Shahad Alaa Tamkeen ¹* , Murtada M. Jawad ²*
Author Affiliations
Background & objective: Hypertensive nephropathy is a major global health concern, contributing to significant morbidity and mortality. Early detection of kidney damage using specific biomarkers can aid in timely intervention and improved patient outcomes. This study aimed to assess the diagnostic potential of serum Kidney Injury Molecule-1 (KIM-1) as a biomarker for the early detection of nephropathy in hypertensive patients and its role in the progression of the disease.
Materials and Methods: This study comprised 135 participants aged between 35 and 70 years, categorized into three groups: 45 healthy individuals (control group), 45 hypertensive patients without clinically diagnosed nephropathy (HP), and 45 hypertensive patients with clinically diagnosed nephropathy (HNP). The biomarker KIM-1 was measured using enzyme-linked immunosorbent assay (ELISA). The study was conducted at Al-Sadr Teaching Hospital and Al-Najaf Teaching Hospital.
Result: The HNP group had a mean KIM-1 level of 4.44 ± 1.98 ng/mL, significantly higher than both the HP group, which had a mean of 1.55 ± 0.57 ng/mL, and the control group, which had a mean of 1.21 ± 0.26 ng/mL. The comparison yielded a P < 0.001, indicating a significant difference among the three groups, the highest in hypertensive nephropathy compared to hypertension and control groups.
Conclusion: According to our results, KIM-1 is a potential biomarker for early detection and prognosis monitoring of hypertensive nephropathy. Further studies are needed to validate its clinical utility and prognostic value in larger patient cohorts.
Abbreviations: ACR: urine albumin-to-creatinine ratio, CKD: chronic kidney disease, ESRD: end-stage renal disease, HNP: Hypertensive nephropathy, KIM-1: Kidney Injury Molecule-1, TIM: T-cell immunoglobulin and mucin domain
Keywords: Nephropathy; KIM-1, Hypertension; Hypertensive Nephropathy
Citation: Tamkeen SA, Jawad MM. Serum KIM-1 as an early biomarker for hypertensive nephropathy. Anaesth. pain intensive care 2025;29(5):281-287. DOI: 10.35975/apic.v29i5.2852
Received: May 09, 2024; Revised: October 26, 2024; Accepted: January 01, 2025
Hypertensive nephropathy (HNP) refers to kidney damage caused by prolonged hypertension, which might lead to a reduction in its functionality that may lead to end-stage renal disease (ESRD).1 The global prevalence of chronic kidney disease (CKD) among hypertensive individuals is estimated to be approximately 34.97% (2). This prevalence differs by region, with South America experiencing the highest rate at 60.10% and Europe the lowest at 28.23%.2 Hypertension induces structural changes in the kidney, affecting the vasculature, glomeruli, and tubules.3 HN often progresses silently, with many patients remaining asymptomatic and thus not seeking regular medical evaluations. Consequently, by the time of their initial medical examination, a significant number already have CKD in its early stages. Once clinical symptoms or routine test abnormalities become apparent, substantial renal damage may have already occurred. While HN can develop in individuals newly diagnosed with hypertension, its clinical manifestations typically emerge after 10 to 15 years of sustained high blood pressure. Therefore, early detection of HN is crucial to prevent further kidney damage and manage the condition effectively.4
Kidney Injury Molecule-1 (KIM-1), also known as T-cell immunoglobulin and mucin domain 1 (TIM-1) is “a type I transmembrane glycoprotein that belongs to the T-cell immunoglobulin and mucin domain (TIM) family of proteins”. KIM-1 is expressed by both immune and epithelial cells, in contrast to other members of the TIM family. Its expression is mostly noticed in proximal tubular epithelial cells following kidney injury, rendering it a vital biomarker for identifying renal damage.5,6 People with normal renal function do not have KIM-1 in their tubular cells. On the other hand, KIM-1 levels are higher in urine and plasma samples from patients with kidney injury, where the cells of tubular epithelium undergo high differentiation and proliferation.7 KIM-1 is significantly upregulated in both acute kidney injury and chronic kidney disease , where it plays a crucial role in apoptotic cell clearance, reducing inflammation, and contributing to tissue remodeling.5,6 It plays a vital role in the removal of apoptotic cells, lowering inflammation, and promoting tissue remodeling. Recent studies indicate that KIM-1 may also serve as a potential biomarker for predicting how CKD will progress.8
Our study aimed to indicate whether KIM-1 is a predictive diagnostic molecule for nephropathy in hypertensive patients.
This case-control study was conducted at Al-Sadr Teaching Hospital and Al-Najaf Teaching Hospital. The research will begin in September 2024 and go until March 2025. The research, followed a protocol authorized by a local ethics committee at “Al-Furat Al-Awsat Technical University/College of Health and Medical Techniques/Kufa and Al-Najaf Health Directorate” under reference number (34578) dated 2024-9-16.
The study included 135 participants, aged 35 to 70 years, categorized into three groups: 45 healthy individuals (control group), 45 hypertensive patients without clinically diagnosed nephropathy (HP group), and 45 hypertensive patients with clinically diagnosed nephropathy confirmed by nephrologist (HNP group).
