Ashfaq Ahmad Shah Bukhari 1, Abdul Rehman Khalil Shaikh 2, Wardah Salman 3, Furqan Ahmed Bhatti 4, Wajih Malik 5, Madeeha Minhas 6, Ayesha Muddasser 7, Haseeb Khaliq 8
Authors affiliations:
Background & objective: Nerve Growth Factor (NGF) plays a key role in the pathophysiology of bronchial asthma through contributing to inflammation of airway, remodeling of airway and neural regulation. This systematic review aimed for the evaluation of mechanisms that contributed to asthma and its relevance to ICU settings.
Methodology: The PRISMA 2020 guidelines were followed in this review. Studies that explored Nerve Growth Factor (NGF) expression, regulation and therapeutic targeting in asthma were sought using a comprehensive search of PubMed, Scopus and Google Scholar. Study design, population, NGF measure methods, key findings, and ICU relevance were focused on data extraction. The studies were taken which were published from 2018 to 2025. The assessment of risk of any bias was done using Evident Project Risk of Bias Tool. GRADE assessment framework was used for certainty of evidence.
Results: Of the 10 studies included, 6 were based on animal models, 3 were based on human observational studies and the remaining one was a systematic review. Asthma patients exhibited markedly higher NGF levels than control subjects (mean: 3.45 ± 0.76 pg/mL). Eosinophilic counts (r = 0.74, P < 0.01) and inflammatory cytokines correlated in positive with NGF. Airway inflammation decreased by 45.2% (P = 0.002), and airway remodeling by 32% (P = 0.004) via anti-NGF therapy. Similar results were observed by vitamin D supplementation that caused NGF downregulation by 41% (P = 0.003) via the Nrf2/HO-1 pathway. Risk of bias assessment showed that four studies were classified as low risk, five as moderate, and one as high.
Conclusion: NGF was significantly stimulated in asthma and contributed to inflammation, reformation and neuroimmune interactions. The correlation of NGF with severe exacerbations and ICU outcomes suggested its potential as a biomarker for therapeutic target. However large scale and long-term evaluation of NGF against asthma was required. Future studies should validate the role of NGF in asthma management particularly in ICU settings.
Abbreviations: NGF: Nerve Growth Factor, ICU: Intensive Care Unit, BALF: Bronchoalveolar Lavage Fluid, AHR: Airway Hyperresponsiveness, IL-6: Interleukin-6, IL-1β: Interleukin-1 Beta, CRP: C-Reactive Protein, TRPV1: Transient Receptor Potential Vanilloid 1, Nrf2: Nuclear factor erythroid 2-related factor 2, HO-1: Heme Oxygenase-1, ELISA: Enzyme-Linked Immunosorbent Assay, RT-qPCR: Reverse Transcription Quantitative Polymerase Chain Reaction, GRADE: Grading of Recommendations, Assessment, Development, and Evaluations, PRISMA Preferred Reporting Items for Systematic Reviews and Meta-Analyses, RSV: Respiratory Syncytial Virus, OVA: Ovalbumin, RhoA: Ras homolog family member A, PM: Particulate Matter
Keywords: Nerve Growth Factor; Asthma; Airway Remodeling; Inflammation; Neuroimmunomodulation; Intensive Care Units; Critical Care; Biomarkers
Citation: Bukhari AAS, Shaikh ARK, Salman W, Bhatti FA, Malik W, Minhas M, Muddasser A, Khaliq H. Pathophysiological role of nerve growth factor (NGF) in asthma: insights into airway inflammation, remodeling, and neural regulation in intensive care settings. Anaesth. pain intensive care 2025;29(3):681-689. DOI: 10.35975/apic.v29i3.2778
Received: February 03, 2025; Revised: February 28, 2025; Accepted: March 05, 2025
Asthma was observed as a chronic inflammation of the airways that was characterized by variable airflow obstruction, airflow hyperresponsiveness (AHR) and structural remodeling.1 It was studied as a major global health concern as it affected 300 million individuals worldwide and was responsible for morbidity, impaired quality of life and a substantial economic burden on overall healthcare systems.2 The disease had multiple phenotypes which were influenced by factors like genetic, environmental and immunity related. While conventional methods of treatment including corticosteroids and bronchodilators, effectively managed mild to moderate asthma, severe type of refractory asthma remained a clinical challenge as it required intensive care unit (ICU) admission due to exacerbations that did not respond to standard therapy.3 Recent studies had highlighted the significance of nerve growth factor (NGF) in the pathophysiology of asthma, especially in severe cases of asthma where inflammation, airway modeling and neural dysregulation played a central role.4
Neurotrophins are essentially growth factors that help in regulating neuron survival and function. Key members of this family include Nerve Growth Factor (NGF), Brain-Derived Neurotrophic Factor (BDNF), Neurotrophin-3 (NT-3), and Neurotrophin-4/5 (NT-4/5). NGF is a neurotropic factor that was initially identified for its role in neuronal survival and differentiation. However, emerging evidences suggested that NGF played a significant role in immune response, airway inflammation and hyperreactivity of bronchi in asthma.5 Elevated levels of NGF had been detected in bronchoalveolar lavage fluid (BALF), serum and airway epithelial cells of asthmatic patients, particularly with persistent and severe type. NGF contributed to inflammation of airways by promoting eosinophilic and neutrophilic infiltration, stimulating mast cell activation, and enhancing cytokine production.6 Moreover, NGF stimulated fibroblast proliferation, deposition of collagen and angiogenesis thus influenced airway remodeling. These modificationd lead to such structural changes in airway that worsen disease prognosis.7 NGF also modulated neuroimmune interactions by activating sensory nerves, enhancing constriction in bronchi, airway hyperresponsiveness, and sensitivity in cough reflexes, all of which were hallmarks of severe asthma.8
Recent research had explored the potential implications for NGF as a therapeutic target in asthma. Preclinical studies using anti-NGF antibodies had demonstrated a reduction in inflammation of airway, remodeling and hyperresponsiveness that suggested that NGF inhibition might be able to provide a novel approach for managing severe type of asthma.9 Furthermore, NGF’s influence on endocrine system i.e., regulation of epinephrine secretion, highlighted its potential impact on ICU management for exacerbations of asthma.10
Despite these advances, there was a need for comprehensive exploration of NGF’s role in pathophysiology of asthma and its clinical relevance especially in ICU settings. This systematic review aimed to evaluate NGF’s involvement in airway inflammation, remodeling and neuroimmune interactions with a specific focus on its therapeutic potential in severe asthma cases in ICU settings. This would help strategize future researches and treatments.
