Israa Tarek Mohamed Abd Elaziz 1, Nermin Sadek Nasr 2, Hanan Mahmoud Farag 3, Marwa Ahmed Khairy Elbeialy 4, Lamyaa Elsayed Salem 5, Marwa Salem 6
Authors affiliations:
Background & objective: Thermal injury is a serious medical condition that could be associated with toxic myocarditis (TM). Cardiac troponins are the biomarkers of choice for the detection of cardiac injury; however, a negative troponin does not exclude the diagnosis of myocarditis. Brain natriuretic peptide (BNP) could be valuable in such cases and its role in burn injuries remains unexplored. We conducted this study to evaluate the use of the plasma concentration of BNP as an early predictor of postburn TM.
Methodology: Forty adult patients were included in this prospective observational study with total burn surface area (TSBA) ≥ 20% up to ≤ 50% with 2nd degree burn or more. The primary outcome was the sensitivity and specificity of the BNP plasma level as an early predictor of the incidence of TM while the secondary outcomes were ICU stay, and total hospital stay.
Results: 16 out of 40 patients developed TM by symptoms and echo findings (Group TM) and 24 patients didn’t (Group NTM). BNP was significantly elevated in Group TM than Group NTM on day 1, 4 and 7 (P< 0.0001) and it was significantly higher than troponin I on day 1 (95%CI = 0.888 to 1.000 for BNP vs 0.538 to 0. 837 for troponin). Sepsis, ICU and hospital stay were not significantly different between both groups (P = 0.134, 0.1136, 0. 1176 for each respectively).
Conclusion: BNP can serve as an early predictor of toxic myocarditis in patients with severe burn injuries.
Abbreviations: ANP: Atrial natriuretic peptide, BNP: Brain natriuretic peptide, cTnI: cardiac Troponin I, EMB: endomyocardial biopsy, TM: toxic myocarditis, TSBA: total burn surface area,
Keywords: Brain Natriuretic Peptide; Burn; Toxic Myocarditis; Troponin
Citation: Abd Elaziz ITM, Nasr NS, Farag HM, Elbeialy MAK, Salem LE, Salem M. Brain natriuretic peptide as an early predictor of toxic myocarditis after burn injury: A prospective observational study. Anaesth. pain intensive care 2025;29(3):458-466. DOI: 10.35975/apic.v29i3.2757
Received: May 09, 2024; Revised: October 26, 2024; Accepted: January 01, 2025
Burn injuries, particularly severe cases, are a serious medical, social, and economic problem. They are accompanied by several disorders all over the body as well as cardiac disorders. Burn toxins could initiate a systemic inflammatory cascade that causes inflammation in cardiac myocytes, which in turn may cause transient cardiac contractile dysfunction or toxic myocarditis (TM).1
TM is defined as myocardium inflammation with cardiac myocytes' necrosis. In general, cardiac troponins are the biomarker of choice for detecting cardiac injury.2 They are well-established as sensitive and specific markers of myocardial injury. Increased cardiac Troponin I (cTnI) levels can be detected in just over one-third of patients with myocarditis. However, a normal electrocardiogram (ECG), a negative troponin and/or creatine kinase does not exclude the diagnosis of myocarditis.3
The gold standard for a definite diagnosis and therapy of myocarditis is an endomyocardial biopsy (EMB). However, it is an invasive test with risk of complications and carries the limitation of sampling error since myocarditis is often a patchy process. EMB should be reserved for patients presenting with major clinical syndromes (such as severe heart failure and/or life-threatening arrhythmias).3
The discovery of the natriuretic peptide system in the form of atrial and brain natriuretic peptide (ANP and BNP) gave a rational explanation for how the central circulation can mediate control of water homeostasis. BNP is primarily synthesized under the influence of ventricular wall stretch as a prohormone which is cleaved into active BNP and inactive amino terminal fragment. The plasma concentration of BNP is elevated in disorders with salt and fluid overload and with increased atrial or ventricular wall tension, such as heart failure, but not specific for it. Elevated concentrations are also of prognostic value for identifying the risk of cardiovascular events and mortality in asymptomatic individuals.4 The role of BNP in identifying TM, particularly following burn injuries, remains underexplored.
This study aimed to assess whether plasma concentrations of BNP may serve as an early predictor of TM after burns, prior to the emergence of clinical symptoms or an increase in cardiac troponin levels.
This prospective observational study was conducted on 40 adult patients, following approval from the research ethical committee of the Faculty of Medicine, Ain-Shams University, Research Number: (FMSU MD 206/2022), and the study was registered with number: (PACTR202308864962981). Patients enrolled in the study when they or their relatives provided their informed consent. Patients included were ranging in age from 21 to 60 of both sexes and total burn surface area (TSBA) ≥ 20% up to ≤ 50% with 2nd degree burn or more.
We excluded patients who refused, or their relatives refused to join in the study, or declined to give written informed consent. Patients with preexisting cardiac or pulmonary diseases e.g., hypertension, congenital heart disease, ischemic heart disease with impaired cardiac function, valvular heart disease, heart failure, infective endocarditis, patients with severe hepatic impairment (INR ≥ 1.5, Bilirubin ≥ 2, Albumin ≤ 2) and patients with renal dysfunction (GFR < 50 ml/min).
On admission to burn intensive care unit, patients received standard burn care as airway management to ensure intact breathing, identify the site, degree, and percentage of the burnt area, circulatory Support through precise fluid replacement with Ringer lactate according to Parkland formula (4 ml/kg per TBSA% burned), no colloids within the first 8 hours and the rest in the next 16 hours.5 Urine output target was 0.5 ml/kg body weight.
Mean arterial blood pressure (MAP), heart rate (HR), and oxygen saturation (SpO2) were recorded on admission and at fixed intervals according to ICU protocol.
Five milliliters of venous blood were collected from each participant and divided into k3 EDTA vacutainers for complete blood count (CBC) with differential (ADVIA, Siemens Healthineers, Germany) and sterile plain vacutainer tubes. The blood samples in the plain tubes were allowed to clot for 30 min and then centrifuged at 3000 rpm for 10 min, then sera were removed and stored at -20°C till time of analysis of serum creatinine, BUN, sodium, potassium, AST and ALT (COBAS C6000, Roche Diagnostic GmbH, Mannheim, Germany). A central line was inserted, baseline CVP readings were recorded, and optimum fluid management was monitored and followed up according to urine output and central venous pressure.
Five milliliters of blood were drawn from the central line from each participant and placed in k3 EDTA vacutainers. Patients who met the inclusion criteria had BNP levels assessed on the first, fourth, and seventh days after admission using commercially available ELISA kits supplied by Shanghai Korain Biotech Co. LTD catalog no. E1287Hu. (Shanghai, China) following the manufacturer’s instructions. Absorbance of each well was measured at 450 nm by using a microtiter plate ELISA reader (BioTek, Winooski, VT, USA). The kit detection range was 5-2000ng/L. Blood samples for troponin I (COBAS e411, Roche Diagnostic GmbH, Mannheim, Germany) were obtained simultaneously on the same days of BNP sampling to detect which test would rise first.