The exclusion criteria included: patients suffering from diabetes mellitus, autoimmune diseases, cancers, primary parathyroid disorders, pregnant women, or other causes of chronic kidney disease such as polycystic kidney disease, glomerulonephritis, kidney stones, or other secondary causes.
2.1. Sample Collection
After interviewing the participants from the study groups, demographic data were collected using a researcher-designed questionnaire. Each participant had about 5 mL of venous blood drawn from them. Following collection in gel tubes and coagulation time, the blood samples were centrifuged at 3000 rpm for 15 minutes. The resultant serum was subsequently kept in Eppendorf tubes at -20°C for further biochemical analysis.9
Urine samples were collected from participants in sterile containers. The samples were subsequently separated into two sections: one for creatinine analysis and the other for albumin measurement.
2.2. Measurement of Biomarkers
Serum urea and creatinine levels, along with urine creatinine were measured using the Spin 200E analyzer (SPINREACT) following the manufacturer's instructions. Urinary albumin levels were assessed in random spot samples using the Ichroma II device (Boditech Med). An automated sphygmomanometer was used to measure blood pressure.
Using the sandwich enzyme-linked immunosorbent assay (ELISA) method, the serum KIM-1 concentration was determined in accordance with the manufacturer's instructions (BT LAB company, China).
2.3. Statistical Analysis
Categorical variables were expressed as numbers and percentages (%) and evaluated using the chi-square test or Fisher’s exact test, as applicable. Continuous variables were presented as mean ± SD were analysed using the Student’s t-test. One-way analysis of variance (ANOVA) or the Kruskal-Wallis test was conducted for comparisons involving three or more groups. Correlations between biomarkers and clinical parameters were evaluated using Pearson's or Spearman's correlation coefficients. The predictive performance of biomarkers was assessed by calculating the area under the receiver operating characteristic (ROC) curve (AUC). All statistical analyses were conducted using SPSS v.28 (IBM, IL, USA), with statistical significance established at P < 0.05.11
Table 1 presents the demographic and clinical characteristics of HNP, HP, and control groups. There were no statistically significant differences in age (P = 0.715), sex (P = 0.399), smoking status (P = 0.162), or family history (P = 0.197).
Nonetheless, HNP and HP groups showed a significant difference (P = 0.008) in BMI, with higher obesity prevalence as compared to controls. This aligns with existing research showing that obesity is a common comorbidity in individuals with hypertension and chronic kidney disease. Both systolic and diastolic BP were significantly elevated in patient groups (P = 0.0001). With 62.2% of HNP patients having >10 years of hypertension, the duration of hypertension was significantly longer than that of the HP group (20%; P = 0.0001)
Table 2 presents a comparison of kidney function biomarkers among three groups. Significant differences were observed across all parameters (P < 0.001). HNP group patients exhibited elevated serum creatinine, urea, and urine albumin-to-creatinine ratio (ACR) levels, alongside significantly reduced eGFR, indicating impaired kidney function. HP group showed mild or no alterations compared to controls, suggesting an early or less severe impact on renal function.
Table 3 shows a significant difference in KIM-1 levels among HNP, HP, and healthy control groups (P < 0.001). HNP patients had the highest KIM-1 levels, indicating severe kidney injury, while HP individuals showed moderately elevated levels compared to controls, suggesting early signs of kidney involvement. Healthy controls had the lowest levels, confirming normal kidney function. The statistical differences between groups (A, B, C) indicate that KIM-1 can serve as a potential biomarker for early detecting of hypertensive kidney injury and monitoring of disease.
The results demonstrate significant correlations between biomarker and kidney function parameters in both HNP and HP groups. In HNP patients, KIM-1 is positively correlated with S. Creatinine (r = 0.617, p < 0.001), and S. Urea (r = 0.329, P = 0.027), while it shows a negative correlation with eGFR (r = -0.483, P = 0.001). In the HP group, KIM-1 is positively correlated with S. Creatinine (r = 0.443, P = 0.002) but negatively with eGFR (r = -0.488, P = 0.001).
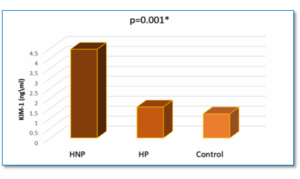
Figure 1. Comparison of KIM- level in HNP, HP, and Healthy controls
The receiver operator characteristic (ROC) are presented in Table 5. The cut-off values for KIM-1 in the diagnosis of hypertensive nephropathy were established through the administration of a ROC curve analysis. The sensitivity was 66%, the specificity was 100%, and the P was 0.0001 for KIM-1 (ng/mL), with an area under the curve (AUC) of 0.82 (95% CI: 0.75–0.89) and a cut-off value of >1.68.
