Hiba I. Abdu Al-Sattar 1 , Muna A. Kashmoola 2
Authors affiliations:
Background & objective: Thrombocytosis is not a very uncommon clinical condition. In the recent past, rapid development has been seen in the identification of the role of various biomarkers in diagnosis as well as prognosis. We evaluated the use of various platelet indices, interleukin-6 (IL-6), and C-reactive protein (CRP) in the differential diagnosis of thrombocytosis (reactive or clonal).
Methodology: This is a case-series study, and we included a total of 94 patients with thrombocytosis of varying etiologies. Serum IL-6 and C-reactive protein levels were measured in every patient.
Results: Patients with reactive thrombocytosis demonstrated substantially elevated inflammatory activity, with IL-6 levels (22.92 ± 23.46 pg/mL), versus low levels in clonal thrombocytosis, 3.72 ± 5.54 pg/mL. The reactive thrombocytosis group exhibited CRP levels of 67.27 ± 68.06 mg/L compared to near-normal CRP levels, 4.28 ± 6.57 mg/L. Patients with clonal thrombocytosis demonstrated significantly elevated platelet counts 801.31 ± 272.98 versus 500.33 ± 6.71 plateletcrit values 0.82 ± 0.24 versus 0.50 ± 0.06. Both patient groups exhibited significant correlations between interleukin-6 and CRP levels. Additionally, reactive thrombocytosis patients showed a meaningful association between CRP levels and platelet counts, which was absent in the clonal group.
Conclusion: Platelet count and plateletcrit measurements can serve as useful discriminatory markers between clonal and reactive thrombocytosis. Similarly, serum interleukin-6 and CRP levels provide valuable diagnostic information for differentiating these two thrombocytosis types.
Abbreviations: CRP: C-reactive protein, IL-6: interleukin-6, PDW: platelet distribution width, TCIPA: tumor cell-induced platelet activation
Keywords: Platelet, Thrombosis, Inteleukine-6, C-reactive protein.
Citation: Al-Sattar HIA, Kashmoola MA. Association of C-reactive protein and Interleukin-6 with plateletcrit in critically ill patients with thrombocytosis. Anaesth. pain intensive care 2025;29(8):875-882. DOI: 10.35975/apic.v29i8.2737
Received: April 10, 2025; Revised: July 24, 2025; Accepted: October 22, 2025
Platelets are the most tiny cellular components of blood. The role of stimulated platelets to adhere to the injured blood vessel surface and create a mesh was initially reported during the 1800s.¹ These cell fragments serve as the primary contributors in initial hemostasis and play necessary roles in clot formation.² Beyond their documented functions in blood clotting and bleeding stoppage, recent studies have demonstrated new insights into platelet biology and their different roles, perhaps due to their rich granular stores of growth factors, cytokines, and various biomolecules that respond to various signals and directs multiple biological pathways, potentially contributing to disease pathogenesis, propagation of inflammation, atherosclerosis initiation, immune responses, cancer progression, and tumor spread.²⁻⁴
Thrombocytosis is a common finding in (1.5-2.2%) of the population aged >40 visiting primary care.5-7 Reactive thrombocytosis is usually identified in routine laboratory results. Among subjects with thrombocytosis (80-90%) is known that to have reactive thrombocytosis.8 Approximately 75-82% of individuals without any prior myeloproliferative disorders progressed to thrombocytosis after splenectomy.9 No race, gender, or age preference exists for reactive thrombocytosis.8 The population annual rate of essential thrombocythemia is nearly 1.5-2.0 new cases for every 100,000 individuals. The age of disease onset is between 50-55 years, followed by a secondary peak in women during their childbearing ages; nevertheless, the condition is uncommon in children.10-13
C-reactive protein (CRP) is a homopentameric acute-phase protein, with a spike phase of 1,000-fold at locations of infection or inflammation. Once inflammation settles, CRP levels exponentially decline within 18-20 hours.14-17 The CRP was initially identified as a factor responsible for the differentiation of activated B-cells into immunoglobulin-secreting cells.¹⁸ Later on, it has been found that CRP works as a proinflammatory factor with various roles, synthesized by various immune cells, including T-cells, B-cells, monocytes, and fibroblasts.
Interleukin-6 has the ability to encourage megakaryocyte development in laboratory conditions, elevate platelet counts in animal models, and modulate immune signaling. Additionally, it is well-known for its role in initiating liver production of acute-phase reactants, such as, CRP.¹⁹ Increased IL-6 levels are reported in various human inflammatory conditions, including rheumatoid arthritis, Castleman's disease, autoimmune disorders, and systemic juvenile idiopathic arthritis.20 The present study aimed to evaluate the use of various platelet indices in the differential diagnosis of thrombocytosis (reactive or clonal) and to evaluate the use of IL-6 and CRP in the differential diagnosis of thrombocytosis (reactive or clonal).
This case series study was done in Nineveh Province at Ibn-Sina teaching hospital in the period between May 2022 and November 2022 and involved the collection of data, blood samples, and laboratory investigations. The study included 94 patients of thrombocytosis of varying aetiologies. All patients presented with platelet counts ≥ 450 x 109/L. This study was approved by the Ministry of Health, Nineveh Health Department (15263/May 10, 2022).
A full history was taken from all patients regarding the following: patient's name, age, and gender; date of diagnosis; clinical symptoms; past medical history; history of any previous bleeding manifestation or thrombosis; past surgical history; family history for any hereditary disease; and drug history.
A general and systemic physical examination was done. The patients underwent hematological and biochemical investigations, including complete blood count, IL-6 level and CRP level estimation. Inclusion criterion was patients with platelet counts ≥ 450 x109/L, aged 18 years or older, from both genders. Blood Sampling: Six milliliters (mL) of peripheral venous blood samples were collected by a suitable venipuncture method from each patient.
Three milliliters of blood were placed in the K3EDTA tube, immediately inverted several times, and then placed on a blood shaker at room temperature to ensure complete mixing with the anticoagulant, and used within less than an hour.
Another three milliliters were placed in the gel and clot activator tube and allowed to clot at room temperature. Then it was centrifuged at 2000 rpm for 15 minutes to separate the serum to be used for biochemical analysis.
Platelet indices:
An automated hematological analyzer (Sysmex/XN-350) is used to perform platelet indices on all patients with EDTA tube samples.
Interleukin-6 Assay:
It is a sandwich chemiluminescence immunoassay performed by using a 300-microliter serum sample using the Maglumi fully-auto chemiluminescence immunoassay analyzer and the IL-6 (CLIA) kit from Clia Maglumi (Snibe).
CRP Assay:
It is a sandwich chemiluminescence immunoassay performed by using a 300-microliter serum sample using the Maglumi fully-auto chemiluminescence immunoassay analyzer and the CRP (CLIA) kit from Clia Maglumi (Snibe)
Statistical Analysis:
This statistical analysis used SPSS V26 (USA) and Minitab V17 (USA) to investigate results through descriptive statistics (frequency counts, percentages, averages, and variability measures) and inferential testing. Statistical significance was assessed using Fisher's exact test and Student's t-test, while associations between variables were evaluated through Pearson correlation coefficients and simple regression analysis. A significance threshold of p ≤ 0.05 was applied to determine meaningful statistical relationships.
This study included a total of 94 patients with a platelet count ≥ 450 ×109 who were observed during the study period from May 2022 to November 2022, with ages ranging from 18-85 years old. Males made up 31/94 cases (33%) and females made up 63/94 cases (67%). Out of 94 cases of thrombocytosis, 38 cases (40.4%) were found to be of clonal etiology with a mean age of (54.27±15.44), and 56 cases (59.6%) of reactive etiology with a mean age of (43.3±16.80).
The most common causes of clonal thrombocytosis in 38 patients were essential thrombocythemia (44.7%), followed by polycythemia vera (39.5%) and chronic myeloid leukemia (10.5%), while in reactive thrombocytosis in 56 patients, the first and second most common causes were both iron-deficiency anemia (21.4%) and inflammatory disease (21.4%), followed by malignancy (19.6%).
In patients with clonal thrombocytosis, the most common presenting features were headache in 10/38 (26.3%), visual changes in 5/38 (13.2%), bleeding manifestation in 5/38 (13.2%), weight loss in 3/38 (7.9%) and erythromelalgia in 3/38 (7.9%), while 5/38 (13.2%) were asymptomatic (found on routine investigations) (Table 1).
Patients with reactive thrombocytosis demonstrated substantially elevated inflammatory activity, with IL-6 levels (22.92± 23.46 pg/mL). In contrast, patients with clonal thrombocytosis showed markedly lower IL-6 concentrations, 3.72 ± 5.54 pg/mL. The reactive thrombocytosis group exhibited CRP levels of 67.27 ± 68.06 mg/L. Conversely, the clonal thrombocytosis group maintained near-normal CRP levels, 4.28 ± 6.57 mg/L (Table 2).
In the clonal thrombocytosis, 32 patients presented with normal IL-6 levels, while 6 patients had elevated IL-6 levels. Interestingly, the platelet counts were remarkably similar between these two subgroups, with patients having normal IL-6 levels showing platelet counts of 801.31 ± 272.98 × 10⁹/L and those with high IL-6 levels displaying 807.00 ± 220.32 × 10⁹/L.
The reactive thrombocytosis group, patients with high IL-6 levels, showed moderately higher platelet counts (570.34 ± 10.2 × 10⁹/L) compared to those with normal IL-6 levels (500.33 ± 6.71 × 10⁹/L).
The platelet distribution width (PDW) remained relatively consistent across all groups, ranging from 11.52 to 12.83 fl, with minimal variation between normal and elevated IL-6 subgroups. Similarly, mean platelet volume (MPV) showed stability across groups, with values between 9.84 and 10.65 fl, suggesting that IL-6 levels do not significantly influence platelet size characteristics.
Within the clonal group, patients with elevated IL-6 showed slightly higher P-LCR values (30.27 ± 8.38%) compared to those with normal IL-6 levels (26.62 ± 5.86%), though this difference was not marked as statistically significant. In clonal thrombocytosis, both normal and elevated IL-6 groups showed comparable PCT values (0.82 ± 0.24% and 0.81 ± 0.18%, respectively). However, when comparing between thrombocytosis types, the clonal thrombocytosis groups demonstrated significantly higher PCT values than the reactive thrombocytosis groups, with the reactive group showing PCT values of 0.50 ± 0.06% for normal IL-6 and 0.56 ± 0.11% for elevated IL-6 levels. This reinforces the fundamental difference in platelet mass between clonal and reactive thrombocytosis, regardless of inflammatory marker status (Table 3).
In the clonal thrombocytosis group, 33 patients had normal CRP levels, while only 5 patients presented with elevated CRP levels. The platelet count showed a notable difference between these subgroups, with patients having normal CRP levels displaying significantly higher platelet counts (823.15 ± 270.03 × 10⁹/L) compared to those with high CRP levels (664 ± 165.59 × 10⁹/L). In clonal thrombocytosis, patients with normal CRP levels had significantly higher PCT values (0.83 ± 0.24%) compared to those with high CRP levels (0.74 ± 0.17%). Additionally, when comparing normal CRP levels between clonal and reactive thrombocytosis, the clonal group showed significantly higher PCT values than the reactive group (0.48 ± 0.03%), indicating fundamental differences in platelet mass between these two thrombocytosis types (Table 4).
There was a significant positive correlation between serum IL-6 level and platelet count in patients with reactive thrombocytosis (P = 0.001; r = 0.444), while there was no correlation between serum IL-6 level and platelet count in patients with clonal thrombocytosis (P = 0.997; r = -0.001) (Figure 1A). There was a significant positive correlation between serum CRP level and platelet count in patients with reactive thrombocytosis (P = 0.001; r = 0.449), while no correlation was found between serum CRP level and platelet count in patients with clonal thrombocytosis (P = 0.275, r = -0.182) (Figure 1B).
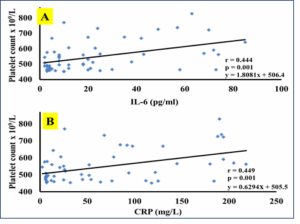
Figure 1: The correlation between inflammatory markers in the patient group
(A) Correlation between serum IL-6 and CRP in reactive thrombocytosis.
(B) Correlation between serum IL-6 and CRP in clonal thrombocytosis.
