Ahmed Elbaiomy Abo ElKheir 1, Mohammed Said ElSharkawy *1, Mohamed Hamed Abd El Aziz 2, Khaled Hamama 1, Marwa Ahmed El Oraby Elfert 1
Authors affiliations:
Background & objectives: Although spinal anesthesia (SA) has largely replaced general anesthesia (GA) for inguinal herniorrhaphy (IH), the anesthetist might be compelled to go for GA in some special cases, including patient refusal to have SA, or neonates, young children or female patients. We conducted this study to compare the efficacy of SA and GA in neonates undergoing inguinal herniorrhaphy (IH).
Methodology: This randomized controlled study was conducted on 74 neonates classified as American Society of Anesthesiology physical status I-II undergoing IH. The patients were randomized equally into two groups. GA group: GA was maintained with 2% sevoflurane. SA group: SA was performed using 0.6 mg/kg of bupivacaine 0.5%. The patients were sedated by administering intermittent injections of ketamine 1 mg, as required. Oxygen was administeredthrough a nasal cannula.
Results: The heart rate measurements were significantly higher, and mean arterial blood pressure measurements were significantly lower at 20, 30, 40, 50, and 60min intraoperatively, 30, 60, 90, and 120min in PACU in the SA in comparison to GA group (P < 0.05). Oxygen (O2) saturation measurements were significantly higher at 30, 60, 90, and 120 min in PACU in the SA in comparison to the GA group (P < 0.05). There was no discernible variation within both groups in the incidence of bradycardia, hypotension, the need for postoperative O2 supplementation, and hospital stays. The incidence of apnea was substantially decreased in the SA group compared to the GA group (12 (32.43%) vs 4 (10.81%), P = 0.045).
Conclusions: SA is a viable alternative for GA in neonates undergoing IH, as it offers better hemodynamic and O2 saturation stability and reduces postoperative apnea.
Abbreviations: IH: inguinal herniorrhaphy, GA: general anesthesia, PACU: post-anesthesia care unit SA: spinal anesthesia,
Keywords: Spinal anesthesia; General anesthesia; Herniorrhaphy; Neonates; Heart rate
Citation: Abo ElKheir AE, ElSharkawy MS, Abd El Aziz MH, Hamama K, Elfert MAE. A randomized controlled trial comparing spinal and general anesthesia in neonates undergoing inguinal herniorrhaphy. Anaesth. pain intensive care 2025;29(3):397-402. DOI: 10.35975/apic.v29i3.2769
Received: December 09, 2024; Revised: February 08, 2025; Accepted: February 08, 2025
Inguinal hernia is a prevalent surgical issue, with a prevalence of 1:3000 births. Early surgery prevents incarceration, bowel ischemia, and gonadal infarction.1
The physiology of neonates is distinguished by a high metabolic rate and a restricted cardiac, pulmonary, renal, and thermoregulatory reserve. The neurocognitive development of the neonate may be adversely affected by these metabolic and hemodynamic alterations in the perioperative period. This immaturity generates differences in the pharmacokinetics and dynamics of the drug compared to older children or adults.2
With general anesthesia (GA), patients may need positive pressure ventilation assistance. Also, GA impairs cardiovascular function. Either inhalational or intravenous drugs can deliver GA. Neonates are more vulnerable to hypotension, which may impair cerebral perfusion. So, maintaining normotension and normocarbia is highly important intraoperatively.3 Also, complications of GA are significantly increased in neonates, including apnea and bradycardia.4,5
In neonates, regional anesthesia (RA) may be beneficial for many clinical reasons. These include the avoidance of respiratory depression or airway manipulation.6 Moreover, RA improves perioperative pain management and decreases the possible neurotoxic effects of the drug used in GA.2
Spinal anesthesia (SA) is a cost-effective, straightforward, and rapid technique primarily employed in pediatric anesthesia, including infants, neonates, and the preterm.7-10 SA facilitates the prevention and mitigation of perioperative complications.11
A minimal risk of postoperative events was observed in a large observational study of SA for infants undergoing inguinal herniorrhaphy (IH).12 Compared to GA, SA reduces respiratory morbidity and oxygen (O2) desaturation in babies who were born prematurely and had IH postoperatively.13 This study aims to compare SA and GA in neonates undergoing IH to broaden our understanding of this concept.
