Rayan A. Qutob, MD*
Author’s affiliation:
*Rayan A Qutob, Department of Internal Medicine, College of Medicine, Imam Mohammad Ibn Saud Islamic University, Riyadh, Saudi Arabia; Email: Dr.rayan.qutob@gmail.com
Correspondence: Dr Rayan A. Qutob, Email: Dr.rayan.qutob@gmail.com, Phone: 00966500175489
Methylene blue is a broad and potent therapeutic substance that is effective in treating various medical conditions in the Intensive Care Unit (ICU). Methylene blue (MB) has a multitarget mode of action that involves inhibiting nitric oxide synthase and guanylate cyclase, as well as serving as a redox agent. This makes it beneficial in treating various disorders in the intensive care unit, including septic and vasoplegic shock, methemoglobinemia, and Ifosfamide-induced encephalopathy. This comprehensive narrative review provides current evidence regarding MB use in the ICU setting, constituents of the chemical, clinical applications, its efficacy, safety, dosing, guidelines, actual recommendations for its usage, and future research directions for use in ICU. MB has demonstrated enhanced results for people who are critically unwell. However, when using MB as a supplementary medication, it is important to carefully examine the dosage and treatment schedule in order to obtain the best possible clinical outcome. This includes closely monitoring and making necessary adjustments, especially for patients with conditions such as renal or hepatic impairment, as well as Glucose-6-Phosphate Dehydrogenase (G6PD) deficiency. Further research and clinical trials are essential for expanding the understanding and application of MB in critical care.
Abbreviations: CPB: cardiopulmonary bypass, G6PD: Glucose-6-Phosphate Dehydrogenase, ICU: Intensive Care Unit, iNOS: inducible nitric oxide synthase, MAOIs: monoamine oxidase inhibitors, MB: Methylene blue, NADH: nicotinamide adenine dinucleotide hydrogen, NO: Nitric oxide, SNRIs: serotonin and norepinephrine reuptake inhibitors, SSRIs: selective serotonin reuptake inhibitors, IV: intravenous, TCAs: tricyclic antidepressants
Keywords: Intensive care unit; Methylene blue; Review
Citation: Qutob RA. Methylene blue in the intensive care unit: A literature review. Anaesth. pain intensive care 2025;29(3):407-417. DOI: 10.35975/apic.v29i3.2829
Received: February 03, 2025; Revised: February 28, 2025; Accepted: March 05, 2025
Heinrich Caro, a German chemist, first synthesized Methylene Blue (MB) in 1876 as a dye from aniline, to dye cotton.1 MB belongs to the phenothiazines family and is a cationic dye.2,3 It is characterized by its tricyclic phenothiazine structure, solubilized in some organic solvents and water,2 and is acknowledged as the first synthetic medicine applied to humans.4 MB has been widely utilized for biological staining in hematology, bacteriology, and histology.3
Paul Ehrlich found that MB could target the malarial parasite during the 1890s. Its first applications resulted in the inclusion of MB in malaria treatments due to growing resistant to different treatments.5-7 Ehrlich's description of the unique properties of MB led to its widespread use in the treatment of a wide range of disorders.8,9
The clinical utility of MB exceeds its historical applications because of its antimicrobial, neuroprotective, antioxidant, and anti-inflammatory properties.10-15 MB is considered a multifunctional drug with postoperative,16,17 curative,18 diagnostic,19,20 and preventive use.13,15 Recognizing its importance in a basic health system, the World Health Organization (WHO) includes MB on its ‘List of Essential Medicines’.21
In the intensive care unit (ICU), MB is utilized for multiple ICU applications.22 MB is even utilized in regulating excessive blood circulation in liver cirrhosis, therapeutic use against low oxygen levels, and addressing low blood pressure associated with different medical conditions.23 The Food and Drug Administration (FDA) in the United States has approved MB for a variety of indications, including cyanide poisoning, ifosfamide-induced encephalopathy, and vasoplegic syndrome.24 Also, a prior meta-analysis has shown that the use of MB can enhance survival in perioperative and critically ill patients and may also diminish the length of hospital stay in the ICU.25
The wide range of applications and critical conditions treated in the ICU demand good knowledge of the evidence supporting the use of MB. Therefore, this literature review will investigate available current evidence regarding MB use in the ICU setting, constituents of the chemical, clinical applications, its efficacy, safety, dosing, guidelines, actual recommendations for its usage, and future research directions for its use in ICU.
This narrative review was performed by collecting original research articles and reviews on the use of MB in the ICU. Articles published in peer-reviewed scientific journals were included. Articles were excluded if they were not written in English or published in peer-reviewed scientific journals.
2.1. Search strategy
Literature searches were performed in PubMed and Web of Science databases from the date of database inception through June 2024. The search included the following keywords: ‘methylene blue’ and ‘intensive care unit’.
2.2. Study selection
Relevant articles were identified through our aforementioned search strategy and selected articles were screened based on the title and abstract. After that, all included articles were carefully discussed in the present review. A total of 104 articles were finally selected and discussed in the review. This narrative review presented pharmacokinetic properties of MB, its clinical indications in ICU, its safety and adverse effects, and dosing and administration procedures.
2.3. Pharmacokinetic Properties
MB shows several important pharmacokinetic properties necessary for its therapeutic use. The half-life of terminal elimination varies from 5 to 14 hours,26-29 depending on the dose and formulation: for instance, prior studies noted an approximate half-life of 5 to 6.5 hours,26-28 while another study determined a half-life of about 14 hours.29 When given orally, MB has high absolute bioavailability, with figures around 48.4% to 96.2%.30
After oral administration, MB is well absorbed; absorption rates range between 53% and 97%.28 The variability of these absorption rates might be due to inter-subject variability and the specific conditions under which the drug was administered. After being absorbed, MB is well distributed in the body,31 and crosses the blood-brain barrier32 due to its lipophilic and cationic properties.1 MB is extensively metabolized in the liver33 and reduced to leukomethylene blue.28 It is excreted in the urine, bile, and feces.28,34
2.4. Clinical indications in ICU
MB is used in numerous clinical situations in the ICU. We review the main conditions where MB has shown effective therapeutic potential.