The pro-inflammatory cytokines, which are associated with rheumatoid arthritis, stimulate megakaryopoiesis and thrombopoiesis; hence could be used as an indicator of disease activity.21 Thrombocytosis occurs in patients with various infectious diseases.22
Inflammatory substances (e.g., IL-6) stimulate increased platelet production when tissues are injured during surgical operations. This elevation in platelet levels may continue for as long as six weeks following tissue damage, with peak platelet counts occurring within the initial two-week period.23 Platelet activation can additionally happen in malignancies. When cancer spreads through the bloodstream, malignant cells may stimulate platelets in circulation, a process termed tumor cell-induced platelet activation (TCIPA). This platelet stimulation subsequently leads to the production of growth factors originating from platelets and forms clots composed of tumor cells, platelets, and fibrin-rich material.23
In the current study, the vaso-occlusive symptoms, including headache, visual changes, erythromelalgia, and bleeding manifestations, were significantly seen in patients with clonal thrombocytosis in comparison with reactive thrombocytosis, which shows a different clinical presentation that is related to the underlying disease rather than the previously mentioned manifestations. In regard to vaso-occlusive symptoms, they were due to microvascular inflammation, platelet aggregation, and arteriolar microthrombi formation,24 and this is in agreement with Buss et al. (1994) and Bleeker et al. (2011) studies.13,25 Also, the clonal group was significantly noticed in routine investigations, and this is in agreement with the study reported by Jensen et al. (2000).26
Megakaryocytopoiesis and thrombopoiesis appear to be regulated by a variety of hematopoietic growth factors, including cytokines.27 The determination of these cytokines in various forms of thrombocytosis may provide insight into the etiology of elevated platelet counts and aid in the differentiation of reactive thrombocytosis from clonal thrombocytosis.28-30
IL-6 has different biological functions, such as, regulation of hemopoiesis and immune responses. Its synthesis could be triggered by pathogens, inflammation, and other cytokines such as interleukin-1, tumour necrotic factor alpha, and platelet-derived growth factor. By promoting the formation of haematopoietic thrombopoietin or megakaryocytopoiesis, it plays a crucial role in platelet generation.31,32 Increased production of IL-6 has also been reported in certain tumor states, where it is important for several systemic manifestations such as the production of acute phase proteins and thrombocytosis.33 The contribution of the present study was the assessment of CRP levels as a possible surrogate for measurement of interleukin-6.CRP is an acute-phase reactant, and its hepatic synthesis may be mediated by interleukin6.34
The data in this study shows the mean serum concentration levels of IL-6 and CRP were significantly higher in reactive thrombocytosis than in uncomplicated clonal thrombocytosis (the latter is an autonomous phenomenon and is not regulated by cytokines that affect megakaryocytopoiesis),29 which is in agreement with the results done by Tefferi et al. (1994),28 Haznedaroglu et al.(1993), 35 Hsu et al. 36 Araneda et al. (1999), 37 and M.G. Alexandrakis et al. (2003), 38
However, normal levels of CRP and IL-6 do not exclude a reactive cause of thrombocytosis, as some patients have normal levels of both or one of the inflammatory markers (IL-6 and CRP). In this study, about 30% of cases of reactive thrombocytosis have normal levels of IL-6, and some of them even have normal levels of CRP. In agreement with these results is the study done by Tefferi et al. (1994).28
Correlation analysis in patients with reactive thrombocytosis showed that IL-6 correlated well with CRP, same as in report from Tefferi et al. (1994), 28 and platelet count as in report from Ünsal et al. 39 However, in contrast to Hsu et al. 36 The study found no correlation with platelet count. Our study also showed a positive correlation between IL-6 and white blood cell counts, the same as that reported by Hsu et al. (1999).36
However, in patients with clonal thrombocytosis, no significant correlation was observed with all mentioned data, as also reported by Hsu et al. 36 apart from significant positive correlation between IL-6 and CRP, which was also reported by Tefferi et al. (1994), and M.G.Alexandrakis et al. (2003).28,38 In regard to CRP correlation analysis, patients with reactive thrombocytosis demonstrated a marked correlation with platelet count, which is in agreement with a study by Ito et al., that reported a significant correlation between CRP and platelet count in patients with renal cell carcinoma,40 and with study by Zareifar et al.(2014), show platelet count was directly correlated with CRP,41 but disagreed with a study by Toprak et al., that reported no correlation between platelet count and CRP in reactive thrombocytosis,42 while no correlation was found between CRP and platelet count in clonal thrombocytosis, as also, reported by Tefferi et al. (1994).28
Platelet indices (platelet count and PCT) were significantly and significantly higher in the clonal group, implying that platelet indices (platelet count and PCT) may aid in distinguishing between reactive and clonal thrombocytosis. Interleukin-6 and CRP were significantly higher in the reactive group. In the reactive group, platelet count and PCT were significantly higher in cases with high serum IL-6 than in cases with normal IL-6 levels, and PCT was also significantly higher in cases with high serum CRP than in cases with normal CRP levels. In patients with reactive thrombocytosis, a positive relationship exists between serum IL-6 and CRP concentrations. A similar positive association between IL-6 and CRP levels was also observed in patients with clonal thrombocytosis. Additionally, serum IL-6 levels showed a meaningful correlation with platelet counts in the reactive thrombocytosis group, but this relationship was not present in patients with clonal thrombocytosis.
Authors affiliations:
- Hiba I. Abdu Al-Sattar, College of Medicine, University of Mosul, Mosul, Iraq; Email; hibaomer641@gmail.com
- Muna A. Kashmoola, College of Medicine, University of Mosul, Mosul, Iraq; Email; mak@uomosul.edu.iq
ABSTRACT
Background & objective: Thrombocytosis is not a very uncommon clinical condition. In the recent past, rapid development has been seen in the identification of the role of various biomarkers in diagnosis as well as prognosis. We evaluated the use of various platelet indices, interleukin-6 (IL-6), and C-reactive protein (CRP) in the differential diagnosis of thrombocytosis (reactive or clonal).
Methodology: This is a case-series study, and we included a total of 94 patients with thrombocytosis of varying etiologies. Serum IL-6 and C-reactive protein levels were measured in every patient.
Results: Patients with reactive thrombocytosis demonstrated substantially elevated inflammatory activity, with IL-6 levels (22.92 ± 23.46 pg/mL), versus low levels in clonal thrombocytosis, 3.72 ± 5.54 pg/mL. The reactive thrombocytosis group exhibited CRP levels of 67.27 ± 68.06 mg/L compared to near-normal CRP levels, 4.28 ± 6.57 mg/L. Patients with clonal thrombocytosis demonstrated significantly elevated platelet counts 801.31 ± 272.98 versus 500.33 ± 6.71 plateletcrit values 0.82 ± 0.24 versus 0.50 ± 0.06. Both patient groups exhibited significant correlations between interleukin-6 and CRP levels. Additionally, reactive thrombocytosis patients showed a meaningful association between CRP levels and platelet counts, which was absent in the clonal group.
