Rabab Mohamed Mohamed 1*, Sameh M. Fathy 2, Attia Gad Anwar 1
Author affiliations:
Objectives: The objective of this research endeavor was to assess the efficacy of three distinct intrathecal concentrations of dexmedetomidine (ITD) in conjunction with 0.5% hyperbaric bupivacaine in TURP.
Methods: This study employed a randomized, double-blind design, ninety patients who were admitted for TURP participated. Six (Group A), eight (Group B), or ten (Group C) patients were administered µg ITD diluted in a solution consisting of 0.5 mL saline 0.9%- and 1.5-mL hyperbaric bupivacaine 0.5%.
Results: An absence of statistically significant variation was noted at the outset latencies of sensory block and motor block between groups A and B. Nevertheless, when comparing Group C to groups A and B, the onset latency was diminished considerably (P < 0.05). Significantly prolonged was the duration of first rescue analgesia in groups C and B than in Group A, and in Group C than in Group B (P < 0.05). Significantly reduced postoperative total morphine consumption and NRS scores at 6h, 8h, and 12h were observed in groups C and B, respectively, compared to Group A and Group B (P < 0.05).
Conclusions: When combined with hyperbaric bupivacaine, 10 µg of ITD significantly prolongs analgesia duration, sensory block, and motor blockade in TURP. This prolongation is accompanied by a reduction in the necessity for postoperative analgesics, and the incidence of adverse effects is not significantly different from lower dosages.
Keywords: Transurethral Resection of the Prostate; Dexmedetomidine; Hyperbaric Bupivacaine; Dose
Citation: Mohamed RM, Fathy SM, Anwar AG. Low dose hyperbaric bupivacaine 0.5% in transurethral resection of the prostate with three different doses of intrathecal dexmedetomidine: a randomized double-blind trial. Anaesth. pain intensive care 2025;29(2):217-225. DOI: 10.35975/apic.v29i2.2707
Received: May 22, 2024; Reviewed: January 26, 2025; Accepted: February 15, 2025
A widely employed technique involves the administration of patients undergoing transurethral resection of the prostate (TURP) are administered spinal anesthesia.1 This procedure is typically administered to elderly patients who frequently suffer from cardiovascular, pulmonary, or hormone-related ailments. Hence, it is imperative to regulate the degree of anesthesia with the intention of minimizing the effect on hemodynamics throughout the procedure.2 Utilizing reduced quantities of local anesthetics facilitates sensation control and accelerates recovery, although this may not always be adequate to anesthetize the area for the duration of the operation.3
In order to augment the efficacy of postoperative anesthesia and pain management, opioids or clonidine are administered in conjunction with intrathecal bupivacaine.4 Commonly administered quantities of clonidine, which is well-tolerated in conjunction with local anesthetics without inducing respiratory distress or pruritus, may induce lethargy, hypotension, or a decelerated heart rate.5
Since its FDA approval for intravenous sedation in 1999, dexmedetomidine, a more selective alpha-2 adrenoceptor agonist than clonidine, has been put to use for its analgesic, blood pressure-lowering, and analgesic properties.6 The primary factor responsible for the analgesic effects of dexmedetomidine at the spinal level is its high-fat solubility. This solubility facilitates rapid permeation into the spinal fluid, thereby enhancing its efficacy at the level of the spinal cord.7
Previous studies have also examined the possible synergy that may exist between dexmedetomidine and local anesthetics. These investigations have shown that dexmedetomidine has the ability to extend the effectiveness of bupivacaine and comparable local anesthetics, with minimal adverse effects.8, 9 However, these mixtures can occasionally result in excessively high concentrations and lengthy periods of recovery. Therefore, determining the optimal, safe, and effective dosage is vital.
The concurrent administration of dexmedetomidine (3–15 µg) and 10–15 mg of bupivacaine effectively prolonged the duration of local anesthetics while ensuring a negligible occurrence of adverse effects.8, 10 A definitive agreement concerning the ideal dosage of intrathecal dexmedetomidine (ITD) remains elusive; notwithstanding the substantial volume of research that has been undertaken in this field. Although higher concentrations improve pain management during and after surgery, they also elevate the risk of compromised hemodynamics.11, 12
The aim of this study was to compare and evaluate the effectiveness of three different concentrations of dexmedetomidine when combined with intrathecal 0.5% hyperbaric bupivacaine for TURP.
Ninety TURP-admitted patients aged 55 to 70 years old and classified as physical activity I-III by ASA participated in the A double-blind, randomized research. The research was conducted utilizing the ethical sanction of the Ethical Committee of Tanta University Hospitals in Egypt. (approval code: 36264PR195/5/23). Written informed consent was obtained from the patient or a representative of their family.
Exclusion criteria included any contraindication to SA, drug hypersensitivity, uncontrolled hypertension, previous congestive heart failure and myocardial infarction, cardiac block, and lesions of fixed cardiac output. Additionally, we excluded cases that required an intraoperative switch to general anesthesia.
A complete medical history, clinical examination, and laboratory investigation were performed on every patient. The patients were briefed on the eleven-point NRS (0 = no pain, 10 = severe pain).
Randomization and blindness
In opaque, sequentially numbered envelopes that were sealed and randomly dispersed into three groups by a computer-generated sequence, ninety patients were categorized into three distinct groups. On the morning of surgery, one of the chief nurses, who did not take part in the study or data collection, manually opened the envelopes and verified the grouping of each patient. Patients received either 6 µg (Group A), 8 µg (Group B) or 10 µg (Group C) dexmedetomidine combined with 0.5% hyperbaric bupivacaine 1.5 mL.
Both outcome assessors and cases were blinded. A dedicated pharmacist, who had no other obligations during the study, devised the study medications. Unaware of the group designation, an additional anesthesiologist evaluated intraoperative and postoperative procedures.
Standard monitors that were employed included non-invasive blood pressure (NIBP), electrocardiography, a temperature instrument, and pulse oximetry.
In order to enable the insertion of an IV, a cannula of 18G diameter was utilized. A preload of 10–20 mL/kg of lactated Ringer's solution was administered to the animal. Seated were the individuals who were undergoing treatment. Sterile techniques were implemented, which involved the use of decontamination solutions that contained povidone iodine as well as surgical drapes. Prior to the procedure, the intervertebral spaces L3/L4 or L4/L5 were identified. It is likely that three milliliters of 2% lidocaine were injected into the cutaneous region identified above the intervertebral space using a 22G hypodermic syringe as the anesthetic. In order to administer the study medication, a 22G pencil-point spinal needle was inserted into the lumbar region. After ten seconds of confirming the presence of cerebrospinal fluid flow, the intrathecal medication was administered methodically via injection. The patients were administered ITD in the following concentrations: 6 µg (Group A), 8 µg (Group B), and 10 µg (Group C). In order to produce a solution for each concentration, 1.5 µL of 0.5% hyperbaric bupivacaine was mixed with 0.5 µL of normal saline.
Considerable factors include the duration of motor and sensory limitations, the time required to reach optimal performance, the amount of morphine consumed postoperatively, and the duration of rescue analgesia administration.
Upon introducing a 25 G hypodermic needle through the mid-clavicular line, sensory obstruction is identified by the lack of sensation to a pinprick. Meticulous observation of this threshold was maintained until the peak level of sensory blockade achieved stability. The initiation of the sensory block was determined at the T10 dermatome level as the duration between the end of the intrathecal injection and the disappearance of pinprick sensation.13 The level apex of sensory blockage is defined as the degree to which the testing procedure achieves sensory blockage following three iterations. The peak sensory level is defined as the duration that elapses from the time intrathecal administration concludes until maximal sensory block is achieved.
In order to assess the motor block, the modified Bromage score was implemented:14 0: absence of a motor block, 1: lack of hip flexion, but knee and ankle mobility, 2: lack of hip and knee flexion, but ankle mobility; and 3: hip, knee, and ankle immobility. The onset of motor block is measured from the moment the intrathecal injection is completed until the Bromage 3 score appears. Duration of motor block is the duration from the end of the intrathecal injection till the Bromage 0 score is reinstated.
Heart rate (HR) and mean arterial blood pressure (MAP) were at 5 min, 10 min, 15 min, 30 min, 45 min, and 60 min intraoperatively, at the conclusion of the procedure, and at 2 h, 4 h, 6 h, 8 h, 12 h, 18 h, and 24 h post-surgery in the post-anesthesia care unit (PACU),
In the PACU, NRS scores were assessed at 2, 4, 6, 8, 12, 18, and 24 h. When the NRS was determined to be ≥ 4, morphine 1 mg IV was administered as a rescue analgesic; repeated in 30 min. The cumulative quantity of morphine ingested by the individual during the initial 24 h following the procedure was recorded.
A decrease in systolic blood pressure of less than 90 mmHg or greater than 30% below baseline was categorized as hypotension. Additional intravenous fluids and 5 mg inj ephedrine were used to treat the condition. Bradycardia, which was defined as a HR < 50/min, was managed with a 0.5 mg atropine intravenous infusion. Local anesthetic systemic toxicity (LAST), hypotension, bradycardia, failed blockade, and urinary retention were documented in terms of their incidence.
The duration of the sensory block was the principal outcome, whereas the secondary outcomes included the time to achieve maximal sensory block level, duration of the motor block, time until first rescue analgesia was administered, total morphine consumption postoperatively, and complications.
Sample size calculation
In order to determine the appropriate sample size, G*Power 3.1.9.2 from Universitat Kiel, Germany, was utilized. A preliminary investigation was initiated, comprising five cases in each cohort. The findings revealed that Group A experienced sensory impairment for an average of 377.4 ± 24.66 hours, Group B for 396.4 ± 23.41 hours, and Group C for 400 ± 24.57 hours. The sample size was determined using 90% study power, a 0.409 effect size, a 95% confidence limit, and a 1:1:1 group ratio. Furthermore, to accommodate for attrition, an additional three cases were appended to each cohort. As a result, thirty patients were assigned to each cohort.
Statistical analysis
The data analysis was conducted using SPSS v27 (IBM, Chicago, IL, USA). To determine whether the data followed a normal distribution, the Shapiro-Wilks test and histograms were utilized. Analyses of variance (ANOVA) (F) and the post hoc test (Tukey) were utilized to examine quantitative parametric data expressed as the mean and standard deviation. The researchers utilized a combination of the Mann-Whitney U test and Kruskal-Wallis's test in order to compare the non-parametric quantitative data of the groups. The values obtained were displayed in the form of the median and interquartile range (IQR). The chi-square test was utilized to assess the qualitative variables, which were expressed as percentages and frequencies. Considered to indicate statistical significance was a two-tailed P < 0.05.
Upon assessing the eligibility of 109 participants for this trial, 11 patients failed to meet the inclusion criteria, while 8 patients declined to participate. The remaining patients were allocated into three equal categories, with 30 patients in each group, through the use of random assignment. All assigned patients were followed up on and statistically analyzed (Figure 1).
Demographic data and duration of surgery were matched among the three groups (Table 1).
There was minimal variation in the time required to attain the maximum level of sensory block among the three groups. The onset times of sensory block and motor block did not differ significantly between Group A and Group B. In contrast, both Group A and Group B demonstrated significantly longer onset times than Group C (P < 0.05). There was no significant difference observed in the duration of motor and sensory blocks between groups A and B. Nevertheless, Group C exhibited a substantially prolonged duration of blocks compared to both groups A and B (P < 0.001). Both Group C and Group B exhibited considerably longer intervals between the initial analgesic request than Group A (P < 0.05), with Group C demonstrating a significantly longer interval than Group B. In comparison to Group A, postoperative total morphine consumption was considerably lower in groups C and B; in fact, it was even lower in Group C than in Group B (P < 0.05) as shown in Table 2.
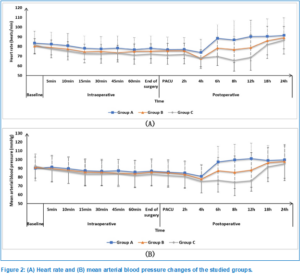
No statistically significant variations were detected in the intraoperative HR and MAP measurements among the three groups. Regarding postoperative HR and MAP measurements at the PACU at 2 h, 4 h, 18 h, and 24 h, no significant differences were observed between the three groups. However, at 6 h, 8 h, and 12 h, these parameters were significantly lower in Group C and Group B compared to Group A and Group C (P < 0.05) (P < 0.05) (Figure 2).
At PACU, after 2, 4, 18, and 24 h, no statistically significant differences were observed in the NRS scores between the three groups. However, at 6, 8, and 12 h, the scores were significantly lower in Groups C and B compared to Group A; and in Group C than in Group B (P < 0.05) (Table 3).
Regarding complications, e.g., hypotension, bradycardia, and urinary retention, there were no statistically significant variations observed across the three cohorts. Every patient had an ineffective obstruction and a deficiency of LAST (Table 4).
SA is the preferred option for TURP on account of its distinctive advantages. Self-administration (SA) during surgery not only facilitates unrestricted respiration but also provides relief from postoperative pain, reduces blood loss, and obviates the necessity for airway obstruction via tracheal intubation, which may result in postoperative hemorrhage and wheezing.15 While regional anesthesia may conceivably provide benefits such as early detection of TURP syndrome in conscious patients, it is not without its limitations, including short duration of anesthesia, patient distress, and discomfort.9
Rather than technical issues, limitations of regional anesthesia, including SA, are frequently ascribed to inadequate sedation.16 In addition to alleviating patient anxiety and tension, effective sedatives in SA increases patient and surgeon satisfaction. Nevertheless, overemphasis on sedation may obscure the symptoms of TURP syndrome and induce delirium subsequent to surgical procedures, especially among the elderly.9patient and surgeon satisfaction. Nevertheless, overemphasis on sedation may obscure the symptoms of TURP syndrome and induce delirium subsequent to surgical procedures, especially among the elderly.9
As a sedative with analgesic properties, dexmedetomidine is an advantageous adjunct to SA for TURP owing to its capacity to compensate for insufficient block height and induce minimal respiratory depression.6
Our research investigated the efficacy of three distinct ITD concentrations (6 µg, 8 µg, and 10 µg) in conjunction with low-dose hyperbaric bupivacaine 0.5% in patients presenting with TURP. Higher concentrations of ITD were associated with a significant and consistent increase in the duration of sensory and motor blockade, in addition to analgesic effects, according to the results of our study. Significantly, these outcomes were attained while maintaining similar profiles of adverse effects and hemodynamic stability. In particular, an increase in the dosage of dexmedetomidine from 6 µg to 10 µg resulted in a commensurate prolongation of both sensory and motor block, in addition to analgesic effects.
Extending the duration of analgesic administration offers a dual benefit: it mitigates the adverse consequences associated with postoperative discomfort, such as impaired immune function, prolonged hospitalization, delayed wound healing, the potential for neurosensitization, and the development of chronic pain. Furthermore, it promotes the progression of motor obstruction, which may reduce mobility and the probability of developing complications such as deep venous thrombosis, pulmonary embolism, and others that are closely related.17
Gupta et al. investigated the dose-response correlation between ITD and SA characteristics.13 Their findings showed that adding 10 µg of ITD, the application of 0.5% hyperbaric bupivacaine, as opposed to 2.5 µg or 5 µg, significantly accelerated the onset of sensory and motor blockade. Furthermore, it resulted in an extended duration of sensory and motor impairment, as well as analgesic properties, while retaining a comparable profile of adverse effects.
Other researchers conducted separate studies comparing different doses of intrathecal dexmedetomidine (ITD), specifically 2 µg vs. 4 µg, 5 µg vs. 10 µg, and 10 µg vs. 15 µg, respectively.18,19,20 In each study, they observed a rise in the sensory block, motor block duration, and analgesic effect that is also dose dependent.
ITD exerts its pain-relieving action through a dual mechanism. Firstly, it inhibits neurotransmitter release by targeting presynaptic α2A receptors. Secondly, it hyperpolarizes the postsynaptic neurons.21 The motor block prolongation could stem from The inhibiting impact of α2 agonists on the dorsal horn motor neurons of the spinal cord.22
It has been suggested that sensitivity to dexmedetomidine may vary depending on the type of nerve fiber, considering that the ED50 for maximal inhibition is 2.5 µg for sensory C fibers and above 10 µg for Aβ motor fibers.23 This led us to utilize a dosage in this range between 6 µg and 10 µg for this trial. While Eid et al. employed an increased dosage of 15 µg of ITD, we chose to exclude it from our dose-response trial design due to the significant increase in sedation scores reported in their study.20 Additionally, our study's relatively short mean length of surgical procedures contributed to this decision. ITD has demonstrated efficacy in managing nociceptive, visceral, and neuropathic pain. Its neurological safety has been established through a follow-up period of up to ten years post-anesthesia.18, 24, 25
In our study, the mean ± SD onset of sensory block time was similar among Groups A and B; however, it was earlier significantly in Group C (4.9 ± 2.14 vs 4.4 ± 1.61 vs 2.9 ± 1.36; P < 0.001 in Groups A, B and C respectively).
These results align with those reported by Halder et al.19, who employed a comparable definition for the onset time of sensory block and observed a significantly earlier onset with 10 µg than 5 µg of ITD. Similarly, Yektas et al. found a substantial rise in the number of sensory segments blocked that depends on dosage when comparing 2 µg and 4 µg of ITD.18 Moreover, Gupta et al. investigated the dose-response correlation between ITD and the onset of both sensory and motor block.13
The use of ITD in conjunction with hyperbaric bupivacaine SA has been associated with a notable benefit which is a decrease in the need for postoperative analgesia.8, 9, 26 We also noted a substantial dose-dependent reduction (P = 0.001) in the 24-hour morphine need as the doses of ITD increased.
The primary and clinically notable adverse effect linked with α2 agonists is hemodynamic disturbance, specifically manifesting as hypotension and bradycardia.27 Most studies did not report any significant rise in the occurrence of hemodynamic adverse events accompanied by the utilization of ITD, regardless of the dosage administered.19, 28, 29
We noted a dose-dependent elevation in hypotension incidence (20% with 6µg, 26.67% with 8µg, and 33.33% with 10µg as well as bradycardia (10%), (13.33%), and (20%)) across groups A, B, and C, respectively, Nevertheless, this rise failed to reach statistical significance.
The potential for further sympatholysis induced by dexmedetomidine may have been constrained in our trial due to the substantial sympatholysis induced by the higher dose and volume of bupivacaine utilized. Our observations are consistent with comparable dose-independent hemodynamic findings reported by other researchers.19, 20 Additionally, side effects such as urinary retention were comparable across all groups. Similar dose-independent side effects were also noted by Yektas et al. and Halder et al.18, 19
Our study is limited by short follow-up time and the absence of a control group, thus future studies with longer monitoring duration in the presence of a control group are needed to generalize our findings.
In TURP, 10 µg of ITD in conjunction with hyperbaric bupivacaine 0.5% considerably extends analgesia duration, sensory block, and motor blockade and is accompanied by a reduction in the need for analgesics used postoperatively, while the incidence of adverse effects does not differ significantly when compared to lesser dosages.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
The authors declare no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
RMM: developed the original idea and the protocol, abstracted and analyzed data, wrote the manuscript, and is a guarantor.
MA: contributed to the development of the protocol, abstracted data.
AGA: prepared the manuscript.
Author affiliations:
- Rabab Mohamed Mohamed, Assistant Professor of Anesthesiology, Surgical Intensive Care and Pain Medicine, Faculty of Medicine, Tanta University, Tanta, Egypt: Email: rabmoh_30@outlook.com
- Sameh M. Fathy, Department of Anesthesiology, Surgical ICU and Pain Management, Faculty of Medicine, Beni-Suef University, Beni-Suef, Egypt; Email: samehsadek923@gmail.com, ORCID: 0009-0003-1117-4276
- Attia Gad Anwar, Department of Anesthesiology, Surgical Intensive Care and Pain Medicine, Faculty of Medicine, Tanta University, Tanta, Egypt: Email: atteiagad@gmail.com
ABSTRACT
Objectives: The objective of this research endeavor was to assess the efficacy of three distinct intrathecal concentrations of dexmedetomidine (ITD) in conjunction with 0.5% hyperbaric bupivacaine in TURP.
Methods: This study employed a randomized, double-blind design, ninety patients who were admitted for TURP participated. Six (Group A), eight (Group B), or ten (Group C) patients were administered µg ITD diluted in a solution consisting of 0.5 mL saline 0.9%- and 1.5-mL hyperbaric bupivacaine 0.5%.
Results: An absence of statistically significant variation was noted at the outset latencies of sensory block and motor block between groups A and B. Nevertheless, when comparing Group C to groups A and B, the onset latency was diminished considerably (P < 0.05). Significantly prolonged was the duration of first rescue analgesia in groups C and B than in Group A, and in Group C than in Group B (P < 0.05). Significantly reduced postoperative total morphine consumption and NRS scores at 6h, 8h, and 12h were observed in groups C and B, respectively, compared to Group A and Group B (P < 0.05).
Conclusions: When combined with hyperbaric bupivacaine, 10 µg of ITD significantly prolongs analgesia duration, sensory block, and motor blockade in TURP. This prolongation is accompanied by a reduction in the necessity for postoperative analgesics, and the incidence of adverse effects is not significantly different from lower dosages.
Keywords: Transurethral Resection of the Prostate; Dexmedetomidine; Hyperbaric Bupivacaine; Dose
Citation: Mohamed RM, Fathy SM, Anwar AG. Low dose hyperbaric bupivacaine 0.5% in transurethral resection of the prostate with three different doses of intrathecal dexmedetomidine: a randomized double-blind trial. Anaesth. pain intensive care 2025;29(2):217-225. DOI: 10.35975/apic.v29i2.2707
Received: May 22, 2024; Reviewed: January 26, 2025; Accepted: February 15, 2025
1. INTRODUCTION
A widely employed technique involves the administration of patients undergoing transurethral resection of the prostate (TURP) are administered spinal anesthesia.1 This procedure is typically administered to elderly patients who frequently suffer from cardiovascular, pulmonary, or hormone-related ailments. Hence, it is imperative to regulate the degree of anesthesia with the intention of minimizing the effect on hemodynamics throughout the procedure.2 Utilizing reduced quantities of local anesthetics facilitates sensation control and accelerates recovery, although this may not always be adequate to anesthetize the area for the duration of the operation.3
In order to augment the efficacy of postoperative anesthesia and pain management, opioids or clonidine are administered in conjunction with intrathecal bupivacaine.4 Commonly administered quantities of clonidine, which is well-tolerated in conjunction with local anesthetics without inducing respiratory distress or pruritus, may induce lethargy, hypotension, or a decelerated heart rate.5
Since its FDA approval for intravenous sedation in 1999, dexmedetomidine, a more selective alpha-2 adrenoceptor agonist than clonidine, has been put to use for its analgesic, blood pressure-lowering, and analgesic properties.6 The primary factor responsible for the analgesic effects of dexmedetomidine at the spinal level is its high-fat solubility. This solubility facilitates rapid permeation into the spinal fluid, thereby enhancing its efficacy at the level of the spinal cord.7
Previous studies have also examined the possible synergy that may exist between dexmedetomidine and local anesthetics. These investigations have shown that dexmedetomidine has the ability to extend the effectiveness of bupivacaine and comparable local anesthetics, with minimal adverse effects.8, 9 However, these mixtures can occasionally result in excessively high concentrations and lengthy periods of recovery. Therefore, determining the optimal, safe, and effective dosage is vital.
The concurrent administration of dexmedetomidine (3–15 µg) and 10–15 mg of bupivacaine effectively prolonged the duration of local anesthetics while ensuring a negligible occurrence of adverse effects.8, 10 A definitive agreement concerning the ideal dosage of intrathecal dexmedetomidine (ITD) remains elusive; notwithstanding the substantial volume of research that has been undertaken in this field. Although higher concentrations improve pain management during and after surgery, they also elevate the risk of compromised hemodynamics.11, 12
The aim of this study was to compare and evaluate the effectiveness of three different concentrations of dexmedetomidine when combined with intrathecal 0.5% hyperbaric bupivacaine for TURP.
2. METHODOLOGY
Ninety TURP-admitted patients aged 55 to 70 years old and classified as physical activity I-III by ASA participated in the A double-blind, randomized research. The research was conducted utilizing the ethical sanction of the Ethical Committee of Tanta University Hospitals in Egypt. (approval code: 36264PR195/5/23). Written informed consent was obtained from the patient or a representative of their family.
Exclusion criteria included any contraindication to SA, drug hypersensitivity, uncontrolled hypertension, previous congestive heart failure and myocardial infarction, cardiac block, and lesions of fixed cardiac output. Additionally, we excluded cases that required an intraoperative switch to general anesthesia.
A complete medical history, clinical examination, and laboratory investigation were performed on every patient. The patients were briefed on the eleven-point NRS (0 = no pain, 10 = severe pain).
Randomization and blindness
In opaque, sequentially numbered envelopes that were sealed and randomly dispersed into three groups by a computer-generated sequence, ninety patients were categorized into three distinct groups. On the morning of surgery, one of the chief nurses, who did not take part in the study or data collection, manually opened the envelopes and verified the grouping of each patient. Patients received either 6 µg (Group A), 8 µg (Group B) or 10 µg (Group C) dexmedetomidine combined with 0.5% hyperbaric bupivacaine 1.5 mL.
Both outcome assessors and cases were blinded. A dedicated pharmacist, who had no other obligations during the study, devised the study medications. Unaware of the group designation, an additional anesthesiologist evaluated intraoperative and postoperative procedures.
Standard monitors that were employed included non-invasive blood pressure (NIBP), electrocardiography, a temperature instrument, and pulse oximetry.
In order to enable the insertion of an IV, a cannula of 18G diameter was utilized. A preload of 10–20 mL/kg of lactated Ringer's solution was administered to the animal. Seated were the individuals who were undergoing treatment. Sterile techniques were implemented, which involved the use of decontamination solutions that contained povidone iodine as well as surgical drapes. Prior to the procedure, the intervertebral spaces L3/L4 or L4/L5 were identified. It is likely that three milliliters of 2% lidocaine were injected into the cutaneous region identified above the intervertebral space using a 22G hypodermic syringe as the anesthetic. In order to administer the study medication, a 22G pencil-point spinal needle was inserted into the lumbar region. After ten seconds of confirming the presence of cerebrospinal fluid flow, the intrathecal medication was administered methodically via injection. The patients were administered ITD in the following concentrations: 6 µg (Group A), 8 µg (Group B), and 10 µg (Group C). In order to produce a solution for each concentration, 1.5 µL of 0.5% hyperbaric bupivacaine was mixed with 0.5 µL of normal saline.
Considerable factors include the duration of motor and sensory limitations, the time required to reach optimal performance, the amount of morphine consumed postoperatively, and the duration of rescue analgesia administration.
Upon introducing a 25 G hypodermic needle through the mid-clavicular line, sensory obstruction is identified by the lack of sensation to a pinprick. Meticulous observation of this threshold was maintained until the peak level of sensory blockade achieved stability. The initiation of the sensory block was determined at the T10 dermatome level as the duration between the end of the intrathecal injection and the disappearance of pinprick sensation.13 The level apex of sensory blockage is defined as the degree to which the testing procedure achieves sensory blockage following three iterations. The peak sensory level is defined as the duration that elapses from the time intrathecal administration concludes until maximal sensory block is achieved.
In order to assess the motor block, the modified Bromage score was implemented:14 0: absence of a motor block, 1: lack of hip flexion, but knee and ankle mobility, 2: lack of hip and knee flexion, but ankle mobility; and 3: hip, knee, and ankle immobility. The onset of motor block is measured from the moment the intrathecal injection is completed until the Bromage 3 score appears. Duration of motor block is the duration from the end of the intrathecal injection till the Bromage 0 score is reinstated.
Heart rate (HR) and mean arterial blood pressure (MAP) were at 5 min, 10 min, 15 min, 30 min, 45 min, and 60 min intraoperatively, at the conclusion of the procedure, and at 2 h, 4 h, 6 h, 8 h, 12 h, 18 h, and 24 h post-surgery in the post-anesthesia care unit (PACU),
In the PACU, NRS scores were assessed at 2, 4, 6, 8, 12, 18, and 24 h. When the NRS was determined to be ≥ 4, morphine 1 mg IV was administered as a rescue analgesic; repeated in 30 min. The cumulative quantity of morphine ingested by the individual during the initial 24 h following the procedure was recorded.

A decrease in systolic blood pressure of less than 90 mmHg or greater than 30% below baseline was categorized as hypotension. Additional intravenous fluids and 5 mg inj ephedrine were used to treat the condition. Bradycardia, which was defined as a HR < 50/min, was managed with a 0.5 mg atropine intravenous infusion. Local anesthetic systemic toxicity (LAST), hypotension, bradycardia, failed blockade, and urinary retention were documented in terms of their incidence.
The duration of the sensory block was the principal outcome, whereas the secondary outcomes included the time to achieve maximal sensory block level, duration of the motor block, time until first rescue analgesia was administered, total morphine consumption postoperatively, and complications.
Sample size calculation
In order to determine the appropriate sample size, G*Power 3.1.9.2 from Universitat Kiel, Germany, was utilized. A preliminary investigation was initiated, comprising five cases in each cohort. The findings revealed that Group A experienced sensory impairment for an average of 377.4 ± 24.66 hours, Group B for 396.4 ± 23.41 hours, and Group C for 400 ± 24.57 hours. The sample size was determined using 90% study power, a 0.409 effect size, a 95% confidence limit, and a 1:1:1 group ratio. Furthermore, to accommodate for attrition, an additional three cases were appended to each cohort. As a result, thirty patients were assigned to each cohort.
| Table 1: Demographic data and duration of surgery of the studied groups | |||||
| Parameter | Group A (n = 30) |
Group B (n = 30) |
Group C (n = 30) |
P | |
| Age (years) | 62.9 ± 3.54 | 64.9 ± 3.67 | 63.8 ± 2.27 | 0.072 | |
| Sex | Male | 24 (80) | 20 (66.67) | 22 (73.33) | 0.506 |
| Female | 6 (20) | 10 (33.33) | 8 (26.67) | ||
| Weight (kg) | 75.1 ± 11.81 | 72.2 ± 12.66 | 76.2 ± 11.19 | 0.415 | |
| Height (m) | 1.66 ± 0.09 | 1.67 ± 0.07 | 1.64 ± 0.09 | 0.544 | |
| BMI (kg/m2) | 27.4 ± 5.04 | 26.2 ± 5.51 | 28.5 ± 5.53 | 0.262 | |
| ASA
physical status |
I | 22 (73.33) | 20 (66.67) | 23 (76.67) | 0.857 |
| II | 6 (20) | 7 (23.33) | 6 (20) | ||
| III | 2 (6.67) | 3 (10) | 1 (3.33) | ||
| Duration of surgery (min) | 99.7 ± 16.34 | 109.8 ± 24.41 | 112 ± 23.69 | 0.070 | |
| Data are presented as mean ± SD or frequency (%), BMI: Body mass index, ASA: American society of anesthesiologists. | |||||
| Table 2: Comparative sensory and motor block characteristics and the rescue analgesia in the studied groups | |||||
| Time measurements | Group A (n = 30) |
Group B (n = 30) |
Group C (n = 30) |
P | Post hoc |
| Onset time of sensory block (min) | 4.9 ± 2.14 | 4.4 ± 1.61 | 2.9 ± 1.36 | < 0.001* | P1 = 0.461 P2 < 0.001* P3 = 0.005* |
| Onset time of motor block (min) | 5.7 ± 2.08 | 5.2 ± 2.28 | 3.9 ± 1.36 | 0.015* | P1 = 0.583 P2 = 0.002* P3 = 0.031* |
| Time to attain peak sensory block level (min) | 7.7 ± 2.23 | 7.3 ± 2.57 | 7.1 ± 2.53 | 0.681 | |
| Motor block duration (min) | 327.47 ± 28.37 | 343.1 ± 31.84 | 374.6 ± 24.18 | < 0.001* | P1 = 0.088 P2 < 0.001* P3 < 0.001* |
| Sensory block duration
(min) |
352.87 ± 27.47 | 368.2 ± 32.66 | 397.8 ± 25.21 | < 0.001* | P1 = 0.102 P2 < 0.001* P3 < 0.001* |
| Time of first rescue analgesia (h) | 6.9 ± 1.01 | 8.1 ± 1.7 | 11.2 ± 2.06 | < 0.001* | P1 = 0.016*
P2 < 0.001* P3 < 0.001* |
| Total morphine consumption postoperative in 1st 24h postoperative (mg) | 7.3 ± 1.53 | 6.2 ± 1.49 | 5.2 ± 1.61 | P1 = 0.024*
P2 < 0.001* P3 = 0.046* |
|
| Data are presented as mean ± SD. * Significant P < 0.05. P1: P between Group A and Group B, P2: P between Group A and Group C, P3: P between Group B and Group C. | |||||
Statistical analysis
The data analysis was conducted using SPSS v27 (IBM, Chicago, IL, USA). To determine whether the data followed a normal distribution, the Shapiro-Wilks test and histograms were utilized. Analyses of variance (ANOVA) (F) and the post hoc test (Tukey) were utilized to examine quantitative parametric data expressed as the mean and standard deviation. The researchers utilized a combination of the Mann-Whitney U test and Kruskal-Wallis's test in order to compare the non-parametric quantitative data of the groups. The values obtained were displayed in the form of the median and interquartile range (IQR). The chi-square test was utilized to assess the qualitative variables, which were expressed as percentages and frequencies. Considered to indicate statistical significance was a two-tailed P < 0.05.
3. RESULTS
Upon assessing the eligibility of 109 participants for this trial, 11 patients failed to meet the inclusion criteria, while 8 patients declined to participate. The remaining patients were allocated into three equal categories, with 30 patients in each group, through the use of random assignment. All assigned patients were followed up on and statistically analyzed (Figure 1).
Demographic data and duration of surgery were matched among the three groups (Table 1).
There was minimal variation in the time required to attain the maximum level of sensory block among the three groups. The onset times of sensory block and motor block did not differ significantly between Group A and Group B. In contrast, both Group A and Group B demonstrated significantly longer onset times than Group C (P < 0.05). There was no significant difference observed in the duration of motor and sensory blocks between groups A and B. Nevertheless, Group C exhibited a substantially prolonged duration of blocks compared to both groups A and B (P < 0.001). Both Group C and Group B exhibited considerably longer intervals between the initial analgesic request than Group A (P < 0.05), with Group C demonstrating a significantly longer interval than Group B. In comparison to Group A, postoperative total morphine consumption was considerably lower in groups C and B; in fact, it was even lower in Group C than in Group B (P < 0.05) as shown in Table 2.
| Table 3: NRS score of the studied patients | |||||
| Time | Group A (n = 30) |
Group B (n = 30) |
Group C (n = 30) |
P | Post hoc |
| At PACU | 0 (0−0) | 0 (0−0) | 0 (0−0) | ---- | |
| 2h | 0 (0−1) | 0 (0−0.75) | 0 (0−1) | 0.855 | |
| 4h | 1 (0−1) | 1 (0−1) | 0.5 (0−1) | 0.391 | |
| 6h | 4 (2−5.75) | 2.5 (2−4) | 1 (1−2) | < 0.001* | P1 = 0.016*
P2 < 0.001* P3 = 0.021* |
| 8h | 3 (3−5.75) | 2 (1−4) | 1 (1−2) | < 0.001* | P1 = 0.031*
P2 < 0.001* P3 = 0.027* |
| 12h | 5 (3−6) | 3 (2−5) | 2 (1−4) | < 0.001* | P1 = 0.036*
P2 < 0.001* P3 = 0.042* |
| 18h | 4 (3−5.75) | 5 (3−5) | 4 (3−4) | 0.054 | |
| 24h | 4 (3.25−5) | 4.5 (4−5) | 4 (3.25−5) | 0.439 | |
| Data are presented as median (IQR). * Significant p value < 0.05. P1: P between Group A and Group B, P2: P between Group A and Group C, P3: P between Group B and Group C. NRS: Numerical rating scale. PACU: Post-Anesthesia Care Unit. | |||||

No statistically significant variations were detected in the intraoperative HR and MAP measurements among the three groups. Regarding postoperative HR and MAP measurements at the PACU at 2 h, 4 h, 18 h, and 24 h, no significant differences were observed between the three groups. However, at 6 h, 8 h, and 12 h, these parameters were significantly lower in Group C and Group B compared to Group A and Group C (P < 0.05) (P < 0.05) (Figure 2).
At PACU, after 2, 4, 18, and 24 h, no statistically significant differences were observed in the NRS scores between the three groups. However, at 6, 8, and 12 h, the scores were significantly lower in Groups C and B compared to Group A; and in Group C than in Group B (P < 0.05) (Table 3).
Regarding complications, e.g., hypotension, bradycardia, and urinary retention, there were no statistically significant variations observed across the three cohorts. Every patient had an ineffective obstruction and a deficiency of LAST (Table 4).
4. DISCUSSION
SA is the preferred option for TURP on account of its distinctive advantages. Self-administration (SA) during surgery not only facilitates unrestricted respiration but also provides relief from postoperative pain, reduces blood loss, and obviates the necessity for airway obstruction via tracheal intubation, which may result in postoperative hemorrhage and wheezing.15 While regional anesthesia may conceivably provide benefits such as early detection of TURP syndrome in conscious patients, it is not without its limitations, including short duration of anesthesia, patient distress, and discomfort.9
Rather than technical issues, limitations of regional anesthesia, including SA, are frequently ascribed to inadequate sedation.16 In addition to alleviating patient anxiety and tension, effective sedatives in SA increases patient and surgeon satisfaction. Nevertheless, overemphasis on sedation may obscure the symptoms of TURP syndrome and induce delirium subsequent to surgical procedures, especially among the elderly.9patient and surgeon satisfaction. Nevertheless, overemphasis on sedation may obscure the symptoms of TURP syndrome and induce delirium subsequent to surgical procedures, especially among the elderly.9
| Table 4: Complications of the studied groups | ||||
| Complication | Group A (n = 30) |
Group B (n = 30) |
Group C (n = 30) |
P |
| Hypotension | 6 (20%) | 8 (26.67%) | 10 (33.33%) | 0.506 |
| Bradycardia | 3 (10%) | 4 (13.33%) | 6 (20%) | 0.533 |
| Failed blockade | 0 (0%) | 0 (0%) | 0 (0%) | --- |
| Urinary retention | 3 (10%) | 5 (16.67%) | 8 (26.67%) | 0.236 |
| LAST | 0 (0%) | 0 (0%) | 0 (0%) | --- |
| Data are presented as frequency (%), LAST: Local anesthetic systemic toxicity. | ||||
As a sedative with analgesic properties, dexmedetomidine is an advantageous adjunct to SA for TURP owing to its capacity to compensate for insufficient block height and induce minimal respiratory depression.6
Our research investigated the efficacy of three distinct ITD concentrations (6 µg, 8 µg, and 10 µg) in conjunction with low-dose hyperbaric bupivacaine 0.5% in patients presenting with TURP. Higher concentrations of ITD were associated with a significant and consistent increase in the duration of sensory and motor blockade, in addition to analgesic effects, according to the results of our study. Significantly, these outcomes were attained while maintaining similar profiles of adverse effects and hemodynamic stability. In particular, an increase in the dosage of dexmedetomidine from 6 µg to 10 µg resulted in a commensurate prolongation of both sensory and motor block, in addition to analgesic effects.
Extending the duration of analgesic administration offers a dual benefit: it mitigates the adverse consequences associated with postoperative discomfort, such as impaired immune function, prolonged hospitalization, delayed wound healing, the potential for neurosensitization, and the development of chronic pain. Furthermore, it promotes the progression of motor obstruction, which may reduce mobility and the probability of developing complications such as deep venous thrombosis, pulmonary embolism, and others that are closely related.17
Gupta et al. investigated the dose-response correlation between ITD and SA characteristics.13 Their findings showed that adding 10 µg of ITD, the application of 0.5% hyperbaric bupivacaine, as opposed to 2.5 µg or 5 µg, significantly accelerated the onset of sensory and motor blockade. Furthermore, it resulted in an extended duration of sensory and motor impairment, as well as analgesic properties, while retaining a comparable profile of adverse effects.
Other researchers conducted separate studies comparing different doses of intrathecal dexmedetomidine (ITD), specifically 2 µg vs. 4 µg, 5 µg vs. 10 µg, and 10 µg vs. 15 µg, respectively.18,19,20 In each study, they observed a rise in the sensory block, motor block duration, and analgesic effect that is also dose dependent.
ITD exerts its pain-relieving action through a dual mechanism. Firstly, it inhibits neurotransmitter release by targeting presynaptic α2A receptors. Secondly, it hyperpolarizes the postsynaptic neurons.21 The motor block prolongation could stem from The inhibiting impact of α2 agonists on the dorsal horn motor neurons of the spinal cord.22
It has been suggested that sensitivity to dexmedetomidine may vary depending on the type of nerve fiber, considering that the ED50 for maximal inhibition is 2.5 µg for sensory C fibers and above 10 µg for Aβ motor fibers.23 This led us to utilize a dosage in this range between 6 µg and 10 µg for this trial. While Eid et al. employed an increased dosage of 15 µg of ITD, we chose to exclude it from our dose-response trial design due to the significant increase in sedation scores reported in their study.20 Additionally, our study's relatively short mean length of surgical procedures contributed to this decision. ITD has demonstrated efficacy in managing nociceptive, visceral, and neuropathic pain. Its neurological safety has been established through a follow-up period of up to ten years post-anesthesia.18, 24, 25
In our study, the mean ± SD onset of sensory block time was similar among Groups A and B; however, it was earlier significantly in Group C (4.9 ± 2.14 vs 4.4 ± 1.61 vs 2.9 ± 1.36; P < 0.001 in Groups A, B and C respectively).
These results align with those reported by Halder et al.19, who employed a comparable definition for the onset time of sensory block and observed a significantly earlier onset with 10 µg than 5 µg of ITD. Similarly, Yektas et al. found a substantial rise in the number of sensory segments blocked that depends on dosage when comparing 2 µg and 4 µg of ITD.18 Moreover, Gupta et al. investigated the dose-response correlation between ITD and the onset of both sensory and motor block.13
The use of ITD in conjunction with hyperbaric bupivacaine SA has been associated with a notable benefit which is a decrease in the need for postoperative analgesia.8, 9, 26 We also noted a substantial dose-dependent reduction (P = 0.001) in the 24-hour morphine need as the doses of ITD increased.
The primary and clinically notable adverse effect linked with α2 agonists is hemodynamic disturbance, specifically manifesting as hypotension and bradycardia.27 Most studies did not report any significant rise in the occurrence of hemodynamic adverse events accompanied by the utilization of ITD, regardless of the dosage administered.19, 28, 29
We noted a dose-dependent elevation in hypotension incidence (20% with 6µg, 26.67% with 8µg, and 33.33% with 10µg as well as bradycardia (10%), (13.33%), and (20%)) across groups A, B, and C, respectively, Nevertheless, this rise failed to reach statistical significance.
The potential for further sympatholysis induced by dexmedetomidine may have been constrained in our trial due to the substantial sympatholysis induced by the higher dose and volume of bupivacaine utilized. Our observations are consistent with comparable dose-independent hemodynamic findings reported by other researchers.19, 20 Additionally, side effects such as urinary retention were comparable across all groups. Similar dose-independent side effects were also noted by Yektas et al. and Halder et al.18, 19
5. LIMITATIONS
Our study is limited by short follow-up time and the absence of a control group, thus future studies with longer monitoring duration in the presence of a control group are needed to generalize our findings.
6. CONCLUSIONS
In TURP, 10 µg of ITD in conjunction with hyperbaric bupivacaine 0.5% considerably extends analgesia duration, sensory block, and motor blockade and is accompanied by a reduction in the need for analgesics used postoperatively, while the incidence of adverse effects does not differ significantly when compared to lesser dosages.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Conflict of interest
The authors declare no conflict of interest.
9. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
10. Authors’ contribution
RMM: developed the original idea and the protocol, abstracted and analyzed data, wrote the manuscript, and is a guarantor.
MA: contributed to the development of the protocol, abstracted data.
AGA: prepared the manuscript.
11. REFERENCES
- Nugroho A, Oktavianto RR. Management of local anesthetics systemic toxicity in a 66-year-old male undergoing nephrostomy percutaneous. Anesth Pain Med. 2022;3:61-3. DOI: 21776/ub.jap.2022.003.03.06
- