil apoptosis and inhibit lymphocyte proliferation and cytokine release, thereby modulating inflammatory responses.2 Although neutrophils are terminally differentiated cells and not expected to rely on cell cycle machinery, these effects suggest a non-traditional role for CDKs in immune regulation. A key feature of inflammation resolution is the removal of neutrophils from affected tissues. Neutrophil apoptosis ensures that their toxic intracellular contents are safely contained and cleared via phagocytosis, typically by macrophages. This process not only eliminates inflammatory cells but also promotes a shift in macrophage activity toward a tissue-repairing, anti-inflammatory phenotype through the release of cytokines such as IL-10 and TGF-β.3-5 One mechanism by which CDK may promote resolution is through downregulation of survival proteins like Mcl-1, which CDK2 transcriptionally controls. Inhibiting CDK2 has been shown to suppress Mcl-1 expression and thus encourage neutrophil apoptosis, contributing to the clearance of inflammation.6,7 Similarly, CDK1 involvement in apoptosis signaling and transcription further supports the rationale for targeting these kinases in inflammatory conditions.
In conclusion, selective inhibition of CDK1 and CDK2 offers a promising therapeutic avenue for controlling inflammation and pain.8 By disrupting key survival pathways in immune cells, CDK may help resolve chronic inflammation and restore immune homeostasis. rescovitine has an anti-inflammatory effect likely originating from its ability to promote apoptosis in neutrophils and their elimination by macrophages, reduces degranulation of eosinophils and promotes their apoptosis, rescovitine displays analgesic properties, which could contribute to the management of CF-associated pain. Reducing acute and neuropathic pain across animal models and orally suitable drug that has already undergone preclinical pharmacological and toxicological studies, and extensive phase I and II clinical trials, in particular against cancer.9-12 Five derivatives of N-((4-cyano-1H-pyrazol-5-yl) carbamothioyl) benzamide to achieve a resemblance to rescovitine in terms of high selectivity and low toxicity, while addressing the limitation of its brief half-life (i.e., 2–5 hours) in humans.13 Consequently, the aforementioned facts lead us to synthesize thiourea derivatives and examine their inhibition activity against the CDK2 enzyme.
This study was reviewed and approved by the Institutional Ethics Committee of the Department of Pharmaceutical Chemistry, College of Pharmacy, University of Basrah, by the ethical standards outlined in the Declaration of Helsinki for research involving human participants. Approval Number: 669/5/3, Date of Approval: August 30, 2023.
The study aimed to identify compounds with superior anti-inflammatory efficacy by targeting CDK2, a key regulator of neutrophil apoptosis and inflammatory resolution, while also offering improved pharmacokinetic properties.
2.1. Chemical
Through a multi-step reaction pathway that involved the formation of acid chlorides, isothiocyanates, and derivatization to produce thiourea derivatives, new chemical compounds were created. The synthesis was meticulously tracked and refined. Melting point determination, thin layer chromatography (TLC), FT-IR, ¹H and ¹³C NMR spectroscopy, and mass spectrometry were among the analytical methods used to confirm the structures and purity of all intermediates and final products. Every compound will be assessed for its ability to inhibit the CDK enzyme.14-17
2.2. Biological
Using a human cell line, the MTT assay was used to assess the cytotoxic effects of Rescovitine and the synthetic compounds as Cyclin-Dependent Kinase inhibitors (CDKIs). For cell cycle progression and proliferation, these cell lines require CDK activity, especially CDK2. After being exposed to progressively higher concentrations of each compound for a full day, the cells were incubated with the MTT reagent to determine their viability. The tested compounds were then added to 96-well plates (three replicates) to achieve eight concentrations for each compound. The decrease in viable cells indicates how well the substances work to stop CDK-driven cell division. Dose-response curves were used to determine the IC₅₀ values, or concentrations needed to inhibit 50% of cell viability. This concentration is equal to the mean concentration. Reduced cell viability in this assay is an indirect sign that the tested compounds are effectively inhibiting CDK, since CDK2 is a crucial regulator of the G1/S transition. This method offers applicable proof that.18-20).