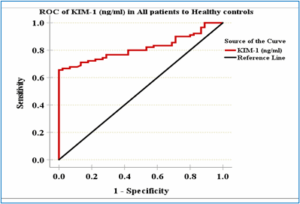
Figure 2. Roc Curve of KIM-1 level in All patients to Healthy control
Chronic hypertension can lead to renal tubular injury through increased intraglomerular pressure, causing hyperfiltration and damage to the renal tubules. This process is further exacerbated by Renin-Angiotensin-Aldosterone System (RAAS) activation, which induces vasoconstriction, sodium retention, and fibrosis, While oxidative stress and inflammation contribute to tubular damage and interstitial fibrosis, ultimately progressing hypertensive nephropathy.12–14 KIM-1 is a protein that is upregulated in proximal tubular cells in response to kidney stress.15 Therefore, a chronic hypertension-induced renal tubular injury will lead to elevated serum levels of KIM-1.
In the present study, patients with HNP exhibited significantly higher serum KIM-1 levels (4.44 ± 1.98 ng/mL) compared to those with hypertension alone (1.55 ± 0.57 ng/mL) and healthy controls (1.21 ± 0.26 ng/mL). These findings align with previous studies have reported substantially increased KIM-1 concentrations in individuals with chronic kidney disease (CKD) relative to non-CKD participants.16,17, reinforcing the possible role of KIM-1 as a kidney injury biomarker.
Experimental evidence also supports the association among KIM-1 elevation and hypertensive kidney injury. A prior study investigating the impact of “angiotensin II type 1A receptor “on KIM-1 expression in a mouse model of renovascular hypertension (2K1C) demonstrated a substantial increase in KIM-1 levels in the affected kidney.18 This observation suggests that KIM-1 upregulation is related to established renal injury in hypertensive conditions, further validating its role in the pathophysiology of hypertensive nephropathy.
Interestingly, studies on kidney transplant dysfunction have shown that upregulated KIM-1 expression may also serve a reparative role in tubular epithelial cells.19 While our study did not assess this aspect directly, KIM-1 upregulation represents an adaptive response to injury. However, as the disease progresses, sustained KIM-1 expression may contribute to chronic inflammation and fibrosis, exacerbating renal damage. This aligns with research indicating that KIM-1 is involved in macrophage recruitment and inflammatory signaling pathways such as STAT3 and ERK activation, which may accelerate kidney injury in hypertensive nephropathy.
Furthermore, the outcomes of this study confirm a substantial correlation among serum KIM-1 levels and established renal function markers. Consistent with previous findings in CKD patients, KIM-1 demonstrated a substantial positive correlation with creatinine and blood urea nitrogen (BUN), while an inverse correlation was observed with estimated glomerular filtration rate (eGFR).17 These associations indicate that KIM-1 not only serves as a marker of tubular injury but also reflects renal function decline in hypertensive nephropathy.20,21
Overall, these findings support the clinical utility of KIM-1 as a biomarker for detecting early kidney injury in hypertensive patients and monitoring disease progression.
This study detects KIM-1 as a potential prognostic and diagnostic biomarker for nephropathy in hypertensive patients. The significant correlation between KIM-1 and kidney function tests suggests that increased KIM-1 expression reflects renal injury severity and dysfunction in hypertensive nephropathy. Furthermore, the study offers valued insights into the clinical utility of KIM-1 for early diagnosis and disease monitoring. However, additional studies with larger and more diverse patient cohorts are required to validate its prognostic value and optimize its application in clinical practice.
6. Availability of data
The data and materials associated with this research will be made available by the corresponding author upon reasonable request.
7. Ethical considerations
The research, dated 2024-9-16, followed a protocol authorized by a local ethics committee at “Al-Furat Al-Awsat Technical University/College of Health and Medical Techniques/Kufa and Al-Najaf Health Directorate” under reference number (34578).
8. Consent for publication
All the authors gave consent for the publication of the work under the Creative Commons Attribution- Non-Commercial 4.0 license.
9. Competing interests
The authors declare that they have no competing interests. No external or industry funding was involved.
10. Acknowledgement
We sincerely thank all the pregnant women who participated in this study for their invaluable contribution. Our deepest appreciation goes to the clinical and laboratory staff at Al-Sadr Teaching Hospital and Al-Najaf Teaching Hospital, whose unwavering support and professionalism were instrumental in completing this research. We are especially grateful to the research coordinators for their meticulous work in data collection and to the laboratory technicians for their expertise in handling and analyzing samples.
11. Author contributions
The author was responsible for all aspects of the study and approved the final version of the paper.
Author Affiliations
- Shahad Alaa Tamkeen, Al-Furat Al-Awsat Technical University /College of Health and Medical Techniques, Department of Medical Laboratory Techniques, Kufa, Iraq; Email: shahadalaatt@gmail.com ; {ORCID:0009-0004-8017-2634}
- Murtada M. Jawad, Al-Furat Al-Awsat Technical University /College of Health and Medical Techniques, Department of Medical Laboratory Techniques, Kufa, Iraq; Email: kuh.dr.mur@atu.edu.iq; {ORCID:0000-0002-7097-1270}
ABSTRACT
Background & objective: Hypertensive nephropathy is a major global health concern, contributing to significant morbidity and mortality. Early detection of kidney damage using specific biomarkers can aid in timely intervention and improved patient outcomes. This study aimed to assess the diagnostic potential of serum Kidney Injury Molecule-1 (KIM-1) as a biomarker for the early detection of nephropathy in hypertensive patients and its role in the progression of the disease.