2.4.1. Methemoglobinemia
Methemoglobinemia is a condition that can be either present from birth or acquired, with life-threatening characteristics.35 It is an uncommon condition wherein the divalent ferrous iron (Fe2+) in hemoglobin is oxidated to ferric iron (Fe3+) in methemoglobin.36 The Fe3+ state induces allosteric transitions that allow irreversible binding with oxygen.37 The diagnosis of methemoglobinemia generally occurs when the concentration of methemoglobin in the blood is more than the normal range, which is 1% to 3%.38,39 This clinically appears with symptoms such as characteristic "chocolate-colored" blood, hypoxia, and cyanosis of the extremities and the lips.40 Severe cases might result in metabolic acidosis and possibly death.22 MB is the antidote of choice for treating symptomatic methemoglobinemia.41 MB acts rapidly within red blood cells to form leukomethylene blue, a reducing agent that converts the Fe3+ in methemoglobin back to its oxygen-carrying iron state Fe2+.42
2.4.2. Septic shock and vasoplegia
Septic shock is the most serious subset of sepsis (a life-threatening condition);43,44 it is characterized by persistent low systemic vascular and low blood pressure (vasoplegia) despite effective fluid resuscitation and necessitating the administration of vasopressors to keep organ perfusion pressure.45,46 Septic shock markedly elevates the mortality risk45 and accounts for most of the non-coronary mortality and morbidity in ICU patients.47,48 Nitric oxide (NO) release is a critical contributor to the cardiovascular dysfunction associated with septic shock.35 MB is effective in treating septic shock by inhibiting the enzyme guanylate cyclase, which nitric oxide targets to cause endothelial relaxation.49 MB blocks soluble guanylate cyclase (sGC) in the smooth muscle cells of the blood vessels and inducible nitricoxide synthase (iNOS),50,51 in turn, enhancing cardiac function and arterial pressure.49
2.4.3. Vasoplegic syndrome and cardiovascular surgeries
Vasoplegic syndrome is a vasodilatory shock commonly seen after patients undergoing major cardiovascular surgeries, specifically cardiopulmonary bypass (CPB); it may affect up to 50% of patients.52-57 Compared with other cardiac surgical patients, patients with vasoplegia sydrome more frequently exhibit increased mortality after surgery, extended stays in the ICU, and higher rates of postoperative complications.58-62 MB was noticed to be an effective treatment of vasoplegic syndrome in cardiovascular surgical settings. The efficiency of MB has been demonstrated to correlate with its ability to block the NO-induced vasodilation pathway and, consequently, decrease the intensity of systemic inflammatory reactions.63,64 The early use of MB decreases the incidence of operative mortality and postoperative kidney failure.65
2.4.4. Ifosfamide-induced encephalopathy
Ifosfamide is an alkylating agent and is used as therapy for multiple types of solid tumors.66,67 Due to the incidence of specific adverse consequences, Ifosfamide usage may be restricted. One such intense adverse consequence is central nervous system depression, an adverse effect called ifosfamide-induced encephalopathy67 (an acute neurologic complication that develops during or immediately after ifosfamide infusion).68 Even though it is reversible under most usual circumstances, it is a severe and life-threatening complication that requires immediate intervention.68 MB has been reported as an effective therapy for ifosfamide-induced encephalopathy. A prior review has revealed that MB is an effective ifosfamide-induced encephalopathy therapy for both prophylactic and treatment.67 MB serves as a substitute electron acceptor, thus reversing the nicotinamide adenine dinucleotide hydrogen (NADH) blockage of hepatic gluconeogenesis.69 It also prevents the multiple activities of amine oxidases, which prevents the production of chloroacetaldehyde (toxic metabolite).69 In addition, it prevents the conversion of chloroethylamine to chloroacetaldehyde.69
2.5. Safety and Adverse Effects
MB is safe when taken at doses of less than 2 mg/kg (therapeutic dose) but has many side effects at doses higher than 7 mg/kg.35 Discoloration of body fluids is the most common of MB side effects.70,71
Other commonly experienced side effects include gastrointestinal effects such as vomiting, and nausea.22 Patients may also experience confusion, headaches, fever, or dizziness.72,73 Figure 1 below presents the most common side effects related to MB. 74
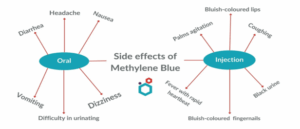
Figure 1: Side effects of methylene blue
More serious adverse effects of MB include hemolytic anemia in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency, neurotoxicity, methemoglobinemia, serotonin syndrome, phototoxicity, anaphylactic reaction, skin discoloration, refractory hypotension, hypertension, dyspnea, and chest pain.28,75-81 Furthermore, MB may also cause false low oxygen saturation values due to interference with the light emission of pulse oximeters.28 Hemolytic anemia, phototoxicity, pulmonary edema, respiratory depression, and hyperbilirubinemia are among the adverse effects reported with the use of methylene blue in neonates.82,83
MB administration is contraindicated in patients with Heinz body anemia, in individuals with severe renal insufficiency, in patients with G6PD because of the risk of severe hemolysis, in patients with a known hypersensitivity to the drug,28,71,75 or during pregnancy as the FDA has classified it as pregnancy category X (because of fetal death and intestinal atresia after intra-amniotic injection in the 2nd trimester).84
Moreover, combining MB with serotonergic drugs (like tricyclic antidepressants (TCAs), monoamine oxidase inhibitors (MAOIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), and selective serotonin reuptake inhibitors (SSRIs)) could lead to serotonin syndrome, therefore, it is contraindicated in patients who are taking serotonergic drugs.71
Maintenance of monitoring when using MB is key to both efficacy and minimization of potential side effects. Monitoring parameters include levels of methemoglobin, renal, hepatic, and pulmonary functions.
4.1. Methemoglobin Levels
Monitoring of methemoglobin levels is necessary in the treatment of methemoglobinemia with MB. Although continuous MB infusion has been used to prevent methemoglobinemia's rebound.85 This rebound methemoglobinemia is evidenced by an increase in methemoglobin levels following the end of therapy, secondary to the reverse of the reduction reaction,37 this should generally be discouraged as standard therapy.86 The proper recommended treatment method is the administration of intermittent bolus doses of MB.87 If methemoglobinemia exacerbates following MB therapy, an exchange transfusion should be performed urgently.37
4.2. Renal Function
MB use should be cautioned in cases of renal failure because it has been reported to cause a reduction in renal blood flow.35 It should be utilized with caution in severe renal insufficiency patients.88 Intensive monitoring of renal function should be maintained to ensure that the dose is well-adjusted to avoid further renal impairment.
4.3. Hepatic Function
Since the liver is the primary site of metabolism for MB, it is imperative to closely monitor for potential drug interactions and toxicities in MB-treated patients suffering from hepatic insufficiency.89 Monitoring the patients is also required for a protracted period post-MB to detect any delayed toxicities.89
4.4. Pulmonary Function
Maintaining proper monitoring of pulmonary function is very important, particularly in severe pulmonary hypertension patients, because MB can significantly raise the pressure in the pulmonary artery, which can compromise gas exchange.90 Patients with pre-existing pulmonary disease should be monitored to prevent potentially harmful effects.
Finally, as MB adversely affects the cardiovascular system91,92 and central nervous system,72 assessment of hemodynamic and neurological parameters for patients may also be required.
The dosing of MB for various ICU conditions has been evaluated in several studies (Table 1). The standard initial dose for methemoglobinemia is 1-2 mg/kg intravenous (IV) over 3 to 5 minutes; followed by 1 mg/kg within 30 minutes if methemoglobin does not resolve.93 For septic shock, MB is administered at 2 mg/kg IV bolus over 15 minutes as an initial dose and then is given as a continuous infusion.94 In vasoplegic syndrome, MB is administered at 1.5-2 mg /kg IV over 20 minutes to 1 hour.59,81,95-97 For cardiac surgery, 2 mg/kg was added to the CPB prime and followed with a constant infusion.98 In ifosfamide-induced encephalopathy, MB treats the condition effectively at 50 mg every 4 hours IV/orally (PO).67 MB across conditions exhibits a favorable safety profile with careful monitoring for potential adverse effects.
Several researchers have discussed the effectiveness, safety, and clinical outcomes of MB among critically ill patients admitted to the ICU with different conditions (Table 2).
A prior study reported that MB was an effective treatment for methemoglobinemia.28 Other studies have reported that MB in patients with septic shock improved hemodynamic stability, reduced vasopressor requirement, and might or might not have led to an increased survival rate.49,94,99,100 Studies reported that MB improved the hemodynamic parameters significantly in cardiac surgery and vasoplegic syndrome, reduced morbidity, improved patient outcomes, decreased use of high dose vasopressors, and had shorter lengths of stay in the ICU.97,101-103 Studies reported for ifosfamide-induced encephalopathy report that MB had prophylactic and treatment effects that lead to a rapid resolution and improved clinical outcomes.67,104 Overall, with proper monitoring and recommended dose alterations, MB was found to be safe.
There are some limitations to this research. First, this study did not include all relevant literature, and there may be missing relevant articles on the use of MB in ICU settings. Therefore, the author believes that more high-quality research on the use of MB in ICU settings is needed.
MB is a multifaceted, effective therapeutic agent in several therapeutic areas within the ICU. A multitarget mechanism of action of MB included inhibition of nitric oxide synthase and guanylate cyclase and acting as a redox agent, making it effective in the management of many ICU conditions, such as septic and vasoplegic shock, methemoglobinemia, and Ifosfamide-induced encephalopathy. MB has shown improved outcomes for critically ill patients. Still, utilizing MB as an adjunct in treatment should consider dosing and regimen practices to achieve optimal clinical benefit with monitoring and adjustments, particularly for specific patients (e.g., renal/hepatic impairment and G6PD deficiency). More research and clinical trials are fundamental to advancing the knowledge and the use of MB in critical care.