CDK2 Inhibition Activity: The compounds VIa- VIe were tested for their ability to inhibit CDK2, a key regulator of the cell cycle. The enzyme inhibition was measured by assessing the compound's ability to bind to the ATP-binding site of CDK2, thereby preventing its interaction with cyclins and halting the cell cycle progression among the compounds tested. VIa, VIe exhibited the most potent CDK2 inhibition, with an IC₅₀ value comparable to Rescovitine. Rescovitine, a known CDK2 inhibitor, serves as a standard in these studies and is widely recognized for its ability to arrest the cell cycle in cancer cells. Compounds VIc and VId also displayed moderate inhibition, while VIb showed the weakest inhibitory effect, according to Table 1 and Figure 3.
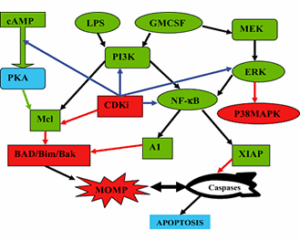
Figure 1: Potential CDK inhibitors affect survival signaling pathways involved in inflammation.
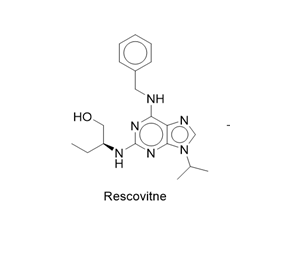
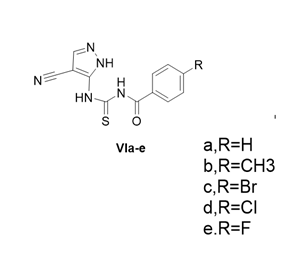
Figure 2: Conversion of Rescovitne intp Via-e
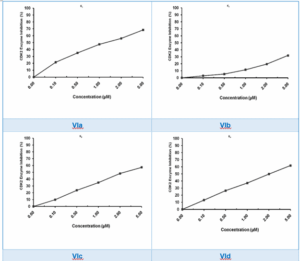
Figure 3: Cytotoxicity of thiourea derivatives against CDK2
This variation in activity suggests that the introduction of different substituents on the benzamide ring significantly influences CDK2 binding, with higher electronegativity groups like F (Fluorine) in VIe (21,22).
The study examined the impact of newly synthesized compounds on Cyclin-Dependent Kinase 2 (CDK2), a key regulator of inflammation and cell cycle progression. The compounds were tested alongside Rescovitine, a well-known CDK inhibitor, using in vitro enzyme inhibition. The results showed that the compounds showed varying degrees of CDK2 inhibition, with some showing comparable or enhanced potency. This suggests their potential as alternative or improved CDK2 inhibitors. Previous research has shown that CDK2 inhibition promotes apoptosis of neutrophils, reducing inflammation and chronic inflammatory responses. The compounds, designed to enhance these effects, were found to inhibit CDK2 activity and reduce cell viability in cancer cell lines. The study also highlighted the potential pleiotropic effects of these compounds in modulating inflammatory pathways and cell survival. These findings align with previous reports indicating that CDK inhibitors can reduce leukocyte recruitment, inflammatory cytokine production, and fibroblast proliferation, which are key factors in inflammatory diseases.
The Swiss ADEM (acute disseminated encephalomyelitis); helps chemists and researchers evaluate a compound's suitability as a drug candidate, filter out toxic or poorly absorbed compounds, and accelerate drug discovery. Recent studies have increasingly emphasized the optimization of absorption, distribution, metabolism, excretion, and toxicity (ADMET) properties, early in the design process. The most recently approved kinase inhibitors are primarily metabolized by CYP3A4, with implications for drug-drug interactions and personalized medicine approaches.
To use a chemical molecule as an oral medication, a drug must meet a few requirements: a molecular weight (MWT) of over 500, more than five H-bond donors (NHD), and ten H-bond acceptors (NHA). The number of rotatable bonds (NRB) is a key factor determining permeability properties. Lipinski's rule of five suggests that the molecular weight of a drug must be less than 500 g/mol to increase its permeability.