Materials and Methods: This study comprised 135 participants aged between 35 and 70 years, categorized into three groups: 45 healthy individuals (control group), 45 hypertensive patients without clinically diagnosed nephropathy (HP), and 45 hypertensive patients with clinically diagnosed nephropathy (HNP). The biomarker KIM-1 was measured using enzyme-linked immunosorbent assay (ELISA). The study was conducted at Al-Sadr Teaching Hospital and Al-Najaf Teaching Hospital.
Result: The HNP group had a mean KIM-1 level of 4.44 ± 1.98 ng/mL, significantly higher than both the HP group, which had a mean of 1.55 ± 0.57 ng/mL, and the control group, which had a mean of 1.21 ± 0.26 ng/mL. The comparison yielded a P < 0.001, indicating a significant difference among the three groups, the highest in hypertensive nephropathy compared to hypertension and control groups.
Conclusion: According to our results, KIM-1 is a potential biomarker for early detection and prognosis monitoring of hypertensive nephropathy. Further studies are needed to validate its clinical utility and prognostic value in larger patient cohorts.
Abbreviations: ACR: urine albumin-to-creatinine ratio, CKD: chronic kidney disease, ESRD: end-stage renal disease, HNP: Hypertensive nephropathy, KIM-1: Kidney Injury Molecule-1, TIM: T-cell immunoglobulin and mucin domain
Keywords: Nephropathy; KIM-1, Hypertension; Hypertensive Nephropathy
Citation: Tamkeen SA, Jawad MM. Serum KIM-1 as an early biomarker for hypertensive nephropathy. Anaesth. pain intensive care 2025;29(5):281-287. DOI: 10.35975/apic.v29i5.2852
Received: May 09, 2024; Revised: October 26, 2024; Accepted: January 01, 2025
1. INTRODUCTION
Hypertensive nephropathy (HNP) refers to kidney damage caused by prolonged hypertension, which might lead to a reduction in its functionality that may lead to end-stage renal disease (ESRD).1 The global prevalence of chronic kidney disease (CKD) among hypertensive individuals is estimated to be approximately 34.97% (2). This prevalence differs by region, with South America experiencing the highest rate at 60.10% and Europe the lowest at 28.23%.2 Hypertension induces structural changes in the kidney, affecting the vasculature, glomeruli, and tubules.3 HN often progresses silently, with many patients remaining asymptomatic and thus not seeking regular medical evaluations. Consequently, by the time of their initial medical examination, a significant number already have CKD in its early stages. Once clinical symptoms or routine test abnormalities become apparent, substantial renal damage may have already occurred. While HN can develop in individuals newly diagnosed with hypertension, its clinical manifestations typically emerge after 10 to 15 years of sustained high blood pressure. Therefore, early detection of HN is crucial to prevent further kidney damage and manage the condition effectively.4
Kidney Injury Molecule-1 (KIM-1), also known as T-cell immunoglobulin and mucin domain 1 (TIM-1) is “a type I transmembrane glycoprotein that belongs to the T-cell immunoglobulin and mucin domain (TIM) family of proteins”. KIM-1 is expressed by both immune and epithelial cells, in contrast to other members of the TIM family. Its expression is mostly noticed in proximal tubular epithelial cells following kidney injury, rendering it a vital biomarker for identifying renal damage.5,6 People with normal renal function do not have KIM-1 in their tubular cells. On the other hand, KIM-1 levels are higher in urine and plasma samples from patients with kidney injury, where the cells of tubular epithelium undergo high differentiation and proliferation.7 KIM-1 is significantly upregulated in both acute kidney injury and chronic kidney disease , where it plays a crucial role in apoptotic cell clearance, reducing inflammation, and contributing to tissue remodeling.5,6 It plays a vital role in the removal of apoptotic cells, lowering inflammation, and promoting tissue remodeling. Recent studies indicate that KIM-1 may also serve as a potential biomarker for predicting how CKD will progress.8
Our study aimed to indicate whether KIM-1 is a predictive diagnostic molecule for nephropathy in hypertensive patients.
2. METHODOLOGY
This case-control study was conducted at Al-Sadr Teaching Hospital and Al-Najaf Teaching Hospital. The research will begin in September 2024 and go until March 2025. The research, followed a protocol authorized by a local ethics committee at “Al-Furat Al-Awsat Technical University/College of Health and Medical Techniques/Kufa and Al-Najaf Health Directorate” under reference number (34578) dated 2024-9-16.
The study included 135 participants, aged 35 to 70 years, categorized into three groups: 45 healthy individuals (control group), 45 hypertensive patients without clinically diagnosed nephropathy (HP group), and 45 hypertensive patients with clinically diagnosed nephropathy confirmed by nephrologist (HNP group).
