Sayed M. Abed 1 , Somaya Elsheikh 2 , Ahmed H. Bakeer 3 , Mohammed Magdy 4 , Mohammed Elsaed Abdelfattah 5
Authors affiliations:
Background & objective: The most frequent malignancy in women is breast cancer (BC), and besides chemotherapy and radiotherapy, partial or total surgical resection is required. Modified Radical Mastectomy (MRM) is a common surgical intervention for BC management. The procedure needs multimodal analgesia to manage the perioperative pain, which is often severe to intolerable.
This study aimed to compare the effect of ultrasound-guided erector spinae plane block (ESPB) and ultrasound-guided serratus anterior plane block (SAPB) on the incidence of Post Mastectomy Pain Syndrome (PMPS) in patients undergoing MRM under general anesthesia.
Methodology: We enrolled 120 female cancer patients, ASA II or III, and randomized into three groups; ESPB group to receive ESPB, SAPB group to receive SAPB, and a control group. The primary outcome was the incidence of PMPS during 24 weeks postoperatively. Secondary outcomes included acute postoperative pain, opioid consumption, quality of life, and the patient satisfaction.
Results: Out of 120 patients, 37 (30.8%) developed PMPS, with 13 (10%) reporting mild, 14 (11%) moderate, and 10 (8%) reporting severe pain. PMPS was developed by 47.5% (95%CI: 31.5%-63.9%) of individuals in the control group and 30% (95%CI: 16.6%-46.5%) and 15% (95%CI: 5.7%-29.3%) of participants in the SAPB and ESPB groups, respectively (P = 0.007).Compared to the control group, the ESPB group showed a significant reduction in PMPS rate. Meanwhile, there was no significant difference between the SAPB group and the control or ESPB groups. Severe PMPS was more common in the control and SAPB groups compared to the ESPB group. ESPB group provided significantly better analgesic efficacy in the acute phase, lower opioid consumption, lower pain scores, improved quality of life and higher patient satisfaction compared to the SAPB group and the control groups.
Conclusion: ESPB significantly reduced PMPS incidence and severity. SAPB improved acute pain but showed no significant PMPS reduction. ESPB is more effective for PMPS prevention.
Abbreviations: ESPB: serratus anterior plane block, LANSS: Leeds Assessment of Neuropathic Symptoms and Signs, PMPS: Post Mastectomy Pain Syndrome, SAPB: serratus anterior plane block,
Keywords: Block; Cancer; Incidence; Mastectomy; Pain; Serratus Anterior Muscle
Citation: Abed SM, Elsheikh S, Bakeer AH, Magdy M, Abdelfattah ME. Effect of erector spinae plane block versus serratus anterior block on post mastectomy pain syndrome: a randomized study. Anaesth. pain intensive care 2025;29(4):299-308. DOI: 10.35975/apic.v29i5.2853
Received: April 24, 2025; Revised: June 12, 2025; Accepted: June 25, 2025
Breast cancer (BC) remains the most prevalent malignancy among women and a leading cause of cancer-related mortality. Modified Radical Mastectomy (MRM) is the surgical intervention for BC treatment.1 A significant proportion of patients who have breast surgery have acute severe postoperative pain, ranging from 40% to 60%. More concerning is the fact that 20% to 50% of these individuals develop persistent pain lasting 6 to 12 months, a condition known as Post-Mastectomy Pain Syndrome (PMPS).2 PMPS is defined by the International Association for the Study of Pain (IASP) as chronic pain that lasts longer than three months in the anterior thorax, axilla, and/or medial upper arm following lumpectomy or mastectomy. Patients often describe the pain as a burning, stabbing, or pulling sensation.3-6
Managing PMPS presents significant challenges, similar to other neuropathic pain conditions.7 Current research focuses on perioperative interventions aimed at preventing the development of PMPS. Effective postoperative pain management is crucial in mitigating the risk of persistent pain following breast surgery, and numerous studies have explored various treatment modalities, with particular emphasis on regional anesthetic techniques.8
Regional anesthetic techniques, such as thoracic paravertebral block, pectoral nerve blocks, and retrolaminar block, have been assessed for their effectiveness in avoiding PMPS.9 the ability of erector spinae plane block (ESPB) and serratus anterior plane block (SAPB) to deliver powerful analgesia has attracted a lot of interest.
This study aimed to compare the effects of ultrasound-guided ESPB and SAPB on the incidence and severity of PMPS in MRM under general anesthesia.
This randomized, double-blind, prospective trial was conducted on consecutive 120 female participants aged 18-65 years who underwent MRM and were classified as class II or III by the American Society of Anesthesiologists at the National Cancer Institute (NCI), Cairo University Hospitals, Egypt, from October 2021 to May 2024. Approval was granted by the NCI, Cairo University ethics committee (AP2110-30111), and the trial was registered on www.clinicaltrials.gov (NCT05201963). Exclusion criteria encompassed patient refusals, pregnancy, ASA physical status IV, BMI less than 20 kg/m² or greater than 35 kg/m², known allergies or contraindications to medications (local anesthetics, opioids), preexisting uncontrolled respiratory, cardiac, hepatic or renal disease, pre-existing peripheral nerve issues, psychological disorders, chronic pain, coagulation abnormalities (INR > 1.5, platelet count < 80,000/µL), and local infections at the site of regional blocks.
The preliminary pilot study was conducted on the first 63 patients, with 21 patients in each group, to estimate the proportion of patients with PMPS. This evaluation was done because prior research has not explored the topic. The results revealed that 61% of patients in the control group, 42% of SAPB, and 28% of ESPB patients, respectively, developed PMPS. We calculated the sample size based on the smaller intergroup difference assuming superiority of either block compared to the control cases. Thus, if 61% of the subjects in the control group and 42% in the SAPB group would experience PMPS, the study would require a sample size of 36 for each group, to achieve a power of 80% and a level of significance of 5%, for declaring that the block is superior to the active control drug at a 15% margin of superiority. The sample was increased by 10% to compensate for dropouts to include 40 patients per group (a total of 120 patients). The sample size was calculated using Statulator: an online statistical calculator. http://statulator.com/SampleSize/ss2P.html
Preoperative management included obtaining consent, fasting according to standard protocols, performing a complete medical history and physical examination, and running standard laboratory tests. Intravenous midazolam (0.01-0.02 mg/kg) was administered for premedication after placing a 20-gauge cannula.
In the operating room, patients were randomly assigned to the ESPB group (n = 40), SAPB group (n = 40), or control group (n = 40) in a 1:1:1 ratio using sealed envelopes containing computer-generated randomization numbers. An investigator not involved in patient care provided these envelopes to the block procedure's anesthetist. Participants in the ESPB group received an ultrasound-guided erector spinae plane block on the same side as the affected area, while those in the SAPB group underwent an ipsilateral ultrasound-guided serratus anterior plane block. Both blocks were done under aseptic conditions after induction of anesthesia. The control group did not receive any block. The anesthesiologist responsible for data collection was blinded to the block administered. To maintain blinding in this study, several measures were implemented to minimize bias. All patients were under general anesthesia during the administration of the regional blocks, ensuring they were unaware of the group assignment. The anesthesiologist performing ESPB or SAPB, was not involved in postoperative data collection or patient follow-up. Instead, an anesthesiologist, blinded to the group allocation, conducted all postoperative assessments and outcome evaluations. Although no sham block or placebo injection was performed in the control group to avoid subjecting patients to unnecessary invasive procedures, all patients, regardless of group, received standardized surgical and anesthetic care to reduce the risk of detection bias.
Continuous monitoring of vital signs, including electrocardiogram (ECG), non-invasive blood pressure (NIBP), end-tidal carbon dioxide EtCO2) levels, and peripheral arterial oxygen saturation (SpO2), was performed during surgery. General anesthesia was induced using fentanyl 1 μg/kg and propofol 2 mg/kg IV, with rocuronium 0.5 mg/kg administered to facilitate tracheal intubation. Blocks were performed using the SonoSite M-Turbo portable ultrasound system's HFL38X linear multi-frequency 6-13 MHz probe.
2.1. ESPB
For the ESPB, the T5 transverse process tip was identified while the patient was in lateral decubitus position, and a parasagittal view was obtained by rotating the probe longitudinally to see erector spinae (ESM), rhomboids, and trapezius muscles posterior to T5 transverse process. A 22-G echogenic spinal needle was inserted in-plane in the cranial side of the probe until it reached the T5 transverse process, 0.5-1 mL of normal saline was injected to confirm the needle tip location by seeing the fluid spread between the anterior fascial plane of ESM and T5 process. Following this, a 30 mL bolus of 0.25% levobupivacaine was administered.
2.2. SAPB
In the SAPB group, the ultrasonic probe was placed on the midaxillary line at the level of the fifth rib to show serratus anterior and latissimus dorsi muscles. A 22-G spinal needle was inserted in-plane at a 45-degree angle towards the fifth rib, and 0.5-1 mL of normal saline was injected to open the interfascial plane between serratus anterior and latissimus dorsi (LD) muscles. The needle was monitored all the time at the US screen to avoid any trauma. A 30 mL injection of 0.25% levobupivacaine was then administered.
Anesthesia was maintained with sevoflurane in oxygen-enriched air at a concentration of 2-2.5% (FiO2 = 0.5), with rocuronium (0.1 mg/kg) administered every half hour. Multimodal analgesia included paracetamol 1 g, and ketorolac 30 mg, with increments of fentanyl 25 μg IV as rescue analgesia if heart rate or mean arterial blood pressure elevated more than 20% of the baseline. Ringer lactate solution was used for fluid management, and mechanical ventilation was adjusted to maintain EtCO2 at 30-35 mmHg. Post-surgery, neostigmine 0.05 mg/kg and atropine 0.02 mg/kg were administered to reverse residual neuromuscular blockade, followed by extubation.
2.3. Postoperative Management
Postoperative management involved meticulous monitoring of vital signs and pain intensity using the Numeric Rating Scale (NRS) upon arrival at the PACU. Rescue analgesia with intravenous boluses of morphine 3 mg was administered, if the NRS score ≥ 4, with a maximum dosage of 0.5 mg/kg/24 hours. Multimodal analgesia included intravenous paracetamol 1 g/8 hours and ketorolac 30 mg/8 hours, if the NRS score was ≥ 3. For the first 24 hours following surgery, NRS scores, and adverse events like vomiting, nausea, sedation, and difficulty breathing were observed. Ondansetron 0.1 mg/kg was administered for moderate to severe nausea and vomiting.10
The follow-up visits were scheduled at 2, 4, 8, 12, and 24 weeks following MRM. PMPS was defined as chronic pain in the anterior thorax, axilla, and/or medial upper arm, characterized by burning, stabbing, or pulling sensations that endure for over three months post-surgery.3-6 In addition, the Barthel Index of Activities of Daily Living (ADL) and the Flanagan Quality of Life Scale (QoLS) after 2, 4, 8, 12, and 24 weeks postmastectomy.11,12
Neuropathic pain was managed with pregabalin 75-600 mg and amitriptyline 10-25 mg. Analgesics, e.g., paracetamol, nonsteroidal anti-inflammatory drugs (NSAIDs), tramadol hydrochloride 100-400 mg daily, and oxycodone 20-60 mg daily were prescribed based on pain severity.
2.4. Outcomes
The primary outcome was set as the incidence of PMPS. Secondary outcomes included PMPS severity, total intraoperative fentanyl consumption, postoperative morphine consumption, time of first rescue analgesia, patient satisfaction, the Flanagan Quality of Life Scale (QoLS) and Barthel Index of Activities of Daily Living (ADL).
2.5. Statistical Analysis
We utilized SPSS version 27 (IBM§, Chicago, IL, USA) to carry out the statistical analysis. Shapiro-Wilks test and histograms were used to see if the data had a normal distribution. Quantitative parametric data, shown as mean ± SD, was evaluated using the ANOVA (F) test. For comparisons made after the fact, the Tukey test was employed. Median and interquartile range (IQR) data sets were utilized to assess quantitative non-parametric data using the Kruskal-Wallis test. When comparing two sets of data, the Mann-Whitney U test was employed. The qualitative variables, presented as frequencies and percentages, were evaluated using the Chi-square test. A P-value with two tails less than 0.05 was considered statistically significant.
Initially, 153 individuals were considered for inclusion; 7 patients declined to participate, and 26 did not meet all criteria. Thus, 120 participants were included and randomized into the three groups (Figure 1).
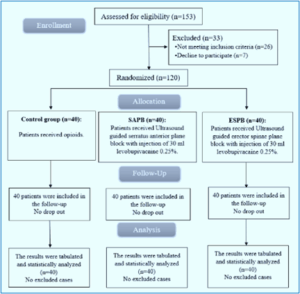
Figure 1: CONSORT flowchart of the enrolled patients.
There were no notable differences between the three groups regarding age, weight, height, BMI, ASA physical status, and surgical side (Table 1).
By the end of the follow-up, 37 participants (30.8%) developed PMPS, with a significant difference between groups (P = 0.007). The rate in the control group was 47.5% (95%CI: 31.5%-63.9%), compared to 30% (95%CI: 16.6%-46.5%) in the SAPB group and 15% (95%CI: 5.7%-29.3%) in the ESPB group. Therefore, compared to the control group, the ESPB group showed a statistically significant reduction in the rate of PMPS development. Meanwhile, there was no significant difference between the SAPB group and the control or ESPB groups. Severe PMPS was more common in the control and SAPB groups compared to the ESPB group (Table 2). Among patients who developed PMPS, ESPB was associated with less pain in the chest wall and axilla, while the major site of pain was in the arm. SAPB demonstrated the lowest frequency of axillary pain among the three groups (Table 2).
Table 3 shows that ESPB was more effective than the control and SAPB groups regarding the reduction of intra- and postoperative opioid consumption and duration of postoperative analgesia. The SAPB group also showed less intraoperative fentanyl consumption and longer analgesia duration than the control group.
Both ESPB and SAPB effectively managed early postoperative pain. However, ESPB demonstrated a statistically significant decrease in pain intensity as determined by the NRS throughout 24 hours at rest (Figure 2A) and during movement (Figure 2B), in comparison to the other two groups (P < 0.005).
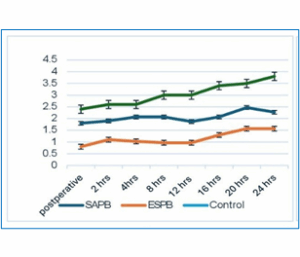
Figure 2: (A) Numeric Pain Rating Scale at rest, P-value < 0.005 significant at all times
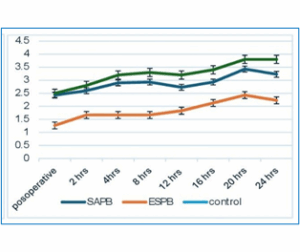
Figure 2: (B) Numeric Pain Rating Scale at movement, P < 0.005 significant at all times
Quality of life (QoL) scores showed no significant intergroup differences up to 12 weeks. Then, ESPB demonstrated significantly higher scores at 24 weeks compared to the control group (Table 4).
Baseline activity levels were comparable between groups (P = 0.881). However, starting from the 2nd week, ESPB showed significantly higher ADL scores than the control group (Table 4). Participants who received ESPB demonstrated better overall satisfaction, which was statistically significant; SAPB offered higher satisfaction rates than the control group, yet it was statistically insignificant (Figure 2C).
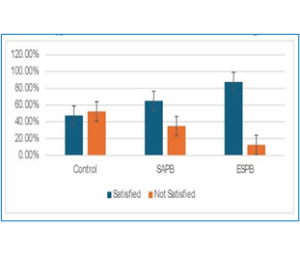
Figure 2: (C) Patient satisfaction in Control, SAPB and ESPB groups
MRM encompasses the breast and axillary area, necessitating extensive surgical incisions, a primary contributor to stress and inflammatory responses. Inadequately managed immediate postoperative pain significantly contributes to persistent pain, stress, and inflammatory reactions.13 The pathogenesis of PMPS remains incompletely elucidated; nonetheless, the predominant theory is intercostobrachial nerve neuralgia supplying cutaneous sensation to the axilla and inner arm. Dissection of axillary lymph nodes or manipulation due to retraction during mastectomy may result in inflammation and nerve damage,14 inducing ectopic neuronal activity at the lesion site and dorsal root ganglion, leading to heightened sensitivity to chemical or mechanical stimuli and an amplified experience of pain.6 Besides, the sensitivity of neuronal cells in the brain may be a contributing factor.15
Regrettably, all these potential causes are mostly unmodifiable within the context of MRM. Conversely, intense acute postoperative pain leads to a higher occurrence of persistent postoperative pain;16 and is a clearly changeable risk factor. Numerous studies indicate that moderate to severe acute postoperative pain significantly increases the incidence of PMPS (odds ratio 1.34–2.8).17,18
Thus, preventative and perioperative analgesia, along with pharmacological medications, may mitigate the onset or progression of PMPS. Because local and regional blocks better control intra- and postoperative pain, the anesthetic literature supports their use in preventing PMPS. Preventive regional analgesia may mitigate the hazards of central sensitization by obstructing the transmission of harmful stimuli.
The results of the current study support the notion that adequate management of acute postoperative pain is an effective measure to reduce the incidence of PMPS. The study revealed that the ESPB group showed a statistically significant reduction in the rate of PMPS development compared to the control group (15% vs. 47.5%). Meanwhile, there was no significant difference between SAPB and the control or ESPB groups in the rate of PMPS. Most cases of severe pain were observed in the control group. This reduction of PMPS rate and severity was associated with better control of acute postoperative pain. ESPB was more effective than the control and SAPB groups regarding the reduction of intra- and postoperative opioid consumption, and postoperative pain intensity, in addition to the longer duration of postoperative analgesia.
These findings are consistent with other recent studies. A recent randomized controlled trial indicated that the ESPB offers enhanced analgesic benefits compared to SAPB in patients having MRM, resulting in less morphine consumption and lower postoperative cortisol levels.19 A further study demonstrated that ESPB is equally effective as TPV block for early postoperative analgesia, diminished stress response, and lowered Leeds Assessment of Neuropathic Symptoms and Signs (LANSS) score at the 6-month follow-up.20
The mechanism of action of the ESPB remains incompletely elucidated, with numerous hypotheses posited throughout the years. It is considered to be multifactorial. Research indicates ESPB at the T5 transverse process effectively inhibits the ipsilateral T3-T9 spinal innervation zone.21 ESPB was thought to work through LA diffusion into the paravertebral space and subsequently into the epidural region to block spinal nerves. However, cadaveric studies have challenged this notion, suggesting that the dye does not rapidly spread into the paravertebral space. 22 The physical distribution of LA related to the ESPB influences the dorsal rami, which innervates the spine and paravertebral tissues.23 Fusco et al. (2024) suggested that the fascia may serve as a target, as prior investigations have shown it to be abundant in free nerve terminals.24 Furthermore, they postulated that LA facilitates muscular relaxation, supported by an initial demonstration using elastosonography.
Although it was shown that SAPB was less effective than ESPB, it still showed some protection against PMPS than the control group. It has shown significantly better analgesic efficacy in the acute phase compared to the control group and superior, yet non-significant, value in reducing PMPS (30% vs. 47.5%). SAPB targets the thoracodorsal nerve, long thoracic nerve, intercostobrachial nerve, and the lateral cutaneous branches of the intercostal nerves (T3-T9). These nerves are located between the midaxillary border and the SA and LD muscles.25 The method by which SAPB effectively prevents PMPS is its capacity to diminish acute postoperative pain and alleviate central sensitization. Research indicates that early intervention with SAPB can interrupt this sequence, thereby diminishing the probability of PMPS.26 Furthermore, the interruption of nociceptive signals, less peripheral and central neuronal sensitization, and attenuated inflammatory responses may facilitate the alleviation of persistent postoperative pain.27
Many studies tested the efficacy of multiple regional block techniques for the prevention of PMPS. Mendonça et al. (2023) investigated the efficacy of pectoserratus plane block (PSPB) for reducing the incidence of PMPS after breast cancer surgery.28 PSPB was associated with a reduction of chronic pain from 65% to 22% and the risk of neuropathic pain from 54% to 18%. In another study, PSPB achieved a comparable effect with ESPB on postoperative opioid consumption and acute and chronic pain scores.29 The pectoral nerve-2 (PECS 2) block was compared to SAPB in a randomized trial. PECS2 block reduced the rate of moderate or severe chronic pain from 33% with SAPB to 10%. The authors observed a superior reduction of postoperative morphine consumption in the PECS block, but acute pain scores were similar.13 In a randomized, double-blinded trial ultrasound-guided multilevel PVB was compared to placebo. PVB reduced the incidence of chronic pain from 51.2% to 34.5% after 3 months and from 37.2% to 22.1% after 6 months.30 A systematic review and network meta-analysis evaluated different interventions for PMPS prevention.31 They found that nerve blocks demonstrated improvements in functional outcomes and quality of life (OR: 0.34; 95% CrI: 0.24–0.46).
Notably, observing muscle hematoma during surgery following SAPB in the current study was a significant complication. This can be attributed to the proximity of the SAPB injection site to the surgical field in MRM procedures, where the resulting muscle edema or hematoma could potentially interfere with the surgical process.
Yet, the research on PMPS is sparse, and there is no consensus on a definitive gold standard of practice. Nonetheless, several high-volume facilities have developed their management practices in recent years. Yang et al. (2021) have introduced an algorithm for the systematic selection of suitable medicines in the management of PMPS.32 Another group has presented an algorithm emphasizing the establishment of an appropriate multidisciplinary treatment team for the management of PMPS. They demonstrated that local blocks can be utilized for both diagnostic and symptom treatment.33 Experience at the National Cancer Institute (NCI) of Cairo University can offer valuable insights into the practical implications of regional techniques for preventing PMPS.
This study's generalizability may be limited due to its single-center design, and the relatively small sample size. Furthermore, the length and dosage of adjuvant therapy (radiation or chemotherapy) given to patients were outside the purview of this investigation.
Erector spinae plane block (ESPB) significantly reduced the incidence and severity of post-mastectomy pain syndrome (PMPS), highlighting its effectiveness for long-term pain prevention. Although serratus anterior plane block (SAPB) improved acute postoperative pain, it did not significantly reduce PMPS. ESPB is a more reliable technique for PMPS prevention.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Acknowledgement
We gratefully thank Faculty of Medicine, National Cancer Institute, Cairo University, Cairo for facilitating this research.