9. Conflict of Interest
None declared by the authors
10. Ethics considerations
Ethical approval not required.
11. Author’s contributions
Rayan A. Qutob is the sole author of this paper, responsible for Conceptualization, investigation, methodology, resources, validation, drafting original draft, review and editing.
Author’s affiliation:
*Rayan A Qutob, Department of Internal Medicine, College of Medicine, Imam Mohammad Ibn Saud Islamic University, Riyadh, Saudi Arabia; Email: Dr.rayan.qutob@gmail.com
Correspondence: Dr Rayan A. Qutob, Email: Dr.rayan.qutob@gmail.com, Phone: 00966500175489
ABSTRACT
Methylene blue is a broad and potent therapeutic substance that is effective in treating various medical conditions in the Intensive Care Unit (ICU). Methylene blue (MB) has a multitarget mode of action that involves inhibiting nitric oxide synthase and guanylate cyclase, as well as serving as a redox agent. This makes it beneficial in treating various disorders in the intensive care unit, including septic and vasoplegic shock, methemoglobinemia, and Ifosfamide-induced encephalopathy. This comprehensive narrative review provides current evidence regarding MB use in the ICU setting, constituents of the chemical, clinical applications, its efficacy, safety, dosing, guidelines, actual recommendations for its usage, and future research directions for use in ICU. MB has demonstrated enhanced results for people who are critically unwell. However, when using MB as a supplementary medication, it is important to carefully examine the dosage and treatment schedule in order to obtain the best possible clinical outcome. This includes closely monitoring and making necessary adjustments, especially for patients with conditions such as renal or hepatic impairment, as well as Glucose-6-Phosphate Dehydrogenase (G6PD) deficiency. Further research and clinical trials are essential for expanding the understanding and application of MB in critical care.
Abbreviations: CPB: cardiopulmonary bypass, G6PD: Glucose-6-Phosphate Dehydrogenase, ICU: Intensive Care Unit, iNOS: inducible nitric oxide synthase, MAOIs: monoamine oxidase inhibitors, MB: Methylene blue, NADH: nicotinamide adenine dinucleotide hydrogen, NO: Nitric oxide, SNRIs: serotonin and norepinephrine reuptake inhibitors, SSRIs: selective serotonin reuptake inhibitors, IV: intravenous, TCAs: tricyclic antidepressants
Keywords: Intensive care unit; Methylene blue; Review
Citation: Qutob RA. Methylene blue in the intensive care unit: A literature review. Anaesth. pain intensive care 2025;29(3):407-417. DOI: 10.35975/apic.v29i3.2829
Received: February 03, 2025; Revised: February 28, 2025; Accepted: March 05, 2025
1. INTRODUCTION
Heinrich Caro, a German chemist, first synthesized Methylene Blue (MB) in 1876 as a dye from aniline, to dye cotton.1 MB belongs to the phenothiazines family and is a cationic dye.2,3 It is characterized by its tricyclic phenothiazine structure, solubilized in some organic solvents and water,2 and is acknowledged as the first synthetic medicine applied to humans.4 MB has been widely utilized for biological staining in hematology, bacteriology, and histology.3
Paul Ehrlich found that MB could target the malarial parasite during the 1890s. Its first applications resulted in the inclusion of MB in malaria treatments due to growing resistant to different treatments.5-7 Ehrlich's description of the unique properties of MB led to its widespread use in the treatment of a wide range of disorders.8,9
The clinical utility of MB exceeds its historical applications because of its antimicrobial, neuroprotective, antioxidant, and anti-inflammatory properties.10-15 MB is considered a multifunctional drug with postoperative,16,17 curative,18 diagnostic,19,20 and preventive use.13,15 Recognizing its importance in a basic health system, the World Health Organization (WHO) includes MB on its ‘List of Essential Medicines’.21
In the intensive care unit (ICU), MB is utilized for multiple ICU applications.22 MB is even utilized in regulating excessive blood circulation in liver cirrhosis, therapeutic use against low oxygen levels, and addressing low blood pressure associated with different medical conditions.23 The Food and Drug Administration (FDA) in the United States has approved MB for a variety of indications, including cyanide poisoning, ifosfamide-induced encephalopathy, and vasoplegic syndrome.24 Also, a prior meta-analysis has shown that the use of MB can enhance survival in perioperative and critically ill patients and may also diminish the length of hospital stay in the ICU.25
The wide range of applications and critical conditions treated in the ICU demand good knowledge of the evidence supporting the use of MB. Therefore, this literature review will investigate available current evidence regarding MB use in the ICU setting, constituents of the chemical, clinical applications, its efficacy, safety, dosing, guidelines, actual recommendations for its usage, and future research directions for its use in ICU.
2. METHODOLOGY
This narrative review was performed by collecting original research articles and reviews on the use of MB in the ICU. Articles published in peer-reviewed scientific journals were included. Articles were excluded if they were not written in English or published in peer-reviewed scientific journals.
2.1. Search strategy
Literature searches were performed in PubMed and Web of Science databases from the date of database inception through June 2024. The search included the following keywords: ‘methylene blue’ and ‘intensive care unit’.
2.2. Study selection
Relevant articles were identified through our aforementioned search strategy and selected articles were screened based on the title and abstract. After that, all included articles were carefully discussed in the present review. A total of 104 articles were finally selected and discussed in the review. This narrative review presented pharmacokinetic properties of MB, its clinical indications in ICU, its safety and adverse effects, and dosing and administration procedures.
2.3. Pharmacokinetic Properties
MB shows several important pharmacokinetic properties necessary for its therapeutic use. The half-life of terminal elimination varies from 5 to 14 hours,26-29 depending on the dose and formulation: for instance, prior studies noted an approximate half-life of 5 to 6.5 hours,26-28 while another study determined a half-life of about 14 hours.29 When given orally, MB has high absolute bioavailability, with figures around 48.4% to 96.2%.30
After oral administration, MB is well absorbed; absorption rates range between 53% and 97%.28 The variability of these absorption rates might be due to inter-subject variability and the specific conditions under which the drug was administered. After being absorbed, MB is well distributed in the body,31 and crosses the blood-brain barrier32 due to its lipophilic and cationic properties.1 MB is extensively metabolized in the liver33 and reduced to leukomethylene blue.28 It is excreted in the urine, bile, and feces.28,34
2.4. Clinical indications in ICU
MB is used in numerous clinical situations in the ICU. We review the main conditions where MB has shown effective therapeutic potential.