The exclusion criteria included: patients suffering from diabetes mellitus, autoimmune diseases, cancers, primary parathyroid disorders, pregnant women, or other causes of chronic kidney disease such as polycystic kidney disease, glomerulonephritis, kidney stones, or other secondary causes.
2.1. Sample Collection
After interviewing the participants from the study groups, demographic data were collected using a researcher-designed questionnaire. Each participant had about 5 mL of venous blood drawn from them. Following collection in gel tubes and coagulation time, the blood samples were centrifuged at 3000 rpm for 15 minutes. The resultant serum was subsequently kept in Eppendorf tubes at -20°C for further biochemical analysis.9
Urine samples were collected from participants in sterile containers. The samples were subsequently separated into two sections: one for creatinine analysis and the other for albumin measurement.
2.2. Measurement of Biomarkers
Serum urea and creatinine levels, along with urine creatinine were measured using the Spin 200E analyzer (SPINREACT) following the manufacturer's instructions. Urinary albumin levels were assessed in random spot samples using the Ichroma II device (Boditech Med). An automated sphygmomanometer was used to measure blood pressure.
Using the sandwich enzyme-linked immunosorbent assay (ELISA) method, the serum KIM-1 concentration was determined in accordance with the manufacturer's instructions (BT LAB company, China).
2.3. Statistical Analysis
Categorical variables were expressed as numbers and percentages (%) and evaluated using the chi-square test or Fisher’s exact test, as applicable. Continuous variables were presented as mean ± SD were analysed using the Student’s t-test. One-way analysis of variance (ANOVA) or the Kruskal-Wallis test was conducted for comparisons involving three or more groups. Correlations between biomarkers and clinical parameters were evaluated using Pearson's or Spearman's correlation coefficients. The predictive performance of biomarkers was assessed by calculating the area under the receiver operating characteristic (ROC) curve (AUC). All statistical analyses were conducted using SPSS v.28 (IBM, IL, USA), with statistical significance established at P < 0.05.11
3. RESULTS
Table 1 presents the demographic and clinical characteristics of HNP, HP, and control groups. There were no statistically significant differences in age (P = 0.715), sex (P = 0.399), smoking status (P = 0.162), or family history (P = 0.197).
| Table 1: Comparative demographic and clinical characteristics of three groupsa | |||||||||
| Variables | HNP group
(n = 45) |
HP group
(n = 45) |
Control group
(n = 45) |
X2 | P-value | ||||
| Age (years) | Mean ± SD | 52.44 ± 10.7 | 51.42 ± 9.09 | 49.96 ± 11.69 | 0.532¥ | ||||
| 35 – 44 | 12 (26.7) | 16 (35.6) | 17 (37.8) | 2.12 | 0.715 | ||||
| 45 – 54 | 16 (35.6) | 17 (37.8) | 14 (31.1) | ||||||
| ≥ 55 | 17 (37.8) | 12 (26.7) | 14 (31.1) | ||||||
| Sex | Male | 20 (44.4) | 21 (46.7) | 26 (57.8) | 1.84 | 0.399 | |||
| Female | 25 (55.6) | 24 (53.3) | 19 (42.2) | ||||||
| BMI (kg/m2) | Mean ± SD | 28.33 ± 4.07 | 29.06 ± 4.87 | 26.39 ± 3.23 | 0.008¥ | ||||
| Normal weight | 9 (20.0) | 9 (20.0) | 22 (48.9) | 13.66 | 0.008* | ||||
| Overweight | 18 (40.0) | 17 (37.8) | 15 (33.3) | ||||||
| Obesity | 18 (40.0) | 19 (42.2) | 8 (17.8) | ||||||
| Smoking history | 16 (35.6) | 12 (26.7) | 8 (17.8) | 3.64 | 0.162 | ||||
| Family history | 10 (22.2) | 13 (28.9) | 6 (13.3) | 3.25 | 0.197 | ||||
| SBP (mmHg) | 147.84 ± 13.31 | 144.78 ± 14.14 | 120.47 ± 4.92 | 0.0001*¥ | |||||
| DBP (mmHg) | 97.44 ± 8.73 | 96.78 ± 9.38 | 78.89 ± 4.49 | 0.0001*¥ | |||||
| BP duration (yr) | Mean ± SD | 13.25 ± 6.42 | 6.87 ± 4.07 | 0 | 0.0001*¥ | ||||
| ≤ 5 yr. | 5 (11.1) | 16 (35.6) | 0 (0.0) | 17.52a | 0.0001* | ||||
| 6-10 yr. | 12 (26.7) | 20 (44.4) | 0 (0.0) | ||||||
| > 10 yr. | 28 (62.2) | 9 (20.0) | 0 (0.0) | ||||||
| Significant differences at P ≤0.05; X2: Chi-Square test; a: Testing between patients only. # Testing between categories of Hyper. NP group only. ¥: ANOVA or Mann Whitney test. | |||||||||
Nonetheless, HNP and HP groups showed a significant difference (P = 0.008) in BMI, with higher obesity prevalence as compared to controls. This aligns with existing research showing that obesity is a common comorbidity in individuals with hypertension and chronic kidney disease. Both systolic and diastolic BP were significantly elevated in patient groups (P = 0.0001). With 62.2% of HNP patients having >10 years of hypertension, the duration of hypertension was significantly longer than that of the HP group (20%; P = 0.0001)
Table 2 presents a comparison of kidney function biomarkers among three groups. Significant differences were observed across all parameters (P < 0.001). HNP group patients exhibited elevated serum creatinine, urea, and urine albumin-to-creatinine ratio (ACR) levels, alongside significantly reduced eGFR, indicating impaired kidney function. HP group showed mild or no alterations compared to controls, suggesting an early or less severe impact on renal function.