We conducted this open-label, randomized controlled study on a sample of 74 neonates, both sexes, American Society of Anesthesiology (ASA) physical status I-II who underwent IH. The study was done at Tanta University Hospitals, Egypt, after approval from the institutional ethical committee and registration at clinicaltrials.gov (NCT06352606) from February 2024 to July 2024. Informed written consent was obtained from the patient.
The exclusion criteria were obstructed hernia, severe chronic pulmonary disease (e.g., a condition that is associated with recurrent hypercapnia or hypoxemia in ambient air), recognized congenital cardiac defect accompanied with symptoms (such as cyanosis or congestive heart failure), or symptomatic disease of the central nervous system (e.g., seizures).
2.1. Randomization and blindness:
Participants were randomly assigned using computer-generated randomization numbers by using an online randomization program (http://www.randomizer.org) to generate a random list; every patient had a secret code number. Patients were assigned randomly to two cohorts using a 1:1 allocation ratio in a parallel manner to receive either GA or SA. Because of the variety of approaches used, the research was classified as open-label.
History-taking, clinical examination, and routine laboratory tests were done preoperatively. Patients fast for solid food for 6 hours for artificial milk, 4 hours for nonclear and breast milk, and 2 hours for clear liquids.
Capnography, ECG, non-invasive blood pressure monitoring, pulse oximetry, and temperature probes were utilized to monitor patients' standards.
2.2. Spinal anesthesia technique:
The neonates' upper extremities were secured with flexible restraints to prevent abrupt movement. Oral sucrose drips were permissible. The patient was sedated by administering intermittent injections of ketamine 1 mg, as required. O2 was administered through a nasal cannula at a rate of two L/min when required. The lateral decubitus position was used to administer SA using a 25-gauge spinal catheter at the L3/4 or L4/5 level, and 0.6 mg/kg of bupivacaine (0.5%) was administered. If GA is required, the patient is excluded and replaced by another.
2.4. General anesthesia technique:
Induction of GA was performed using sevoflurane 6-8% and 80% O2/air. GA was maintained with sevoflurane (2%) and 50% O2/air with assisted spontaneous ventilation.
Opioids were not administered intraoperatively. Paracetamol 15 mg/kg was administered in both groups before skin incision. The surgery was done by expert surgeons using the same technique.
The mean arterial blood pressure (MAP), heart rate (HR), and O2 saturation were recorded at baseline, every 10 min intraoperatively, and then every 30 min in the post-anesthesia care unit (PACU). Babies were carefully observed and continuously observed in the PACU for two hours following the operation. The need for O2 supplementation was recorded.
The term "apnea" refers to a respiratory delay that exceeds 15 sec or 10 sec if it is accompanied by bradycardia (a 20% decrease in HR) or O2 saturation below 80%.
The interventions required for postoperative apnea included; tactile stimulation, caffeine administration, and the provision of O2 via a respirator (both with and without positive pressure ventilation).
The primary outcome was hemodynamic parameters including HR and MAP, duration of surgery, incidence of adverse events (bradycardia, hypotension, postoperative apnea), need for postoperative O2 supplementation, and length of hospital stay.
2.4. Sample Size Calculation:
We calculated the size of the sample utilizing G*Power 3.1.9.2 (Universitat Kiel, Germany). Based on a prior study [13], the mean ± standard deviation of postoperative HR (the primary outcome) was 108.1 ± 29.7 beats/min with GA and 124.6 ± 15.2 beats/min with SA. The following parameters were used: 95% confidence limit, 0.699 effect size, 80% power of the study, a 1:1 group ratio, and three cases were incorporated into each group to address the drop-out issue. Therefore, we included 37 patients in each cohort.
2.5. Statistical analysis
We utilized SPSS v27 (IBM©, Armonk, NY, USA) for statistical analysis. The Shapiro-Wilks test and histograms were implemented to assess the normality of the data distribution. The quantitative parametric data presented as the mean and standard deviation (SD) were analyzed using the unpaired student t-test. The qualitative variables, which were presented as frequency (%), were analyzed using the Chi-square test or Fisher's exact test. A two-tailed P less than 0.05 was used to define statistical significance.
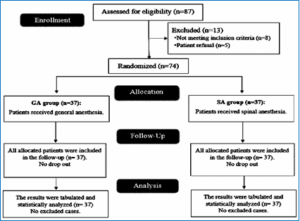
1: CONSORT flowchart of the enrolled Figure patients
Eighty-seven patients were evaluated for eligibility in this study; 8 did not satisfy the criteria, and the
guardians of the 5 decided not to participate. All other babies were randomly assigned to one of the two groups, each consisting of 37 babies. The follow-up and statistical analysis of all allocated patients were conducted and there was no drop-outs (Figure 1).