2.4.1. Methemoglobinemia
Methemoglobinemia is a condition that can be either present from birth or acquired, with life-threatening characteristics.35 It is an uncommon condition wherein the divalent ferrous iron (Fe2+) in hemoglobin is oxidated to ferric iron (Fe3+) in methemoglobin.36 The Fe3+ state induces allosteric transitions that allow irreversible binding with oxygen.37 The diagnosis of methemoglobinemia generally occurs when the concentration of methemoglobin in the blood is more than the normal range, which is 1% to 3%.38,39 This clinically appears with symptoms such as characteristic "chocolate-colored" blood, hypoxia, and cyanosis of the extremities and the lips.40 Severe cases might result in metabolic acidosis and possibly death.22 MB is the antidote of choice for treating symptomatic methemoglobinemia.41 MB acts rapidly within red blood cells to form leukomethylene blue, a reducing agent that converts the Fe3+ in methemoglobin back to its oxygen-carrying iron state Fe2+.42
2.4.2. Septic shock and vasoplegia
Septic shock is the most serious subset of sepsis (a life-threatening condition);43,44 it is characterized by persistent low systemic vascular and low blood pressure (vasoplegia) despite effective fluid resuscitation and necessitating the administration of vasopressors to keep organ perfusion pressure.45,46 Septic shock markedly elevates the mortality risk45 and accounts for most of the non-coronary mortality and morbidity in ICU patients.47,48 Nitric oxide (NO) release is a critical contributor to the cardiovascular dysfunction associated with septic shock.35 MB is effective in treating septic shock by inhibiting the enzyme guanylate cyclase, which nitric oxide targets to cause endothelial relaxation.49 MB blocks soluble guanylate cyclase (sGC) in the smooth muscle cells of the blood vessels and inducible nitricoxide synthase (iNOS),50,51 in turn, enhancing cardiac function and arterial pressure.49
2.4.3. Vasoplegic syndrome and cardiovascular surgeries
Vasoplegic syndrome is a vasodilatory shock commonly seen after patients undergoing major cardiovascular surgeries, specifically cardiopulmonary bypass (CPB); it may affect up to 50% of patients.52-57 Compared with other cardiac surgical patients, patients with vasoplegia sydrome more frequently exhibit increased mortality after surgery, extended stays in the ICU, and higher rates of postoperative complications.58-62 MB was noticed to be an effective treatment of vasoplegic syndrome in cardiovascular surgical settings. The efficiency of MB has been demonstrated to correlate with its ability to block the NO-induced vasodilation pathway and, consequently, decrease the intensity of systemic inflammatory reactions.63,64 The early use of MB decreases the incidence of operative mortality and postoperative kidney failure.65
2.4.4. Ifosfamide-induced encephalopathy
Ifosfamide is an alkylating agent and is used as therapy for multiple types of solid tumors.66,67 Due to the incidence of specific adverse consequences, Ifosfamide usage may be restricted. One such intense adverse consequence is central nervous system depression, an adverse effect called ifosfamide-induced encephalopathy67 (an acute neurologic complication that develops during or immediately after ifosfamide infusion).68 Even though it is reversible under most usual circumstances, it is a severe and life-threatening complication that requires immediate intervention.68 MB has been reported as an effective therapy for ifosfamide-induced encephalopathy. A prior review has revealed that MB is an effective ifosfamide-induced encephalopathy therapy for both prophylactic and treatment.67 MB serves as a substitute electron acceptor, thus reversing the nicotinamide adenine dinucleotide hydrogen (NADH) blockage of hepatic gluconeogenesis.69 It also prevents the multiple activities of amine oxidases, which prevents the production of chloroacetaldehyde (toxic metabolite).69 In addition, it prevents the conversion of chloroethylamine to chloroacetaldehyde.69
2.5. Safety and Adverse Effects
MB is safe when taken at doses of less than 2 mg/kg (therapeutic dose) but has many side effects at doses higher than 7 mg/kg.35 Discoloration of body fluids is the most common of MB side effects.70,71
Other commonly experienced side effects include gastrointestinal effects such as vomiting, and nausea.22 Patients may also experience confusion, headaches, fever, or dizziness.72,73 Figure 1 below presents the most common side effects related to MB. 74

Figure 1: Side effects of methylene blue
More serious adverse effects of MB include hemolytic anemia in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency, neurotoxicity, methemoglobinemia, serotonin syndrome, phototoxicity, anaphylactic reaction, skin discoloration, refractory hypotension, hypertension, dyspnea, and chest pain.28,75-81 Furthermore, MB may also cause false low oxygen saturation values due to interference with the light emission of pulse oximeters.28 Hemolytic anemia, phototoxicity, pulmonary edema, respiratory depression, and hyperbilirubinemia are among the adverse effects reported with the use of methylene blue in neonates.82,83
3. CONTRAINDICATIONS
MB administration is contraindicated in patients with Heinz body anemia, in individuals with severe renal insufficiency, in patients with G6PD because of the risk of severe hemolysis, in patients with a known hypersensitivity to the drug,28,71,75 or during pregnancy as the FDA has classified it as pregnancy category X (because of fetal death and intestinal atresia after intra-amniotic injection in the 2nd trimester).84
Moreover, combining MB with serotonergic drugs (like tricyclic antidepressants (TCAs), monoamine oxidase inhibitors (MAOIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), and selective serotonin reuptake inhibitors (SSRIs)) could lead to serotonin syndrome, therefore, it is contraindicated in patients who are taking serotonergic drugs.71
4. MONITORING
Maintenance of monitoring when using MB is key to both efficacy and minimization of potential side effects. Monitoring parameters include levels of methemoglobin, renal, hepatic, and pulmonary functions.
4.1. Methemoglobin Levels
Monitoring of methemoglobin levels is necessary in the treatment of methemoglobinemia with MB. Although continuous MB infusion has been used to prevent methemoglobinemia's rebound.85 This rebound methemoglobinemia is evidenced by an increase in methemoglobin levels following the end of therapy, secondary to the reverse of the reduction reaction,37 this should generally be discouraged as standard therapy.86 The proper recommended treatment method is the administration of intermittent bolus doses of MB.87 If methemoglobinemia exacerbates following MB therapy, an exchange transfusion should be performed urgently.37
4.2. Renal Function
MB use should be cautioned in cases of renal failure because it has been reported to cause a reduction in renal blood flow.35 It should be utilized with caution in severe renal insufficiency patients.88 Intensive monitoring of renal function should be maintained to ensure that the dose is well-adjusted to avoid further renal impairment.
4.3. Hepatic Function
Since the liver is the primary site of metabolism for MB, it is imperative to closely monitor for potential drug interactions and toxicities in MB-treated patients suffering from hepatic insufficiency.89 Monitoring the patients is also required for a protracted period post-MB to detect any delayed toxicities.89
4.4. Pulmonary Function
Maintaining proper monitoring of pulmonary function is very important, particularly in severe pulmonary hypertension patients, because MB can significantly raise the pressure in the pulmonary artery, which can compromise gas exchange.90 Patients with pre-existing pulmonary disease should be monitored to prevent potentially harmful effects.
Finally, as MB adversely affects the cardiovascular system91,92 and central nervous system,72 assessment of hemodynamic and neurological parameters for patients may also be required.
5. DOSING AND ADMINISTRATION
The dosing of MB for various ICU conditions has been evaluated in several studies (Table 1). The standard initial dose for methemoglobinemia is 1-2 mg/kg intravenous (IV) over 3 to 5 minutes; followed by 1 mg/kg within 30 minutes if methemoglobin does not resolve.93 For septic shock, MB is administered at 2 mg/kg IV bolus over 15 minutes as an initial dose and then is given as a continuous infusion.94 In vasoplegic syndrome, MB is administered at 1.5-2 mg /kg IV over 20 minutes to 1 hour.59,81,95-97 For cardiac surgery, 2 mg/kg was added to the CPB prime and followed with a constant infusion.98 In ifosfamide-induced encephalopathy, MB treats the condition effectively at 50 mg every 4 hours IV/orally (PO).67 MB across conditions exhibits a favorable safety profile with careful monitoring for potential adverse effects.
| Table 1: Summary MB doses used in ICU conditions. | |||
| Condition | Initial Dose | Repeat Dose/Infusion | References |
| Methemoglobinemia | 1-2 mg/kg IV over 3-5 minutes | 1 mg/kg within 30 minutes if needed (methemoglobin does not resolve) | Wright et al., 199993 |
| Septic shock | 2 mg/kg IV bolus over 15 minutes | 1-h infusions of 0.25, 0.5, 1, and 2 mg/kg/hr, respectively | Kirov et al., 200194 |
| Vasoplegic syndrome | 1.5-2 mg /kg IV over 20 minutes to 1hr | N/A | Egi et al., 2007, Kofidis et al., 2001, Levin et al., 2004, Leyh et al., 2003, Ozal et al., 200559,81,95-97 |
| Cardiac surgery (During CPB) | 2 mg/kg added to CPB prime | Continuous infusion at 0.25-2 mg/kg/hr | Grayling and Deakin, 200398 |
| Ifosfamide-induced Encephalopathy | 50 mg every 4 hrs IV/PO | Prophylaxis 50 mg every 6 hours IV/PO | Pelgrims et al., 200067 |
| IV, intravenous; N/A, not available; hr, hour; CPB, cardiopulmonary bypass; PO, orally. | |||
6. LITERATURE REVIEW
Several researchers have discussed the effectiveness, safety, and clinical outcomes of MB among critically ill patients admitted to the ICU with different conditions (Table 2).