The study identified five organic compounds suitable for oral drugs, thanks to their water solubility and effective absorption in the gastrointestinal tract, without crossing the blood-brain barrier. Compared to Rescovitine, some of our compounds may offer improved selectivity or potency, warranting further in vivo studies to validate. We tested compounds VIa–VIe to see if they could stop CDK2, which is an important regulator of the cell cycle. The inhibition assay looked at how well the compounds could bind to the ATP-binding site of CDK2, which would stop it from interacting with cyclins and slowing down the cell cycle. VIe and VIa were the most effective at stopping CDK2 from working, with IC₅₀ values similar to those of Rescovitine - a well-known CDK2 inhibitor that is used as a reference standard. VIc and VId had moderate effects on inhibition, while VIb, which had a methyl group, had the weakest effects. These results show how important the electronic properties of substituents are for the benzamide moiety. For example, fluorine in VIe makes binding affinity and activity stronger.23-25
This study synthesized five new thiourea derivatives (VIa–VIe) and evaluated their inhibitory activity against Cyclin-Dependent Kinase 2 (CDK2), a key target in inflammation. The biological tests, which involved CDK2 inhibition, showed that compounds VIa and VIe were the most powerful, with IC₅₀ values close to those of the reference compound Rescovitine. The fluorine group that pulls electrons away from VIe makes it more active. This probably makes it easier for VIe to bind to the CDK2 ATP-binding site. These results support the idea that changing the structure of the thiourea scaffold can make it better at blocking CDK2, which could make it better at fighting inflammation. Some of the tested compounds work just as well or better than Rescovitine and may be able to get around problems like a short half-life and off-target effects. The results also support the idea that blocking CDK2 helps neutrophils die, and stops inflammation, which is in line with what other studies have found about the many anti-inflammatory effects of CDK inhibitors. In general, these compounds are good candidates for more research in the lab to see if they could be used as drugs to treat inflammatory and proliferative diseases by targeting CDK2.
6. Data availability
The numerical data generated during this research is available with the authors.
7. Conflict of interest
All authors declare that there was no conflict of interest.
8. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
9. Acknowledgment:
The authors express gratitude to the College of Pharmacy, University of Basrah, Iraq, for their support and resources.
10. Authors’ contribution
HN and MA: designed the research and supervised the project.
KT: carried out the synthesis and biological evaluation.
Authors affiliations:
- Karrar Talib Awad, College of Science, University of Thi Qar, Thi Qar. Iraq; Email: Karrar.talib.aa.30@gmail.com; {ORCID:0009-0006-5159-1623}
- Hussein Nasser AL-Salman, Basrah University, Basrah, Iraq.
- Mazin Abed Al ghani Najm, College of Science, University of Thi Qar, Thi Qar. Iraq.
ABSTRACT
Background & objective: Cyclin-dependent kinases (CDKs), particularly CDK2, are essential regulators of cell cycle progression and have recently been implicated in inflammation and immune modulation. Inhibitors like Rescovitine promote neutrophil apoptosis, reduce cytokine release, and show anti-inflammatory. However, its short half-life and off-target effects limit its clinical utility.
To synthesize and evaluate five novel thiourea derivatives of N-((4-cyano-1H-pyrazol-5-yl) carbamothioyl) benzamide that not only structurally resemble Rescovitine but also exhibit enhanced biological activity—particularly in inflammation control—through potent and selective inhibition of CDK2. This study aims to identify compounds with superior anti-inflammatory efficacy by targeting CDK2, a key regulator of neutrophil apoptosis and inflammatory resolution, while also offering improved pharmacokinetic properties.
Methodology: The derivatives VIa–VIe were synthesized through a multi-step reaction sequence, and their CDK2 inhibitory activity was evaluated in vitro using the MTT assay on human cell lines. The IC₅₀ values were determined for each compound.
Results: All compounds showed varying levels of CDK2 inhibition. VIa and VIe demonstrated the highest inhibitory activity with IC₅₀ values of 1.28 µM and 0.94 µM, respectively, comparable to Rescovitine (0.60 µM). VId and VId had moderate effects, while VIb was the least active. Fluorine—substitution in VIe enhanced activity, suggesting a key role of electronic substituents in CDK2 binding.
Conclusion: VIa and VIe are biologically promising anti-inflammatory candidates, and their ability to inhibit CDK2 could make them useful in treating inflammatory and proliferative diseases, where CDK2 plays a critical pathological role. Further preclinical studies could confirm their therapeutic value.