Conclusion: Platelet count and plateletcrit measurements can serve as useful discriminatory markers between clonal and reactive thrombocytosis. Similarly, serum interleukin-6 and CRP levels provide valuable diagnostic information for differentiating these two thrombocytosis types.
Abbreviations: CRP: C-reactive protein, IL-6: interleukin-6, PDW: platelet distribution width, TCIPA: tumor cell-induced platelet activation
Keywords: Platelet, Thrombosis, Inteleukine-6, C-reactive protein.
Citation: Al-Sattar HIA, Kashmoola MA. Association of C-reactive protein and Interleukin-6 with plateletcrit in critically ill patients with thrombocytosis. Anaesth. pain intensive care 2025;29(8):875-882. DOI: 10.35975/apic.v29i8.2737
Received: April 10, 2025; Revised: July 24, 2025; Accepted: October 22, 2025
1. INTRODUCTION
Platelets are the most tiny cellular components of blood. The role of stimulated platelets to adhere to the injured blood vessel surface and create a mesh was initially reported during the 1800s.¹ These cell fragments serve as the primary contributors in initial hemostasis and play necessary roles in clot formation.² Beyond their documented functions in blood clotting and bleeding stoppage, recent studies have demonstrated new insights into platelet biology and their different roles, perhaps due to their rich granular stores of growth factors, cytokines, and various biomolecules that respond to various signals and directs multiple biological pathways, potentially contributing to disease pathogenesis, propagation of inflammation, atherosclerosis initiation, immune responses, cancer progression, and tumor spread.²⁻⁴
Thrombocytosis is a common finding in (1.5-2.2%) of the population aged >40 visiting primary care.5-7 Reactive thrombocytosis is usually identified in routine laboratory results. Among subjects with thrombocytosis (80-90%) is known that to have reactive thrombocytosis.8 Approximately 75-82% of individuals without any prior myeloproliferative disorders progressed to thrombocytosis after splenectomy.9 No race, gender, or age preference exists for reactive thrombocytosis.8 The population annual rate of essential thrombocythemia is nearly 1.5-2.0 new cases for every 100,000 individuals. The age of disease onset is between 50-55 years, followed by a secondary peak in women during their childbearing ages; nevertheless, the condition is uncommon in children.10-13
C-reactive protein (CRP) is a homopentameric acute-phase protein, with a spike phase of 1,000-fold at locations of infection or inflammation. Once inflammation settles, CRP levels exponentially decline within 18-20 hours.14-17 The CRP was initially identified as a factor responsible for the differentiation of activated B-cells into immunoglobulin-secreting cells.¹⁸ Later on, it has been found that CRP works as a proinflammatory factor with various roles, synthesized by various immune cells, including T-cells, B-cells, monocytes, and fibroblasts.
Interleukin-6 has the ability to encourage megakaryocyte development in laboratory conditions, elevate platelet counts in animal models, and modulate immune signaling. Additionally, it is well-known for its role in initiating liver production of acute-phase reactants, such as, CRP.¹⁹ Increased IL-6 levels are reported in various human inflammatory conditions, including rheumatoid arthritis, Castleman's disease, autoimmune disorders, and systemic juvenile idiopathic arthritis.20 The present study aimed to evaluate the use of various platelet indices in the differential diagnosis of thrombocytosis (reactive or clonal) and to evaluate the use of IL-6 and CRP in the differential diagnosis of thrombocytosis (reactive or clonal).
2. METHODOLOGY
This case series study was done in Nineveh Province at Ibn-Sina teaching hospital in the period between May 2022 and November 2022 and involved the collection of data, blood samples, and laboratory investigations. The study included 94 patients of thrombocytosis of varying aetiologies. All patients presented with platelet counts ≥ 450 x 109/L. This study was approved by the Ministry of Health, Nineveh Health Department (15263/May 10, 2022).
A full history was taken from all patients regarding the following: patient's name, age, and gender; date of diagnosis; clinical symptoms; past medical history; history of any previous bleeding manifestation or thrombosis; past surgical history; family history for any hereditary disease; and drug history.
A general and systemic physical examination was done. The patients underwent hematological and biochemical investigations, including complete blood count, IL-6 level and CRP level estimation. Inclusion criterion was patients with platelet counts ≥ 450 x109/L, aged 18 years or older, from both genders. Blood Sampling: Six milliliters (mL) of peripheral venous blood samples were collected by a suitable venipuncture method from each patient.
Three milliliters of blood were placed in the K3EDTA tube, immediately inverted several times, and then placed on a blood shaker at room temperature to ensure complete mixing with the anticoagulant, and used within less than an hour.
Another three milliliters were placed in the gel and clot activator tube and allowed to clot at room temperature. Then it was centrifuged at 2000 rpm for 15 minutes to separate the serum to be used for biochemical analysis.
Platelet indices:
An automated hematological analyzer (Sysmex/XN-350) is used to perform platelet indices on all patients with EDTA tube samples.
Interleukin-6 Assay:
It is a sandwich chemiluminescence immunoassay performed by using a 300-microliter serum sample using the Maglumi fully-auto chemiluminescence immunoassay analyzer and the IL-6 (CLIA) kit from Clia Maglumi (Snibe).
CRP Assay:
It is a sandwich chemiluminescence immunoassay performed by using a 300-microliter serum sample using the Maglumi fully-auto chemiluminescence immunoassay analyzer and the CRP (CLIA) kit from Clia Maglumi (Snibe)
Statistical Analysis:
This statistical analysis used SPSS V26 (USA) and Minitab V17 (USA) to investigate results through descriptive statistics (frequency counts, percentages, averages, and variability measures) and inferential testing. Statistical significance was assessed using Fisher's exact test and Student's t-test, while associations between variables were evaluated through Pearson correlation coefficients and simple regression analysis. A significance threshold of p ≤ 0.05 was applied to determine meaningful statistical relationships.
3. RESULTS
This study included a total of 94 patients with a platelet count ≥ 450 ×109 who were observed during the study period from May 2022 to November 2022, with ages ranging from 18-85 years old. Males made up 31/94 cases (33%) and females made up 63/94 cases (67%). Out of 94 cases of thrombocytosis, 38 cases (40.4%) were found to be of clonal etiology with a mean age of (54.27±15.44), and 56 cases (59.6%) of reactive etiology with a mean age of (43.3±16.80).
The most common causes of clonal thrombocytosis in 38 patients were essential thrombocythemia (44.7%), followed by polycythemia vera (39.5%) and chronic myeloid leukemia (10.5%), while in reactive thrombocytosis in 56 patients, the first and second most common causes were both iron-deficiency anemia (21.4%) and inflammatory disease (21.4%), followed by malignancy (19.6%).