Demographic data and surgery duration did not exhibit statistically significant differences between the two groups. Table 1
HR and MAP measurements did not vary considerably at baseline and 10 min within the grouping. HR measurements were significantly higher, and MAP measurements were significantly lower at 20, 30, 40, 50, 60 min intraoperatively, 30, 60, 90, and 120 min in PACU in the SA in comparison to the GA group (P < 0.05). Figure 2
The O2 saturation measurements were not significantly different at baseline, 10, 20, 30, 40, 50, and 60 min intraoperatively between both groups and were markedly more elevated at 30, 60, 90, and 120 min in PACU in the SA in comparison to in GA group (P < 0.05).
The two groups did not significantly differ in bradycardia, hypotension, the need for postoperative O2 supplementation, and hospital stays. The SA group exhibited a substantially lower incidence of apnea than the GA group (12 (32.43%) vs 4 (10.81%), P = 0.045). Table 2
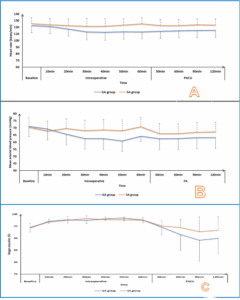
Figure 2: (A) Heart rate; (B) Mean arterial blood pressure, and (C) SpO2 changes of the studied groups
IH repair is frequently performed on preterm and term infants.14,15 SA is being considered as a potential substitute for safeguarding against GA during the early phases of life.16
HR measurements were significantly higher, and MAP measurements were significantly lower at 20, 30, 40, 50, and 60 min intraoperatively, 30, 60, 90, and 120 min in PACU in the SA in comparison to the GA group. O2 saturation measurements were markedly more elevated at 30, 60, 90, and 120 min in PACU in the SA compared to the GA group. Similarly to what we discovered, Kim et al. illustrated that the HR and O2 saturation were notably increased in the SA in comparison to the GA group, while herniorrhaphy in babies that were born prematurely.13 Also, Davidson et al. informed that the intraoperative heart rate in the RA group (spinal alone, spinal plus caudal, spinal plus ilioinguinal, or caudal alone) was marginally higher than the GA group for IH.17
Moreover, Ali et al.,13 found that blood pressure was notably reduced in the SA in comparison to the GA group in neonates undergoing infra umbilical surgeries. However, the spinal group exhibited significantly reduced heart rate and O2 saturation than the GA group. This difference is due to our study's high incidence of apnea, which affects HR and O2 saturation.
In addition, the meta-analysis by Jones et al. explained that O2 saturation was considerably lower in those who were administered SA than those who were administered GA in preterm neonates undergoing IH in early infancy.18
Our results revealed that the incidence of bradycardia, hypotension, and hospital stays was not significantly different in both groups. Aligned with our results, Kim et al. found that the SA and GA groups exhibited no significant differences in bradycardia or hospital stay.13 Moreover, Lambertz et al. found that hospital stay was insignificantly different between SA and GA groups for IH repair in infants.19 Also, Jones et al. reported the duration of hospitalization without a substantial distinction between SA and GA.18 Conversely, Somri et al. discovered that there was a substantial prolonged hospital stay in GA compared to SA with herniorrhaphy in newborns at high risk.20 This difference may be due to the inclusion of only high-risk (ASA physical status II or greater) infants who suffer from complications other than IH, which may extend the hospital stay compared to our study, which included full-term and preterm neonates.
In our study, the SA group exhibited a significantly reduced incidence of apnea than the GA group, and the need for postoperative O2 supplementation was insignificant between both groups. This is supported by Frumiento et al.,21 who showed that the documented apnea rates in neonates after GA are considerably higher than the potential for apnea in premature newborns after SA. In addition, Jones et al.,18 revealed that the possibility of apnea after surgery was diminished by 47% for preterm infants who did not receive any sedation when SA was used instead of GA. Additionally, the need for respiratory support was reduced in preterm infants who received SA, although this difference was not statistically significant. Furthermore, Davidson et al. demonstrated that the apnea rate was greatly lower in the RA in comparison to the GA group.17 In contrast, postoperative O2 supplementation was insignificantly different between the RA and GA groups. This difference may be due to their larger sample size.
Our trial has had few limitations including a relatively limited sample size, and it was conducted at a single center. Also, we could not record postoperative pain scores in the neonates, and did not record postoperative analgesic requirements. Further studies are required using various additives, concentrations, volumes, and blocks.
Spinal anesthesia is a viable substitute for general anesthesia in neonates undergoing inguinal herniorrhaphy, as it offers hemodynamic stability, satisfactory oxygenation and reduced postoperative apnea.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
AEAE and KH: Contributed to the development of the protocol, abstracted data.
MSE and MAEOE: Developed the original idea and the protocol, abstracted and analysed data, wrote the manuscript, and is a guarantor.
MHAE: Prepared the manuscript.
Authors affiliations:
- Ahmed Elbaiomy Abo ElKheir, Lecturer, Department of Anesthesia, ICU & Pain Management, Faculty of Medicine, Tanta University, Tanta, Egypt; Email: ahmed_baiomy@med.tanta.edu.eg
- Mohammed Said ElSharkawy, , Lecturer, Department of Anesthesia, ICU & Pain Management, Faculty of Medicine, Tanta University, Tanta, Egypt; Email: mselsharkawy@med.tanta.edu.eg
- Mohamed Hamed Abd El Aziz, Lecturer, Department of Anesthesia, ICU & Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: ert.bas.mh@gmail.com
- Khaled Hamama, , Lecturer, Department of Anesthesia, ICU & Pain Management, Faculty of Medicine, Tanta University, Tanta, Egypt; Email: khaled.hamama@med.tanta.edu.eg
- Marwa Ahmed El Oraby Elfert, , Lecturer, Department of Anesthesia, ICU & Pain Management, Faculty of Medicine, Tanta University, Tanta, Egypt; Email: dr.marwaoraby5@gmail.com
ABSTRACT
Background & objectives: Although spinal anesthesia (SA) has largely replaced general anesthesia (GA) for inguinal herniorrhaphy (IH), the anesthetist might be compelled to go for GA in some special cases, including patient refusal to have SA, or neonates, young children or female patients. We conducted this study to compare the efficacy of SA and GA in neonates undergoing inguinal herniorrhaphy (IH).
Methodology: This randomized controlled study was conducted on 74 neonates classified as American Society of Anesthesiology physical status I-II undergoing IH. The patients were randomized equally into two groups. GA group: GA was maintained with 2% sevoflurane. SA group: SA was performed using 0.6 mg/kg of bupivacaine 0.5%. The patients were sedated by administering intermittent injections of ketamine 1 mg, as required. Oxygen was administeredthrough a nasal cannula.
Results: The heart rate measurements were significantly higher, and mean arterial blood pressure measurements were significantly lower at 20, 30, 40, 50, and 60min intraoperatively, 30, 60, 90, and 120min in PACU in the SA in comparison to GA group (P < 0.05). Oxygen (O2) saturation measurements were significantly higher at 30, 60, 90, and 120 min in PACU in the SA in comparison to the GA group (P < 0.05). There was no discernible variation within both groups in the incidence of bradycardia, hypotension, the need for postoperative O2 supplementation, and hospital stays. The incidence of apnea was substantially decreased in the SA group compared to the GA group (12 (32.43%) vs 4 (10.81%), P = 0.045).
Conclusions: SA is a viable alternative for GA in neonates undergoing IH, as it offers better hemodynamic and O2 saturation stability and reduces postoperative apnea.
Abbreviations: IH: inguinal herniorrhaphy, GA: general anesthesia, PACU: post-anesthesia care unit SA: spinal anesthesia,
Keywords: Spinal anesthesia; General anesthesia; Herniorrhaphy; Neonates; Heart rate
Citation: Abo ElKheir AE, ElSharkawy MS, Abd El Aziz MH, Hamama K, Elfert MAE. A randomized controlled trial comparing spinal and general anesthesia in neonates undergoing inguinal herniorrhaphy. Anaesth. pain intensive care 2025;29(3):397-402. DOI: 10.35975/apic.v29i3.2769
Received: December 09, 2024; Revised: February 08, 2025; Accepted: February 08, 2025
1. INTRODUCTION
Inguinal hernia is a prevalent surgical issue, with a prevalence of 1:3000 births. Early surgery prevents incarceration, bowel ischemia, and gonadal infarction.1
The physiology of neonates is distinguished by a high metabolic rate and a restricted cardiac, pulmonary, renal, and thermoregulatory reserve. The neurocognitive development of the neonate may be adversely affected by these metabolic and hemodynamic alterations in the perioperative period. This immaturity generates differences in the pharmacokinetics and dynamics of the drug compared to older children or adults.2
With general anesthesia (GA), patients may need positive pressure ventilation assistance. Also, GA impairs cardiovascular function. Either inhalational or intravenous drugs can deliver GA. Neonates are more vulnerable to hypotension, which may impair cerebral perfusion. So, maintaining normotension and normocarbia is highly important intraoperatively.3 Also, complications of GA are significantly increased in neonates, including apnea and bradycardia.4,5
In neonates, regional anesthesia (RA) may be beneficial for many clinical reasons. These include the avoidance of respiratory depression or airway manipulation.6 Moreover, RA improves perioperative pain management and decreases the possible neurotoxic effects of the drug used in GA.2
Spinal anesthesia (SA) is a cost-effective, straightforward, and rapid technique primarily employed in pediatric anesthesia, including infants, neonates, and the preterm.7-10 SA facilitates the prevention and mitigation of perioperative complications.11
A minimal risk of postoperative events was observed in a large observational study of SA for infants undergoing inguinal herniorrhaphy (IH).12 Compared to GA, SA reduces respiratory morbidity and oxygen (O2) desaturation in babies who were born prematurely and had IH postoperatively.13 This study aims to compare SA and GA in neonates undergoing IH to broaden our understanding of this concept.
2. METHODOLOGY
We conducted this open-label, randomized controlled study on a sample of 74 neonates, both sexes, American Society of Anesthesiology (ASA) physical status I-II who underwent IH. The study was done at Tanta University Hospitals, Egypt, after approval from the institutional ethical committee and registration at clinicaltrials.gov (NCT06352606) from February 2024 to July 2024. Informed written consent was obtained from the patient.
The exclusion criteria were obstructed hernia, severe chronic pulmonary disease (e.g., a condition that is associated with recurrent hypercapnia or hypoxemia in ambient air), recognized congenital cardiac defect accompanied with symptoms (such as cyanosis or congestive heart failure), or symptomatic disease of the central nervous system (e.g., seizures).
2.1. Randomization and blindness:
Participants were randomly assigned using computer-generated randomization numbers by using an online randomization program (http://www.randomizer.org) to generate a random list; every patient had a secret code number. Patients were assigned randomly to two cohorts using a 1:1 allocation ratio in a parallel manner to receive either GA or SA. Because of the variety of approaches used, the research was classified as open-label.
History-taking, clinical examination, and routine laboratory tests were done preoperatively. Patients fast for solid food for 6 hours for artificial milk, 4 hours for nonclear and breast milk, and 2 hours for clear liquids.
Capnography, ECG, non-invasive blood pressure monitoring, pulse oximetry, and temperature probes were utilized to monitor patients' standards.
2.2. Spinal anesthesia technique:
The neonates' upper extremities were secured with flexible restraints to prevent abrupt movement. Oral sucrose drips were permissible. The patient was sedated by administering intermittent injections of ketamine 1 mg, as required. O2 was administered through a nasal cannula at a rate of two L/min when required. The lateral decubitus position was used to administer SA using a 25-gauge spinal catheter at the L3/4 or L4/5 level, and 0.6 mg/kg of bupivacaine (0.5%) was administered. If GA is required, the patient is excluded and replaced by another.
2.4. General anesthesia technique:
Induction of GA was performed using sevoflurane 6-8% and 80% O2/air. GA was maintained with sevoflurane (2%) and 50% O2/air with assisted spontaneous ventilation.
Opioids were not administered intraoperatively. Paracetamol 15 mg/kg was administered in both groups before skin incision. The surgery was done by expert surgeons using the same technique.
The mean arterial blood pressure (MAP), heart rate (HR), and O2 saturation were recorded at baseline, every 10 min intraoperatively, and then every 30 min in the post-anesthesia care unit (PACU). Babies were carefully observed and continuously observed in the PACU for two hours following the operation. The need for O2 supplementation was recorded.
The term "apnea" refers to a respiratory delay that exceeds 15 sec or 10 sec if it is accompanied by bradycardia (a 20% decrease in HR) or O2 saturation below 80%.
The interventions required for postoperative apnea included; tactile stimulation, caffeine administration, and the provision of O2 via a respirator (both with and without positive pressure ventilation).
The primary outcome was hemodynamic parameters including HR and MAP, duration of surgery, incidence of adverse events (bradycardia, hypotension, postoperative apnea), need for postoperative O2 supplementation, and length of hospital stay.