| Table 2: Summary of studies on MB use in ICU. | |||
| Study | ICU Condition | Efficacy and Clinical Outcomes | Safety |
| Clifton and Leikin, 200328 | Methemoglobinemia | Reduce back to hemoglobin | Generally safe at IV dose of 1 to 2 mg/kg. |
| Preiser et al., 199549 | Septic shock | Increase mean arterial pressure (MAP). Improve cardiac function. Does not raise cellular oxygen. |
No adverse effect was reported. |
| Kirov et al., 200194 | Septic shock | MB reduced the plasma concentration of nitrites/ nitrates, the body temperature, and the catecholamines requirement. Increased survival. |
No significant adverse effects were reported. |
| Kwok and Howes, 200699 | Septic shock | Increased systemic vascular resistance. Increased MAP. Unknown effect on mortality and oxygen delivery. |
No adverse effect was reported. |
| Ballarin et al., 2024100 | Septic shock | Reduced days on mechanical ventilation, length of ICU stay, and time to vasopressor discontinuation. | No abnormal levels of methemoglobinemia were reported. |
| Zhao et al., 2022101 | Vasodilatory shock | MB along with vasopressors improved survival, improved hemodynamics, decreased lactate levels, and reduced vasopressor requirements. | No serious side effects were reported. |
| Huang et al., 2024102 | Vasodilatory shock | Reduced the length of hospital stay, length of ICU stays, and duration of mechanical ventilation. Use of MB may not decrease mortality. |
N/A |
| Levin et al., 200497 | Vasoplegic syndrome after heart surgery | Mortality reduction. Shorten the length of vasoplegia. |
No adverse effects, except for blue or green coloring of the urine. |
| Evora et al., 2009103 | Vasoplegic syndrome in heart surgery | Facilitate the norepinephrine vasoconstrictor effect Cheapest and best option. |
Safest option. |
| Park et al., 2005104 | Ifosfamide-induced encephalopathy | The patient recovered completely. | N/A |
| Pelgrims et al., 200067 | Ifosfamide-induced encephalopathy | MB is an effective therapy (shortens the duration of ifosfamide-induced encephalopathy). MB may also be used for ifosfamide-induced encephalopathy prevention. |
N/A |
| IV, intravenous; MAP, mean arterial pressure; MB, methylene blue; N/A, not available. | |||
A prior study reported that MB was an effective treatment for methemoglobinemia.28 Other studies have reported that MB in patients with septic shock improved hemodynamic stability, reduced vasopressor requirement, and might or might not have led to an increased survival rate.49,94,99,100 Studies reported that MB improved the hemodynamic parameters significantly in cardiac surgery and vasoplegic syndrome, reduced morbidity, improved patient outcomes, decreased use of high dose vasopressors, and had shorter lengths of stay in the ICU.97,101-103 Studies reported for ifosfamide-induced encephalopathy report that MB had prophylactic and treatment effects that lead to a rapid resolution and improved clinical outcomes.67,104 Overall, with proper monitoring and recommended dose alterations, MB was found to be safe.
7. LIMITATIONS
There are some limitations to this research. First, this study did not include all relevant literature, and there may be missing relevant articles on the use of MB in ICU settings. Therefore, the author believes that more high-quality research on the use of MB in ICU settings is needed.
8. CONCLUSION
MB is a multifaceted, effective therapeutic agent in several therapeutic areas within the ICU. A multitarget mechanism of action of MB included inhibition of nitric oxide synthase and guanylate cyclase and acting as a redox agent, making it effective in the management of many ICU conditions, such as septic and vasoplegic shock, methemoglobinemia, and Ifosfamide-induced encephalopathy. MB has shown improved outcomes for critically ill patients. Still, utilizing MB as an adjunct in treatment should consider dosing and regimen practices to achieve optimal clinical benefit with monitoring and adjustments, particularly for specific patients (e.g., renal/hepatic impairment and G6PD deficiency). More research and clinical trials are fundamental to advancing the knowledge and the use of MB in critical care.
9. Conflict of Interest
None declared by the authors
10. Ethics considerations
Ethical approval not required.
11. Author’s contributions
Rayan A. Qutob is the sole author of this paper, responsible for Conceptualization, investigation, methodology, resources, validation, drafting original draft, review and editing.
12. REFERENCES
- Oz M, Lorke DE, Hasan M, Petroianu GA. Cellular and molecular actions of Methylene Blue in the nervous system. Med Res Rev. 2011;31(1):93-117. PubMed DOI: 10.1002/med.20177
- Silva M, Caro V, Guzmán C, Perry G, Areche C, Cornejo A. Chapter 1 - α-Synuclein and tau, two targets for dementia. In: Atta ur R, ed. Studies in Natural Products Chemistry. Vol 67. Elsevier; 2020:1-25. DOI: 10.1016/B978-0-12-819483-6.00001-1
- Kristiansen JE. Dyes, antipsychotic drugs, and antimicrobial activity. Fragments of a development, with special reference to the influence of Paul Ehrlich. Dan Med Bull. 1989;36(2):178-185. PubMed
- Saha BK, Burns SL. The Story of Nitric Oxide, Sepsis and Methylene Blue: A Comprehensive Pathophysiologic Review. Am J Med Sci. 2020;360(4):329-337. PubMed DOI: 10.1016/j.amjms.2020.06.007
- Wainwright M, Amaral L. The phenothiazinium chromophore and the evolution of antimalarial drugs. Trop Med Int Health. 2005;10(6):501-511. PubMed DOI: 10.1111/j.1365-3156.2005.01417.x
- Schirmer RH, Coulibaly B, Stich A, Scheiwein M, Merkle H, Eubel J, et al. Methylene blue as an antimalarial agent. Redox Rep. 2003;8(5):272-275. PubMed DOI: 10.1179/135100003225002899
- Krafts K, Hempelmann E, Skórska-Stania A. From methylene blue to chloroquine: a brief review of the development of an antimalarial therapy. Parasitol Res. 2012;111(1):1-6. PubMed DOI: 10.1007/s00436-012-2886-x
- The Collected Papers of Paul Ehrlich. Proc R Soc Med. 1957;50(3):210. DOI: 10.1177/003591575705000326. PMC: 1889126.