In patients with clonal thrombocytosis, the most common presenting features were headache in 10/38 (26.3%), visual changes in 5/38 (13.2%), bleeding manifestation in 5/38 (13.2%), weight loss in 3/38 (7.9%) and erythromelalgia in 3/38 (7.9%), while 5/38 (13.2%) were asymptomatic (found on routine investigations) (Table 1).
| Table 1: Comparison between reactive and clonal thrombocytosis based on clinical findings. | |||
| Clinical Findings | Reactive Thrombocytosis
(n = 56) |
Clonal Thrombocytosis
(n = 38) |
|
| Headache | 0 (0) | 10 (26.3) | |
| visual changes | 0 (0) | 5 (13.2) | |
| pruritus | 0 (0) | 2 (5.3) | |
| Erythromelalgia | 0 (0) | 3 (7.8) | |
| On routine investigation | 0 (0) | 5 (13.2) | |
| Abdominal distention | 0 (0) | 1 (2.6) | |
| vomiting | 0 (0) | 1 (2.6) | |
| Stroke | 0 (0) | 2 (5.3) | |
| Fever | 7 (12.5) | 0 (0) | |
| Fatigue | 1 (1.8) | 0 (0) | |
| Pallor | 10 (17.9) | 1 (2.6) | |
| palpitation | 6 (10.7) | 0 (0) | |
| Weight loss | 0 (0) | 3 (7.9) | |
| Lymph node enlargement | 8 (14.3) | 0 (0) | |
| Loin pain | 1 (1.8) | 0 (0) | |
| Joint pain | 10 (17.9) | 0 (0) | |
| Back pain | 1 (1.8) | 0 (0) | |
| Chest pain | 7 (12.5) | 0 (0) | |
| Bleeding manifestation | 2 (3.6) | 5 (13.2) | |
| Abdominal pain and Jaundice | 1 (1.8) | 0 (0) | |
| Dyspnea | 2 (3.6) | 0 (0) | |
| Data presented as n (%) | |||
Patients with reactive thrombocytosis demonstrated substantially elevated inflammatory activity, with IL-6 levels (22.92± 23.46 pg/mL). In contrast, patients with clonal thrombocytosis showed markedly lower IL-6 concentrations, 3.72 ± 5.54 pg/mL. The reactive thrombocytosis group exhibited CRP levels of 67.27 ± 68.06 mg/L. Conversely, the clonal thrombocytosis group maintained near-normal CRP levels, 4.28 ± 6.57 mg/L (Table 2).
| Table 2. Proinflammatory markers in reactive thrombocytosis and clonal thrombocytosis | |||
| Parameters | Reactive thrombocytosis
(n = 56) |
Clonal thrombocytosis
(n = 38) |
P- value |
| IL-6 pg/mL | 22.92 ± 23.46 | 3.72 ± 5.54 | 0.0001 |
| CRP mg/L | 67.27 ± 68.06 | 4.28 ± 6.57 | 0.0001 |
| Data are given as mean ± SD; P < 0.05 considered significant | |||
In the clonal thrombocytosis, 32 patients presented with normal IL-6 levels, while 6 patients had elevated IL-6 levels. Interestingly, the platelet counts were remarkably similar between these two subgroups, with patients having normal IL-6 levels showing platelet counts of 801.31 ± 272.98 × 10⁹/L and those with high IL-6 levels displaying 807.00 ± 220.32 × 10⁹/L.
The reactive thrombocytosis group, patients with high IL-6 levels, showed moderately higher platelet counts (570.34 ± 10.2 × 10⁹/L) compared to those with normal IL-6 levels (500.33 ± 6.71 × 10⁹/L).
The platelet distribution width (PDW) remained relatively consistent across all groups, ranging from 11.52 to 12.83 fl, with minimal variation between normal and elevated IL-6 subgroups. Similarly, mean platelet volume (MPV) showed stability across groups, with values between 9.84 and 10.65 fl, suggesting that IL-6 levels do not significantly influence platelet size characteristics.
Within the clonal group, patients with elevated IL-6 showed slightly higher P-LCR values (30.27 ± 8.38%) compared to those with normal IL-6 levels (26.62 ± 5.86%), though this difference was not marked as statistically significant. In clonal thrombocytosis, both normal and elevated IL-6 groups showed comparable PCT values (0.82 ± 0.24% and 0.81 ± 0.18%, respectively). However, when comparing between thrombocytosis types, the clonal thrombocytosis groups demonstrated significantly higher PCT values than the reactive thrombocytosis groups, with the reactive group showing PCT values of 0.50 ± 0.06% for normal IL-6 and 0.56 ± 0.11% for elevated IL-6 levels. This reinforces the fundamental difference in platelet mass between clonal and reactive thrombocytosis, regardless of inflammatory marker status (Table 3).
| Table 3: Comparison of platelet parameters between patients with high and normal IL-6 levels in thrombocytosis. | ||||
| Platelet parameter | Clonal Thrombocytosis | Reactive Thrombocytosis | ||
| Normal IL-6 level
(n = 32) |
High IL-6 level
(n = 6) |
Normal IL-6 level
(n = 18) |
High IL-6 level
(n = 38) |
|
| Platelet count 109/l | 801.31 ± 272.98* | 807.00 ± 220.32# | 500.33 ± 6.71 | 570.34 ± 10.2 |
| PDW fl | 12.47 ± 2.10 | 12.83 ± 2.60 | 11.79 ± 2.51 | 11.52 ± 2.37 |
| MPV fl | 10.20 ± 0.92 | 10.65 ± 1.12 | 9.92 ± 0.92 | 9.84 ± 1.10 |
| P-LCR % | 26.62 ± 5.86 | 30.27 ± 8.38 | 25.09 ± 7.11 | 24.74 ± 7.62 |
| PCT % | 0.82 ± 0.24* | 0.81 ± 0.18# | 0.50 ± 0.06 | 0.56 ± 0.11* |
| *significant difference at p value 0.05 using two two-sample t-test comparing normal CPR in clonal versus reactive thrombocytosis
#significant difference at p value 0.05 using two-sample t-test comparing normal CPR in clonal versus reactive thrombocytosis |
||||
In the clonal thrombocytosis group, 33 patients had normal CRP levels, while only 5 patients presented with elevated CRP levels. The platelet count showed a notable difference between these subgroups, with patients having normal CRP levels displaying significantly higher platelet counts (823.15 ± 270.03 × 10⁹/L) compared to those with high CRP levels (664 ± 165.59 × 10⁹/L). In clonal thrombocytosis, patients with normal CRP levels had significantly higher PCT values (0.83 ± 0.24%) compared to those with high CRP levels (0.74 ± 0.17%). Additionally, when comparing normal CRP levels between clonal and reactive thrombocytosis, the clonal group showed significantly higher PCT values than the reactive group (0.48 ± 0.03%), indicating fundamental differences in platelet mass between these two thrombocytosis types (Table 4).