Abbreviations: CDK: Cyclin-dependent kinase, CDKI: CDK inhibitor, IL-10: Interleukin-10, MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide, TGF-β: Transforming growth factor beta, TLC: thin layer chromatography, C NMR: Computational nuclear magnetic resonance
Keywords: Anti-inflammatory agents; CDK2 inhibition; Inflammation; Kinase inhibitors; Rescovitine; Thiourea derivatives
Citation: Awad KT, AL-Salman HN, Najm MAA. Synthesis and CDK2 inhibition profiling of novel pyrazolyl-thiourea derivatives as potential anti-inflammatory agents. Anaesth. pain intensive care 2025;29(6):594-599; DOI: 10.35975/apic.v29i6.2855
Received: July 31, 2025; Revised: August 05, 2024; Accepted: August 05, 2025
1. INTRODUCTION
Cyclin-dependent kinases (CDKs) are critical enzymes belonging to the serine/threonine kinase family, primarily known for their role in orchestrating the progression of the cell cycle through interactions with cyclin proteins. While their classical function involves cell proliferation, research has uncovered broader roles for certain CDKs—particularly CDK1 and CDK2—in transcription regulation, apoptosis, and immune cell function.1 Recent interest has emerged around the use of CDK inhibitors (CDKIs) as potential anti-inflammatory agents. Compounds such as Rrescovitine have demonstrated the capacity to accelerate neutrophil apoptosis and inhibit lymphocyte proliferation and cytokine release, thereby modulating inflammatory responses.2 Although neutrophils are terminally differentiated cells and not expected to rely on cell cycle machinery, these effects suggest a non-traditional role for CDKs in immune regulation. A key feature of inflammation resolution is the removal of neutrophils from affected tissues. Neutrophil apoptosis ensures that their toxic intracellular contents are safely contained and cleared via phagocytosis, typically by macrophages. This process not only eliminates inflammatory cells but also promotes a shift in macrophage activity toward a tissue-repairing, anti-inflammatory phenotype through the release of cytokines such as IL-10 and TGF-β.3-5 One mechanism by which CDK may promote resolution is through downregulation of survival proteins like Mcl-1, which CDK2 transcriptionally controls. Inhibiting CDK2 has been shown to suppress Mcl-1 expression and thus encourage neutrophil apoptosis, contributing to the clearance of inflammation.6,7 Similarly, CDK1 involvement in apoptosis signaling and transcription further supports the rationale for targeting these kinases in inflammatory conditions.
In conclusion, selective inhibition of CDK1 and CDK2 offers a promising therapeutic avenue for controlling inflammation and pain.8 By disrupting key survival pathways in immune cells, CDK may help resolve chronic inflammation and restore immune homeostasis. rescovitine has an anti-inflammatory effect likely originating from its ability to promote apoptosis in neutrophils and their elimination by macrophages, reduces degranulation of eosinophils and promotes their apoptosis, rescovitine displays analgesic properties, which could contribute to the management of CF-associated pain. Reducing acute and neuropathic pain across animal models and orally suitable drug that has already undergone preclinical pharmacological and toxicological studies, and extensive phase I and II clinical trials, in particular against cancer.9-12 Five derivatives of N-((4-cyano-1H-pyrazol-5-yl) carbamothioyl) benzamide to achieve a resemblance to rescovitine in terms of high selectivity and low toxicity, while addressing the limitation of its brief half-life (i.e., 2–5 hours) in humans.13 Consequently, the aforementioned facts lead us to synthesize thiourea derivatives and examine their inhibition activity against the CDK2 enzyme.
2. METHODOLOGY
This study was reviewed and approved by the Institutional Ethics Committee of the Department of Pharmaceutical Chemistry, College of Pharmacy, University of Basrah, by the ethical standards outlined in the Declaration of Helsinki for research involving human participants. Approval Number: 669/5/3, Date of Approval: August 30, 2023.
The study aimed to identify compounds with superior anti-inflammatory efficacy by targeting CDK2, a key regulator of neutrophil apoptosis and inflammatory resolution, while also offering improved pharmacokinetic properties.