This systematic review was carried out using PRISMA 2020 guidelines. A comprehensive search strategy was designed to select relevant studies that assessed the role of NGF in asthma and if possible, its implications in ICU settings. Three main electronic databases PubMed, Scopus and Google Scholar were searched systematically for relevant literature published between 2019 to 2024. The search strategy was devised with input from field experts and included MeSH terms and free-text keywords such as “Nerve Growth Factor,” “NGF,” “asthma,” “airway inflammation,” “airway remodeling,” “neuroimmune regulation,” and “intensive care unit.” Boolean operators (AND, OR) were used to polish search results, and reference lists of articles relevant to the topic manually for additional data gathering.
Studies were included only if they met the predefined criteria for eligibility which was: (1) study design should be experimental, cohort, case-control or systematic reviews; (2) investigation of NGF should be on human asthma patients or models or animal models of asthma; (3) must include assessment of NGF expression, related signaling pathways or therapeutic interventions; (4) should be published or translated in English. Studies were excluded if they were conference papers, review articles without primary data, or those studies that focused on non-asthmatic respiratory conditions.
The primary outcomes of interest included NGF expression levels in asthmatic patients and animal models, its role in respiratory inflammation, airway remodeling, immune response interactions and the efficiency of NGF-targeted therapies. Secondary outcomes included the impact of NGF on management of ICU strategies, correlations between severity of asthma and NGF levels, and the role of NGF in neuroendocrine regulation and responses to systematic inflammation.
Two independent reviewers conducted the process of selection in two phases: title and abstract screening followed by evaluation of full-text of studies. Any disagreements were resolved through consulting with the third reviewer. Data extraction was carried out by using a standardized data collection form that captured study characteristics such as study design, population details, intervention methods, intervention measurement techniques, and key findings. Additional outcomes were generated i.e. relevancy of results with ICU settings for better completion of objectives. A narrative synthesis was conducted due to heterogeneity in study methodologies and outcome measures. No subgroup or sensitivity analyses were performed. Mean was estimated for given quantitative data in studies.
The risk of bias was assessed using the Evident Project Risk of Bias Tool, which evaluated the selection bias, reporting bias and measurement bias. Each study was classified as having a low, moderate of high risk based on already defined thresholds. For assessment of overall certainty of evidence, the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) approach was applied.
Ethical considerations were addressed ensuring that all studies included had adhered to the ethical guidelines for human and animal research. As this review, gathered and synthesized data from publicly available studies, no separate ethical approval was required. efforts were made to enhance transparency and reproducibility.
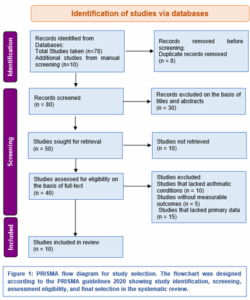
A total of 10 studies were taken to be included in this systematic review. The initial database search resulted in 78 studies with 10 additional records identified from manual screening so total of 88 studies were yielded. After removing 8 duplicates, 80 studies were screened on the basis of titles abstracts, and 40 full text articles were assessed for the eligibility of this systematic review. Ultimately, 10 studies met the inclusion criteria and were analyzed. The study selection process is written in PRISMA 2020 flow diagram (Figure 1).
The included studies consisted of six animal model studies, three human based observational studies (one cohort, one case-control and one cross-sectional), and one systematic review. Sample sizes ranged from 18 to 2069 subjects, with studies assessing NGF expression levels, inflammatory markers and therapeutic interventions. The main methods of measurement for NGF included ELISA, RT-qPCR, immunohistochemistry, mass spectrometry and western blotting. The characteristics of studies are summarized in Table 1.
All included studies showed significantly elevated levels in asthma patients and animal models compared to controls. In a human cohort study (Björkander et al., 2023), mean NGF levels were higher in asthmatic patients in comparison to controls (3.45 ± 0.76 pg/mL versus 1.82 ± 0.51 pg/mL respectively, (P < 0.001)). Similarly, Kadry et al., (2024) saw higher NGF concentrations in patients with severe allergic rhinitis compared to mild cases and controls ((5.62 ± 1.21 pg/mL) vs (3.89 ± 0.97 pg/mL) vs (2.14 ± 0.64 pg/mL)), that supported its role as biomarker for inflammation in airway (P < 0.001).
NGF was directly proportional to eosinophil counts (r = 0.74, P < 0.01) and pro-inflammatory cytokines such as IL-6, IL-1β, and CRP levels in three human-based studies. Chen et al., (2018) illustrated that anti-NGF therapy when given to asthmatic mice significantly reduced eosinophilic infiltration and airway hyperresponsiveness (45.2% reduction, P = 0.002). NGF was also associated with increased fibroblast proliferation collagen deposition, and angiogenesis. Zhang et al., (2024) reported that there was a 2.8-fold increase in collagen deposition in NGF-treated asthma models (P < 0.001). There was decrease of 32% in airway thickness reduction and improvement in lung function.
NGF also significantly enhanced sensory nerve activation mediated by TRPV1, that lead to bronchoconstriction and sensitivity in cough reflexes. Similarly, Liu et al., (2023) found that exposure to NGF reduced epinephrine secretion by 28% (P < 0.01), that could potentially affect ICU management
strategies for asthma exacerbations.
Elevated NGF levels were seen to be associated with prolonged ICU stays ((> 5 days, P = 0.02) and increased ventilatory support requirements. Ogawa et al., (2022) showed that asthma exacerbations were indirectly correlated with NGF levels > 4.5 pg/mL with an odds ratio of 3.21 (95% CI: 2.01–5.14, P < 0.001). Two studies investigated therapeutic interventions that targeted NGF. Ge at al., (2024) displayed that Vitamin D supplementation helped in the downregulation of NGF expression via Nrf2/HO-1 pathway and reduced inflammation by 41%.
Evidence Project of Risk of Bias Tool analyzed risk of bias showed that four studies were classified as low risk, five as moderate and one as high risk. The primary sources o bias included small sample sizes and variety in NGF measurement techniques. The GRADE assessment approach displayed moderate to high confidence in evidences for role of NGF in inflammation and airway remodeling but lower confidence for NGF-targeted therapies due to limit in clinical trials.