| Table 4. Comparison of platelet parameters between patients with high and normal CRP levels | ||||
| Platelet parameter | Clonal Thrombocytosis | Reactive Thrombocytosis | ||
| Normal CRP level
(n = 33) |
High CRP level
(n = 5) |
Normal CRP level
(n = 8) |
High CRP level
(n = 48) |
|
| Platelet count (109/L) | 823.15 ± 270.03* | 664 ± 165.59# | 502.38 ± 38.79 | 555.42 ± 100.18 |
| PDW fl | 12.50 ± 2.15 | 12.74 ± 2.36 | 10.95 ± 1.90 | 11.72 ± 2.47 |
| MPV fl | 10.20 ± 0.95 | 10.74 ± 0.93 | 9.48 ± 0.82 | 9.93 ± 1.06 |
| P-LCR % | 26.65 ± 6.25 | 30.76 ± 6.31 | 21.90 ± 5.41 | 25.35 ± 7.61 |
| PCT % | 0.83 ± 0.24* | 0.74 ± 0.17# | 0.48 ± 0.03 | 0.55 ± 0.10* |
| *significant difference at p value 0.05 using two-sample t-test comparing normal CPR in clonal versus reactive thrombocytosis
#significant difference at p value 0.05 using two-sample t-test comparing normal CPR in clonal versus reactive thrombocytosis |
||||
4. DISCUSSION
There was a significant positive correlation between serum IL-6 level and platelet count in patients with reactive thrombocytosis (P = 0.001; r = 0.444), while there was no correlation between serum IL-6 level and platelet count in patients with clonal thrombocytosis (P = 0.997; r = -0.001) (Figure 1A). There was a significant positive correlation between serum CRP level and platelet count in patients with reactive thrombocytosis (P = 0.001; r = 0.449), while no correlation was found between serum CRP level and platelet count in patients with clonal thrombocytosis (P = 0.275, r = -0.182) (Figure 1B).

Figure 1: The correlation between inflammatory markers in the patient group
(A) Correlation between serum IL-6 and CRP in reactive thrombocytosis.
(B) Correlation between serum IL-6 and CRP in clonal thrombocytosis.
The pro-inflammatory cytokines, which are associated with rheumatoid arthritis, stimulate megakaryopoiesis and thrombopoiesis; hence could be used as an indicator of disease activity.21 Thrombocytosis occurs in patients with various infectious diseases.22
Inflammatory substances (e.g., IL-6) stimulate increased platelet production when tissues are injured during surgical operations. This elevation in platelet levels may continue for as long as six weeks following tissue damage, with peak platelet counts occurring within the initial two-week period.23 Platelet activation can additionally happen in malignancies. When cancer spreads through the bloodstream, malignant cells may stimulate platelets in circulation, a process termed tumor cell-induced platelet activation (TCIPA). This platelet stimulation subsequently leads to the production of growth factors originating from platelets and forms clots composed of tumor cells, platelets, and fibrin-rich material.23
In the current study, the vaso-occlusive symptoms, including headache, visual changes, erythromelalgia, and bleeding manifestations, were significantly seen in patients with clonal thrombocytosis in comparison with reactive thrombocytosis, which shows a different clinical presentation that is related to the underlying disease rather than the previously mentioned manifestations. In regard to vaso-occlusive symptoms, they were due to microvascular inflammation, platelet aggregation, and arteriolar microthrombi formation,24 and this is in agreement with Buss et al. (1994) and Bleeker et al. (2011) studies.13,25 Also, the clonal group was significantly noticed in routine investigations, and this is in agreement with the study reported by Jensen et al. (2000).26
Megakaryocytopoiesis and thrombopoiesis appear to be regulated by a variety of hematopoietic growth factors, including cytokines.27 The determination of these cytokines in various forms of thrombocytosis may provide insight into the etiology of elevated platelet counts and aid in the differentiation of reactive thrombocytosis from clonal thrombocytosis.28-30
IL-6 has different biological functions, such as, regulation of hemopoiesis and immune responses. Its synthesis could be triggered by pathogens, inflammation, and other cytokines such as interleukin-1, tumour necrotic factor alpha, and platelet-derived growth factor. By promoting the formation of haematopoietic thrombopoietin or megakaryocytopoiesis, it plays a crucial role in platelet generation.31,32 Increased production of IL-6 has also been reported in certain tumor states, where it is important for several systemic manifestations such as the production of acute phase proteins and thrombocytosis.33 The contribution of the present study was the assessment of CRP levels as a possible surrogate for measurement of interleukin-6.CRP is an acute-phase reactant, and its hepatic synthesis may be mediated by interleukin6.34
The data in this study shows the mean serum concentration levels of IL-6 and CRP were significantly higher in reactive thrombocytosis than in uncomplicated clonal thrombocytosis (the latter is an autonomous phenomenon and is not regulated by cytokines that affect megakaryocytopoiesis),29 which is in agreement with the results done by Tefferi et al. (1994),28 Haznedaroglu et al.(1993), 35 Hsu et al. 36 Araneda et al. (1999), 37 and M.G. Alexandrakis et al. (2003), 38
However, normal levels of CRP and IL-6 do not exclude a reactive cause of thrombocytosis, as some patients have normal levels of both or one of the inflammatory markers (IL-6 and CRP). In this study, about 30% of cases of reactive thrombocytosis have normal levels of IL-6, and some of them even have normal levels of CRP. In agreement with these results is the study done by Tefferi et al. (1994).28
Correlation analysis in patients with reactive thrombocytosis showed that IL-6 correlated well with CRP, same as in report from Tefferi et al. (1994), 28 and platelet count as in report from Ünsal et al. 39 However, in contrast to Hsu et al. 36 The study found no correlation with platelet count. Our study also showed a positive correlation between IL-6 and white blood cell counts, the same as that reported by Hsu et al. (1999).36
However, in patients with clonal thrombocytosis, no significant correlation was observed with all mentioned data, as also reported by Hsu et al. 36 apart from significant positive correlation between IL-6 and CRP, which was also reported by Tefferi et al. (1994), and M.G.Alexandrakis et al. (2003).28,38 In regard to CRP correlation analysis, patients with reactive thrombocytosis demonstrated a marked correlation with platelet count, which is in agreement with a study by Ito et al., that reported a significant correlation between CRP and platelet count in patients with renal cell carcinoma,40 and with study by Zareifar et al.(2014), show platelet count was directly correlated with CRP,41 but disagreed with a study by Toprak et al., that reported no correlation between platelet count and CRP in reactive thrombocytosis,42 while no correlation was found between CRP and platelet count in clonal thrombocytosis, as also, reported by Tefferi et al. (1994).28
5. CONCLUSION
Platelet indices (platelet count and PCT) were significantly and significantly higher in the clonal group, implying that platelet indices (platelet count and PCT) may aid in distinguishing between reactive and clonal thrombocytosis. Interleukin-6 and CRP were significantly higher in the reactive group. In the reactive group, platelet count and PCT were significantly higher in cases with high serum IL-6 than in cases with normal IL-6 levels, and PCT was also significantly higher in cases with high serum CRP than in cases with normal CRP levels. In patients with reactive thrombocytosis, a positive relationship exists between serum IL-6 and CRP concentrations. A similar positive association between IL-6 and CRP levels was also observed in patients with clonal thrombocytosis. Additionally, serum IL-6 levels showed a meaningful correlation with platelet counts in the reactive thrombocytosis group, but this relationship was not present in patients with clonal thrombocytosis.