2.1. Chemical
Through a multi-step reaction pathway that involved the formation of acid chlorides, isothiocyanates, and derivatization to produce thiourea derivatives, new chemical compounds were created. The synthesis was meticulously tracked and refined. Melting point determination, thin layer chromatography (TLC), FT-IR, ¹H and ¹³C NMR spectroscopy, and mass spectrometry were among the analytical methods used to confirm the structures and purity of all intermediates and final products. Every compound will be assessed for its ability to inhibit the CDK enzyme.14-17
2.2. Biological
Using a human cell line, the MTT assay was used to assess the cytotoxic effects of Rescovitine and the synthetic compounds as Cyclin-Dependent Kinase inhibitors (CDKIs). For cell cycle progression and proliferation, these cell lines require CDK activity, especially CDK2. After being exposed to progressively higher concentrations of each compound for a full day, the cells were incubated with the MTT reagent to determine their viability. The tested compounds were then added to 96-well plates (three replicates) to achieve eight concentrations for each compound. The decrease in viable cells indicates how well the substances work to stop CDK-driven cell division. Dose-response curves were used to determine the IC₅₀ values, or concentrations needed to inhibit 50% of cell viability. This concentration is equal to the mean concentration. Reduced cell viability in this assay is an indirect sign that the tested compounds are effectively inhibiting CDK, since CDK2 is a crucial regulator of the G1/S transition. This method offers applicable proof that.18-20).
3. RESULTS
CDK2 Inhibition Activity: The compounds VIa- VIe were tested for their ability to inhibit CDK2, a key regulator of the cell cycle. The enzyme inhibition was measured by assessing the compound's ability to bind to the ATP-binding site of CDK2, thereby preventing its interaction with cyclins and halting the cell cycle progression among the compounds tested. VIa, VIe exhibited the most potent CDK2 inhibition, with an IC₅₀ value comparable to Rescovitine. Rescovitine, a known CDK2 inhibitor, serves as a standard in these studies and is widely recognized for its ability to arrest the cell cycle in cancer cells. Compounds VIc and VId also displayed moderate inhibition, while VIb showed the weakest inhibitory effect, according to Table 1 and Figure 3.
| Table 1: Cytotoxicity against CDK2 comparable to IC50 | |||
| I.D | CDK2 Inhibition (%) | IC50 |
SD IC50 |
| VIa | 68.42 | 1.28 | 0.23 |
| VIb | 31.94 | >5 | >5 weak |
| VIc | 57.18 | 2.39 | 0.93 |
| VId | 61.89 | 2.04 | 0.67 |
| VIe | 75.36 | 0.94 | 0.09 |
| Rescovitine | 84.52 | 0.60 | 0.04 |

Figure 1: Potential CDK inhibitors affect survival signaling pathways involved in inflammation.


Figure 2: Conversion of Rescovitne intp Via-e

Figure 3: Cytotoxicity of thiourea derivatives against CDK2
This variation in activity suggests that the introduction of different substituents on the benzamide ring significantly influences CDK2 binding, with higher electronegativity groups like F (Fluorine) in VIe (21,22).
4. DISCUSSION
The study examined the impact of newly synthesized compounds on Cyclin-Dependent Kinase 2 (CDK2), a key regulator of inflammation and cell cycle progression. The compounds were tested alongside Rescovitine, a well-known CDK inhibitor, using in vitro enzyme inhibition. The results showed that the compounds showed varying degrees of CDK2 inhibition, with some showing comparable or enhanced potency. This suggests their potential as alternative or improved CDK2 inhibitors. Previous research has shown that CDK2 inhibition promotes apoptosis of neutrophils, reducing inflammation and chronic inflammatory responses. The compounds, designed to enhance these effects, were found to inhibit CDK2 activity and reduce cell viability in cancer cell lines. The study also highlighted the potential pleiotropic effects of these compounds in modulating inflammatory pathways and cell survival. These findings align with previous reports indicating that CDK inhibitors can reduce leukocyte recruitment, inflammatory cytokine production, and fibroblast proliferation, which are key factors in inflammatory diseases.
The Swiss ADEM (acute disseminated encephalomyelitis); helps chemists and researchers evaluate a compound's suitability as a drug candidate, filter out toxic or poorly absorbed compounds, and accelerate drug discovery. Recent studies have increasingly emphasized the optimization of absorption, distribution, metabolism, excretion, and toxicity (ADMET) properties, early in the design process. The most recently approved kinase inhibitors are primarily metabolized by CYP3A4, with implications for drug-drug interactions and personalized medicine approaches.
To use a chemical molecule as an oral medication, a drug must meet a few requirements: a molecular weight (MWT) of over 500, more than five H-bond donors (NHD), and ten H-bond acceptors (NHA). The number of rotatable bonds (NRB) is a key factor determining permeability properties. Lipinski's rule of five suggests that the molecular weight of a drug must be less than 500 g/mol to increase its permeability.