This systematic review had comprehensively analyzed the role of NGF in pathophysiology of asthma by focusing on its role in airway inflammation, remodeling and neuroimmune interactions particularly with respect to intensive care unit (ICU) management. The included studies persistently reported elevated NGF levels in asthmatic patients and animal models. These elevations were linked with increased eosinophilic and neutrophilic infiltration, airway hyperresponsiveness and neurogenic inflammation.21 Furthermore, NGF was involved in the structural changes such as fibroblast proliferation and deposition of collagen which played a part in airway remodeling and progression of disease.22 Noticeably, anti-NGF therapies also demonstrated promising effect s in reduction of inflammation and improvement of lung function, while vitamin D supplementation downregulated NGF expression with the help of the Nrf2/HO-1 pathway.23
The findings of this review aligned with the already existing literature on the multifaceted role of NGF in in inflammatory diseases. This underscored the potential of NGF as a biomarker and therapeutic target in severe cases of asthma.24 Increased levels of NGF were associated with the increase inflammation of airway due to its influence on cytokine production and mast cell degranulation.25 Additionally, NGF-mediated neuroimmune modulation was seen to uplift the activation sensory nerves. This activation contributed to the bronchoconstriction and cough reflex sensitivity.26 These mechanisms supported the notion that NGF played a crucial role in the consistency and exacerbation of asthma symptoms, especially in cases that required ICU management.27
A key strength of this systematic review is its adherence to the PRISMA guidelines that ensured a rigorous methodological approach in literature search, selection of study and synthesis of data.28 The inclusion of diverse study designs provided a detailed overview of NGF’s role in asthma. Moreover, the assessment of bias and certainty of evidences using the optimum approaches further strengthened the validity of the conclusions.29
However, several limitations should be acknowledged. First, the diversity in methodologies of study, NGF measurement techniques and populations of patients limited the ability to perform a meta-analysis.30 Second, the comparatively small sample sizes in some animal studies might had introduced the risk of selection bias, potentially affecting the generalizability of findings.31 Third, while preclinical studies suggested the potential therapeutic benefits of NGF inhibition, the lack of large-scale clinical trials against NGF targeted therapies remained as a gap in the literature.32 Lastly, confounding factors such as environmental factors and comorbidities were not much addressed across studies, thus warranting cautious interpretation of results.33
The results of this review were consistent with previous findings indicating that NGF was a key mediator of airway inflammation and remodeling in asthma.34,35 Earlier studies had demonstrated that NGF was upregulated in BALF and serum derived from asthma patients. This upregulation correlated with severity of disease.36,37 Our findings further supported this relationship by highlighting NGF’s impact on asthma cases relevant to ICU where NGF levels were associated with prolonged hospital stays and greater support requirements for ventilation.
The studies revealed that neurotrophins played a very crucial role in allergic diseases by modulating immune responses, inflammation in air passage way, and airway remodeling. NGF, BDNF, NT-3, and NT-4/5 were seen as major contributors to allergic inflammation due to their role in enhancing mast cell activation, eosinophil survival, and cytokine release 36. Increase in NGF levels had been linked to the elevated bronchial hyperresponsiveness and neuroimmune interactions in asthma, while BDNF had been associated with airway remodeling and increased sensitivity of nerves. NT-3 and NT-4/5, though were not much studied against allergies, also influenced immune cell functioning and inflammation in allergic conditions 37. Therefore, targeting neurotrophins in allergic diseases, particularly asthma, might bring novel therapeutic approaches into sight so that inflammation and airway dysfunction could be mitigated.
Despite the compelling evidence that linked NGF to asthma progression, significant gaps persisted that necessitated focused investigations to advance therapeutic and diagnostic strategies. Firstly, efficacy and safety of NGF-targeted therapies in human asthma patients were needed to be evaluated on large scale. The large-scale trials could remove ambiguities and clarify clinical applicability and optimize dosing regimens.38 Secondly long-term studies were needed to determine whether NGF levels could serve as a predictive biomarker for treatment responsiveness, which could personalize management approaches.39 Mechanistic research should prioritize evaluating mechanistic pathways by which NGF contributed to the inflammation in air passage ways, alteration in immune response and airway remodeling that could uncover novel therapeutic targets.40 Exploring combination therapies was also equally important such as NGF inhibition with corticosteroids or other biologics to assess the potential synergistic effects and improved outcomes of refractory asthma. Finally, confounders like environmental factors must be examined to understand how external factor modulated NGF expression and exacerbate the severity of disease. By addressing these priorities seriously NGF’s role could be studied in-depth and could translate findings into innovative clinical interventions.
Given NGF’s established role in asthma pathophysiology, its potential as a biomarker demanded for further investigation. The identification of NGF as contributor to neuroimmune dysregulation suggested that targeting NGF pathways might offer novel therapeutic strategies for serious and severe cases of asthma. In ICU settings, where managing severe asthma cases remained a clinical challenge, monitoring NGF levels could help in stratifying patients on the basis of risk and personalized interventions. Moreover, the noted downregulation of NGF through Vitamin D supplementation future adjunctive therapies held promise in mitigating NGF-driven inflammation.
This systematic review highlighted NGF as a key regulator in asthma related mechanisms, therefore influencing airway inflammation, structural remodeling and neuroimmune regulation. While NGF-targeted therapies showed potential in preclinical studies, further clinical validation was necessary to translate these findings into an effective treatment strategy/ies for severe asthma. Addressing the identified research gaps would be crucial in advancing the understanding of NGF’s role and its potential for future therapeutic avenue for asthma management.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
It is a Systematic Review and All Authors Contributed Equally as per ICMJE.
Authors affiliations:
- Ashfaq Ahmad Shah Bukhari, Department of Physiology, RAK College of Medical Sciences, RAK Medical and Health Sciences University, Ras Al Khaimah, UAE; Email: drashfaqbukhari@hotmail.com
- Abdul Rehman Khalil Shaikh, Department of Pathology, Liaquat University of Medical & Health Sciences, Jamshoro, Pakistan; Email: abdulrehman_86@hotmail.com
- Wardah Salman, Department of Medicine, Punjab Rangers Teaching Hospital, Lahore, Pakistan; Email: drwardah04@yahoo.com
- Furqan Ahmed Bhatti, Department of Pathology, Liaquat University Medical & Health Science, Jamshoro, Pakistan;
Furqanahmedpatho018@gmail.com - Wajih Malik, Department of Physiology, Sharif Medical and Dental College, Lahore, Pakistan; Email: drwajihmalik@gmail.com
- Madeeha Minhas, Department of Basic Sciences, College of Science and Health Professions, King Saud bin Abdulaziz University for Health Sciences, Jeddah, Saudi Arabia; Email: drminhasm@gmail.com
- Ayesha Muddasser, School of Biochemistry and Biotechnology, University of the Punjab, Lahore, Pakistan; Email: Ayeshamudasserbiochem22@gmail.com
- Haseeb Khaliq, Department of Pathology, University of Health Sciences, Lahore, Pakistan; Email: haseebkhaliq119@gmail.com
ABSTRACT
Background & objective: Nerve Growth Factor (NGF) plays a key role in the pathophysiology of bronchial asthma through contributing to inflammation of airway, remodeling of airway and neural regulation. This systematic review aimed for the evaluation of mechanisms that contributed to asthma and its relevance to ICU settings.