- Deeds F, Stockton A, THOMAS JO. Studies on phenothiazine VIII. Antiseptic value of phenothiazine in urinary tract infections. J Pharmacol Experimental Therapeutics. 1939;65(4):353-371. DOI: 10.1016/S0022-3565(25)08193-5
- Schirmer RH, Adler H, Pickhardt M, Mandelkow E. "Lest we forget you--methylene blue...". Neurobiol Aging. 2011;32(12):2325.e2327-2316. PubMed DOI: 10.1016/j.neurobiolaging.2010.12.012
- Bountogo M, Zoungrana A, Coulibaly B, Klose C, Mansmann U, Mockenhaupt FP, et al. Efficacy of methylene blue monotherapy in semi-immune adults with uncomplicated falciparum malaria: a controlled trial in Burkina Faso. Trop Med Int Health. 2010;15(6):713-717. PubMed DOI: 10.1111/j.1365-3156.2010.02526.x
- Liu JJ, Lu L, Hu FQ, et al. Methylene blue attenuates renal ischemia-reperfusion injury by negative regulation of NLRP3 signaling pathway. Eur Rev Med Pharmacol Sci. 2018;22(9):2847-2853. PubMed DOI: 10.26355/eurrev_201805_14986
- Zoellner LA, Telch M, Foa EB, et al. Enhancing Extinction Learning in Posttraumatic Stress Disorder With Brief Daily Imaginal Exposure and Methylene Blue: A Randomized Controlled Trial. J Clin Psychiatry. 2017;78(7):e782-e789. PubMed DOI: 10.4088/JCP.16m10936
- Rodriguez P, Zhou W, Barrett DW, Altmeyer W, Gutierrez JE, Li J, et al. Multimodal Randomized Functional MR Imaging of the Effects of Methylene Blue in the Human Brain. Radiology. 2016;281(2):516-526. PubMed DOI: 10.1148/radiol.2016152893
- Alda M, McKinnon M, Blagdon R, et al. Methylene blue treatment for residual symptoms of bipolar disorder: randomised crossover study. Br J Psychiatry. 2017;210(1):54-60. PubMed DOI: 10.1192/bjp.bp.115.173930
- Farrokhi MR, Lotfi M, Masoudi MS, Gholami M. Effects of methylene blue on postoperative low-back pain and functional outcomes after lumbar open discectomy: a triple-blind, randomized placebo-controlled trial. J Neurosurg Spine. 2016;24(1):7-15. PubMed DOI: 10.3171/2015.3.SPINE141172
- Sim HL, Tan KY. Randomized single-blind clinical trial of intradermal methylene blue on pain reduction after open diathermy haemorrhoidectomy. Colorectal Dis. 2014;16(8):O283-287. PubMed DOI: 10.1111/codi.12587
- Woo KY, Heil J. A prospective evaluation of methylene blue and gentian violet dressing for management of chronic wounds with local infection. Int Wound J. 2017;14(6):1029-1035. PubMed DOI: 10.1111/iwj.12753
- Christensen RD. 81 - Neonatal Erythrocyte Disorders. In: Gleason CA, Juul SE, eds. Avery's Diseases of the Newborn. 10th Ed. Philadelphia: Elsevier; 2018. p.1152-1179.e1154.
- Glader B. Chapter 72 - Other Hereditary Red Blood Cell Disorders. In: Rimoin D, Pyeritz R, Korf B, eds. Emery and Rimoin's Principles and Practice of Medical Genetics. 6th Ed. Oxford: Academic Press; 2013. p.1-25.
- Réunion OmdlsCdedlseludme, Organization WH. The Selection and Use of Essential Medicines: Report of the WHO Expert Committee, 2015 (including the 19th WHO Model List of Essential Medicines and the 5th WHO Model List of Essential Medicines for Children). Vol 994: World Health Organization; 2015.
- Venkategowda PM, Raut MK, Yarramalle SP, Yogesh R. Methylene blue and its role in ICU patients. A Review. International J Adv Health Science. 2015;2(4):426-433. Full Text
- Information NCfB. PubChem compound summary. In: National Center for Biotechnology Information Bethesda, MD, USA; 2021.
- U.S. Food and Drug Administration. FDA drug safety communication: serious CNS reactions possible when methylene blue is given to patients taking certain psychiatric medications; 2011. Full Text
- Pruna A, Bonaccorso A, Belletti A, Turi S, Di Prima AL, D'amico F, et al. Methylene Blue Reduces Mortality in Critically Ill and Perioperative Patients: A Meta-Analysis of Randomized Trials. Journal of Cardiothoracic and Vascular Anesthesia. 2024;38(1):268-274. PubMed DOI: 10.1053/j.jvca.2023.09.037
- Peter C, Hongwan D, Küpfer A, Lauterburg BH. Pharmacokinetics and organ distribution of intravenous and oral methylene blue. Eur J Clin Pharmacol. 2000;56(3):247-250. PubMed DOI: 10.1007/s002280000124
- Nguyen LP, Gerstein NS. Chapter 11 - Cardiovascular Pharmacology in Noncardiac Surgery. In: Kaplan JA, Cronin B, Maus TM, eds. Essentials of Cardiac Anesthesia for Noncardiac Surgery. New York: Elsevier; 2019. p.247-288.
- Clifton J, 2nd, Leikin JB. Methylene blue. Am J Ther. 2003;10(4):289-291. PubMed DOI: 10.1097/00045391-200307000-00009
- Chuang ST, Papp H, Kuczmog A, et al. Methylene Blue Is a Nonspecific Protein-Protein Interaction Inhibitor with Potential for Repurposing as an Antiviral for COVID-19. Pharmaceuticals (Basel). 2022;15(5). PubMed DOI: 10.3390/ph15050621
- Walter-Sack I, Rengelshausen J, Oberwittler H, et al. High absolute bioavailability of methylene blue given as an aqueous oral formulation. Eur J Clin Pharmacol. 2009;65(2):179-189. PubMed DOI: 10.1007/s00228-008-0563-x
- Li Z, Lang Y, Sakamuru S, Samrat S, Trudeau N, Kuo L, et al. Methylene blue is a potent and broad-spectrum inhibitor against Zika virus in vitro and in vivo. Emerg Microbes Infect. 2020;9(1):2404-2416. PubMed DOI: 10.1080/22221751.2020.1838954
- Annadurai N, Das V. Chapter 20 - Microtubule affinity regulating kinase 4: A potential drug target from cancers to neurodegenerative diseases. In: Hassan MI, Noor S, eds. Protein Kinase Inhibitors. Academic Press; 2022. p.571-596.
- Drugs.com. Methylene Blue Dosage; 2023. Available from: https://www.drugs.com/dosage/methylene-blue.html#:~:text=Methylene%20blue%20is%20extensively%20metabolized,an%20extended%20period%20following%20therapy
- Allegaert K, Miserez M, Lerut T, Naulaers G, Vanhole C, Devlieger H. Methemoglobinemia and hemolysis after enteral administration of methylene blue in a preterm infant: relevance for pediatric surgeons. J Pediatr Surg. 2004;39(1):E35-37. PubMed DOI: 10.1016/j.jpedsurg.2003.09.045
- Ginimuge PR, Jyothi SD. Methylene blue: revisited. J Anaesthesiol Clin Pharmacol. 2010;26(4):517-520. PubMed
- Prchal J. Methemoglobinemia and other dyshemoglobinemias. Williams Hematology 10th ed New York: McGraw Hill. 2021.