The study identified five organic compounds suitable for oral drugs, thanks to their water solubility and effective absorption in the gastrointestinal tract, without crossing the blood-brain barrier. Compared to Rescovitine, some of our compounds may offer improved selectivity or potency, warranting further in vivo studies to validate. We tested compounds VIa–VIe to see if they could stop CDK2, which is an important regulator of the cell cycle. The inhibition assay looked at how well the compounds could bind to the ATP-binding site of CDK2, which would stop it from interacting with cyclins and slowing down the cell cycle. VIe and VIa were the most effective at stopping CDK2 from working, with IC₅₀ values similar to those of Rescovitine - a well-known CDK2 inhibitor that is used as a reference standard. VIc and VId had moderate effects on inhibition, while VIb, which had a methyl group, had the weakest effects. These results show how important the electronic properties of substituents are for the benzamide moiety. For example, fluorine in VIe makes binding affinity and activity stronger.23-25
5. CONCLUSION
This study synthesized five new thiourea derivatives (VIa–VIe) and evaluated their inhibitory activity against Cyclin-Dependent Kinase 2 (CDK2), a key target in inflammation. The biological tests, which involved CDK2 inhibition, showed that compounds VIa and VIe were the most powerful, with IC₅₀ values close to those of the reference compound Rescovitine. The fluorine group that pulls electrons away from VIe makes it more active. This probably makes it easier for VIe to bind to the CDK2 ATP-binding site. These results support the idea that changing the structure of the thiourea scaffold can make it better at blocking CDK2, which could make it better at fighting inflammation. Some of the tested compounds work just as well or better than Rescovitine and may be able to get around problems like a short half-life and off-target effects. The results also support the idea that blocking CDK2 helps neutrophils die, and stops inflammation, which is in line with what other studies have found about the many anti-inflammatory effects of CDK inhibitors. In general, these compounds are good candidates for more research in the lab to see if they could be used as drugs to treat inflammatory and proliferative diseases by targeting CDK2.
6. Data availability
The numerical data generated during this research is available with the authors.
7. Conflict of interest
All authors declare that there was no conflict of interest.
8. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
9. Acknowledgment:
The authors express gratitude to the College of Pharmacy, University of Basrah, Iraq, for their support and resources.
10. Authors’ contribution
HN and MA: designed the research and supervised the project.
KT: carried out the synthesis and biological evaluation.
11. REFERENCES
- Meijer L, Raymond E. Rescovitine and other purines as kinase inhibitors. From starfish oocytes to clinical trials. Acc Chem Res. 2003;36(6):417–25. PubMed DOI: 1021/ar0201198
- Borgne A, Golsteyn RM. The role of cyclin-dependent kinases in apoptosis. Prog Cell Cycle Res. 2003;5:453-9. PubMed
- Rossi AG, Sawatzky DA, Walker A, Ward C, Sheldrake TA, Riley NA, et al. Cyclin-dependent kinase inhibitors enhance the resolution of inflammation by promoting inflammatory cell apoptosis. Nat Med. 2006;12(9):1056–64. PubMed DOI: 1038/nm1468
- Obligado SH, Goldfarb DS, Panico F, Ravi R, Goligorsky MS. Cyclin-dependent kinase 2 activation in cisplatin-induced acute kidney injury. Am J Physiol Renal Physiol. 2008;294(4): F721–9.
- Fadok VA, Bratton DL, Guthrie L, Henson PM. Differential effects of apoptotic versus lysed cells on macrophage production of cytokines: role of proteases. J Immunol. 2001;166(11):6847–54. PubMed DOI: 4049/jimmunol.166.11.6847
- Faber AC, Chiles TC. Inhibition of cyclin-dependent kinase activity and cell-cycle progression by flavopiridol in mantle cell lymphoma cells. Cell Cycle. 2007;6(24):3030–8.