9. Conflict of interest
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
SMA: Study concept and design, data collection, statistical analysis, manuscript revision.
SE: Study concept and design, manuscript editing and critical revision.
AHB: Data analysis and interpretation, manuscript review.
MEA: Drafting of the manuscript, critical revision for intellectual content.
MMO: Literature review, data collection assistance, manuscript proofreading.
Authors affiliations:
- Sayed M. Abed, Assistant Professor, Department of Anesthesia, Surgical Critical Care & Pain Management, National Cancer Institute, Cairo University, Cairo, Egypt; Email: sayed.abed@nci.cu.edu.eg; {ORCID:0000-0003-2362-5508}
- Somaya Elsheikh, Professor, Department of Anesthesia, Surgical Critical Care & Pain Management, National Cancer Institute, Cairo University, Cairo, Egypt; Email: [somaya_elshaikh@outlook.com
- Ahmed H. Bakeer, Professor, Department of Anesthesia, Surgical Critical Care & Pain Management, National Cancer Institute, Cairo University, Cairo, Egypt; Email: ahmed_bakir77@yahoo.com
- Mohammed Magdy, Lecturer, Department of Anesthesia, Surgical Critical Care & Pain Management, National Cancer Institute, Cairo University, Cairo, Egypt; Email: mohammed.magdy@nci.cu.edu.eg
- Mohammed Elsaed Abdelfattah, Lecturer, Department of Anesthesia, Surgical Critical Care & Pain Management, National Cancer Institute, Cairo University, Cairo, Egypt; Email: mohamed-elsaid@cu.edu.eg
ABSTRACT
Background & objective: The most frequent malignancy in women is breast cancer (BC), and besides chemotherapy and radiotherapy, partial or total surgical resection is required. Modified Radical Mastectomy (MRM) is a common surgical intervention for BC management. The procedure needs multimodal analgesia to manage the perioperative pain, which is often severe to intolerable.
This study aimed to compare the effect of ultrasound-guided erector spinae plane block (ESPB) and ultrasound-guided serratus anterior plane block (SAPB) on the incidence of Post Mastectomy Pain Syndrome (PMPS) in patients undergoing MRM under general anesthesia.
Methodology: We enrolled 120 female cancer patients, ASA II or III, and randomized into three groups; ESPB group to receive ESPB, SAPB group to receive SAPB, and a control group. The primary outcome was the incidence of PMPS during 24 weeks postoperatively. Secondary outcomes included acute postoperative pain, opioid consumption, quality of life, and the patient satisfaction.
Results: Out of 120 patients, 37 (30.8%) developed PMPS, with 13 (10%) reporting mild, 14 (11%) moderate, and 10 (8%) reporting severe pain. PMPS was developed by 47.5% (95%CI: 31.5%-63.9%) of individuals in the control group and 30% (95%CI: 16.6%-46.5%) and 15% (95%CI: 5.7%-29.3%) of participants in the SAPB and ESPB groups, respectively (P = 0.007).Compared to the control group, the ESPB group showed a significant reduction in PMPS rate. Meanwhile, there was no significant difference between the SAPB group and the control or ESPB groups. Severe PMPS was more common in the control and SAPB groups compared to the ESPB group. ESPB group provided significantly better analgesic efficacy in the acute phase, lower opioid consumption, lower pain scores, improved quality of life and higher patient satisfaction compared to the SAPB group and the control groups.
Conclusion: ESPB significantly reduced PMPS incidence and severity. SAPB improved acute pain but showed no significant PMPS reduction. ESPB is more effective for PMPS prevention.
Abbreviations: ESPB: serratus anterior plane block, LANSS: Leeds Assessment of Neuropathic Symptoms and Signs, PMPS: Post Mastectomy Pain Syndrome, SAPB: serratus anterior plane block,
Keywords: Block; Cancer; Incidence; Mastectomy; Pain; Serratus Anterior Muscle
Citation: Abed SM, Elsheikh S, Bakeer AH, Magdy M, Abdelfattah ME. Effect of erector spinae plane block versus serratus anterior block on post mastectomy pain syndrome: a randomized study. Anaesth. pain intensive care 2025;29(4):299-308. DOI: 10.35975/apic.v29i5.2853
Received: April 24, 2025; Revised: June 12, 2025; Accepted: June 25, 2025
1. INTRODUCTION
Breast cancer (BC) remains the most prevalent malignancy among women and a leading cause of cancer-related mortality. Modified Radical Mastectomy (MRM) is the surgical intervention for BC treatment.1 A significant proportion of patients who have breast surgery have acute severe postoperative pain, ranging from 40% to 60%. More concerning is the fact that 20% to 50% of these individuals develop persistent pain lasting 6 to 12 months, a condition known as Post-Mastectomy Pain Syndrome (PMPS).2 PMPS is defined by the International Association for the Study of Pain (IASP) as chronic pain that lasts longer than three months in the anterior thorax, axilla, and/or medial upper arm following lumpectomy or mastectomy. Patients often describe the pain as a burning, stabbing, or pulling sensation.3-6
Managing PMPS presents significant challenges, similar to other neuropathic pain conditions.7 Current research focuses on perioperative interventions aimed at preventing the development of PMPS. Effective postoperative pain management is crucial in mitigating the risk of persistent pain following breast surgery, and numerous studies have explored various treatment modalities, with particular emphasis on regional anesthetic techniques.8
Regional anesthetic techniques, such as thoracic paravertebral block, pectoral nerve blocks, and retrolaminar block, have been assessed for their effectiveness in avoiding PMPS.9 the ability of erector spinae plane block (ESPB) and serratus anterior plane block (SAPB) to deliver powerful analgesia has attracted a lot of interest.
This study aimed to compare the effects of ultrasound-guided ESPB and SAPB on the incidence and severity of PMPS in MRM under general anesthesia.
2. METHODOLOGY
This randomized, double-blind, prospective trial was conducted on consecutive 120 female participants aged 18-65 years who underwent MRM and were classified as class II or III by the American Society of Anesthesiologists at the National Cancer Institute (NCI), Cairo University Hospitals, Egypt, from October 2021 to May 2024. Approval was granted by the NCI, Cairo University ethics committee (AP2110-30111), and the trial was registered on www.clinicaltrials.gov (NCT05201963). Exclusion criteria encompassed patient refusals, pregnancy, ASA physical status IV, BMI less than 20 kg/m² or greater than 35 kg/m², known allergies or contraindications to medications (local anesthetics, opioids), preexisting uncontrolled respiratory, cardiac, hepatic or renal disease, pre-existing peripheral nerve issues, psychological disorders, chronic pain, coagulation abnormalities (INR > 1.5, platelet count < 80,000/µL), and local infections at the site of regional blocks.
The preliminary pilot study was conducted on the first 63 patients, with 21 patients in each group, to estimate the proportion of patients with PMPS. This evaluation was done because prior research has not explored the topic. The results revealed that 61% of patients in the control group, 42% of SAPB, and 28% of ESPB patients, respectively, developed PMPS. We calculated the sample size based on the smaller intergroup difference assuming superiority of either block compared to the control cases. Thus, if 61% of the subjects in the control group and 42% in the SAPB group would experience PMPS, the study would require a sample size of 36 for each group, to achieve a power of 80% and a level of significance of 5%, for declaring that the block is superior to the active control drug at a 15% margin of superiority. The sample was increased by 10% to compensate for dropouts to include 40 patients per group (a total of 120 patients). The sample size was calculated using Statulator: an online statistical calculator. http://statulator.com/SampleSize/ss2P.html
Preoperative management included obtaining consent, fasting according to standard protocols, performing a complete medical history and physical examination, and running standard laboratory tests. Intravenous midazolam (0.01-0.02 mg/kg) was administered for premedication after placing a 20-gauge cannula.