| Table 2: Comparative biochemical parameters of the kidney functions | ||||
| Parameters | HNP group | HP group | Control group | P-value |
| S. creatinine (mg/dL) | 2.72 ± 1.22 A | 0.69 ± 0.13 B | 0.63 ± 0.14 B | < 0.001* |
| S. urea (mg/dL) | 85.56 ± 26.4 A | 35 ± 8.83 B | 29.98 ± 7.19 B | < 0.001* |
| ACR (mg/g) | 445.03 ± 304.09 A | 12.28 ± 7.06 B | 6.19 ± 2.49 C | < 0.001* |
| eGFR (mL/min/1.73 m²) | 31.78 ± 17.06 A | 95.27 ± 9.21 B | 113.51 ± 12.25 C | < 0.001* |
| aSignificant differences at P ≤ 0.05; All data expressed as mean ± SD; Different capital letters show significant difference by ANOVA with (Tukey's post hoc) or Kruskal test; ACR: urine albumin-to-creatinine ratio | ||||
Table 3 shows a significant difference in KIM-1 levels among HNP, HP, and healthy control groups (P < 0.001). HNP patients had the highest KIM-1 levels, indicating severe kidney injury, while HP individuals showed moderately elevated levels compared to controls, suggesting early signs of kidney involvement. Healthy controls had the lowest levels, confirming normal kidney function. The statistical differences between groups (A, B, C) indicate that KIM-1 can serve as a potential biomarker for early detecting of hypertensive kidney injury and monitoring of disease.
| Table 3: Comparison of biomarkers in three groups a | ||||
| Parameters | HNP group | HP group | Control group | P-value |
| KIM-1 (ng/mL) | 4.44 ± 1.98 A | 1.55 ± 0.57 B | 1.21 ± 0.26 C | < 0.001* |
| aSignificant differences at P ≤ 0.05; All data expressed as mean ± SD (standard division); Different capital letters show significant difference by ANOVA with (Tukey's post hoc) or Kruskal test. | ||||
The results demonstrate significant correlations between biomarker and kidney function parameters in both HNP and HP groups. In HNP patients, KIM-1 is positively correlated with S. Creatinine (r = 0.617, p < 0.001), and S. Urea (r = 0.329, P = 0.027), while it shows a negative correlation with eGFR (r = -0.483, P = 0.001). In the HP group, KIM-1 is positively correlated with S. Creatinine (r = 0.443, P = 0.002) but negatively with eGFR (r = -0.488, P = 0.001).
| Table 4: Correlations of KIM-1 with kidney function parameters in both HNP and HP group patients a | |||
| Parameters | HPN | HP | |
| S. Creatinine (mg/dL) | r | 0.617** | 0.443** |
| p | 0.0001 | 0.002 | |
| S. Urea (mg/dL) | r | 0.329* | 0.014 |
| p | 0.027 | 0.925 | |
| ACR (mg/g) | r | 0.443** | 0.350* |
| p | 0.002 | 0.019 | |
| aCorrelation is significant at the P ≤ 0.05*, and < 0.01** level
ACR: urine albumin-to-creatinine ratio |
|||

Figure 1. Comparison of KIM- level in HNP, HP, and Healthy controls
The receiver operator characteristic (ROC) are presented in Table 5. The cut-off values for KIM-1 in the diagnosis of hypertensive nephropathy were established through the administration of a ROC curve analysis. The sensitivity was 66%, the specificity was 100%, and the P was 0.0001 for KIM-1 (ng/mL), with an area under the curve (AUC) of 0.82 (95% CI: 0.75–0.89) and a cut-off value of >1.68.
| Table 5: ROC analysis of biomarkers for hypertensive nephropathy diagnosis in all patients to healthy control. | |||||||
| Biomarkers | Area | P-value | Asymptotic 95% Confidence Interval | Cutoff | Sensitivity | Specificity | |
| Lower Bound | Upper Bound | ||||||
| KIM-1 (ng/mL) | 0.82 | 0.0001 | 0.75 | 0.89 | >1.68 | 0.66 | 1.00 |

Figure 2. Roc Curve of KIM-1 level in All patients to Healthy control
4. DISCUSSION
Chronic hypertension can lead to renal tubular injury through increased intraglomerular pressure, causing hyperfiltration and damage to the renal tubules. This process is further exacerbated by Renin-Angiotensin-Aldosterone System (RAAS) activation, which induces vasoconstriction, sodium retention, and fibrosis, While oxidative stress and inflammation contribute to tubular damage and interstitial fibrosis, ultimately progressing hypertensive nephropathy.12–14 KIM-1 is a protein that is upregulated in proximal tubular cells in response to kidney stress.15 Therefore, a chronic hypertension-induced renal tubular injury will lead to elevated serum levels of KIM-1.