- Mangla B, Mittal P, Kumar P, Aggarwal G. Multifaceted role of erlotinib in various cancer: nanotechnology intervention, patent landscape, and advancements in clinical trials. Med Oncol. 2024;41(7). PubMed DOI: 1007/s12032-024-02414-5
- Valavanidis A, Vlachogianni T, Fiotakis K. 8-hydroxy-2′-deoxyguanosine (8-OHdG): a critical biomarker of oxidative stress and carcinogenesis. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev. 2009;27(2):120–39. PubMed DOI: 1080/10590500902885684
- Brenner DJ, Hall EJ. Computed Tomography — An Increasing Source of Radiation Exposure. N Engl J Med. 2007;357(22):2277–84. PubMed DOI: 1056/NEJMra072149
- Ewald PW. Symbionts and immunopathology in chronic diseases: insights from evolution. Clin Exp Immunol. 2010;159(1):27. PubMed DOI: 1111/j.1365-2249.2010.04127.x
- Malumbres M, Barbacid M. To cycle or not to cycle: A critical decision in cancer. Nat Rev Cancer. 2001;1(3):222–31. PubMed DOI: 1038/35106065
- Berndt N, Karim RM, Schönbrunn E. Advances of small molecule targeting of kinases. Curr Opin Chem Biol. 2017;39:126–32. PubMed DOI: 1016/j.cbpa.2017.06.015
- Schwartz PA, Murray BW. Protein kinase biochemistry and drug discovery. Bioorg Chem. 2011;39(5–6):192–210. PubMed DOI: 1016/j.bioorg.2011.07.004
- Cicenas J, Kalyan K, Sorokinas A, Stankunas E, Levy J, Meskinyte I, et al. Roscovitine in cancer and other diseases. Ann Transl Med. 2015;3(10):135. PubMed DOI: 3978/j.issn.2305-5839.2015.03.61
- Pang X, Zhao Y, Li G, Liu J, Yan A. A SAR and QSAR study on cyclin dependent kinase 4 inhibitors using machine learning methods. Digit Discov. 2023;2(4). DOI: 1039/D2DD00143H
- Alhussona AJ, Al-Musawi AH, Jalood HH, Hamzah SS. A study of ATPase gene variants of mitochondrial DNA in patients with Type 2 diabetes mellitus. Biomedicine. 2023;43(1 Suppl):225–9. Full Text
- Das RR, Kabra SK, Singh M. Treatment of Pseudomonas and Staphylococcus bronchopulmonary infection in patients with cystic fibrosis. Sci World J. 2013;2013:645653. PubMed DOI: 1155/2013/645653
- Guha M. Blockbuster dreams for Pfizer’s CDK inhibitor. Nat Biotechnol. 2013;31(3):187. PubMed DOI: 1038/nbt0313-187a
- Carruthers W, Coldham I. Modern Methods of Organic Synthesis. 4th ed. Cambridge: Cambridge University Press; 2004.
- Ali M, Mohasein RJ, Al-Salman HNK. Study of the biological activity of pyrazoline derivatives with fusidic acid and ZnO as an antibacterial and antioxidant. J Kufa Chem Sci. 2023;2(10):231. DOI: 36329/jkcm/2023/v2.i10.13888
- Ali ET, Al-Salman HNK, Rasool KH, Jabir MS, Ghimire TR, Shari FH, et al. 2-(Benzhydryl sulfinyl)-N-sec-butylacetamide isolated from fig augmented trastuzumab-triggered phagocytic killing of cancer cells through interface with Fcγ receptor. Nat Prod Res. 2023;37(21):3815–23. PubMed DOI: 1080/14786419.2023.2169861
- Neacsu A, Badiceanu C, Stoicescu C, Chihaia V. DFT Studies on Physicochemical Properties and Spectral Data of 2-Thiophene Carboxylic Acid Thiourea Derivatives. Chem Proc. 2024;16(1):27. DOI: 3390/ecsoc-28-20214
- Chen X, Li H, Tian L, Li Q, Luo J, Zhang Y. Analysis of the Physicochemical Properties of Acaricides Based on Lipinski’s Rule of Five. J Comput Biol. 2020;27(9):1397–406. PubMed DOI: 1089/cmb.2019.0323
- Byth KF, Culshaw JD, Green S, Oakes SE, Thomas AP. Imidazo[1,2-a]pyridines. Part 2: SAR and optimization of a potent and selective class of cyclin-dependent kinase inhibitors. Bioorg Med Chem Lett. 2004;14(9):2245–8. PubMed DOI: 1016/j.bmcl.2004.02.015
- Oyebamiji AK, Akintelu SA, Odelade KA, Adetuyi B, Akintayo ET, Akintayo CO, et al. Electron withdrawing group effect on biological activities of pyrimidine hybrids as potential anti-matrix metalloproteinase-7. Quim Nova. 2023 Oct;46(8):791–7. DOI: 21577/0100-4042.20230055