In the operating room, patients were randomly assigned to the ESPB group (n = 40), SAPB group (n = 40), or control group (n = 40) in a 1:1:1 ratio using sealed envelopes containing computer-generated randomization numbers. An investigator not involved in patient care provided these envelopes to the block procedure's anesthetist. Participants in the ESPB group received an ultrasound-guided erector spinae plane block on the same side as the affected area, while those in the SAPB group underwent an ipsilateral ultrasound-guided serratus anterior plane block. Both blocks were done under aseptic conditions after induction of anesthesia. The control group did not receive any block. The anesthesiologist responsible for data collection was blinded to the block administered. To maintain blinding in this study, several measures were implemented to minimize bias. All patients were under general anesthesia during the administration of the regional blocks, ensuring they were unaware of the group assignment. The anesthesiologist performing ESPB or SAPB, was not involved in postoperative data collection or patient follow-up. Instead, an anesthesiologist, blinded to the group allocation, conducted all postoperative assessments and outcome evaluations. Although no sham block or placebo injection was performed in the control group to avoid subjecting patients to unnecessary invasive procedures, all patients, regardless of group, received standardized surgical and anesthetic care to reduce the risk of detection bias.
Continuous monitoring of vital signs, including electrocardiogram (ECG), non-invasive blood pressure (NIBP), end-tidal carbon dioxide EtCO2) levels, and peripheral arterial oxygen saturation (SpO2), was performed during surgery. General anesthesia was induced using fentanyl 1 μg/kg and propofol 2 mg/kg IV, with rocuronium 0.5 mg/kg administered to facilitate tracheal intubation. Blocks were performed using the SonoSite M-Turbo portable ultrasound system's HFL38X linear multi-frequency 6-13 MHz probe.
2.1. ESPB
For the ESPB, the T5 transverse process tip was identified while the patient was in lateral decubitus position, and a parasagittal view was obtained by rotating the probe longitudinally to see erector spinae (ESM), rhomboids, and trapezius muscles posterior to T5 transverse process. A 22-G echogenic spinal needle was inserted in-plane in the cranial side of the probe until it reached the T5 transverse process, 0.5-1 mL of normal saline was injected to confirm the needle tip location by seeing the fluid spread between the anterior fascial plane of ESM and T5 process. Following this, a 30 mL bolus of 0.25% levobupivacaine was administered.
2.2. SAPB
In the SAPB group, the ultrasonic probe was placed on the midaxillary line at the level of the fifth rib to show serratus anterior and latissimus dorsi muscles. A 22-G spinal needle was inserted in-plane at a 45-degree angle towards the fifth rib, and 0.5-1 mL of normal saline was injected to open the interfascial plane between serratus anterior and latissimus dorsi (LD) muscles. The needle was monitored all the time at the US screen to avoid any trauma. A 30 mL injection of 0.25% levobupivacaine was then administered.
Anesthesia was maintained with sevoflurane in oxygen-enriched air at a concentration of 2-2.5% (FiO2 = 0.5), with rocuronium (0.1 mg/kg) administered every half hour. Multimodal analgesia included paracetamol 1 g, and ketorolac 30 mg, with increments of fentanyl 25 μg IV as rescue analgesia if heart rate or mean arterial blood pressure elevated more than 20% of the baseline. Ringer lactate solution was used for fluid management, and mechanical ventilation was adjusted to maintain EtCO2 at 30-35 mmHg. Post-surgery, neostigmine 0.05 mg/kg and atropine 0.02 mg/kg were administered to reverse residual neuromuscular blockade, followed by extubation.
2.3. Postoperative Management
Postoperative management involved meticulous monitoring of vital signs and pain intensity using the Numeric Rating Scale (NRS) upon arrival at the PACU. Rescue analgesia with intravenous boluses of morphine 3 mg was administered, if the NRS score ≥ 4, with a maximum dosage of 0.5 mg/kg/24 hours. Multimodal analgesia included intravenous paracetamol 1 g/8 hours and ketorolac 30 mg/8 hours, if the NRS score was ≥ 3. For the first 24 hours following surgery, NRS scores, and adverse events like vomiting, nausea, sedation, and difficulty breathing were observed. Ondansetron 0.1 mg/kg was administered for moderate to severe nausea and vomiting.10
The follow-up visits were scheduled at 2, 4, 8, 12, and 24 weeks following MRM. PMPS was defined as chronic pain in the anterior thorax, axilla, and/or medial upper arm, characterized by burning, stabbing, or pulling sensations that endure for over three months post-surgery.3-6 In addition, the Barthel Index of Activities of Daily Living (ADL) and the Flanagan Quality of Life Scale (QoLS) after 2, 4, 8, 12, and 24 weeks postmastectomy.11,12
Neuropathic pain was managed with pregabalin 75-600 mg and amitriptyline 10-25 mg. Analgesics, e.g., paracetamol, nonsteroidal anti-inflammatory drugs (NSAIDs), tramadol hydrochloride 100-400 mg daily, and oxycodone 20-60 mg daily were prescribed based on pain severity.
2.4. Outcomes
The primary outcome was set as the incidence of PMPS. Secondary outcomes included PMPS severity, total intraoperative fentanyl consumption, postoperative morphine consumption, time of first rescue analgesia, patient satisfaction, the Flanagan Quality of Life Scale (QoLS) and Barthel Index of Activities of Daily Living (ADL).
2.5. Statistical Analysis
We utilized SPSS version 27 (IBM§, Chicago, IL, USA) to carry out the statistical analysis. Shapiro-Wilks test and histograms were used to see if the data had a normal distribution. Quantitative parametric data, shown as mean ± SD, was evaluated using the ANOVA (F) test. For comparisons made after the fact, the Tukey test was employed. Median and interquartile range (IQR) data sets were utilized to assess quantitative non-parametric data using the Kruskal-Wallis test. When comparing two sets of data, the Mann-Whitney U test was employed. The qualitative variables, presented as frequencies and percentages, were evaluated using the Chi-square test. A P-value with two tails less than 0.05 was considered statistically significant.
3. RESULTS
Initially, 153 individuals were considered for inclusion; 7 patients declined to participate, and 26 did not meet all criteria. Thus, 120 participants were included and randomized into the three groups (Figure 1).

Figure 1: CONSORT flowchart of the enrolled patients.