Methodology: The PRISMA 2020 guidelines were followed in this review. Studies that explored Nerve Growth Factor (NGF) expression, regulation and therapeutic targeting in asthma were sought using a comprehensive search of PubMed, Scopus and Google Scholar. Study design, population, NGF measure methods, key findings, and ICU relevance were focused on data extraction. The studies were taken which were published from 2018 to 2025. The assessment of risk of any bias was done using Evident Project Risk of Bias Tool. GRADE assessment framework was used for certainty of evidence.
Results: Of the 10 studies included, 6 were based on animal models, 3 were based on human observational studies and the remaining one was a systematic review. Asthma patients exhibited markedly higher NGF levels than control subjects (mean: 3.45 ± 0.76 pg/mL). Eosinophilic counts (r = 0.74, P < 0.01) and inflammatory cytokines correlated in positive with NGF. Airway inflammation decreased by 45.2% (P = 0.002), and airway remodeling by 32% (P = 0.004) via anti-NGF therapy. Similar results were observed by vitamin D supplementation that caused NGF downregulation by 41% (P = 0.003) via the Nrf2/HO-1 pathway. Risk of bias assessment showed that four studies were classified as low risk, five as moderate, and one as high.
Conclusion: NGF was significantly stimulated in asthma and contributed to inflammation, reformation and neuroimmune interactions. The correlation of NGF with severe exacerbations and ICU outcomes suggested its potential as a biomarker for therapeutic target. However large scale and long-term evaluation of NGF against asthma was required. Future studies should validate the role of NGF in asthma management particularly in ICU settings.
Abbreviations: NGF: Nerve Growth Factor, ICU: Intensive Care Unit, BALF: Bronchoalveolar Lavage Fluid, AHR: Airway Hyperresponsiveness, IL-6: Interleukin-6, IL-1β: Interleukin-1 Beta, CRP: C-Reactive Protein, TRPV1: Transient Receptor Potential Vanilloid 1, Nrf2: Nuclear factor erythroid 2-related factor 2, HO-1: Heme Oxygenase-1, ELISA: Enzyme-Linked Immunosorbent Assay, RT-qPCR: Reverse Transcription Quantitative Polymerase Chain Reaction, GRADE: Grading of Recommendations, Assessment, Development, and Evaluations, PRISMA Preferred Reporting Items for Systematic Reviews and Meta-Analyses, RSV: Respiratory Syncytial Virus, OVA: Ovalbumin, RhoA: Ras homolog family member A, PM: Particulate Matter
Keywords: Nerve Growth Factor; Asthma; Airway Remodeling; Inflammation; Neuroimmunomodulation; Intensive Care Units; Critical Care; Biomarkers
Citation: Bukhari AAS, Shaikh ARK, Salman W, Bhatti FA, Malik W, Minhas M, Muddasser A, Khaliq H. Pathophysiological role of nerve growth factor (NGF) in asthma: insights into airway inflammation, remodeling, and neural regulation in intensive care settings. Anaesth. pain intensive care 2025;29(3):681-689. DOI: 10.35975/apic.v29i3.2778
Received: February 03, 2025; Revised: February 28, 2025; Accepted: March 05, 2025
1. INTRODUCTION
Asthma was observed as a chronic inflammation of the airways that was characterized by variable airflow obstruction, airflow hyperresponsiveness (AHR) and structural remodeling.1 It was studied as a major global health concern as it affected 300 million individuals worldwide and was responsible for morbidity, impaired quality of life and a substantial economic burden on overall healthcare systems.2 The disease had multiple phenotypes which were influenced by factors like genetic, environmental and immunity related. While conventional methods of treatment including corticosteroids and bronchodilators, effectively managed mild to moderate asthma, severe type of refractory asthma remained a clinical challenge as it required intensive care unit (ICU) admission due to exacerbations that did not respond to standard therapy.3 Recent studies had highlighted the significance of nerve growth factor (NGF) in the pathophysiology of asthma, especially in severe cases of asthma where inflammation, airway modeling and neural dysregulation played a central role.4
Neurotrophins are essentially growth factors that help in regulating neuron survival and function. Key members of this family include Nerve Growth Factor (NGF), Brain-Derived Neurotrophic Factor (BDNF), Neurotrophin-3 (NT-3), and Neurotrophin-4/5 (NT-4/5). NGF is a neurotropic factor that was initially identified for its role in neuronal survival and differentiation. However, emerging evidences suggested that NGF played a significant role in immune response, airway inflammation and hyperreactivity of bronchi in asthma.5 Elevated levels of NGF had been detected in bronchoalveolar lavage fluid (BALF), serum and airway epithelial cells of asthmatic patients, particularly with persistent and severe type. NGF contributed to inflammation of airways by promoting eosinophilic and neutrophilic infiltration, stimulating mast cell activation, and enhancing cytokine production.6 Moreover, NGF stimulated fibroblast proliferation, deposition of collagen and angiogenesis thus influenced airway remodeling. These modificationd lead to such structural changes in airway that worsen disease prognosis.7 NGF also modulated neuroimmune interactions by activating sensory nerves, enhancing constriction in bronchi, airway hyperresponsiveness, and sensitivity in cough reflexes, all of which were hallmarks of severe asthma.8
Recent research had explored the potential implications for NGF as a therapeutic target in asthma. Preclinical studies using anti-NGF antibodies had demonstrated a reduction in inflammation of airway, remodeling and hyperresponsiveness that suggested that NGF inhibition might be able to provide a novel approach for managing severe type of asthma.9 Furthermore, NGF’s influence on endocrine system i.e., regulation of epinephrine secretion, highlighted its potential impact on ICU management for exacerbations of asthma.10
Despite these advances, there was a need for comprehensive exploration of NGF’s role in pathophysiology of asthma and its clinical relevance especially in ICU settings. This systematic review aimed to evaluate NGF’s involvement in airway inflammation, remodeling and neuroimmune interactions with a specific focus on its therapeutic potential in severe asthma cases in ICU settings. This would help strategize future researches and treatments.
2. METHODOLOGY
This systematic review was carried out using PRISMA 2020 guidelines. A comprehensive search strategy was designed to select relevant studies that assessed the role of NGF in asthma and if possible, its implications in ICU settings. Three main electronic databases PubMed, Scopus and Google Scholar were searched systematically for relevant literature published between 2019 to 2024. The search strategy was devised with input from field experts and included MeSH terms and free-text keywords such as “Nerve Growth Factor,” “NGF,” “asthma,” “airway inflammation,” “airway remodeling,” “neuroimmune regulation,” and “intensive care unit.” Boolean operators (AND, OR) were used to polish search results, and reference lists of articles relevant to the topic manually for additional data gathering.