Daily follow-up of total leucocyte count (TLC) and body temperature to detect early signs of sepsis where the patient was considered feverish if the temperature was above 37.2oC, and cold foments were applied and paracetamol (10-15 mg/kg) was given to control fever. Blood, urine, sputum, and wound cultures were sampled before starting broad-spectrum antibiotics. Furthermore, daily measurements of vital signs (heart rate, systolic and diastolic blood pressure, SpO2, CVP) were recorded every hour. Also, chest examination for early signs of chest infection and cardiac examination for heart sounds and murmurs were performed daily.
The patient’s cardiac condition was checked daily for signs and symptoms of TM, which included unexplained tachycardia > 120 bpm that was not related to fever or infection, a drop in MAP > 20% of the baseline reading, a rise in CVP > 12 mmHg, and signs of pulmonary congestion, such as dyspnea or fine basal crepitation in the lower lobes of the lungs. Transthoracic echocardiography was performed simultaneously with BNP and Troponin I sampling on day 1, 4 and 7. Ejection fraction abnormalities were recorded. Patients were divided into two groups according to the development of TM. Group TM: patients who developed TM and Group NTM: patients who didn’t develop TM.
The primary outcome was the sensitivity and specificity of the BNP plasma level as an early predictor of the incidence of TM while the secondary outcomes were the incidence of other complications such as sepsis and other organ failure as well as ICU stay, and total hospital stay.
Sample size calculation
By using PASS 11 for sample size calculation, setting power at 99%, alpha error at 5% and after reviewing previous study results (Ashry et al., 2018) showed that the level of BNP was higher in patients with cardiac troponin positive than cardiac troponin negative patients (1801.54 ± 812.15 versus 770.94 ± 655.95 respectively); based on that, a sample size of at least 40 patients with burn were sufficient to achieve study objective.
Statistical analysis
Data were analysed using Statistical Package for Social Science (SPSS) version 27.0., Quantitative data were expressed as mean ± SD or Median (IQR) where indicated. Qualitative data were expressed as frequency and percentages. Independent-samples t-test of significance was used when comparing between two means. Chi-square (ꭓ2) test of significance was used in order to compare proportions between two qualitative parameters.
ROC analysis
An ROC curve is a plot of the true positive rate (sensitivity) in function of the false positive rate (100-specificity) for different cut-off points of a parameter. Each point on the ROC curve represents a sensitivity/specificity pair corresponding to a particular decision threshold. The Area Under the ROC curve (AUC) is a measure of how well a parameter can distinguish between two diagnostic groups (diseased/normal). The confidence interval was set to 95% and the margin of error accepted was set to 5%. So, P-value < 0.05 was considered significant.
Diagnostic characteristics were calculated as follows:
- Sensitivity = (True positive test / Total positive golden) x 100
- Specificity = (True negative test / Total negative golden) x 100
- Predictive positive value = (True positive test / Total positive test) x 100
- Predictive negative value = (True negative test / Total negative test) x 100
- Positive likelihood ratio LR+ = (sensitivity/ 1-specificity)
- Negative likelihood ratio LR- = (1-sensitivity / specificity)
Fifty-one patients were screened for eligibility; six individuals did not meet the criteria, while five individuals refused to participate in our study, leaving 40 patients to be enrolled in our trial, 16 patients developed TM.
3.1. Patients’ characteristics
The patient demographic data (age, sex), associated comorbidities and the mode of burn showed no statistically significant differences between the groups (P > 0.05) (Table 1, 2). There was a statistically significant difference between groups in terms of TSBA where it was higher in Group TM than Group NTM (P < 0.0001) (Table 2).
3.2. The cardiac biomarkers
Groups were compared regarding the BNP and troponin (Trop) levels at days 1, 4, and 7. Patients among the Group TM showed higher BNP levels on days 1, 4, and 7 than patients among the Group NTM, which was statistically significant (P > 0.001). Group TM showed similar results for Trop level (P > 0.001) (Table 3).
3.3. ROC analysis
Receiver operating characteristic (ROC) curve was used for detection of optimum cutoff value of diagnostic performance for BNP to predict TM. On the first day, BNP had an AUC of 0.987 (P < 0.001), with a cut-off value >87 pg/mL yielding 100% sensitivity and 91.7% specificity. BNP levels on the fourth and seventh day (BNP 4 and BNP 7) achieved an AUC of 1.000, indicating perfect diagnostic accuracy (P < 0.001). Troponin showed lower sensitivity and specificity across all time points (Figure 1, Table 4).
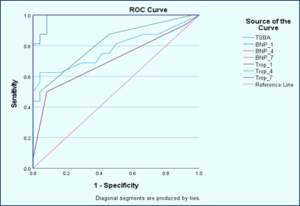
Figure 1: ROC curve for TBSA and cardiac markers to predict TM. TBSA= total burn surface area, BNP= brain natriuretic peptide, Trop= troponin
BNP and Trop showed significant pairwise comparison with AUC for BNP (0.987 and 95% CI = 0.888 to 1.0) higher than Trop (0.703 & 95% CI = 0.538 to 0.837) to predict TM at day 1. The difference between areas is 0.284 with P = 0.0004.
3.4. Hemodynamic data and temperature
Regarding the hemodynamic data (in terms of systolic blood pressure (SBP), diastolic blood pressure (DBP), mean blood pressure (MAP), heart rate (HR), oxygen saturation (SO2), and CVP) and temperature (temp), there were no statistically significant differences between groups at day 1. However, SBP, DBP, MBP, HR, and CVP were significantly lower among patients with TM at days 4 and 7
and SO2 at day 7. While no significant differences were detected between groups regarding the temp at day 4 or 7 (Figures 2, 3, 4, 5 and 6).
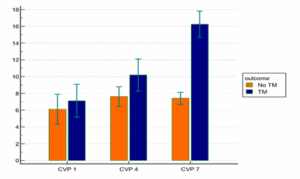
Figure 2: Comparative NIBP measurements in groups at different
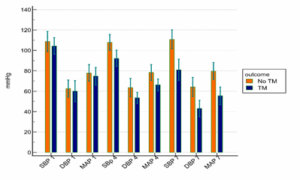
Figure 3: Comparative CVP measurements between groups at different times
4. DISCUSSION
BNP was found to be elevated earlier than the elevation in cardiac troponin in those patients and even before the occurrence of clinical signs and sympt oms. These results were found to be 100% sensitive and 91% specific for most of the patients, which makes the BNP a valuable predictor of TM post-burn.