In the present study, patients with HNP exhibited significantly higher serum KIM-1 levels (4.44 ± 1.98 ng/mL) compared to those with hypertension alone (1.55 ± 0.57 ng/mL) and healthy controls (1.21 ± 0.26 ng/mL). These findings align with previous studies have reported substantially increased KIM-1 concentrations in individuals with chronic kidney disease (CKD) relative to non-CKD participants.16,17, reinforcing the possible role of KIM-1 as a kidney injury biomarker.
Experimental evidence also supports the association among KIM-1 elevation and hypertensive kidney injury. A prior study investigating the impact of “angiotensin II type 1A receptor “on KIM-1 expression in a mouse model of renovascular hypertension (2K1C) demonstrated a substantial increase in KIM-1 levels in the affected kidney.18 This observation suggests that KIM-1 upregulation is related to established renal injury in hypertensive conditions, further validating its role in the pathophysiology of hypertensive nephropathy.
Interestingly, studies on kidney transplant dysfunction have shown that upregulated KIM-1 expression may also serve a reparative role in tubular epithelial cells.19 While our study did not assess this aspect directly, KIM-1 upregulation represents an adaptive response to injury. However, as the disease progresses, sustained KIM-1 expression may contribute to chronic inflammation and fibrosis, exacerbating renal damage. This aligns with research indicating that KIM-1 is involved in macrophage recruitment and inflammatory signaling pathways such as STAT3 and ERK activation, which may accelerate kidney injury in hypertensive nephropathy.
Furthermore, the outcomes of this study confirm a substantial correlation among serum KIM-1 levels and established renal function markers. Consistent with previous findings in CKD patients, KIM-1 demonstrated a substantial positive correlation with creatinine and blood urea nitrogen (BUN), while an inverse correlation was observed with estimated glomerular filtration rate (eGFR).17 These associations indicate that KIM-1 not only serves as a marker of tubular injury but also reflects renal function decline in hypertensive nephropathy.20,21
Overall, these findings support the clinical utility of KIM-1 as a biomarker for detecting early kidney injury in hypertensive patients and monitoring disease progression.
5. CONCLUSION
This study detects KIM-1 as a potential prognostic and diagnostic biomarker for nephropathy in hypertensive patients. The significant correlation between KIM-1 and kidney function tests suggests that increased KIM-1 expression reflects renal injury severity and dysfunction in hypertensive nephropathy. Furthermore, the study offers valued insights into the clinical utility of KIM-1 for early diagnosis and disease monitoring. However, additional studies with larger and more diverse patient cohorts are required to validate its prognostic value and optimize its application in clinical practice.
6. Availability of data
The data and materials associated with this research will be made available by the corresponding author upon reasonable request.
7. Ethical considerations
The research, dated 2024-9-16, followed a protocol authorized by a local ethics committee at “Al-Furat Al-Awsat Technical University/College of Health and Medical Techniques/Kufa and Al-Najaf Health Directorate” under reference number (34578).
8. Consent for publication
All the authors gave consent for the publication of the work under the Creative Commons Attribution- Non-Commercial 4.0 license.
9. Competing interests
The authors declare that they have no competing interests. No external or industry funding was involved.
10. Acknowledgement
We sincerely thank all the pregnant women who participated in this study for their invaluable contribution. Our deepest appreciation goes to the clinical and laboratory staff at Al-Sadr Teaching Hospital and Al-Najaf Teaching Hospital, whose unwavering support and professionalism were instrumental in completing this research. We are especially grateful to the research coordinators for their meticulous work in data collection and to the laboratory technicians for their expertise in handling and analyzing samples.
11. Author contributions
The author was responsible for all aspects of the study and approved the final version of the paper.