- Data availability
- Conflict of interest
- Funding
- Authors’ contribution
10. REFERENCES
- Hvas AM. Crucial stepping stones in platelet history. Semin Thromb Hemost 2023; 49(03): 272-278. DOI: 1055/s-0042-1758119
- Chaudhary PK, Kim S, Kim S. An insight into recent advances on platelet function in health and disease. Int J Mol Sci. 2022;23(11):6022. DOI: 1055/s-0042-1758119 .
- Gremmel T, Frelinger AL, Michelson AD. Platelet physiology. Semin Thromb Hemost. 2016 Apr 1;42(3):191–204. DOI: 1055/s-0035-1564835
- Amable PR, Carias RBV, Teixeira MVT, Da Cruz Pacheco Í, Corrêa Do Amaral RJF, Granjeiro JM, et al. Platelet-rich plasma preparation for regenerative medicine: Optimization and quantification of cytokines and growth factors. Stem cell res ther. 2013;4(3):1–13.
- Bailey SER, Ukoumunne OC, Shephard EA, Hamilton W. Clinical relevance of thrombocytosis in primary care: a prospective cohort study of cancer incidence using English electronic medical records and cancer registry data. Br J Gen Pract. 2017;67(659):e405–13. PMCID: PMC5442956 DOI: 3399/bjgp17X691109 .
- Shephard E, Neal R, Rose P, Walter F, Hamilton WT. Clinical features of kidney cancer in primary care: a case-control study using primary care records. Br J Gen Pract. 2013;63(609):e250. PMCID: PMC3609472 DOI: 3399/bjgp13X665215 .
- Hamilton W, Peters TJ, Round A, Sharp D. What are the clinical features of lung cancer before the diagnosis is made? A population based case-control study. Thorax. 2005;60(12):1059–65. PMCID: PMC1747254 DOI: 1136/thx.2005.045880
- Wang R, Xie Y, Xie L, Liu J, Jia J, Chen X, Wu Q. Platelet-rich plasma versus corticosteroid in the treatment of knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Georgian Med News. 2024; (349):169-82. PMID: 38963222.
- Khan PN, Nair RJ, Olivares J, Tingle LE, Li Z. Postsplenectomy reactive thrombocytosis. Proceedings. InBaylor University Medical Center Proceedings. 2009;22(1):9–12. PMCID: PMC2626351 DOI: 1080/08998280.2009.11928458 .
- Hassan SS, Alkhalidi EF, Hamed RS. Factors Involved in Megakaryocytosis and Platelet Origin. J Nat Sc Biol Med. 2024;15(2):324. DOI:10.4103/jnsbm.JNSBM_15_2_17.
- Zaveri F, Naser S. Prolonged Dual Antiplatelet Therapy in High-Risk ACS Patients: Insights from the OPT-CAD Study. Texila Int J Pub Heal, 2025, 13(1):864-868. DOI: 21522/TIJPH.2013.13.01.ART080
- Papen M, Ghijselings S, Vles G. Reactive Thrombocytosis Leading to Recurrent Arterial Thrombosis Reversed by Management of a Prosthetic Joint Infection of the Hip. Cureus. 2022;14(4). PMCID: PMC9012589 DOI: 7759/cureus.24166 .
- Bleeker JS, Hogan WJ. Thrombocytosis: Diagnostic Evaluation, Thrombotic Risk Stratification, and Risk-Based Management Strategies. Thrombosis. 2011;2011:1–16. PMCID: PMC3200282 DOI: 1155/2011/536062 .
- Tillett WS, Francis T. Serological reactions in pneumonia with a non-protein somatic fraction of pneumococcus. J Exp Med. 1930;52(4):561–71. PMCID: PMC2131884 DOI: 1084/jem.52.4.561 .
- Boras E, Slevin M, Alexander MY, Aljohi A, Gilmore W, Ashworth J, et al. Monomeric C-reactive protein and Notch-3 co-operatively increase angiogenesis through PI3K signalling pathway. Cytokine. 2014;69(2):165–79. DOI: DOI: 1016/j.cyto.2014.05.027
- Ciubotaru I, Potempa LA, Wander RC. Production of modified C-reactive protein in U937-derived macrophages. Exp Biol Med. 2005;230(10):762-70. https://DOI.org/101177/153537020523001010.
- Hage FG, Szalai AJ. C-Reactive Protein Gene Polymorphisms, C-Reactive Protein Blood Levels, and Cardiovascular Disease Risk. J Am Coll Cardiol. 2007;50(12):1115–22. DOI: 1016/j.jacc.2007.06.012.
- Hirano T, Yasukawa K, Harada H, Taga T, Watanabe Y, Matsuda T, et al. Complementary DNA for a novel human interleukin (BSF-2) that induces B lymphocytes to produce immunoglobulin. Nature. 1986;324(6092):73–76. DOI: 1038/324073a0 .
- Sproston NR, Ashworth JJ. Role of C-reactive protein at sites of inflammation and infection. Front. Immunol. 2018;9(4):754760. PMCID: PMC5908901 DOI: 3389/fimmu.2018.00754 .