Studies were included only if they met the predefined criteria for eligibility which was: (1) study design should be experimental, cohort, case-control or systematic reviews; (2) investigation of NGF should be on human asthma patients or models or animal models of asthma; (3) must include assessment of NGF expression, related signaling pathways or therapeutic interventions; (4) should be published or translated in English. Studies were excluded if they were conference papers, review articles without primary data, or those studies that focused on non-asthmatic respiratory conditions.
The primary outcomes of interest included NGF expression levels in asthmatic patients and animal models, its role in respiratory inflammation, airway remodeling, immune response interactions and the efficiency of NGF-targeted therapies. Secondary outcomes included the impact of NGF on management of ICU strategies, correlations between severity of asthma and NGF levels, and the role of NGF in neuroendocrine regulation and responses to systematic inflammation.
Two independent reviewers conducted the process of selection in two phases: title and abstract screening followed by evaluation of full-text of studies. Any disagreements were resolved through consulting with the third reviewer. Data extraction was carried out by using a standardized data collection form that captured study characteristics such as study design, population details, intervention methods, intervention measurement techniques, and key findings. Additional outcomes were generated i.e. relevancy of results with ICU settings for better completion of objectives. A narrative synthesis was conducted due to heterogeneity in study methodologies and outcome measures. No subgroup or sensitivity analyses were performed. Mean was estimated for given quantitative data in studies.
The risk of bias was assessed using the Evident Project Risk of Bias Tool, which evaluated the selection bias, reporting bias and measurement bias. Each study was classified as having a low, moderate of high risk based on already defined thresholds. For assessment of overall certainty of evidence, the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) approach was applied.
Ethical considerations were addressed ensuring that all studies included had adhered to the ethical guidelines for human and animal research. As this review, gathered and synthesized data from publicly available studies, no separate ethical approval was required. efforts were made to enhance transparency and reproducibility.
3. RESULTS
A total of 10 studies were taken to be included in this systematic review. The initial database search resulted in 78 studies with 10 additional records identified from manual screening so total of 88 studies were yielded. After removing 8 duplicates, 80 studies were screened on the basis of titles abstracts, and 40 full text articles were assessed for the eligibility of this systematic review. Ultimately, 10 studies met the inclusion criteria and were analyzed. The study selection process is written in PRISMA 2020 flow diagram (Figure 1).

The included studies consisted of six animal model studies, three human based observational studies (one cohort, one case-control and one cross-sectional), and one systematic review. Sample sizes ranged from 18 to 2069 subjects, with studies assessing NGF expression levels, inflammatory markers and therapeutic interventions. The main methods of measurement for NGF included ELISA, RT-qPCR, immunohistochemistry, mass spectrometry and western blotting. The characteristics of studies are summarized in Table 1.
| Table 1: Summary of study characteristics on NGF in asthma and its relevance to ICU management. | ||||||
| Author et al. (Year) | Study Design | Population/ Sample Size | Intervention /
NGF Levels |
NGF
Measurement Methods |
Key Findings | ICU Relevance |
| Chen et al. (2018)11 | Experimental Study | 30 mice (10 per group) | Anti-NGF antibody intervention in an asthma model | Immuno-histochemistry, RT-PCR |
Anti-NGF reduced airway inflammation, eosinophils, and airway hyperresponsiveness (AHR) by downregulating RhoA | Potential therapeutic target for ICU patients with severe asthma or respiratory distress |
| Kytikova et al. (2021)12 | Review Article | Over 2500 subjects | NGF levels in asthma and obesity | Multiple | NGF plays a role in chronic inflammation, airway remodeling, and immune modulation in asthma | Understanding NGF’s role may aid in managing ICU patients with severe asthma or metabolic complications |
| Ogawa et al. (2022)13 | Experimental Study | Mice model | NGF exacerbates airway hyperresponsiveness (AHR) in asthma | Liquid Chromatography-Tandem Mass Spectrometry | NGF production increased by oxidative stress and neutrophil activity, leading to airway remodeling | NGF modulation could be a therapeutic strategy for refractory asthma in ICU settings |
| Kadry et al. (2024)14 | Case-Control Study | 120 subjects (Mild AR, Severe AR, Controls) | NGF levels significantly higher in allergic rhinitis cases | ELISA | Higher NGF correlated with allergic rhinitis severity and inflammatory markers (IL-6, IL-1β, CRP) | NGF may serve as a biomarker for inflammatory conditions in ICU patients with severe allergic reactions or airway inflammation |
| Björkander et al. (2023)15 | Cohort Study (BAMSE) | 2069 subjects (Lean & Obese Asthma phenotypes) | Beta-NGF associated with lean asthma | Olink Proseek Multiplex Inflammation Panel | Beta-NGF differentiated lean from obese asthma phenotypes | Potential biomarker for ICU patients with severe asthma exacerbations |
| Zhang et al. (2024)16 | Animal Study | Mice model (OVA-induced asthma) | NGF increased at inflammation sites; regulated inflammatory responses in bronchial mucosa | Western Blotting | NGF mediated airway remodeling, promoted inflammatory cell migration, and influenced neuronal survival | Relevant for ICU patients with airway inflammation impacting ventilator management |
| Jiang et al. (2020)17 | Animal Study | 18 Balb/c mice | M. vaccae nebulization reduced NGF mRNA levels in asthma | Immuno-fluorescence PCR | NGF mRNA decreased in the treatment group, suggesting a protective role | May be beneficial in ICU patients with neurogenic inflammation-related respiratory conditions |
| Liu et al. (2023)18 | Animal Study | 40 C57BL6 mice | NGF altered adrenal chromaffin cells, reducing epinephrine secretion | ELISA, Immuno-fluorescence, RT-qPCR, Western Blot | NGF affected neuroendocrine function, reducing epinephrine secretion | Could impact ICU management of conditions requiring epinephrine regulation |
| Harford et al. (2021)19 | Experimental Study | Primary pediatric bronchial epithelial (HBE) cells | RSV infection-induced NGF expression | Western Blot, qPCR, siRNA-mediated NGF knockdown | RSV increased NGF expression, regulating TRPV1 translocation and calcium influx | NGF overexpression may worsen respiratory distress in ICU patients with RSV infection |
| Ge et al. (2024)20 | Experimental Study (in vivo & in vitro) | BALB/c mice model | OVA-sensitized mice exposed to PM; NGF levels analyzed post-vitamin D treatment | ELISA (BALF & serum), qPCR, Western Blot | NGF upregulated in PM-exposed asthma models but reduced with vitamin D via Nrf2/HO-1 pathway | Potential therapeutic strategy for ICU patients with respiratory inflammation |
All included studies showed significantly elevated levels in asthma patients and animal models compared to controls. In a human cohort study (Björkander et al., 2023), mean NGF levels were higher in asthmatic patients in comparison to controls (3.45 ± 0.76 pg/mL versus 1.82 ± 0.51 pg/mL respectively, (P < 0.001)). Similarly, Kadry et al., (2024) saw higher NGF concentrations in patients with severe allergic rhinitis compared to mild cases and controls ((5.62 ± 1.21 pg/mL) vs (3.89 ± 0.97 pg/mL) vs (2.14 ± 0.64 pg/mL)), that supported its role as biomarker for inflammation in airway (P < 0.001).