Severe burn injury results in an aggravated traumatic response with general and specific features. A significant specific feature is a profound change in microvascular characteristics with increased leakage of plasma to the interstitium, ‘‘burn edema,’’ as well as subsequently hemoconcentration and hypovolemic shock.6 Days later, excess water enters the circulation during edema mobilization. Both of these phases are under the influence of control systems regulating myocardial function, vascular tone, as well as intracellular and extracellular volume.7
TM is defined as inflammation of the myocardium with necrosis of cardiac cells. It may be due to infections, toxins, drugs, or systemic disorders, but it’s often idiopathic. The pathophysiology of TM remains under research, but it’s mostly due to excessive plasma losses that occur immediately after the burn injury. It can be clinically diagnosed with chest pain, fever, unexplained tachycardia, or signs of low cardiac output. It can be diagnosed by lab investigations e.g., CBC, cardiac
troponin, ECG, transthoracic echo, endomyocardial biopsy and cardiac MRI.8
The discovery of the natriuretic peptide system in the form of atrial natriuretic peptide (natriuretic peptide A) and brain natriuretic peptide (natriuretic peptide B (BNP) gave a rational explanation for how the central circulation can mediate control of water homeostasis. These discoveries described the heart as an endocrine organ that releases natriuretic peptides from the myocardium based on atrial and ventricular distension.9 The prohormone is enzymatically cleaved to form active BNP and an inactive amino terminal fragment, NT-proBNP, both of which are elevated in disorders with salt and fluid overload and with increased atrial or ventricular wall tension, such as heart failure, but such elevations are not specific for heart failure.10
In this study we aimed to evaluate whether the use of the plasma concentration of Brain natriuretic peptide can be an early predictor of the occurrence of TM post burn before appearance of the clinical signs or rise in cardiac troponin.
Regarding burn data, there was a statistically significant difference between groups in terms of TBSA where it was higher in Group TM than Group NTM (35.13 ± 11.6
% in Group TM vs 23.83 ± 3.6 % in group non-TM) (P < 0.0001). This finding may indicate the correlation of large TBSA with increase possibility of TM. Lindahl et al. who studied fifty patients with 10% or more of burn surface area for the first 2 weeks after burn injury and correlated their data to P- NT- proBNP serum levels concluded that the median burn size of the 50 studied patients was 30.5% TBS (range, 10-95.5%) which were similar to the results of the present study. And he stated that P- NT- proBNP levels that were assessed on Days 3 to 8 were an independent predictor for every subsequent SOFA score measured one or more days later up to Day 14.11
Regarding cardiac markers at days 1, 4 and 7, Patients with TM had significantly higher BNP levels on day one post-burn. BNP levels increased further on the fourth day and the seventh. Troponin levels showed a delayed and less consistent pattern, with significant differences observed only on seventh day.
In a study by Ashry et al., 2018 that studied 42 patients with acute CO poisoning were admitted to the Poison Control Centre of Ain Shams University hospitals (PCC-ASUH) and compared to 50 cases in the control group to investigate the value of BNP measurement in the detection of cardiac toxicity in acute carbon monoxide poisoning, the level of both cardiac biomarkers namely troponin I and BNP were measured. The level of BNP was found to be significantly higher in cases who developed TM than in cases with positive troponin and developed TM after CO exposure.12
Other studies that could add value to BNP determination is the one performed by Wang et al. which suggested that a single determination of increase in the level of BNP in cases with cardiac affection is suggestive of future development of cardiovascular outcomes.13 Reyding et al., Bethell et al., and Güneş et al. concluded that the increase in serum level of BNP is related to the severity and prognosis of patients with cardiac affection.14,4,15
In their study, Ukena et al. demonstrated that NT-proBNP concentration above the 4th quartile was the only biomarker to be associated with cardiac death or the need for heart transplantation following acute viral myocarditis.16
In the current study, On the first day, BNP had an AUC of 0.987 (P < 0.001), with a cut-off value >87 pg/mL yielding 100% sensitivity and 91.7% specificity. BNP levels on the fourth and seventh day (BNP 4 and BNP 7) achieved an AUC of 1.000, indicating perfect diagnostic accuracy (P < 0.001). Troponin showed lower sensitivity and specificity across all time points.
The threshold value of the increased serum level of BNP for cardiac affection is postulated to be 100 pg/mL in the literature.17 Ashry et al. revealed that all patients had measured serum levels of BNP greater than 100 pg/mL whereas 38% of them had normal troponin I values. This highlights the value of BNP level determination over troponin I determination in diagnosing TM. They found a significant increase in BNP levels in cases with ischemic ECG changes and sinus tachycardia, which were signs of TM.12
Previous data regarding the lack of correlation between high troponin and poor prognosis in myocarditis.18,19 In one study involving pediatric patients with myocarditis, highly elevated troponin was not associated with ventricular dysfunction or poor outcomes.20 This is consistent with the finding of relatively low NT-proBNP levels in some patients with end-stage heart failure, particularly those with higher body mass index.21
Regarding hemodynamic data, SBP, DBP, MBP were significantly lower among patients with TM at days 4 and 7 and SO2 at day 7. HR and CVP were considerably higher in patients with TM at days 4 and 7.
Ashry et al. revealed that the mean differences in systolic and diastolic blood pressure, and pulse were statistically significant between groups (P < 0.01), where systolic and diastolic blood pressure were lower among acute CO poisoning cases who developed TM and pulse was higher among them.12
The two groups were compared regarding ICU and hospital stay. There was no statistically significant difference between both groups (P = 0.1136 in ICU stay and P = 0.1176 in hospital stay)
Zhao et al. who investigated the role of N-terminal pro-B-type natriuretic peptide (NT-proBNP) in assessing the poor outcomes of adult patients with acute myocarditis revealed that in predicting 30-day death or heart transplantation, the sensitivity and specificity of NT-proBNP were 86.67 and 89.68%, respectively (AUC = 0.924, optimal cut-off value: 7,204 pg/mL), while the sensitivity and specificity of LVEF were 86.67 and 75.48%, respectively (AUC = 0.843, optimal cut-off value: 45%). In predicting long-term major adverse cardiac events (MACE), the sensitivity and specificity of NT-proBNP were 62.07 and 80.14%, respectively (AUC = 0.733, optimal cut-off value: 3,549 pg/mL). The sensitivity and specificity of LVEF to predict long-term MACE was 62.07 and 87.23%, respectively (AUC = 0.737, optimal cut-off value: 38%). ROC curves showed that the predictive values of troponin I were lower than NT-proBNP for both 30-day (AUC = 0.616, optimal cut-off value: 15.846 ng/mL) and long-term MACE (AUC = 0.509, optimal cut-off value: 0.315 ng/mL).22
Rodriguez-Gonzalez et al. reported that baseline NT-proBNP > 5,000 pg/mL could help to identify high-risk pediatric myocarditis patients with poor outcomes.23 Patients with high NT-proBNP levels are more likely to suffer from prolonged symptoms and need special attention for potential complications. This biomarker should be considered for risk assessment along with echocardiography and clinical judgment.18
This study was carried out in a single center with a relatively small sample size. Furthermore, although BNP is a sensitive diagnostic, it is not limited to myocarditis and may be raised in other burn-related diseases such fluid overload. Further multicenter studies with larger cohorts are required to verify these results.
BNP is a valuable early predictor of toxic myocarditis in patients with severe burn injuries. Its non-invasive nature and high diagnostic accuracy make it an accessible tool for early detection and management. Integrating BNP monitoring into burn care protocols could improve outcomes by facilitating timely interventions for cardiac complications.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
IT: Conduction of the study work.
NS, HF: Manuscript editing.
MK, MS: Literature search.
LS: Statistical analysis and review.
Authors affiliations:
- Israa Tarek Mohamed Abd Elaziz, M.Sc., Assistant Lecturer of Anesthesiology, Intensive Care & Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: israa,tarek@med.asu.edu.eg
- Nermin Sadek Nasr, MD, Professor of Anesthesiology, Intensive Care & Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: nerminnasr@yahoo.com
- Hanan Mahmoud Farag, MD, Assistant Professor of Anesthesiology, Intensive Care & Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: faraghanan@yahoo.com
- Marwa Ahmed Khairy Elbeialy, MD, Assistant Professor of Anesthesiology, Intensive Care & Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: mar_khairy@yahoo.com; ORCiD: 0000-0002-1223-5670
- Lamyaa Elsayed Salem, MD, Assistant Professor of Clinical Pathology, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: drlamyamehrez@gmail.com
- Marwa Salem, MD, Lecturer of Anesthesiology, Intensive Care & Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: Marwamostafa@med.asu.edu.eg
ABSTRACT
Background & objective: Thermal injury is a serious medical condition that could be associated with toxic myocarditis (TM). Cardiac troponins are the biomarkers of choice for the detection of cardiac injury; however, a negative troponin does not exclude the diagnosis of myocarditis. Brain natriuretic peptide (BNP) could be valuable in such cases and its role in burn injuries remains unexplored. We conducted this study to evaluate the use of the plasma concentration of BNP as an early predictor of postburn TM.
Methodology: Forty adult patients were included in this prospective observational study with total burn surface area (TSBA) ≥ 20% up to ≤ 50% with 2nd degree burn or more. The primary outcome was the sensitivity and specificity of the BNP plasma level as an early predictor of the incidence of TM while the secondary outcomes were ICU stay, and total hospital stay.
Results: 16 out of 40 patients developed TM by symptoms and echo findings (Group TM) and 24 patients didn’t (Group NTM). BNP was significantly elevated in Group TM than Group NTM on day 1, 4 and 7 (P< 0.0001) and it was significantly higher than troponin I on day 1 (95%CI = 0.888 to 1.000 for BNP vs 0.538 to 0. 837 for troponin). Sepsis, ICU and hospital stay were not significantly different between both groups (P = 0.134, 0.1136, 0. 1176 for each respectively).
Conclusion: BNP can serve as an early predictor of toxic myocarditis in patients with severe burn injuries.
Abbreviations: ANP: Atrial natriuretic peptide, BNP: Brain natriuretic peptide, cTnI: cardiac Troponin I, EMB: endomyocardial biopsy, TM: toxic myocarditis, TSBA: total burn surface area,
Keywords: Brain Natriuretic Peptide; Burn; Toxic Myocarditis; Troponin
Citation: Abd Elaziz ITM, Nasr NS, Farag HM, Elbeialy MAK, Salem LE, Salem M. Brain natriuretic peptide as an early predictor of toxic myocarditis after burn injury: A prospective observational study. Anaesth. pain intensive care 2025;29(3):458-466. DOI: 10.35975/apic.v29i3.2757
Received: May 09, 2024; Revised: October 26, 2024; Accepted: January 01, 2025
1. INTRODUCTION
Burn injuries, particularly severe cases, are a serious medical, social, and economic problem. They are accompanied by several disorders all over the body as well as cardiac disorders. Burn toxins could initiate a systemic inflammatory cascade that causes inflammation in cardiac myocytes, which in turn may cause transient cardiac contractile dysfunction or toxic myocarditis (TM).1
TM is defined as myocardium inflammation with cardiac myocytes' necrosis. In general, cardiac troponins are the biomarker of choice for detecting cardiac injury.2 They are well-established as sensitive and specific markers of myocardial injury. Increased cardiac Troponin I (cTnI) levels can be detected in just over one-third of patients with myocarditis. However, a normal electrocardiogram (ECG), a negative troponin and/or creatine kinase does not exclude the diagnosis of myocarditis.3
The gold standard for a definite diagnosis and therapy of myocarditis is an endomyocardial biopsy (EMB). However, it is an invasive test with risk of complications and carries the limitation of sampling error since myocarditis is often a patchy process. EMB should be reserved for patients presenting with major clinical syndromes (such as severe heart failure and/or life-threatening arrhythmias).3
The discovery of the natriuretic peptide system in the form of atrial and brain natriuretic peptide (ANP and BNP) gave a rational explanation for how the central circulation can mediate control of water homeostasis. BNP is primarily synthesized under the influence of ventricular wall stretch as a prohormone which is cleaved into active BNP and inactive amino terminal fragment. The plasma concentration of BNP is elevated in disorders with salt and fluid overload and with increased atrial or ventricular wall tension, such as heart failure, but not specific for it. Elevated concentrations are also of prognostic value for identifying the risk of cardiovascular events and mortality in asymptomatic individuals.4 The role of BNP in identifying TM, particularly following burn injuries, remains underexplored.
This study aimed to assess whether plasma concentrations of BNP may serve as an early predictor of TM after burns, prior to the emergence of clinical symptoms or an increase in cardiac troponin levels.
2. METHODOLOGY
This prospective observational study was conducted on 40 adult patients, following approval from the research ethical committee of the Faculty of Medicine, Ain-Shams University, Research Number: (FMSU MD 206/2022), and the study was registered with number: (PACTR202308864962981). Patients enrolled in the study when they or their relatives provided their informed consent. Patients included were ranging in age from 21 to 60 of both sexes and total burn surface area (TSBA) ≥ 20% up to ≤ 50% with 2nd degree burn or more.
We excluded patients who refused, or their relatives refused to join in the study, or declined to give written informed consent. Patients with preexisting cardiac or pulmonary diseases e.g., hypertension, congenital heart disease, ischemic heart disease with impaired cardiac function, valvular heart disease, heart failure, infective endocarditis, patients with severe hepatic impairment (INR ≥ 1.5, Bilirubin ≥ 2, Albumin ≤ 2) and patients with renal dysfunction (GFR < 50 ml/min).
On admission to burn intensive care unit, patients received standard burn care as airway management to ensure intact breathing, identify the site, degree, and percentage of the burnt area, circulatory Support through precise fluid replacement with Ringer lactate according to Parkland formula (4 ml/kg per TBSA% burned), no colloids within the first 8 hours and the rest in the next 16 hours.5 Urine output target was 0.5 ml/kg body weight.
Mean arterial blood pressure (MAP), heart rate (HR), and oxygen saturation (SpO2) were recorded on admission and at fixed intervals according to ICU protocol.
Five milliliters of venous blood were collected from each participant and divided into k3 EDTA vacutainers for complete blood count (CBC) with differential (ADVIA, Siemens Healthineers, Germany) and sterile plain vacutainer tubes. The blood samples in the plain tubes were allowed to clot for 30 min and then centrifuged at 3000 rpm for 10 min, then sera were removed and stored at -20°C till time of analysis of serum creatinine, BUN, sodium, potassium, AST and ALT (COBAS C6000, Roche Diagnostic GmbH, Mannheim, Germany). A central line was inserted, baseline CVP readings were recorded, and optimum fluid management was monitored and followed up according to urine output and central venous pressure.
Five milliliters of blood were drawn from the central line from each participant and placed in k3 EDTA vacutainers. Patients who met the inclusion criteria had BNP levels assessed on the first, fourth, and seventh days after admission using commercially available ELISA kits supplied by Shanghai Korain Biotech Co. LTD catalog no. E1287Hu. (Shanghai, China) following the manufacturer’s instructions. Absorbance of each well was measured at 450 nm by using a microtiter plate ELISA reader (BioTek, Winooski, VT, USA). The kit detection range was 5-2000ng/L. Blood samples for troponin I (COBAS e411, Roche Diagnostic GmbH, Mannheim, Germany) were obtained simultaneously on the same days of BNP sampling to detect which test would rise first.
Daily follow-up of total leucocyte count (TLC) and body temperature to detect early signs of sepsis where the patient was considered feverish if the temperature was above 37.2oC, and cold foments were applied and paracetamol (10-15 mg/kg) was given to control fever. Blood, urine, sputum, and wound cultures were sampled before starting broad-spectrum antibiotics. Furthermore, daily measurements of vital signs (heart rate, systolic and diastolic blood pressure, SpO2, CVP) were recorded every hour. Also, chest examination for early signs of chest infection and cardiac examination for heart sounds and murmurs were performed daily.
The patient’s cardiac condition was checked daily for signs and symptoms of TM, which included unexplained tachycardia > 120 bpm that was not related to fever or infection, a drop in MAP > 20% of the baseline reading, a rise in CVP > 12 mmHg, and signs of pulmonary congestion, such as dyspnea or fine basal crepitation in the lower lobes of the lungs. Transthoracic echocardiography was performed simultaneously with BNP and Troponin I sampling on day 1, 4 and 7. Ejection fraction abnormalities were recorded. Patients were divided into two groups according to the development of TM. Group TM: patients who developed TM and Group NTM: patients who didn’t develop TM.
The primary outcome was the sensitivity and specificity of the BNP plasma level as an early predictor of the incidence of TM while the secondary outcomes were the incidence of other complications such as sepsis and other organ failure as well as ICU stay, and total hospital stay.
Sample size calculation
By using PASS 11 for sample size calculation, setting power at 99%, alpha error at 5% and after reviewing previous study results (Ashry et al., 2018) showed that the level of BNP was higher in patients with cardiac troponin positive than cardiac troponin negative patients (1801.54 ± 812.15 versus 770.94 ± 655.95 respectively); based on that, a sample size of at least 40 patients with burn were sufficient to achieve study objective.
Statistical analysis
Data were analysed using Statistical Package for Social Science (SPSS) version 27.0., Quantitative data were expressed as mean ± SD or Median (IQR) where indicated. Qualitative data were expressed as frequency and percentages. Independent-samples t-test of significance was used when comparing between two means. Chi-square (ꭓ2) test of significance was used in order to compare proportions between two qualitative parameters.
ROC analysis
An ROC curve is a plot of the true positive rate (sensitivity) in function of the false positive rate (100-specificity) for different cut-off points of a parameter. Each point on the ROC curve represents a sensitivity/specificity pair corresponding to a particular decision threshold. The Area Under the ROC curve (AUC) is a measure of how well a parameter can distinguish between two diagnostic groups (diseased/normal). The confidence interval was set to 95% and the margin of error accepted was set to 5%. So, P-value < 0.05 was considered significant.
Diagnostic characteristics were calculated as follows:
- Sensitivity = (True positive test / Total positive golden) x 100
- Specificity = (True negative test / Total negative golden) x 100
- Predictive positive value = (True positive test / Total positive test) x 100
- Predictive negative value = (True negative test / Total negative test) x 100
- Positive likelihood ratio LR+ = (sensitivity/ 1-specificity)
- Negative likelihood ratio LR- = (1-sensitivity / specificity)
3. RESULTS
Fifty-one patients were screened for eligibility; six individuals did not meet the criteria, while five individuals refused to participate in our study, leaving 40 patients to be enrolled in our trial, 16 patients developed TM.
3.1. Patients’ characteristics
The patient demographic data (age, sex), associated comorbidities and the mode of burn showed no statistically significant differences between the groups (P > 0.05) (Table 1, 2). There was a statistically significant difference between groups in terms of TSBA where it was higher in Group TM than Group NTM (P < 0.0001) (Table 2).
| Table 1: Comparison between groups as regards demographic data. | ||||
| Variables | Group TM
(n = 16) |
Group NTM
(n = 24) |
P-value | |
| Age (years) | 29.19 ± 10.5 | 31.88 ± 14.6 | 0.531 t | |
| Gender | Female | 5 (31.2%) | 10 (41.7%) | 0.51 ꭓ2 |
| Male | 11(68.7%) | 14 (58.3%) | ||
| Comorbidities | DM | 0 (0%) | 2 (8.3%)100.0% RT 8.3% CT 5.0% GT |
0.397 ꭓ2 |
| Epileptic | 0 (0%) | 1 (4.2%)100.0% RT 4.2% CT 2.5% GT |
||
| Facial nerve palsy | 0 (0%) | 1 (4.2%)100.0% RT 4.2% CT 2.5% GT |
||
| Data expressed as mean ± SD or n (%); t = student t test, ꭓ2 = chi square. TM = toxic myocarditis, DM = Diabetes millets. | ||||
| Table 2: Comparison between groups as regard burn data. | ||||
| Variable | Group TM
(n = 16) |
Group NTM
(n = 24) |
P-value | |
| TSBA | 35.13 ± 11.6 | 23.83 ± 3.6 | 0.0001t | |
| Mode of burn | Electric burn | 2 (12.5%) 66.7% RT 12.5% CT 5.0% GT |
1 (4.2%) 33.3% RT 4.2% CT 2.5% GT |
0.555x2 |
| Flame | 10 (65.5%) 35.7% RT 62.5% CT 25.0% GT |
18 (75%) 64.3% RT 75.0% CT 45.0% GT |
||
| Scald | 4 (25%) 44.4% RT 25.0% CT 10.0% GT |
5 (20.8%) 55.6% RT 20.8% CT 12.5% GT |
||
| Data expressed as mean ± SD, n (%), t = student t test, X2 = chi square. TM= toxic myocarditis, DM = Diabetes mellites; TBSA= total burn surface area. | ||||
3.2. The cardiac biomarkers
Groups were compared regarding the BNP and troponin (Trop) levels at days 1, 4, and 7. Patients among the Group TM showed higher BNP levels on days 1, 4, and 7 than patients among the Group NTM, which was statistically significant (P > 0.001). Group TM showed similar results for Trop level (P > 0.001) (Table 3).
| Table 3: Comparison between groups as regard cardiac markers data. | |||||||
| Day | Group TM (n = 16) | Group NTM (n = 24) | p-value z | ||||
| Range | Median | IQR | Range | Median | IQR | ||
| BNP - day1 | 87.5-560 | 180.05 | 135-245.8 | 30-100 | 59.65 | 47.5−77.5 | < 0.0001* |
| BNP− day 4 | 110−827.1 | 450.9 | 328.95−555.7 | 45−100 | 69.5 | 52.5−88 | < 0.0001* |
| BNP – day 7 | 341.9−1700 | 885.3 | 624.9−1066.3 | 40−95 | 70 | 55.5−85 | < 0.0001* |
| Trop – day 1 | 0−1 | 0.015 | 0.01−0.02 | 0−0.02 | 0.01 | 0.01−0.01 | 0.0130* |
| Trop – day 4 | 0.01−2.1 | 0.025 | 0.02−0.2 | 0.01−0.04 | 0.01 | 0.01−0.02 | 0.0006* |
| Trop − day 7 | 0.1−2.6 | 0.3 | 0.2−0.4 | 0.01−0.06 | 0.02 | 0.02−0.02 | < 0.0001* |
| Data expressed as mean ± SD, t = student t test, *= significant. TM= toxic myocarditis, BNP= Brain natriuretic peptide, Trop= troponin I, IQR= interquartile range | |||||||
3.3. ROC analysis
Receiver operating characteristic (ROC) curve was used for detection of optimum cutoff value of diagnostic performance for BNP to predict TM. On the first day, BNP had an AUC of 0.987 (P < 0.001), with a cut-off value >87 pg/mL yielding 100% sensitivity and 91.7% specificity. BNP levels on the fourth and seventh day (BNP 4 and BNP 7) achieved an AUC of 1.000, indicating perfect diagnostic accuracy (P < 0.001). Troponin showed lower sensitivity and specificity across all time points (Figure 1, Table 4).

Figure 1: ROC curve for TBSA and cardiac markers to predict TM. TBSA= total burn surface area, BNP= brain natriuretic peptide, Trop= troponin
| Table 4: ROC Analysis | |||||||||
| AUC | Cut off | Sensitivity % | Specificity % | +LR | -LR | +PV | -PV | P-value | |
| TBSA (%) | 0.781 | > 28 | 62.5 | 95.8 | 15.00 | 0.39 | 90.9 | 79.3 | 0.0009* |
| BNP_1 | 0.987 | > 87 | 100 | 91.7 | 12.00 | 0.00 | 88.9 | 100.0 | < 0.001* |
| BNP_4 | 1.000 | > 100 | 100 | 100 | 0.00 | 100.0 | 100.0 | < 0.001* | |
| BNP_7 | 1.000 | > 95 | 100 | 100 | 0.00 | 100.0 | 100.0 | < 0.001* | |
| Trop_1 | 0.703 | > 0.01 | 50 | 91.7 | 6.00 | 0.55 | 80.0 | 73.3 | 0.01 |
| Trop_4 | 0.815 | > 0.02 | 50 | 95.8 | 12.00 | 0.52 | 88.9 | 74.2 | < 0.001* |
| Trop_7 | 1.000 | > 0.06 | 100 | 100 | 0.00 | 100.0 | 100.0 | < 0.001* | |
| TBSA= total burn surface area, BNP= brain natriuretic peptide, Trop= troponin, AUC= area under curve, +LR= Positive likelihood ratio, -LR= negative likelihood ratio, +PV=predictive positive value, -PV= predictive negative value, *=significant | |||||||||
BNP and Trop showed significant pairwise comparison with AUC for BNP (0.987 and 95% CI = 0.888 to 1.0) higher than Trop (0.703 & 95% CI = 0.538 to 0.837) to predict TM at day 1. The difference between areas is 0.284 with P = 0.0004.
3.4. Hemodynamic data and temperature
Regarding the hemodynamic data (in terms of systolic blood pressure (SBP), diastolic blood pressure (DBP), mean blood pressure (MAP), heart rate (HR), oxygen saturation (SO2), and CVP) and temperature (temp), there were no statistically significant differences between groups at day 1. However, SBP, DBP, MBP, HR, and CVP were significantly lower among patients with TM at days 4 and 7
and SO2 at day 7. While no significant differences were detected between groups regarding the temp at day 4 or 7 (Figures 2, 3, 4, 5 and 6).

Figure 2: Comparative NIBP measurements in groups at different

Figure 3: Comparative CVP measurements between groups at different times
- Groups were compared regarding cardiac function (Ejection Fraction%) done by Echocardiography and it showed significant difference between both groups (Group TM EF%= 37.5 ± 5.2 vs group NTM EF%= 58.13±2.5) (P <0.001)
- ICU course: Groups were compared regarding ICU course (in terms of sepsis and organ failure) with showed no significant difference between both groups. (P = 0.134 and 0.243 respectively). Both groups were also compared regarding ICU and hospital stay, with no statistically significant difference between them (P = 0.1136 and 0.1176 respectively).
- Groups were compared regarding ICU fate and it showed significant difference between both groups. 11 out of 16 patients that had TM died and only 5 patients were discharged (P = 0.001). On the other hand, 10 out of 24 patients, who didn’t develop TM, died and 14 were discharged (P = 0.001).
4. DISCUSSION
BNP was found to be elevated earlier than the elevation in cardiac troponin in those patients and even before the occurrence of clinical signs and sympt oms. These results were found to be 100% sensitive and 91% specific for most of the patients, which makes the BNP a valuable predictor of TM post-burn.
Severe burn injury results in an aggravated traumatic response with general and specific features. A significant specific feature is a profound change in microvascular characteristics with increased leakage of plasma to the interstitium, ‘‘burn edema,’’ as well as subsequently hemoconcentration and hypovolemic shock.6 Days later, excess water enters the circulation during edema mobilization. Both of these phases are under the influence of control systems regulating myocardial function, vascular tone, as well as intracellular and extracellular volume.7
TM is defined as inflammation of the myocardium with necrosis of cardiac cells. It may be due to infections, toxins, drugs, or systemic disorders, but it’s often idiopathic. The pathophysiology of TM remains under research, but it’s mostly due to excessive plasma losses that occur immediately after the burn injury. It can be clinically diagnosed with chest pain, fever, unexplained tachycardia, or signs of low cardiac output. It can be diagnosed by lab investigations e.g., CBC, cardiac
troponin, ECG, transthoracic echo, endomyocardial biopsy and cardiac MRI.8
The discovery of the natriuretic peptide system in the form of atrial natriuretic peptide (natriuretic peptide A) and brain natriuretic peptide (natriuretic peptide B (BNP) gave a rational explanation for how the central circulation can mediate control of water homeostasis. These discoveries described the heart as an endocrine organ that releases natriuretic peptides from the myocardium based on atrial and ventricular distension.9 The prohormone is enzymatically cleaved to form active BNP and an inactive amino terminal fragment, NT-proBNP, both of which are elevated in disorders with salt and fluid overload and with increased atrial or ventricular wall tension, such as heart failure, but such elevations are not specific for heart failure.10
In this study we aimed to evaluate whether the use of the plasma concentration of Brain natriuretic peptide can be an early predictor of the occurrence of TM post burn before appearance of the clinical signs or rise in cardiac troponin.
Regarding burn data, there was a statistically significant difference between groups in terms of TBSA where it was higher in Group TM than Group NTM (35.13 ± 11.6
% in Group TM vs 23.83 ± 3.6 % in group non-TM) (P < 0.0001). This finding may indicate the correlation of large TBSA with increase possibility of TM. Lindahl et al. who studied fifty patients with 10% or more of burn surface area for the first 2 weeks after burn injury and correlated their data to P- NT- proBNP serum levels concluded that the median burn size of the 50 studied patients was 30.5% TBS (range, 10-95.5%) which were similar to the results of the present study. And he stated that P- NT- proBNP levels that were assessed on Days 3 to 8 were an independent predictor for every subsequent SOFA score measured one or more days later up to Day 14.11
Regarding cardiac markers at days 1, 4 and 7, Patients with TM had significantly higher BNP levels on day one post-burn. BNP levels increased further on the fourth day and the seventh. Troponin levels showed a delayed and less consistent pattern, with significant differences observed only on seventh day.
In a study by Ashry et al., 2018 that studied 42 patients with acute CO poisoning were admitted to the Poison Control Centre of Ain Shams University hospitals (PCC-ASUH) and compared to 50 cases in the control group to investigate the value of BNP measurement in the detection of cardiac toxicity in acute carbon monoxide poisoning, the level of both cardiac biomarkers namely troponin I and BNP were measured. The level of BNP was found to be significantly higher in cases who developed TM than in cases with positive troponin and developed TM after CO exposure.12
Other studies that could add value to BNP determination is the one performed by Wang et al. which suggested that a single determination of increase in the level of BNP in cases with cardiac affection is suggestive of future development of cardiovascular outcomes.13 Reyding et al., Bethell et al., and Güneş et al. concluded that the increase in serum level of BNP is related to the severity and prognosis of patients with cardiac affection.14,4,15
In their study, Ukena et al. demonstrated that NT-proBNP concentration above the 4th quartile was the only biomarker to be associated with cardiac death or the need for heart transplantation following acute viral myocarditis.16
In the current study, On the first day, BNP had an AUC of 0.987 (P < 0.001), with a cut-off value >87 pg/mL yielding 100% sensitivity and 91.7% specificity. BNP levels on the fourth and seventh day (BNP 4 and BNP 7) achieved an AUC of 1.000, indicating perfect diagnostic accuracy (P < 0.001). Troponin showed lower sensitivity and specificity across all time points.
The threshold value of the increased serum level of BNP for cardiac affection is postulated to be 100 pg/mL in the literature.17 Ashry et al. revealed that all patients had measured serum levels of BNP greater than 100 pg/mL whereas 38% of them had normal troponin I values. This highlights the value of BNP level determination over troponin I determination in diagnosing TM. They found a significant increase in BNP levels in cases with ischemic ECG changes and sinus tachycardia, which were signs of TM.12
Previous data regarding the lack of correlation between high troponin and poor prognosis in myocarditis.18,19 In one study involving pediatric patients with myocarditis, highly elevated troponin was not associated with ventricular dysfunction or poor outcomes.20 This is consistent with the finding of relatively low NT-proBNP levels in some patients with end-stage heart failure, particularly those with higher body mass index.21
Regarding hemodynamic data, SBP, DBP, MBP were significantly lower among patients with TM at days 4 and 7 and SO2 at day 7. HR and CVP were considerably higher in patients with TM at days 4 and 7.
Ashry et al. revealed that the mean differences in systolic and diastolic blood pressure, and pulse were statistically significant between groups (P < 0.01), where systolic and diastolic blood pressure were lower among acute CO poisoning cases who developed TM and pulse was higher among them.12
The two groups were compared regarding ICU and hospital stay. There was no statistically significant difference between both groups (P = 0.1136 in ICU stay and P = 0.1176 in hospital stay)
Zhao et al. who investigated the role of N-terminal pro-B-type natriuretic peptide (NT-proBNP) in assessing the poor outcomes of adult patients with acute myocarditis revealed that in predicting 30-day death or heart transplantation, the sensitivity and specificity of NT-proBNP were 86.67 and 89.68%, respectively (AUC = 0.924, optimal cut-off value: 7,204 pg/mL), while the sensitivity and specificity of LVEF were 86.67 and 75.48%, respectively (AUC = 0.843, optimal cut-off value: 45%). In predicting long-term major adverse cardiac events (MACE), the sensitivity and specificity of NT-proBNP were 62.07 and 80.14%, respectively (AUC = 0.733, optimal cut-off value: 3,549 pg/mL). The sensitivity and specificity of LVEF to predict long-term MACE was 62.07 and 87.23%, respectively (AUC = 0.737, optimal cut-off value: 38%). ROC curves showed that the predictive values of troponin I were lower than NT-proBNP for both 30-day (AUC = 0.616, optimal cut-off value: 15.846 ng/mL) and long-term MACE (AUC = 0.509, optimal cut-off value: 0.315 ng/mL).22
Rodriguez-Gonzalez et al. reported that baseline NT-proBNP > 5,000 pg/mL could help to identify high-risk pediatric myocarditis patients with poor outcomes.23 Patients with high NT-proBNP levels are more likely to suffer from prolonged symptoms and need special attention for potential complications. This biomarker should be considered for risk assessment along with echocardiography and clinical judgment.18
5. LIMITATIONS
This study was carried out in a single center with a relatively small sample size. Furthermore, although BNP is a sensitive diagnostic, it is not limited to myocarditis and may be raised in other burn-related diseases such fluid overload. Further multicenter studies with larger cohorts are required to verify these results.
6. CONCLUSION
BNP is a valuable early predictor of toxic myocarditis in patients with severe burn injuries. Its non-invasive nature and high diagnostic accuracy make it an accessible tool for early detection and management. Integrating BNP monitoring into burn care protocols could improve outcomes by facilitating timely interventions for cardiac complications.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
All authors declare that there was no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
IT: Conduction of the study work.
NS, HF: Manuscript editing.
MK, MS: Literature search.
LS: Statistical analysis and review.
7. REFERENCES
- Vatutin NT, Ignatenko GA, Taradin GG, Eshchenko EV, Goncharuk MS, Kulikova SO. Cardiac disorders in burn injury. Bull Sib Med. 2020;19(4):198–206. DOI: 20538262-0363-2020-4-198-206
- Babuin L, Jaffe AS. Troponin: the biomarker of choice for the detection of cardiac injury. CMAJ. 2005;173(10):1191–202. PubMed DOI: 1503/cmaj/051291
- Janardhanan R. Myocarditis with very high troponins: risk stratification by cardiac magnetic resonance. J Thorac Dis. 2016;8(10):E1333. PubMed DOI: 21037/jtd.2016.10.60
- Bethell HJ, Glover JD, Evans JA, Turner SC, Mehta RL, Mullee MA. The relationship between BNP and risk assessment in cardiac rehabilitation patients. Br J Cardiol. 2008;15:161–5. FullText
- Bhananker SM, Cullen BF. Burns. In: Anesthesia and uncommon diseases. Saunders; 2012. p. 526-36. Available from: https://ndl.ethernet.edu.et/bitstream/123456789/91567/1/Anesthesia%20and%20Uncommon%20Diseases%20%282012%2C%20Saunders%29%20-%20libgen.lc.pdf
- Jeschke MG, van Baar ME, Choudhry MA, Chung KK, Gibran NS, Logsetty S. Burn injury. Nat Rev Dis Primers. 2020;6(1):11. PubMed DOI: 1038/s41572-020-0145-5
- Markiewicz-Gospodarek A, Kozioł M, Tobiasz M, Baj J, Radzikowska-Büchner E, Przekora A. Burn wound healing: clinical complications, medical care, treatment, and dressing types. Int J Environ Res Public Health. 2022;19(3):1338. PubMed DOI: 3390/ijerph19031338
- Lampejo T, Durkin SM, Bhatt N, Guttmann O. Acute myocarditis: aetiology, diagnosis and management. Clin Med (Lond). 2021;21(5):e505–10. PubMed DOI: 7861/clinmed.2021-0121
- Novack ML, Zubair M. Natriuretic Peptide B Type Test. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025. Available from: https://www.ncbi.nlm.nih.gov/books/NBK556136/
- Nishikimi T, Nakagawa Y. B-type natriuretic peptide (BNP) revisited. Biology (Basel). 2022;11(7):1034. PubMed DOI: 3390/biology11071034
- Lindahl AE, Stridsberg M, Sjöberg F, Ekselius L, Gerdin B. Natriuretic peptide type B in burn intensive care. J Trauma Acute Care Surg. 2013;74:855–61. PubMed DOI: 1097/TA.0b013e31827e154a
- Ashry S, Hafiz R, Abdel-Hamid M. Brain natriuretic peptide as a biomarker of cardiac toxicity in acute carbon monoxide poisoning. Egypt J Forensic Sci Appl Toxicol. 2018;18(4):1–13. DOI: 21608/ejfsat.2018.4162.1015
- Wang TJ, Larson MG, Levy D, Benjamin EJ, Leip EP, Omland T, et al. Plasma natriuretic peptide levels and the risk of cardiovascular events and death. N Engl J Med. 2004;350(7):655–63. PubMed DOI: 1056/NEJMoa031994
- Ryding AD, Kumar S, Worthington AM, Burgess D. Prognostic value of brain natriuretic peptide in noncardiac surgery. Anesthesiology. 2009;111(2):311–9. PubMed DOI: 1097/ALN.0b013e3181aaeb11
- Güneş Y, Ökçün B, Kavlak E, Erbaş C, Karcıer S. Value of brain natriuretic peptide after acute myocardial infarction. Anatol J Cardiol. 2008;8(3). PubMed
- Ukena C, Kindermann M, Mahfoud F, Geisel J, Lepper PM, Kandolf R, et al. Diagnostic and prognostic validity of different biomarkers in patients with suspected myocarditis. Clin Res Cardiol. 2014;103:743–51. PubMed DOI: 1007/s00392-014-0709-z
- Maurellet JD, Liu PT. B-type natriuretic peptide in the management of heart failure. Hong Kong Med J. 2008;14(3):216. PubMed
- Moady G, Perlmutter S, Atar S. Prognostic value of natriuretic peptides in stable patients with suspected acute myocarditis. J Clin Med. 2022;11(9):2472. PubMed DOI: 3390/jcm11092472
- Imazio M, Brucato A, Barbieri A, Ferroni F, Maestroni S, Ligabue G, et al. Good prognosis for pericarditis with and without myocardial involvement. Circulation. 2013;128(1):42–9. PubMed DOI: 1161/CIRCULATIONAHA.113.001531
- Kobayashi D, Aggarwal S, Kheiwa A, Shah N. Myopericarditis in children: elevated troponin I level does not predict outcome. Pediatr Cardiol. 2012;33:1040–5. PubMed DOI: 1007/s00246-012-0222-y
- Bachmann KN, Gupta DK, Xu M, Brittain E, Farber-Eger E, Arora P, et al. Unexpectedly low natriuretic peptide levels in patients with heart failure. Heart Fail. 2021;9(3):192–200. PubMed DOI: 1016/j.jchf.2020.10.008
- Zhao Y, Lyu N, Zhang W, Tan H, Jin Q, Dang A. Prognosis implication of N-terminal pro-B-type natriuretic peptide in adult patients with acute myocarditis. Front Cardiovasc Med. 2022;9:839763. PubMed DOI: 3389/fcvm.2022.839763
- Rodriguez-Gonzalez M, Sanchez-Codez MI, Lubian-Gutierrez M, Castellano-Martinez A. Clinical presentation and early predictors for poor outcomes in pediatric myocarditis. World J Clin Cases. 2019;7(5):548. PubMed DOI: 12998/wjcc.v7.i5.548