- Iolascon A, Bianchi P, Andolfo I, Russo R, Barcellini W, Fermo E, et al. Recommendations for diagnosis and treatment of methemoglobinemia. Am J Hematol. 2021;96(12):1666-1678. PubMed DOI: 10.1002/ajh.26340
- Ashurst J, Wasson M. Methemoglobinemia: a systematic review of the pathophysiology, detection, and treatment. Del Med J. 2011;83(7):203-208. PubMed
- Skold A, Cosco DL, Klein R. Methemoglobinemia: pathogenesis, diagnosis, and management. South Med J. 2011;104(11):757-761. PubMed DOI: 10.1097/SMJ.0b013e318232139f
- do Nascimento TS, Pereira RO, de Mello HL, Costa J. Methemoglobinemia: from diagnosis to treatment. Rev Bras Anestesiol. 2008;58(6):651-664. PubMed DOI: 10.1590/s0034-70942008000600011
- Kakhandki S, Yahya M, Praveen M. Acute methaemoglobinaemia initially treated as organophosphate poisoning leading to atropine toxicity. Indian J Anaesth. 2012;56(4):397-400. PubMed DOI: 10.4103/0019-5049.100836
- Boylston M, Beer D. Methemoglobinemia: a case study. Crit Care Nurse. 2002;22(4):50-55. PubMed
- Cohen J. Sepsis. Medicine. 2009;37(10):562-565. DOI: 10.1016/j.mpmed.2009.07.007
- Jarczak D, Kluge S, Nierhaus A. Sepsis-Pathophysiology and Therapeutic Concepts. Front Med (Lausanne). 2021;8:628302. PubMed DOI: 10.3389/fmed.2021.628302
- Hotchkiss RS, Moldawer LL, Opal SM, Reinhart K, Turnbull IR, Vincent JL. Sepsis and septic shock. Nat Rev Dis Primers. 2016;2:16045. PubMed DOI: 10.1038/nrdp.2016.45
- Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). Jama. 2016;315(8):801-810. PubMed DOI: 10.1001/jama.2016.0287
- Aguilar Arzápalo MF, López Avendaño VG, Escalante Castillo A, Góngora Mukul JJ, Franco Herrera B, Cetina Cámara MA. Eficacia del azul de metileno como coadyuvante en el tratamiento de pacientes con choque séptico. Medicina crítica (Colegio Mexicano de Medicina Crítica). 2016;30(2):102-110. Full Text
- Juffermans NP, Vervloet MG, Daemen-Gubbels CR, Binnekade JM, de Jong M, Groeneveld AB. A dose-finding study of methylene blue to inhibit nitric oxide actions in the hemodynamics of human septic shock. Nitric Oxide. 2010;22(4):275-280. PubMed DOI: 10.1016/j.niox.2010.01.006
- Preiser JC, Lejeune P, Roman A, Carlier E, De Backer D, Leeman M, et al. Methylene blue administration in septic shock: a clinical trial. Crit Care Med. 1995;23(2):259-264. PubMed DOI: 10.1097/00003246-199502000-00010
- Paya D, Gray GA, Stoclet JC. Effects of methylene blue on blood pressure and reactivity to norepinephrine in endotoxemic rats. J Cardiovasc Pharmacol. 1993;21(6):926-930. PubMed DOI: 10.1097/00005344-199306000-00012
- Mayer B, Brunner F, Schmidt K. Inhibition of nitric oxide synthesis by methylene blue. Biochem Pharmacol. 1993;45(2):367-374. PubMed DOI: 10.1016/0006-2952(93)90072-5
- Shaefi S, Mittel A, Klick J, Evans A, Ivascu NS, Gutsche J, et al. Vasoplegia After Cardiovascular Procedures-Pathophysiology and Targeted Therapy. J Cardiothorac Vasc Anesth. 2018;32(2):1013-1022. PubMed DOI: 10.1053/j.jvca.2017.10.032
- Omar S, Zedan A, Nugent K. Cardiac vasoplegia syndrome: pathophysiology, risk factors and treatment. Am J Med Sci. 2015;349(1):80-88. PubMed DOI: 10.1097/MAJ.0000000000000341
- Levin MA, Lin HM, Castillo JG, Adams DH, Reich DL, Fischer GW. Early on-cardiopulmonary bypass hypotension and other factors associated with vasoplegic syndrome. Circulation. 2009;120(17):1664-1671. PubMed DOI: 10.1161/CIRCULATIONAHA.108.814533
- Byrne JG, Leacche M, Paul S, Mihaljevic T, Rawn JD, Shernan SK, et al. Risk factors and outcomes for 'vasoplegia syndrome' following cardiac transplantation. Eur J Cardiothorac Surg. 2004;25(3):327-332. PubMed DOI: 10.1016/j.ejcts.2003.11.032
- Mets B, Michler RE, Delphin ED, Oz MC, Landry DW. Refractory vasodilation after cardiopulmonary bypass for heart transplantation in recipients on combined amiodarone and angiotensin-converting enzyme inhibitor therapy: a role for vasopressin administration. J Cardiothorac Vasc Anesth. 1998;12(3):326-329. PubMed DOI: 10.1016/s1053-0770(98)90017-9
- Gomes WJ, Carvalho AC, Palma JH, Teles CA, Branco JN, Silas MG, et al. Vasoplegic syndrome after open heart surgery. J Cardiovasc Surg (Torino). 1998;39(5):619-623. PubMed
- Mazzeffi M, Zivot J, Buchman T, Halkos M. In-hospital mortality after cardiac surgery: patient characteristics, timing, and association with postoperative length of intensive care unit and hospital stay. Ann Thorac Surg. 2014;97(4):1220-1225. PubMed DOI: 10.1016/j.athoracsur.2013.10.040
- Leyh RG, Kofidis T, Strüber M, Fischer S, Knobloch K, Wachsmann B, et al. Methylene blue: the drug of choice for catecholamine-refractory vasoplegia after cardiopulmonary bypass? J Thorac Cardiovasc Surg. 2003;125(6):1426-1431. PubMed DOI: 10.1016/s0022-5223(02)73284-4
- Hansen LS, Hjortdal VE, Andreasen JJ, Mortensen PE, Jakobsen CJ. 30-day mortality after coronary artery bypass grafting and valve surgery has greatly improved over the last decade, but the 1-year mortality remains constant. Ann Card Anaesth. 2015;18(2):138-142. PubMed DOI: 10.4103/0971-9784.154462
- Cremer J, Martin M, Redl H, Bahrami S, Abraham C, Graeter T, et al. Systemic inflammatory response syndrome after cardiac operations. Ann Thorac Surg. 1996;61(6):1714-1720. PubMed DOI: 10.1016/0003-4975(96)00055-0
- Hajjar LA, Vincent JL, Barbosa Gomes Galas FR, Rhodes A, Landoni G, Osawa EA, et al. Vasopressin versus Norepinephrine in Patients with Vasoplegic Shock after Cardiac Surgery: The VANCS Randomized Controlled Trial. Anesthesiology. 2017;126(1):85-93. PubMed DOI: 10.1097/ALN.0000000000001434
- Dagenais F, Mathieu P. Rescue therapy with methylene blue in systemic inflammatory response syndrome after cardiac surgery. Can J Cardiol. 2003;19(2):167-169. PubMed
- Werner I, Guo F, Bogert NV, Stock UA, Meybohm P, Moritz A, et al. Methylene blue modulates transendothelial migration of peripheral blood cells. PLoS One. 2013;8(12):e82214. PubMed DOI: 10.1371/journal.pone.0082214
- Mehaffey JH, Johnston LE, Hawkins RB, Charles EJ, Yarboro L, Kern JA, et al. Methylene Blue for Vasoplegic Syndrome After Cardiac Operation: Early Administration Improves Survival. Ann Thorac Surg. 2017;104(1):36-41. PubMed DOI: 10.1016/j.athoracsur.2017.02.057
- Gangireddy M, Patel P, Nookala V. Ifosfamide. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK542169/
- Pelgrims J, De Vos F, Van den Brande J, Schrijvers D, Prové A, Vermorken JB. Methylene blue in the treatment and prevention of ifosfamide-induced encephalopathy: report of 12 cases and a review of the literature. Br J Cancer. 2000;82(2):291-294. PubMed DOI: 10.1054/bjoc.1999.0917
- Bijivemula NSR, Vaddeboina N, Dattatreya P, Suresh A, Vamsy CM. Ifosfamide induced encephalopathy: A rare case presentation and management with review of the literature. Oncology Journal of India. 2021;5(3):123-125. DOI: 10.4103/oji.oji_32_21
- Alici-Evcimen Y, Breitbart WS. Ifosfamide neuropsychiatric toxicity in patients with cancer. Psychooncology. 2007;16(10):956-960. PubMed DOI: 10.1002/pon.1161
- Gachot B, Bedos JP, Veber B, Wolff M, Regnier B. Short-term effects of methylene blue on hemodynamics and gas exchange in humans with septic shock. Intensive Care Med. 1995;21(12):1027-1031. PubMed DOI: 10.1007/BF01700666
- Bistas E, Sanghavi DK. Methylene blue. In: StatPearls. StatPearls Publishing; 2023. Available from: https://www.ncbi.nlm.nih.gov/books/NBK557593/
- Sweet G, Standiford SB. Methylene-blue-associated encephalopathy. J Am Coll Surg. 2007;204(3):454-458. PubMed DOI: 10.1016/j.jamcollsurg.2006.12.030
- Nadler JE, Green H, Rosenbaum A. Intravenous injection of methylene blue in man with reference to its toxic symptoms and effect on the electrocardiogram. Am J Med Sci. 1934;188(1). Full Text
- MACSEN LABS. Methylene Blue Dye | Uses & Side effects; 2024; Available from: https://www.macsenlab.com/blog/methylene-blue-chemistry-uses-side-effects/. Accessed June 30, 2024.