- Mihara M, Hashizume M, Yoshida H, Suzuki M, Shiina M. IL-6/IL-6 receptor system and its role in physiological and pathological conditions. Clin Sci. 2012;122(4):143–59. DOI: 1042/CS20110340
- Ertenli I, Kiraz S, Öztürk MA, Haznedaroǧlu IC, Çelik I, Çalgüneri M. Pathologic thrombopoiesis of rheumatoid arthritis. Rheumatol Int. 2003;23(2):49–60. DOI: 1007/S00296-003-0289-0.
- Prina E, Ferrer M, Ranzani OT, Polverino E, Cillóniz C, Moreno E, et al. Thrombocytosis is a marker of poor outcome in community-acquired pneumonia. Chest. 2013;143(3):767–75. DOI: 1378/chest.12-1235.
- Choe EI, Kasabian AK, Kolker AR, Karp NS, Zhang L, Bass LS, Nardi M, Josephson G, Karpatkin M. Thrombocytosis after major lower extremity trauma: Mechanism and possible role in free flap failure. Ann Plast Surg. 1996;36(5):489-94.
- Van Genderen PJJ, Michiels JJ. Erythromelalgia: A pathognomonic microvascular thrombotic complication in essential thrombocythemia and polycythemia vera. Semin Thromb Hemost. 1997;23(4):357–63. DOI: 1055/s-2007-996109 .
- Buss DH, Cashell AW, O’Connor ML, Richards F, Case LD. Occurrence, etiology, and clinical significance of extreme thrombocytosis: a study of 280 cases. Am J Med. 1994;96(3):247–53. DOI: 10.10DOI: 1016/0002-9343(94)90150-3
- Jensen MK, De Nully Brown P, Nielsen OJ, Hasselbalch HC. Incidence, clinical features and outcome of essential thrombocythaemia in a well defined geographical area. Eur J Haematol. 2000;65(2):132–9. DOI: 1034/j.1600-0609.2000.90236.x .
- Hassan SS, Alkhalidi EF, Hamed RS. Platelet concentrates a new renaissance in tissue regeneration. Pharmacy Prac. 2025;23(1):1-1. DOI: 10.18549/PharmPract.2025.1.3104.
- Tefferi A, Ho TC, Ahmann GJ, Katzmann JA, Greipp PR. Plasma interleukin-6 and C-reactive protein levels in reactive versus clonal thrombocytosis. Am J Med. 1994;97(4):374–8. DOI: 1016/0002-9343(94)90306-9
- Haznedaroğlu İC, Ertenli İ, Özcebe O, Kiraz S, Özdemir O, Sayinalp NM, et al. Megakaryocyte-related interleukins in reactive thrombocytosis versus autonomous thrombocythemia. Acta Haematol. 1996;95(2):107–11. DOI: 1159/000203857 .
- Kutti J, Wadenvik H. Diagnostic and differential criteria of essential thrombocythemia and reactive thrombocytosis. Leuk Lymphoma. 1996;22 (1):41–5. DOI: 3109/10428199609074359
- Williams N, Bertoncello I, Jackson H, Arnold J, Kavnoudias H. The Role of Interleukin 6 in Megakaryocyte Formation, Megakaryocyte Development and Platelet Production. Ciba Found Symp. 1992;167:160–73. DOI: DOI: 1002/9780470514269.ch10
- Tanaka T, Narazaki M, Kishimoto T. IL-6 in Inflammation, Immunity, and Disease. Cold Spring Harb Perspect Biol. 2014;6(10):16295–6. PMCID: PMC4176007 DOI: 1101/cshperspect.a016295
- Fisher DT, Appenheimer MM, Evans SS. The two faces of IL-6 in the tumor microenvironment. Semin immunol. 2014;26(1):38–47. Tanaka T, Kishi PMCID: PMC3970580
- DOI: 1016/j.smim.2014.01.008 oto T. The biology and medical implications of interleukin-6. Cancer immunology research. 2014;2(4):288–94. DOI: 10.1158/2326-6066.CIR-14-00 DOI: 10.1158/2326-6066.CIR-14-0022 22.
- Sayinalp N, Haznedaroǧlu İC, Osman OÖ, Özcebe, Dündar S, Kirazli S. Interleukin‐1 β and interleukin‐6 in clonal versus reactive thrombocytosis. Eur J Haematol. 1995;55(5):339–40. DOI: 1111/j.1600-0609.1995.tb00708.x .
- Hsu HC, Tsai WH, Jiang ML, Ho CH, Hsu ML, Ho CK, et al. Circulating levels of thrombopoietic and inflammatory cytokines in patients with clonal and reactive thrombocytosis. J Lab Clin Med. 1999;134(4):392–7. DOI: 1016/s0022-2143(99)90154-3 .
- Araneda M, Krishnan V, Hall K, Kalbfleisch J, Krishnaswamy G, Krishnan K. Reactive and clonal thrombocytosis: proinflammatory and hematopoietic cytokines and acute phase proteins. South Med J. 2001;94(4):417–20. DOI: 10.1097/00007611-200104000-00012.
- Alexandrakis MG, Passam FH, Moschandrea IA, Christophoridou A V., Pappa CA, Coulocheri SA, et al. Levels of serum cytokines and acute phase proteins in patients with essential and cancer-related thrombocytosis. Am J Clin Oncol. 2003; 26(2):135–40.DOI: 1097/00000421-200304000-00007.
- Ünsal E, Aksaray S, Köksal D, Şipit T. Potential role of interleukin 6 in reactive thrombocytosis and acute phase response in pulmonary tuberculosis. Postgrad Med J. 2004: 604–7. PMCID: PMC1743352 DOI: 1136/pgmj.2004.030544 .
- Ito K, Asano T, Yoshii H, Satoh A, Sumitomo M, Hayakawa M. Impact of thrombocytosis and C-reactive protein elevation on the prognosis for patients with renal cell carcinoma. Int J Urol . 2006;13(11):1365–70. DOI: 1111/j.1442-2042.2006.01563.x
- Zareifar S, Farahmand Far MR, Golfeshan F, Cohan N. Changes in platelet count and mean platelet volume during infectious and inflammatory disease and their correlation with ESR and CRP. J Clin Lab Anal. 2014;28(3):245–8. DOI: 10.1002/JCLA.21673.
- Al-Tameemi WF, Noori AK. The impact of platelet indices in the evaluation of different causes of platelet count disorder. Iraqi J Hematol. 2022;11(1):32. DOI: 10.4103/IJH.IJH_47_21. http://www.ijhonline.org on Monday, July 4, 2022, IP: 109.224.55.22]