There were no notable differences between the three groups regarding age, weight, height, BMI, ASA physical status, and surgical side (Table 1).
| Table 1: Demographic data and side of surgery of the studied groups | |||
| Variable | Control Group
(n = 40) |
SAPB Group
(n = 40) |
ESPB Group
(n = 40) |
| Age (years) | 47.7 ± 5.4 | 48.5 ± 5.3 | 49.2 ± 5.1 |
| Weight (kg) | 68.3 ± 6.0 | 66.9 ± 5.2 | 70.5 ± 6.3 |
| Height (m) | 1.66 ± 0.04 | 1.68 ± 0.04 | 1.68 ± 0.03 |
| BMI (kg/m²) | 25.1 ± 2.1 | 23.9 ± 1.9 | 25.1 ± 2.0 |
| ASA II / III (n) | 38 / 2 | 36 / 4 | 37 / 3 |
| Surgical Side (Right / Left) | 19 / 21 | 23 / 17 | 25 / 15 |
| Data presented as mean ± SD or number (n). | |||
By the end of the follow-up, 37 participants (30.8%) developed PMPS, with a significant difference between groups (P = 0.007). The rate in the control group was 47.5% (95%CI: 31.5%-63.9%), compared to 30% (95%CI: 16.6%-46.5%) in the SAPB group and 15% (95%CI: 5.7%-29.3%) in the ESPB group. Therefore, compared to the control group, the ESPB group showed a statistically significant reduction in the rate of PMPS development. Meanwhile, there was no significant difference between the SAPB group and the control or ESPB groups. Severe PMPS was more common in the control and SAPB groups compared to the ESPB group (Table 2). Among patients who developed PMPS, ESPB was associated with less pain in the chest wall and axilla, while the major site of pain was in the arm. SAPB demonstrated the lowest frequency of axillary pain among the three groups (Table 2).
| Table 2: Incidence, severity, and site of post-mastectomy pain syndrome in the groups | ||||
| Variables | Control Group
(n = 40) |
SAPB Group
(n = 40) |
ESPB Group
(n = 40) |
P-value |
| Incidence of PMPS | ||||
| Developed PMPS (%) | 19 (47.5) | 12 (30.0) | 6 (15.0) | 0.007 |
| 95% Confidence Interval | 31.5-63.9 | 16.6-46.5 | 5.7-29.3% | |
| NO PMPS (%) | 21 (52.5) | 28 (70) | 34 (85) | |
| Severity of PMPS | ||||
| Mild | 5 (26) | 5 (41) | 3 (50) | * |
| Moderate | 8 (40) | 4 (33) | 2 (33) | |
| Severe | 6 (32) | 3 (25 ) | 1 (16.5) | |
| Site of pain | ||||
| Chest wall | 6 (32) | 5 (42) | 1 (16.6) | * |
| Arm | 4 (21) | 5 (42) | 4 (66.6) | |
| Axilla | 9 (47) | 2 (16) | 1 (16.6) | |
| Data were presented as number (%); PMPS: post-mastectomy pain syndrome; * No P-value due to small numbers in subgroups | ||||
Table 3 shows that ESPB was more effective than the control and SAPB groups regarding the reduction of intra- and postoperative opioid consumption and duration of postoperative analgesia. The SAPB group also showed less intraoperative fentanyl consumption and longer analgesia duration than the control group.
| Table 3: Intraoperative fentanyl consumption, morphine consumption at 1st 24h postoperatively, and time of first rescue analgesia of the studied groups | |||||
| Variable | Control
(n = 40) |
SAPB
(n = 40) |
ESPB
(n = 40) |
P-value* | Post-hoc Comparisons |
| Fentanyl (μg/kg) | 187 ± 25 | 163 ± 22 | 131 ± 20 | < 0.001 | P1 = 0.001, P2 < 0.001, P3 = 0.003 |
| Morphine (mg, 1st 24h) | 7.8 ± 2.1 | 4.7 ± 1.8 | 2.2 ± 1.1 | < 0.001 | P1 = 0.164, P2 < 0.001, P3 = 0.008 |
| Time to Rescue Analgesia (hrs) | 5.2 ± 2.4 | 9.3 ± 3.0 | 13.0 ± 3.2 | < 0.001 | P1 = 0.032, P 2 < 0.001, P3 = 0.047 |
| Data presented as mean ± SD. Post-hoc: P1 = Control vs SAPB, P2 = Control vs ESPB, P3 = SAPB vs ESPB. * P-value of ANOVA test | |||||
Both ESPB and SAPB effectively managed early postoperative pain. However, ESPB demonstrated a statistically significant decrease in pain intensity as determined by the NRS throughout 24 hours at rest (Figure 2A) and during movement (Figure 2B), in comparison to the other two groups (P < 0.005).

Figure 2: (A) Numeric Pain Rating Scale at rest, P-value < 0.005 significant at all times

Figure 2: (B) Numeric Pain Rating Scale at movement, P < 0.005 significant at all times
Quality of life (QoL) scores showed no significant intergroup differences up to 12 weeks. Then, ESPB demonstrated significantly higher scores at 24 weeks compared to the control group (Table 4).
| Table 4: Flanagan Quality of Life Scale and Barthel activities of daily living scale of the studied groups | ||||
| Time Point | Control (n = 40) | SAPB (n = 40) | ESPB (n = 40) | P value |
| Baseline | 82.38 ± 12.74 | 82.68 ± 12.64 | 85.03 ± 13.59 | 0.538 |
| 2 weeks | 78.25 ± 12.27 | 79.48 ± 12.92 | 83.73 ± 12.98 | 0.082 |
| 4 weeks | 77.43 ± 13.47 | 77.98 ± 13.57 | 80.18 ± 13.62 | 0.563 |
| 8 weeks | 75.25 ± 13.32 | 76.33 ± 13.33 | 79.73 ± 13.50 | 0.221 |
| 12 weeks | 73.10 ± 13.38 | 74.65 ± 12.97 | 79.23 ± 13.29 | 0.050 |
| 24 weeks | 70.90 ± 13.96 | 73.80 ± 13.22 | 79.73 ± 13.11 | 0.005* |
| ADL Score | ||||
| Baseline | 19.03 ± 1.35 | 19.00 ± 1.43 | 19.15 ± 1.49 | 0.853 |
| 2 weeks | 17.23 ± 1.67 | 18.18 ± 1.62 | 18.25 ± 1.68 | 0.003* |
| 4 weeks | 16.68 ± 1.64 | 17.25 ± 1.92 | 17.85 ± 1.96 | 0.007* |
| 8 weeks | 15.98 ± 1.98 | 16.88 ± 2.21 | 17.83 ± 2.16 | < 0.001* |
| 12 weeks | 15.23 ± 2.69 | 16.38 ± 2.41 | 17.98 ± 2.11 | < 0.001* |
| 24 weeks | 14.58 ± 2.76 | 16.00 ± 2.35 | 18.15 ± 1.98 | < 0.001* |
| Data are presented as Mean ± SD. *Significant as P value ≤ 0.05. | ||||
Baseline activity levels were comparable between groups (P = 0.881). However, starting from the 2nd week, ESPB showed significantly higher ADL scores than the control group (Table 4). Participants who received ESPB demonstrated better overall satisfaction, which was statistically significant; SAPB offered higher satisfaction rates than the control group, yet it was statistically insignificant (Figure 2C).

Figure 2: (C) Patient satisfaction in Control, SAPB and ESPB groups
4. DISCUSSION
MRM encompasses the breast and axillary area, necessitating extensive surgical incisions, a primary contributor to stress and inflammatory responses. Inadequately managed immediate postoperative pain significantly contributes to persistent pain, stress, and inflammatory reactions.13 The pathogenesis of PMPS remains incompletely elucidated; nonetheless, the predominant theory is intercostobrachial nerve neuralgia supplying cutaneous sensation to the axilla and inner arm. Dissection of axillary lymph nodes or manipulation due to retraction during mastectomy may result in inflammation and nerve damage,14 inducing ectopic neuronal activity at the lesion site and dorsal root ganglion, leading to heightened sensitivity to chemical or mechanical stimuli and an amplified experience of pain.6 Besides, the sensitivity of neuronal cells in the brain may be a contributing factor.15
Regrettably, all these potential causes are mostly unmodifiable within the context of MRM. Conversely, intense acute postoperative pain leads to a higher occurrence of persistent postoperative pain;16 and is a clearly changeable risk factor. Numerous studies indicate that moderate to severe acute postoperative pain significantly increases the incidence of PMPS (odds ratio 1.34–2.8).17,18
Thus, preventative and perioperative analgesia, along with pharmacological medications, may mitigate the onset or progression of PMPS. Because local and regional blocks better control intra- and postoperative pain, the anesthetic literature supports their use in preventing PMPS. Preventive regional analgesia may mitigate the hazards of central sensitization by obstructing the transmission of harmful stimuli.