2.4. Sample Size Calculation:
We calculated the size of the sample utilizing G*Power 3.1.9.2 (Universitat Kiel, Germany). Based on a prior study [13], the mean ± standard deviation of postoperative HR (the primary outcome) was 108.1 ± 29.7 beats/min with GA and 124.6 ± 15.2 beats/min with SA. The following parameters were used: 95% confidence limit, 0.699 effect size, 80% power of the study, a 1:1 group ratio, and three cases were incorporated into each group to address the drop-out issue. Therefore, we included 37 patients in each cohort.
2.5. Statistical analysis
We utilized SPSS v27 (IBM©, Armonk, NY, USA) for statistical analysis. The Shapiro-Wilks test and histograms were implemented to assess the normality of the data distribution. The quantitative parametric data presented as the mean and standard deviation (SD) were analyzed using the unpaired student t-test. The qualitative variables, which were presented as frequency (%), were analyzed using the Chi-square test or Fisher's exact test. A two-tailed P less than 0.05 was used to define statistical significance.

1: CONSORT flowchart of the enrolled Figure patients
3. RESULTS
Eighty-seven patients were evaluated for eligibility in this study; 8 did not satisfy the criteria, and the
guardians of the 5 decided not to participate. All other babies were randomly assigned to one of the two groups, each consisting of 37 babies. The follow-up and statistical analysis of all allocated patients were conducted and there was no drop-outs (Figure 1).
Demographic data and surgery duration did not exhibit statistically significant differences between the two groups. Table 1
| Table 1: Demographic data and duration of surgery of the studied groups | ||||
| Parameters | GA group (n = 37) |
SA group (n = 37) |
P | |
| Gestational age (weeks) | 36.78 ± 2.37 | 37.41 ± 2.19 | 0.245 | |
| Gender | Male | 32 (86.49%) | 34 (91.89%) | 0.710 |
| Female | 5 (13.51%) | 3 (8.11%) | ||
| ASA physical status | I | 30 (81.08%) | 33 (89.19%) | 0.515 |
| II | 7 (18.92%) | 4 (10.81%) | ||
| Duration of surgery (min) | 42.57 ± 12.17 | 43.92 ± 13.08 | 0.647 | |
| Data are presented as mean ± SD or frequency (%); P < 0.05 considered as significant. | ||||
The O2 saturation measurements were not significantly different at baseline, 10, 20, 30, 40, 50, and 60 min intraoperatively between both groups and were markedly more elevated at 30, 60, 90, and 120 min in PACU in the SA in comparison to in GA group (P < 0.05).
The two groups did not significantly differ in bradycardia, hypotension, the need for postoperative O2 supplementation, and hospital stays. The SA group exhibited a substantially lower incidence of apnea than the GA group (12 (32.43%) vs 4 (10.81%), P = 0.045). Table 2
| Table 2: Comparative postoperative status and hospital stays of the studied groups | ||||
| Postoperative status | GA group (n = 37) |
SA group (n = 37) |
P | |
| Adverse events | Bradycardia | 4 (10.81%) | 1 (2.7%) | 0.357 |
| Hypotension | 7 (18.92%) | 3 (8.11%) | 0.308 | |
| Apnea | 12 (32.43%) | 4 (10.81%) | 0.045 | |
| Need for postoperative O2 supplementation | 7 (18.92%) | 2 (5.41%) | 0.152 | |
| Hospital stays (days) | 6 (5−15) | 6 (3−14) | 0.154 | |
| Data presented as median (IQR) or frequency (%); P < 0.05 considered as significant | ||||

Figure 2: (A) Heart rate; (B) Mean arterial blood pressure, and (C) SpO2 changes of the studied groups
DISCUSSION
IH repair is frequently performed on preterm and term infants.14,15 SA is being considered as a potential substitute for safeguarding against GA during the early phases of life.16
HR measurements were significantly higher, and MAP measurements were significantly lower at 20, 30, 40, 50, and 60 min intraoperatively, 30, 60, 90, and 120 min in PACU in the SA in comparison to the GA group. O2 saturation measurements were markedly more elevated at 30, 60, 90, and 120 min in PACU in the SA compared to the GA group. Similarly to what we discovered, Kim et al. illustrated that the HR and O2 saturation were notably increased in the SA in comparison to the GA group, while herniorrhaphy in babies that were born prematurely.13 Also, Davidson et al. informed that the intraoperative heart rate in the RA group (spinal alone, spinal plus caudal, spinal plus ilioinguinal, or caudal alone) was marginally higher than the GA group for IH.17
Moreover, Ali et al.,13 found that blood pressure was notably reduced in the SA in comparison to the GA group in neonates undergoing infra umbilical surgeries. However, the spinal group exhibited significantly reduced heart rate and O2 saturation than the GA group. This difference is due to our study's high incidence of apnea, which affects HR and O2 saturation.