12. REFERENCES
- Kao TW, Huang CC. Blood pressure management and renal protection: revisiting hypertensive nephropathy. J Chin Med Assoc. 2021;84(10):911–6. [[PubMed]] DOI: 1097/JCMA.0000000000000600
- Seid K, Lakew G, Bogale SK, Desalew EG, Andarge GA, Belayneh AG, et al. Prevalence of chronic kidney disease and associated factors among hypertensive patients in Ethiopia: a systematic review and meta-analysis. 2024. DOI: 10.21203/rs.3.rs-4398810/v1
- Seccia TM, Caroccia B, Calò LA. Hypertensive nephropathy: moving from classic to emerging pathogenetic mechanisms. J Hypertens. 2017;35(2):205–12. [PubMed] DOI: 1097/HJH.0000000000001170
- Kostovska I, Trajkovska KT, Labudović D, Kostovski O, Spasovski G. Urinary nephrin as an early biomarker of hypertensive nephropathy. Acta Clin Croat. 2023;62(4):635–43. [PubMed] DOI: 20471/acc.2023.62.04.8
- Tutunea-Fatan E, Arumugarajah S, Suri RS, Edgar CR, Hon I, Dikeakos JD, et al. Sensing dying cells in health and disease: the importance of kidney injury molecule-1. J Am Soc Nephrol. 2024;10:1681. [PubMed] DOI: 1681/ASN.0000000000000334
- Gohda T, Kamei N, Koshida T, Kubota M, Tanaka K, Yamashita Y, et al. Circulating kidney injury molecule‐1 as a biomarker of renal parameters in diabetic kidney disease. J Diabetes Investig. 2020;11(2):435–40. [PubMed] DOI: 1111/jdi.13139
- Ostovar T, Rezaei H, Reza JZ. Assessment of the diagnostic validities of serum NGAL, KIM-1, and L-FABP in patients with chronic kidney disease. Int J Basic Sci Med. 2020;5(2):48–53. Full Text
- Griffin BR, You Z, Noureddine L, Gitomer B, Perrenoud L, Wang W, et al. KIM-1 and kidney disease progression in autosomal dominant polycystic kidney disease: HALT-PKD results. Am J Nephrol. 2020;51(6):473–9. [PubMed] DOI: 1159/000508051
- Giavarina D, Lippi G. Blood venous sample collection: recommendations overview and a checklist to improve quality. Clin Biochem. 2017;50(10–11):568–73. [PubMed] DOI: 1016/j.clinbiochem.2017.02.021
- Miller WG, Kaufman HW, Levey AS, Straseski JA, Wilhelms KW, Yu HYE, et al. National Kidney Foundation Laboratory Engagement Working Group recommendations for implementing the CKD-EPI 2021 race-free equations. Clin Chem. 2022;68(4):511–20. [PubMed] DOI: 1093/clinchem/hvab278
- Sullivan LM. Essentials of biostatistics in public health. Burlington: Jones & Bartlett Learning; 2023.
- Hsu CN, Tain YL. Targeting the renin–angiotensin–aldosterone system to prevent hypertension and kidney disease of developmental origins. Int J Mol Sci. 2021;22(5):2298. [PubMed] DOI: 3390/ijms22052298
- Arendshorst WJ, Vendrov AE, Kumar N, Ganesh SK, Madamanchi NR. Oxidative stress in kidney injury and hypertension. Antioxidants. 2024;13(12):1454. [PubMed] DOI: 3390/antiox13121454
- Sun D, Wang J, Shao W, Wang J, Yao L, Li Z, et al. Pathogenesis and damage targets of hypertensive kidney injury. J Transl Intern Med. 2020;8(4):205–9. [PubMed] DOI: 2478/jtim-2020-0033
- Schmidt IM, Srivastava A, Sabbisetti V, McMahon GM, He J, Chen J, et al. Plasma kidney injury molecule 1 in CKD: findings from the Boston Kidney Biopsy Cohort and CRIC studies. Am J Kidney Dis. 2022;79(2):231–43. [PubMed] DOI: 1053/j.ajkd.2021.05.013
- Bansal A, Nigoskar S. Determination of serum KIM-1 in patients with chronic kidney injury. Asian J Med Sci. 2023;14(8):56–9. DOI: 71152/ajms.v14i8.3669
- Hasan KY, Al Ammar HAJ. Relevance of KIM-1 and NGAL biomarkers in the diagnosis of persistent kidney failure. Anaesth Pain Intensive Care. 2024;28(3):472–80. DOI: 35975/apic.v28i3.2463
- Alawi LF, Dhakal S, Emberesh SE, Sawant H, Hosawi A, Thanekar U, et al. Effects of angiotensin II type 1A receptor on ACE2, neprilysin and KIM-1 in 2K1C model of renovascular hypertension. Front Pharmacol. 2021;11:602985. [PubMed] DOI: 3389/fphar.2020.602985
- Nogare AL, Veronese FV, Carpio VN, Montenegro RM, Pedroso JA, Pegas KL, et al. Kidney injury molecule-1 expression in kidney transplants with interstitial fibrosis and tubular atrophy. BMC Nephrol. 2015;16(1):1–7. [PubMed] DOI: 1186/s12882-015-0011-y
- Lee SH, Kim KH, Lee SM, Park SJ, Lee S, Cha RH, et al. STAT3 blockade ameliorates LPS-induced kidney injury through macrophage-driven inflammation. Cell Commun Signal. 2024;22(1):476. [PubMed] DOI: 1186/s12964-024-01841-1
- Humphreys BD, Xu F, Sabbisetti V, Grgic I, Naini SM, Wang N, et al. Chronic epithelial kidney injury molecule-1 expression causes murine kidney fibrosis. J Clin Invest. 2013;123(9):4023–35. [PubMed] DOI: 1172/JCI45361