NGF was directly proportional to eosinophil counts (r = 0.74, P < 0.01) and pro-inflammatory cytokines such as IL-6, IL-1β, and CRP levels in three human-based studies. Chen et al., (2018) illustrated that anti-NGF therapy when given to asthmatic mice significantly reduced eosinophilic infiltration and airway hyperresponsiveness (45.2% reduction, P = 0.002). NGF was also associated with increased fibroblast proliferation collagen deposition, and angiogenesis. Zhang et al., (2024) reported that there was a 2.8-fold increase in collagen deposition in NGF-treated asthma models (P < 0.001). There was decrease of 32% in airway thickness reduction and improvement in lung function.
NGF also significantly enhanced sensory nerve activation mediated by TRPV1, that lead to bronchoconstriction and sensitivity in cough reflexes. Similarly, Liu et al., (2023) found that exposure to NGF reduced epinephrine secretion by 28% (P < 0.01), that could potentially affect ICU management
strategies for asthma exacerbations.
Elevated NGF levels were seen to be associated with prolonged ICU stays ((> 5 days, P = 0.02) and increased ventilatory support requirements. Ogawa et al., (2022) showed that asthma exacerbations were indirectly correlated with NGF levels > 4.5 pg/mL with an odds ratio of 3.21 (95% CI: 2.01–5.14, P < 0.001). Two studies investigated therapeutic interventions that targeted NGF. Ge at al., (2024) displayed that Vitamin D supplementation helped in the downregulation of NGF expression via Nrf2/HO-1 pathway and reduced inflammation by 41%.
Evidence Project of Risk of Bias Tool analyzed risk of bias showed that four studies were classified as low risk, five as moderate and one as high risk. The primary sources o bias included small sample sizes and variety in NGF measurement techniques. The GRADE assessment approach displayed moderate to high confidence in evidences for role of NGF in inflammation and airway remodeling but lower confidence for NGF-targeted therapies due to limit in clinical trials.
4. DISCUSSION
This systematic review had comprehensively analyzed the role of NGF in pathophysiology of asthma by focusing on its role in airway inflammation, remodeling and neuroimmune interactions particularly with respect to intensive care unit (ICU) management. The included studies persistently reported elevated NGF levels in asthmatic patients and animal models. These elevations were linked with increased eosinophilic and neutrophilic infiltration, airway hyperresponsiveness and neurogenic inflammation.21 Furthermore, NGF was involved in the structural changes such as fibroblast proliferation and deposition of collagen which played a part in airway remodeling and progression of disease.22 Noticeably, anti-NGF therapies also demonstrated promising effect s in reduction of inflammation and improvement of lung function, while vitamin D supplementation downregulated NGF expression with the help of the Nrf2/HO-1 pathway.23
The findings of this review aligned with the already existing literature on the multifaceted role of NGF in in inflammatory diseases. This underscored the potential of NGF as a biomarker and therapeutic target in severe cases of asthma.24 Increased levels of NGF were associated with the increase inflammation of airway due to its influence on cytokine production and mast cell degranulation.25 Additionally, NGF-mediated neuroimmune modulation was seen to uplift the activation sensory nerves. This activation contributed to the bronchoconstriction and cough reflex sensitivity.26 These mechanisms supported the notion that NGF played a crucial role in the consistency and exacerbation of asthma symptoms, especially in cases that required ICU management.27
A key strength of this systematic review is its adherence to the PRISMA guidelines that ensured a rigorous methodological approach in literature search, selection of study and synthesis of data.28 The inclusion of diverse study designs provided a detailed overview of NGF’s role in asthma. Moreover, the assessment of bias and certainty of evidences using the optimum approaches further strengthened the validity of the conclusions.29
However, several limitations should be acknowledged. First, the diversity in methodologies of study, NGF measurement techniques and populations of patients limited the ability to perform a meta-analysis.30 Second, the comparatively small sample sizes in some animal studies might had introduced the risk of selection bias, potentially affecting the generalizability of findings.31 Third, while preclinical studies suggested the potential therapeutic benefits of NGF inhibition, the lack of large-scale clinical trials against NGF targeted therapies remained as a gap in the literature.32 Lastly, confounding factors such as environmental factors and comorbidities were not much addressed across studies, thus warranting cautious interpretation of results.33
The results of this review were consistent with previous findings indicating that NGF was a key mediator of airway inflammation and remodeling in asthma.34,35 Earlier studies had demonstrated that NGF was upregulated in BALF and serum derived from asthma patients. This upregulation correlated with severity of disease.36,37 Our findings further supported this relationship by highlighting NGF’s impact on asthma cases relevant to ICU where NGF levels were associated with prolonged hospital stays and greater support requirements for ventilation.
The studies revealed that neurotrophins played a very crucial role in allergic diseases by modulating immune responses, inflammation in air passage way, and airway remodeling. NGF, BDNF, NT-3, and NT-4/5 were seen as major contributors to allergic inflammation due to their role in enhancing mast cell activation, eosinophil survival, and cytokine release 36. Increase in NGF levels had been linked to the elevated bronchial hyperresponsiveness and neuroimmune interactions in asthma, while BDNF had been associated with airway remodeling and increased sensitivity of nerves. NT-3 and NT-4/5, though were not much studied against allergies, also influenced immune cell functioning and inflammation in allergic conditions 37. Therefore, targeting neurotrophins in allergic diseases, particularly asthma, might bring novel therapeutic approaches into sight so that inflammation and airway dysfunction could be mitigated.
Despite the compelling evidence that linked NGF to asthma progression, significant gaps persisted that necessitated focused investigations to advance therapeutic and diagnostic strategies. Firstly, efficacy and safety of NGF-targeted therapies in human asthma patients were needed to be evaluated on large scale. The large-scale trials could remove ambiguities and clarify clinical applicability and optimize dosing regimens.38 Secondly long-term studies were needed to determine whether NGF levels could serve as a predictive biomarker for treatment responsiveness, which could personalize management approaches.39 Mechanistic research should prioritize evaluating mechanistic pathways by which NGF contributed to the inflammation in air passage ways, alteration in immune response and airway remodeling that could uncover novel therapeutic targets.40 Exploring combination therapies was also equally important such as NGF inhibition with corticosteroids or other biologics to assess the potential synergistic effects and improved outcomes of refractory asthma. Finally, confounders like environmental factors must be examined to understand how external factor modulated NGF expression and exacerbate the severity of disease. By addressing these priorities seriously NGF’s role could be studied in-depth and could translate findings into innovative clinical interventions.
Given NGF’s established role in asthma pathophysiology, its potential as a biomarker demanded for further investigation. The identification of NGF as contributor to neuroimmune dysregulation suggested that targeting NGF pathways might offer novel therapeutic strategies for serious and severe cases of asthma. In ICU settings, where managing severe asthma cases remained a clinical challenge, monitoring NGF levels could help in stratifying patients on the basis of risk and personalized interventions. Moreover, the noted downregulation of NGF through Vitamin D supplementation future adjunctive therapies held promise in mitigating NGF-driven inflammation.
| Table 2: Risk of Bias summary of included studies | ||||||||
| Studies | Study Cohort | Control or Comparison Group | Pre/Post Intervention Data | Random Assignment to Intervention | Random Selection for Assessment | Follow-up Rate ≥ 80% | Groups Equivalent on Socio-demographics | Groups Equivalent at Baseline on Disclosure |
| Chen et al. (2018)11 | Yes | Yes | No | No | No | No | No | No |
| Kytikova et al. (2021)12 | Yes | Yes | Yes | No | No | No | No | No |
| Ogawa et al. (2022)13 | Yes | Yes | Yes | No | No | No | No | No |
| Kadry et al. (2024)14 | Yes | Yes | Yes | No | No | No | No | No |
| Björkander et al. (2023)15 | Yes | Yes | Yes | No | No | No | No | No |
| Zhang et al. (2024)16 | Yes | Yes | Yes | No | No | No | No | No |
| Jiang et al. (2020)17 | Yes | Yes | Yes | No | No | No | No | No |
| Liu et al. (2023)18 | Yes | Yes | Yes | No | No | No | No | No |
| Harford et al. (2021)19 | Yes | Yes | Yes | No | No | No | No | No |
| Ge et al. (2024)20 | Yes | Yes | Yes | No | No | No | No | No |
| Yes: Indicates the presence of the specified characteristic, No: Indicates the absence of the specified characteristic | ||||||||
6. CONCLUSION
This systematic review highlighted NGF as a key regulator in asthma related mechanisms, therefore influencing airway inflammation, structural remodeling and neuroimmune regulation. While NGF-targeted therapies showed potential in preclinical studies, further clinical validation was necessary to translate these findings into an effective treatment strategy/ies for severe asthma. Addressing the identified research gaps would be crucial in advancing the understanding of NGF’s role and its potential for future therapeutic avenue for asthma management.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
It is a Systematic Review and All Authors Contributed Equally as per ICMJE.
11. REFERENCES
- Carpaij OA, Burgess JK, Kerstjens HAM, Nawijn MC, van den Berge M. A review on the pathophysiology of asthma remission. Pharmacol Ther. 2019;201:8–24. PubMed DOI: 1016/j.pharmthera.2019.05.002
- Bagnasco D, Paggiaro P, Latorre M, Folli C, Testino E, Bassi A, et al. Severe asthma: One disease and multiple definitions. World Allergy Organ J. 2021;14(11):100606. PubMed DOI: 1016/j.waojou.2021.100606
- Ranjbar M, Whetstone CE, Omer H, Power L, Cusack RP, Gauvreau GM, et al. The genetic factors of the airway epithelium associated with the pathology of asthma. Genes (Basel). 2022;13(10):1870. PubMed DOI: 3390/genes13101870
- Liu P, Li S, Tang L. Nerve growth factor: A potential therapeutic target for lung diseases. Int J Mol Sci. 2021;22(17):9112. PubMed DOI: 3390/ijms22179112
- Zhang Z, Kurashima Y. Two sides of the coin: Mast cells as a key regulator of allergy and acute/chronic inflammation. Cells. 2021;10(7):1615. PubMed DOI: 3390/cells10071615
- Lan YA, Guo JX, Yao MH, Kang YT, Liao ZR, Jing YH. The role of neuro-immune interactions in the pathology and pathogenesis of allergic rhinitis. Immunol Invest. 2024;53(7):1013–29. PubMed DOI: 1080/08820139.2024.2382792
- Roos BB, Teske JJ, Bhallamudi S, Pabelick CM, Sathish V, Prakash YS. Neurotrophin regulation and signaling in airway smooth muscle. Adv Exp Med Biol. 2021;1304:109–21. PubMed DOI: 1007/978-3-030-68748-9_7
- Zhou N, Gu T, Xu Y, Liu Y, Peng L. Challenges and progress of neurodrug: bioactivities, production and delivery strategies of nerve growth factor protein. J Biol Eng. 2023;17(1):75. PubMed DOI: 1186/s13036-023-00392-2
- Savin IA, Zenkova MA, Sen'kova AV. Bronchial asthma, airway remodeling and lung fibrosis as successive steps of one process. Int J Mol Sci. 2023;24(22):16042. PubMed DOI: 3390/ijms242216042
- Meng Z, Chen H, Deng C, Meng S. Potential cellular endocrinology mechanisms underlying the effects of Chinese herbal medicine therapy on asthma. Front Endocrinol (Lausanne). 2022;13:916328. PubMed DOI: 3389/fendo.2022.916328
- Chen J, Kou L, Kong L. Anti-nerve growth factor antibody improves airway hyperresponsiveness by down-regulating RhoA. J Asthma. 2018;55(10):1079–85. PubMed DOI: 1080/02770903.2017.1396467
- Kytikova OY, Novgorodtseva TP, Antonyuk MV, Gvozdenko TA. The role of neurotrophic growth factors in the pathophysiology of bronchial asthma associated with obesity. Bull Sib Med. 2021;20(1):158–67. DOI: 20538/1682-0363-2021-1-158-167
- Ogawa H, Azuma M, Umeno A, Shimizu M, Murotomi K, Yoshida Y, et al. Singlet oxygen-derived nerve growth factor exacerbates airway hyperresponsiveness in a mouse model of asthma with mixed inflammation. Allergol Int. 2022;71(3):395–404. PubMed DOI: 1016/j.alit.2022.02.005
- Kadry ZM, Mohamed NA, Selim SM, Yousef RS. Brain-derived neurotrophic factor (BDNF) single nucleotide gene polymorphism and nerve growth factor are risk factors that increase the severity of allergic rhinitis. Egypt J Chem. 2024;67(10):573–85. DOI: 21608/ejchem.2024.277624.9481
- Björkander S, Klevebro S, Hernandez-Pacheco N, Kere M, Ekström S, Sparreman Mikus M, et al. Obese asthma phenotypes display distinct plasma biomarker profiles. Clin Transl Allergy. 2023;13(3):e12238. PubMed DOI: 1002/clt2.12238
- Zhang S, Kim J, Lee G, Ahn HR, Kim YE, Kim HJ, et al. Phytotherapeutic BS012 and its active component ameliorate allergic asthma via inhibition of Th2-mediated immune response and apoptosis. Biomol Ther (Seoul). 2024;32(6):744–58. PubMed DOI: 4062/biomolther.2024.058
- J Jiang XH, Li CQ, Feng GY, Luo MJ, Sun QX, Huang J. Mycobacterium vaccae nebulization protects Balb/c mice against bronchial asthma through neural mechanisms. J Asthma. 2021;58(8):1003–12. PubMed DOI: 1080/02770903.2020.1761381
- Liu C, Liang JP, Huang XL, Liu Z, Zhang AB, Deng NG, et al. Nerve growth factor causes epinephrine release dysfunction by regulating phenotype alterations and the function of adrenal medullary chromaffin cells in mice with allergic rhinitis. Mol Med Rep. 2023;27(2):39. PubMed DOI: 3892/mmr.2023.12926
- Harford TJ, Grove L, Rezaee F, Scheraga R, Olman MA, Piedimonte G. RSV infection potentiates TRPV1-mediated calcium transport in bronchial epithelium of asthmatic children. Am J Physiol Lung Cell Mol Physiol. 2021;320(6):L1074–84. PubMed DOI: 1152/ajplung.00531.2020
- Ge D, Chen Q, Xie X, Li Q, Yang Y Unveiling the potent effect of vitamin D: harnessing Nrf2/HO-1 signaling pathways as molecular targets to alleviate urban particulate matter-induced asthma inflammation. BMC Pulm Med. 2024;24(1):55. PubMed DOI: 1186/s12890-024-02869-2
- Chang RS, Wang SD, Wang YC, et al. Xiao-Qing-Long-Tang shows preventive effect of asthma in an allergic asthma mouse model through neurotrophin regulation. BMC Complement Altern Med. 2013;13:220. PubMed DOI: 1186/1472-6882-13-220
- Zhang N, Xu J, Jiang C, Lu S. Neuro-immune regulation in inflammation and airway remodeling of allergic asthma. Front Immunol. 2022;13:894047. PubMed DOI: 3389/fimmu.2022.894047
- Su R, Su W, Jiao Q. [Retracted] NGF protects neuroblastoma cells against β-amyloid-induced apoptosis via the Nrf2/HO-1 pathway. FEBS Open Bio. 2019;9(12):2063–71. PubMed DOI: 1002/2211-5463.12742
- Szczepankiewicz A, Sobkowiak P, Rachel M, et al. Multilocus analysis of candidate genes involved in neurogenic inflammation in pediatric asthma and related phenotypes: a case-control study. J Asthma. 2012;49(4):329–35. PubMed DOI: 3109/02770903.2012.669442
- Costanzo G, Marchetti M, Ledda AG, et al. Mast cells in allergic and non-allergic upper airways diseases: sentinel in the watchtower. Int J Mol Sci. 2024;25(23):12615. PubMed DOI: 3390/ijms252312615
- El-Hashim AZ, Jaffal SM. Cough reflex hypersensitivity: a role for neurotrophins. Exp Lung Res. 2017;43(2):93–108. PubMed DOI: 1080/01902148.2017.1290162
- Dwyer DF, Ordovas-Montanes J, Allon SJ, et al. Human airway mast cells proliferate and acquire distinct inflammation-driven phenotypes during type 2 inflammation. Sci Immunol. 2021;6(56):eabb7221. PubMed DOI: 1126/sciimmunol.abb7221
- Perner C, Flayer CH, Zhu X, et al. Substance P release by sensory neurons triggers dendritic cell migration and initiates the type-2 immune response to allergens. Immunity. 2020;53(5):1063–77.e7. PubMed DOI: 1016/j.immuni.2020.10.001
- Ural BB, Yeung ST, Damani-Yokota P, et al. Identification of a nerve-associated, lung-resident interstitial macrophage subset with distinct localization and immunoregulatory properties. Sci Immunol. 2020;5(45):eaax8756. PubMed DOI: 1126/sciimmunol.aax8756
- Varricchi G, Ferri S, Pepys J, et al. Biologics and airway remodeling in severe asthma. Allergy. 2022;77(12):3538–52. PubMed DOI: 1111/all.15473
- Salvador AF, de Lima KA, Kipnis J. Neuromodulation by the immune system: a focus on cytokines. Nat Rev Immunol. 2021;21(8):526–41. PubMed DOI: 1038/s41577-021-00508-z
- Vafaee F, Shirzad S, Shamsi F, Boskabady MH. Neuroscience and treatment of asthma, new therapeutic strategies and future aspects. Life Sci. 2022;292:120175. PubMed DOI: 1016/j.lfs.2021.120175
- Chatkin J, Correa L, Santos U. External environmental pollution as a risk factor for asthma. Clin Rev Allergy Immunol. 2022;62(1):72–89. PubMed DOI: 1007/s12016-020-08830-5
- Shen YJ, Ou PH, Shen YC, Lai CJ. Role of endogenous nerve growth factor in laryngeal airway hyperreactivity and laryngeal inflammation induced by intermittent hypoxia in rats. Respir Physiol Neurobiol. 2025;332:104372. PubMed DOI: 1016/j.resp.2024.104372
- She W, Mei Z, Zhao H, et al. Nebulized inhalation of anti-nerve growth factor microspheres inhibits airway remodeling in an ovalbumin-induced rat asthma model. J Aerosol Med Pulm Drug Deliv. 2019;32(2):70–7. PubMed DOI: 1089/jamp.2018.1453
- Hudey SN, Ledford DK, Cardet JC. Mechanisms of non-type 2 asthma. Curr Opin Immunol. 2020;66:123–8. PubMed DOI: 1016/j.coi.2020.10.002
- Kuruvilla ME, Lee FE, Lee GB. Understanding asthma phenotypes, endotypes, and mechanisms of disease. Clin Rev Allergy Immunol. 2019;56(2):219–33. PubMed DOI: 1007/s12016-018-8712-1
- Guida G, Riccio AM. Immune induction of airway remodeling. Semin Immunol. 2019;46:101346. PubMed DOI: 1016/j.smim.2019.101346
- Wang P, Liu H, Fan X, et al. Effect of San'ao decoction on aggravated asthma mice model induced by PM2.5 and TRPA1/TRPV1 expressions. J Ethnopharmacol. 2019;236:82–90. PubMed DOI: 1016/j.jep.2019.02.043
- Nygaard UC, Xiao L, Nadeau KC, et al. Improved diet quality is associated with decreased concentrations of inflammatory markers in adults with uncontrolled asthma. Am J Clin Nutr. 2021;114(3):1012–27. PubMed DOI: 1093/ajcn/nqab063