In addition, the meta-analysis by Jones et al. explained that O2 saturation was considerably lower in those who were administered SA than those who were administered GA in preterm neonates undergoing IH in early infancy.18
Our results revealed that the incidence of bradycardia, hypotension, and hospital stays was not significantly different in both groups. Aligned with our results, Kim et al. found that the SA and GA groups exhibited no significant differences in bradycardia or hospital stay.13 Moreover, Lambertz et al. found that hospital stay was insignificantly different between SA and GA groups for IH repair in infants.19 Also, Jones et al. reported the duration of hospitalization without a substantial distinction between SA and GA.18 Conversely, Somri et al. discovered that there was a substantial prolonged hospital stay in GA compared to SA with herniorrhaphy in newborns at high risk.20 This difference may be due to the inclusion of only high-risk (ASA physical status II or greater) infants who suffer from complications other than IH, which may extend the hospital stay compared to our study, which included full-term and preterm neonates.
In our study, the SA group exhibited a significantly reduced incidence of apnea than the GA group, and the need for postoperative O2 supplementation was insignificant between both groups. This is supported by Frumiento et al.,21 who showed that the documented apnea rates in neonates after GA are considerably higher than the potential for apnea in premature newborns after SA. In addition, Jones et al.,18 revealed that the possibility of apnea after surgery was diminished by 47% for preterm infants who did not receive any sedation when SA was used instead of GA. Additionally, the need for respiratory support was reduced in preterm infants who received SA, although this difference was not statistically significant. Furthermore, Davidson et al. demonstrated that the apnea rate was greatly lower in the RA in comparison to the GA group.17 In contrast, postoperative O2 supplementation was insignificantly different between the RA and GA groups. This difference may be due to their larger sample size.
5. LIMITATIONS
Our trial has had few limitations including a relatively limited sample size, and it was conducted at a single center. Also, we could not record postoperative pain scores in the neonates, and did not record postoperative analgesic requirements. Further studies are required using various additives, concentrations, volumes, and blocks.
6. CONCLUSIONS
Spinal anesthesia is a viable substitute for general anesthesia in neonates undergoing inguinal herniorrhaphy, as it offers hemodynamic stability, satisfactory oxygenation and reduced postoperative apnea.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
AEAE and KH: Contributed to the development of the protocol, abstracted data.
MSE and MAEOE: Developed the original idea and the protocol, abstracted and analysed data, wrote the manuscript, and is a guarantor.
MHAE: Prepared the manuscript.
11. REFERENCES
- Alrzoq RA, Alhaji AMA, Alolayt IYI, Almulhim AI, Althobaiti SA, Alzahrani IA, et al. The incidence of inguinal hernia among children. Egypt J Hosp Med. 2018;70:483-6. Full Text
- McCann ME, Schouten AN. Beyond survival; influences of blood pressure, cerebral perfusion and anesthesia on neurodevelopment. Paediatr Anaesth. 2014;24:68-73. PubMedDOI: 10.1111/pan.12310
- Ali MH, EL Taher E, Lamei M, Al-Touny SA. Spinal anesthesia versus general anesthesia in neonates undergoing infraumbilical surgeries. SCUMJ. 2022;25:36-47. DOI: 10.21608/scumj.2022.266179
- Dohms K, Hein M, Rossaint R, Coburn M, Stoppe C, Ehret CB, et al. Inguinal hernia repair in preterm neonates: is there evidence that spinal or general anaesthesia is the better option regarding intraoperative and postoperative complications? A systematic review and meta-analysis. BMJ Open. 2019;19:728-9. PubMedDOI: 10.1136/bmjopen-2018-028728