- Dewachter P, Mouton-Faivre C, Tréchot P, Lleu JC, Mertes PM. Severe anaphylactic shock with methylene blue instillation. Anesth Analg. 2005;101(1):149-150, table of contents. PubMed DOI: 10.1213/01.ANE.0000153497.60047.80
- Harvey JW, Keitt AS. Studies of the efficacy and potential hazards of methylene blue therapy in aniline-induced methaemoglobinaemia. Br J Haematol. 1983;54(1):29-41. PubMed DOI: 10.1111/j.1365-2141.1983.tb02064.x
- Porat R, Gilbert S, Magilner D. Methylene blue-induced phototoxicity: an unrecognized complication. Pediatrics. 1996;97(5):717-721. PubMed
- Liao YP, Hung DZ, Yang DY. Hemolytic anemia after methylene blue therapy for aniline-induced methemoglobinemia. Vet Hum Toxicol. 2002;44(1):19-21. PubMed
- Evora PR. Should methylene blue be the drug of choice to treat vasoplegias caused by cardiopulmonary bypass and anaphylactic shock? J Thorac Cardiovasc Surg. 2000;119(3):632-634. PubMed DOI: 10.1016/s0022-5223(00)70152-8
- Vutskits L, Briner A, Klauser P, Gascon E, Dayer AG, Kiss JZ, et al. Adverse effects of methylene blue on the central nervous system. Anesthesiology. 2008;108(4):684-692. PubMed DOI: 10.1097/ALN.0b013e3181684be4
- Ozal E, Kuralay E, Yildirim V, Kilic S, Bolcal C, Kücükarslan N, et al. Preoperative methylene blue administration in patients at high risk for vasoplegic syndrome during cardiac surgery. Ann Thorac Surg. 2005;79(5):1615-1619. PubMed DOI: 10.1016/j.athoracsur.2004.10.038
- Crooks J. Haemolytic jaundice in a neonate after intra-amniotic injection of methylene blue. Arch Dis Child. 1982;57(11):872-873. PubMed DOI: 10.1136/adc.57.11.872
- Cragan JD. Teratogen update: methylene blue. Teratology. 1999;60(1):42-48. PubMed DOI: 10.1002/(SICI)1096-9926(199907)60:1<42::AID-TERA12>3.0.CO;2-Z
- Kidd SA, Lancaster PA, Anderson JC, Boogert A, Fisher CC, Robertson R, et al. Fetal death after exposure to methylene blue dye during mid-trimester amniocentesis in twin pregnancy. Prenat Diagn. 1996;16(1):39-47. PubMed DOI: 10.1002/(SICI)1097-0223(199601)16:1<39::AID-PD789>3.0.CO;2-P
- Berlin G, Brodin B, Hilden JO, Mårtensson J. Acute dapsone intoxication: a case treated with continuous infusion of methylene blue, forced diuresis and plasma exchange. J Toxicol Clin Toxicol. 1984;22(6):537-548. PubMed DOI: 10.3109/15563658408992583
- Price D, Nelson L, Howland M, Lewin N, Smith S. Goldfrank's Toxicologic Emergencies. 11th Ed; 2019.
- Senthilkumaran S, David SS, Manikam R, Thirumalaikolundusubramanian P. Continuous versus intermittent methylene blue administration: which spin will win? Indian J Crit Care Med. 2014;18(6):407-408. PubMed DOI: 10.4103/0972-5229.133953
- Messmer AS, Nickel CH, Bareiss D. P-chloroaniline poisoning causing methemoglobinemia: a case report and review of the literature. Case Rep Emerg Med. 2015;2015:208732. PubMedDOI: 10.1155/2015/208732
- Drugs.com. Methylene Blue Injection Prescribing Information; 2024. Available from: https://www.drugs.com/pro/methylene-blue-injection.html
- Hajj-Chahine J, Jayle C, Corbi P. Methylene blue in patients with severe pulmonary hypertension. J Thorac Cardiovasc Surg. 2013;145(3):898. PubMedDOI: 10.1016/j.jtcvs.2012.11.091
- American College of Chest Physicians. American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference: definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Crit Care Med. 1992;20(6):864-874. PubMed
- Almeida JA, Riordan SM, Liu J, Galhenage S, Kim R, Bihari D, et al. Deleterious effect of nitric oxide inhibition in chronic hepatopulmonary syndrome. Eur J Gastroenterol Hepatol. 2007;19(4):341-346. PubMed DOI: 10.1097/MEG.0b013e328014a3bf
- Wright RO, Lewander WJ, Woolf AD. Methemoglobinemia: etiology, pharmacology, and clinical management. Ann Emerg Med. 1999;34(5):646-656. PubMed DOI: 10.1016/s0196-0644(99)70167-8
- Kirov MY, Evgenov OV, Evgenov NV, Egorina EM, Sovershaev MA, Sveinbjørnsson B, et al. Infusion of methylene blue in human septic shock: a pilot, randomized, controlled study. Crit Care Med. 2001;29(10):1860-1867. PubMed DOI: 10.1097/00003246-200110000-00002
- Egi M, Bellomo R, Langenberg C, Haase M, Haase A, Doolan L, et al. Selecting a vasopressor drug for vasoplegic shock after adult cardiac surgery: a systematic literature review. Ann Thorac Surg. 2007;83(2):715-723. PubMed DOI: 10.1016/j.athoracsur.2006.08.041
- Kofidis T, Strüber M, Wilhelmi M, Anssar M, Simon A, Harringer W, et al. Reversal of severe vasoplegia with single-dose methylene blue after heart transplantation. J Thorac Cardiovasc Surg. 2001;122(4):823-824. PubMed DOI: 10.1067/mtc.2001.115153
- Levin RL, Degrange MA, Bruno GF, Del Mazo CD, Taborda DJ, Griotti JJ, et al. Methylene blue reduces mortality and morbidity in vasoplegic patients after cardiac surgery. Ann Thorac Surg. 2004;77(2):496-499. PubMed DOI: 10.1016/S0003-4975(03)01510-8
- Grayling M, Deakin CD. Methylene blue during cardiopulmonary bypass to treat refractory hypotension in septic endocarditis. J Thorac Cardiovasc Surg. 2003;125(2):426-427. PubMed DOI: 10.1067/mtc.2003.140
- Kwok ES, Howes D. Use of methylene blue in sepsis: a systematic review. J Intensive Care Med. 2006;21(6):359-363. PubMed DOI: 10.1177/0885066606290671
- Ballarin RS, Lazzarin T, Zornoff L, Azevedo PS, Pereira FWL, Tanni SE, et al. Methylene blue in sepsis and septic shock: a systematic review and meta-analysis. Front Med (Lausanne). 2024;11:1366062. PubMed DOI: 10.3389/fmed.2024.1366062
- Zhao CC, Zhai YJ, Hu ZJ, Huo Y, Li ZQ, Zhu GJ. Efficacy and safety of methylene blue in patients with vasodilatory shock: A systematic review and meta-analysis. Front Med (Lausanne). 2022;9:950596. PubMed DOI: 10.3389/fmed.2022.950596
- Huang X, Yan W, Chen Z, Qian Y. Effect of methylene blue on outcomes in patients with distributive shock: a meta-analysis of randomised controlled trials. BMJ Open. 2024;14(1):e080065. PubMed DOI: 10.1136/bmjopen-2023-080065
- Evora PR, Ribeiro PJ, Vicente WV, Reis CL, Rodrigues AJ, Menardi AC, et al. Methylene blue for vasoplegic syndrome treatment in heart surgery: fifteen years of questions, answers, doubts and certainties. Rev Bras Cir Cardiovasc. 2009;24(3):279-288. PubMed DOI: 10.1590/s0102-76382009000400005
- Park IS, Lee HJ, Lee YS, Hwang JS, Lee MS. Ifosfamide-induced encephalopathy with or without using methylene blue. Int J Gynecol Cancer. 2005;15(5):807-810. PubMed DOI: 10.1111/j.1525-1438.2005.00140.x