The results of the current study support the notion that adequate management of acute postoperative pain is an effective measure to reduce the incidence of PMPS. The study revealed that the ESPB group showed a statistically significant reduction in the rate of PMPS development compared to the control group (15% vs. 47.5%). Meanwhile, there was no significant difference between SAPB and the control or ESPB groups in the rate of PMPS. Most cases of severe pain were observed in the control group. This reduction of PMPS rate and severity was associated with better control of acute postoperative pain. ESPB was more effective than the control and SAPB groups regarding the reduction of intra- and postoperative opioid consumption, and postoperative pain intensity, in addition to the longer duration of postoperative analgesia.
These findings are consistent with other recent studies. A recent randomized controlled trial indicated that the ESPB offers enhanced analgesic benefits compared to SAPB in patients having MRM, resulting in less morphine consumption and lower postoperative cortisol levels.19 A further study demonstrated that ESPB is equally effective as TPV block for early postoperative analgesia, diminished stress response, and lowered Leeds Assessment of Neuropathic Symptoms and Signs (LANSS) score at the 6-month follow-up.20
The mechanism of action of the ESPB remains incompletely elucidated, with numerous hypotheses posited throughout the years. It is considered to be multifactorial. Research indicates ESPB at the T5 transverse process effectively inhibits the ipsilateral T3-T9 spinal innervation zone.21 ESPB was thought to work through LA diffusion into the paravertebral space and subsequently into the epidural region to block spinal nerves. However, cadaveric studies have challenged this notion, suggesting that the dye does not rapidly spread into the paravertebral space. 22 The physical distribution of LA related to the ESPB influences the dorsal rami, which innervates the spine and paravertebral tissues.23 Fusco et al. (2024) suggested that the fascia may serve as a target, as prior investigations have shown it to be abundant in free nerve terminals.24 Furthermore, they postulated that LA facilitates muscular relaxation, supported by an initial demonstration using elastosonography.
Although it was shown that SAPB was less effective than ESPB, it still showed some protection against PMPS than the control group. It has shown significantly better analgesic efficacy in the acute phase compared to the control group and superior, yet non-significant, value in reducing PMPS (30% vs. 47.5%). SAPB targets the thoracodorsal nerve, long thoracic nerve, intercostobrachial nerve, and the lateral cutaneous branches of the intercostal nerves (T3-T9). These nerves are located between the midaxillary border and the SA and LD muscles.25 The method by which SAPB effectively prevents PMPS is its capacity to diminish acute postoperative pain and alleviate central sensitization. Research indicates that early intervention with SAPB can interrupt this sequence, thereby diminishing the probability of PMPS.26 Furthermore, the interruption of nociceptive signals, less peripheral and central neuronal sensitization, and attenuated inflammatory responses may facilitate the alleviation of persistent postoperative pain.27
Many studies tested the efficacy of multiple regional block techniques for the prevention of PMPS. Mendonça et al. (2023) investigated the efficacy of pectoserratus plane block (PSPB) for reducing the incidence of PMPS after breast cancer surgery.28 PSPB was associated with a reduction of chronic pain from 65% to 22% and the risk of neuropathic pain from 54% to 18%. In another study, PSPB achieved a comparable effect with ESPB on postoperative opioid consumption and acute and chronic pain scores.29 The pectoral nerve-2 (PECS 2) block was compared to SAPB in a randomized trial. PECS2 block reduced the rate of moderate or severe chronic pain from 33% with SAPB to 10%. The authors observed a superior reduction of postoperative morphine consumption in the PECS block, but acute pain scores were similar.13 In a randomized, double-blinded trial ultrasound-guided multilevel PVB was compared to placebo. PVB reduced the incidence of chronic pain from 51.2% to 34.5% after 3 months and from 37.2% to 22.1% after 6 months.30 A systematic review and network meta-analysis evaluated different interventions for PMPS prevention.31 They found that nerve blocks demonstrated improvements in functional outcomes and quality of life (OR: 0.34; 95% CrI: 0.24–0.46).
Notably, observing muscle hematoma during surgery following SAPB in the current study was a significant complication. This can be attributed to the proximity of the SAPB injection site to the surgical field in MRM procedures, where the resulting muscle edema or hematoma could potentially interfere with the surgical process.
Yet, the research on PMPS is sparse, and there is no consensus on a definitive gold standard of practice. Nonetheless, several high-volume facilities have developed their management practices in recent years. Yang et al. (2021) have introduced an algorithm for the systematic selection of suitable medicines in the management of PMPS.32 Another group has presented an algorithm emphasizing the establishment of an appropriate multidisciplinary treatment team for the management of PMPS. They demonstrated that local blocks can be utilized for both diagnostic and symptom treatment.33 Experience at the National Cancer Institute (NCI) of Cairo University can offer valuable insights into the practical implications of regional techniques for preventing PMPS.
5. LIMITATIONS
This study's generalizability may be limited due to its single-center design, and the relatively small sample size. Furthermore, the length and dosage of adjuvant therapy (radiation or chemotherapy) given to patients were outside the purview of this investigation.
6. CONCLUSION
Erector spinae plane block (ESPB) significantly reduced the incidence and severity of post-mastectomy pain syndrome (PMPS), highlighting its effectiveness for long-term pain prevention. Although serratus anterior plane block (SAPB) improved acute postoperative pain, it did not significantly reduce PMPS. ESPB is a more reliable technique for PMPS prevention.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Acknowledgement
We gratefully thank Faculty of Medicine, National Cancer Institute, Cairo University, Cairo for facilitating this research.
9. Conflict of interest
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
SMA: Study concept and design, data collection, statistical analysis, manuscript revision.
SE: Study concept and design, manuscript editing and critical revision.
AHB: Data analysis and interpretation, manuscript review.
MEA: Drafting of the manuscript, critical revision for intellectual content.
MMO: Literature review, data collection assistance, manuscript proofreading.
11. REFERENCES
- Garg R, Bhan S, Vig S. Newer regional analgesia interventions (fascial plane blocks) for breast surgeries: review of literature. Indian J Anaesth. 2018;62(4):254–62. [PubMed] DOI: 4103/ija.IJA_46_18
- Gong Y, Tan Q, Qin Q, Wei C. Prevalence of postmastectomy pain syndrome and associated risk factors: a large single-institution cohort study. Medicine (Baltimore). 2020;99(20):e19834. [PubMed] DOI: 1097/MD.0000000000019834
- Capuco A, Urits I, Orhurhu V, Chun R, Shukla B, Burke M, et al. A comprehensive review of the diagnosis, treatment, and management of postmastectomy pain syndrome. Curr Pain Headache Rep. 2020;24(6):41. [PubMed] DOI: 1007/s11916-020-00876-6
- Couceiro TC, Menezes TC, Valença MM. Post-mastectomy pain syndrome: the magnitude of the problem. Rev Bras Anestesiol. 2009;59(3):358–65. [PubMed] DOI: 1590/s0034-70942009000300012
- Cui L, Fan P, Qiu C, Hong Y. Single institution analysis of incidence and risk factors for post-mastectomy pain syndrome. Sci Rep. 2018;8:11494. [PubMed] DOI: 1038/s41598-018-29946-x
- Chappell AG, Bai J, Yuksel S, Ellis MF. Post-mastectomy pain syndrome: defining perioperative etiologies to guide new methods of prevention for plastic surgeons. World J Plast Surg. 2020;9(3):247–53. [PubMed] DOI: 29252/wjps.9.3.247
- Larsson IM, Sørensen JA, Bille C. The post-mastectomy pain syndrome—a systematic review of the treatment modalities. Breast J. 2017;23(3):338–43. [PubMed] DOI: 1111/tbj.12739
- Tait RC, Zoberi K, Ferguson M, Levenhagen K, Luebbert RA, Rowland K, et al. Persistent post-mastectomy pain: risk factors and current approaches to treatment. J Pain. 2018;19(12):1367–83. [PubMed] DOI: 1016/j.jpain.2018.06.002
- Polite BN, Adams-Campbell LL, Brawley OW, Bickell N, Carethers JM, Flowers CR, et al. Charting the future of cancer health disparities research. J Clin Oncol. 2017;35(26):3075–82. [PubMed] DOI: 1200/JCO.2017.73.6546
- Tong L, Erdmann C, Daldalian M, Li J, Esposito T. Comparison of predictive modeling approaches for 30-day all-cause non-elective readmission risk. BMC Med Res Methodol. 2016;16:26. [PubMed] DOI: 1186/s12874-016-0128-0
- Sinoff G, Ore L. The Barthel activities of daily living index: self-reporting versus actual performance in the old-old (≥75 years). J Am Geriatr Soc. 1997;45(7):832–6. [PubMed] DOI: 1111/j.1532-5415.1997.tb01510.x
- Burckhardt CS, Anderson KL. The Quality of Life Scale (QOLS): reliability, validity, and utilization. Health Qual Life Outcomes. 2003;1:60. [PubMed] DOI: 1186/1477-7525-1-60
- Fujii T, Shibata Y, Akane A, Aoki W, Sekiguchi A, Takahashi K, et al. PECS 2 vs. serratus plane block for chronic pain after mastectomy: RCT. Anaesthesia. 2019;74(12):1558–62. [PubMed] DOI: 1111/anae.14856
- Chappell AG, Yuksel S, Sasson DC, Wescott AB, Connor LM, Ellis MF. Post-mastectomy pain syndrome: an up-to-date review of treatment outcomes. JPRAS Open. 2021;30:97–109. [PubMed] DOI: 1016/j.jpra.2021.07.006
- Chaparro LE, Smith SA, Moore RA, Wiffen PJ, Gilron I. Pharmacotherapy for prevention of chronic pain after surgery in adults. Cochrane Database Syst Rev. 2013;2013(7):CD008307. [PubMed] DOI: 1002/14651858.CD008307.pub2
- Woolf CJ. Central sensitization: implications for diagnosis and treatment of pain. Pain. 2011;152(3 Suppl):S2–15. [PubMed] DOI: 1016/j.pain.2010.09.030
- Hickey OT, Burke SM, Hafeez P, Mudrakouski AL, Hayes ID, Shorten GD. Severity of acute pain after breast surgery and persistent pain development. Clin J Pain. 2010;26(6):556–60. [PubMed] DOI: 1097/AJP.0b013e3181dee988
- Vadivelu N, Schreck M, Lopez J, Kodumudi G, Narayan D. Pain after mastectomy and breast reconstruction. Am Surg. 2008;74(4):285–96. [PubMed]
- Bedewy AE, Mohamed MS, Sultan HM, Khalil MS. ESP vs. serratus anterior plane block after mastectomy: RCT. Anesth Pain Med. 2024;14(2):e142189. [PubMed] DOI: 5812/aapm-142189
- Amr SA, Othman AH, Ahmed EH, Naeem RG, Kamal SM. ESP vs. paravertebral block on postmastectomy pain: RCT. BMC Anesthesiol. 2024;24(1):420. [PubMed] DOI: 1186/s12871-024-02810-4
- Forero M, Rajarathinam M, Adhikary S, Chin KJ. Continuous ESP block for rescue analgesia after epidural failure: case report. A A Case Rep. 2017;8(10):254–6. [PubMed] DOI: 1213/XAA.0000000000000478
- Sørenstua M, Zantalis N, Raeder J, Vamnes JS, Leonardsen ACL. Spread of local anesthetics after ESP block: MRI study. Reg Anesth Pain Med. 2023;48(2):74–9. [PubMed] DOI: 1136/rapm-2022-104012
- Singh S, Choudhary NK, Lalin D, Verma VK. Bilateral ultrasound-guided ESP block for lumbar spine surgery: RCT. J Neurosurg Anesthesiol. 2020;32(4):330–4. [PubMed] DOI: 1097/ANA.0000000000000603
- Fusco P, Stecco C, Petroni GM, Ciaschi W, Marinangeli F. ESP block and chronic pain: the dark side. Minerva Anestesiol. 2022;88(5):528–9. [PubMed] DOI: 23736/S0375-9393.22.16282-6
- Zocca JA, Chen GH, Puttanniah VG, Hung JC, Gulati A. Ultrasound-guided serratus plane block for postmastectomy pain: case series. Pain Pract. 2017;17(1):141–6. [PubMed] DOI: 1111/papr.12482
- Qian B, Huang S, Liao X, Wu J, Lin Q, Lin Y. SAPB reduces chronic postsurgical pain after mastectomy: A randomized controlled trial. J Clin Anesth. 2021;74:110410. [PubMed] DOI: 1016/j.jclinane.2021.110410
- Glare P, Aubrey KR, Myles PS. Transition from acute to chronic pain after surgery. Lancet. 2019;393(10180):1537–46. [PubMed] DOI: 1016/S0140-6736(19)30352-6
- Mendonça FT, Nascimento LFC, Veloso NM, Basto GCP. Long-term efficacy of PSPB for postmastectomy pain: Extended Follow-up From a Randomized Controlled Trial. Clin J Pain. 2023;39(7):334–9. [PubMed] DOI: 1097/AJP.0000000000001118
- Genc C, Kaya C, Bilgin S, Dost B, Ustun YB, Koksal E. PSPB vs. ESP block for opioid use and pain after breast surgery: RCT. J Clin Anesth. 2022;79:110691. [PubMed] DOI: 1016/j.jclinane.2022.110691
- Qian B, Fu S, Yao Y, Lin D, Huang L. Paravertebral blocks reduce postmastectomy chronic pain: RCT. J Pain Res. 2019;12:597–603. [PubMed] DOI: 2147/JPR.S190201
- Dzhantemirova N, Akhmedin D, Khasenov D, Khakimova G, Khakimova S, Bekisheva A, et al. Prevention of breast cancer surgery complications: review and meta-analysis. Asian Pac J Cancer Prev. 2024;25(1):9–23. [PubMed] DOI: 31557/APJCP.2024.25.1.9
- Yang A, Nadav D, Legler A, Chen GH, Hingula L, Puttanniah V, et al. Interventional pain algorithm for postmastectomy pain: retrospective review. Pain Med. 2021;22(4):677–86. [PubMed] DOI: 1093/pm/pnaa343
- Beederman M, Bank J. Post-breast surgery pain syndrome: shifting a surgical paradigm. Plast Reconstr Surg Glob Open. 2021;9(7):e3720. [PubMed] DOI: 1097/GOX.0000000000003720