- Picache D, Gluskin D, Noor A, Senken B, Fiorito T, Akerman M, et al. Respiratory syncytial virus infection and apnea risk as criteria for hospitalization in full term healthy infants. Cureus. 2024;16:214-8. PubMedDOI: 10.7759/cureus.53845
- Wingert TEA, Hekmat D, Ayad I. Regional Anesthesia for Neonates. Neoreviews. 2023;24:626-41. PubMedDOI: 10.1542/neo.24-10-e626
- López T, Sánchez FJ, Garzón JC, Muriel C. Spinal anesthesia in pediatric patients. Minerva Anestesiol. 2012;78:78-87. PubMed DOI: 10.1111/j.1460-9592.2011.03769.x
- Frawley G, Ingelmo P. Spinal anaesthesia in the neonate. Best Pract Res Clin Anaesthesiol. 2010;24:337-51. PubMed DOI: 10.1016/j.bpa.2010.02.018
- Randriamizao HM, Rakotondrainibe A, Razafindrabekoto LD, Ravoaviarivelo PF, Rajaonera AT, Andriamanarivo ML. Use of spinal anaesthesia in neonates and infants in antananarivo, madagascar: A retrospective descriptive study. BMC Research Notes. 2020;13:1-6. PubMed DOI: 10.1186/s13104-020-05330-9
- Frawley G. Second infant spinal anesthetic: Incidence, dose modification, and adverse events after initial failure. Paediatr Anaesth. 2024;34:324-31. PubMed DOI: 10.1111/pan.14831
- Verma D, Naithani U, Gokula C. Spinal anesthesia in infants and children: a one year prospective audit. Anesth Essays Res. 2014;8:324-9. PubMed DOI: 10.4103/0259-1162.143124
- Yildirim D, Hut A, Uzman S, Kocakusak A, Demiryas S, Cakir M, et al. Spinal anesthesia is safe in laparoscopic total extraperitoneal inguinal hernia repair. A retrospective clinical trial. Wideochir Inne Tech Maloinwazyjne. 2017;12:417-27. PubMed DOI: 10.5114/wiitm.2017.72325
- Kim GS, Song JG, Gwak MS, Yang M. Postoperative outcome in formerly premature infants undergoing herniorrhaphy: comparison of spinal and general anesthesia. J Korean Med Sci. 2003;18:691-5. PubMed DOI: 10.3346/jkms.2003.18.5.691
- Lamiri R, Chebab F, Kechiche N, Mani S, Hmida HB, El Ouaer M, et al. Inguinal hernia repair in newborns: A systematic literature review. J Neonatal Surg. 2024;13:23-. DOI: 10.52783/jns.v13.1253
- Cho YJ, Kwon H, Ha S, Kim SC, Kim DY, Namgoong JM, et al. Optimal timing for inguinal hernia repair in premature infants: surgical issues for inguinal hernia in premature infants. Ann Surg Treat Res. 2023;104:296-301. PubMed DOI: 10.4174/astr.2023.104.5.296
- Ceccanti S, Cervellone A, Pesce MV, Cozzi DA. Feasibility, safety and outcome of inguinal hernia repair under spinal versus general anesthesia in preterm and term infants. J Pediatr Surg. 2021;56:1057-61. PubMed DOI: 10.1016/j.jpedsurg.2020.09.064
- Davidson AJ, Morton NS, Arnup SJ, De Graaff JC, Disma N, Withington DE, et al. Apnea after awake regional and general anesthesia in infants: the general anesthesia compared to spinal anesthesia study—comparing apnea and neurodevelopmental outcomes, a randomized controlled trial. Anesthesiology. 2015;123:38-54. PubMed DOI: 10.1097/ALN.0000000000000709
- Jones LJ, Craven PD, Lakkundi A, Foster JP, Badawi N. Regional (spinal, epidural, caudal) versus general anaesthesia in preterm infants undergoing inguinal herniorrhaphy in early infancy. Cochrane Database Syst Rev. 2015;2015:36-69. PubMed DOI: 10.1002/14651858.CD003669.pub2
- Lambertz A, Schälte G, Winter J, Röth A, Busch D, Ulmer T, et al. Spinal anesthesia for inguinal hernia repair in infants: a feasible and safe method even in emergency cases. Pediatr Surg Int. 2014;30:1069-73. PubMed DOI: 10.1007/s00383-014-3590-y
- Somri M, Gaitini L, Vaida S, Collins G, Sabo E, Mogilner G. Postoperative outcome in high-risk infants undergoing herniorrhaphy: comparison between spinal and general anaesthesia. Anaesthesia. 1998;53:762-6. PubMed DOI: 10.1046/j.1365-2044.1998.00431.x
- Frumiento C, Abajian JC, Vane DW. Spinal anesthesia for preterm infants undergoing inguinal hernia repair. Arch Surg. 2000;135:445-51. PubMed DOI: 10.1001/archsurg.135.4.445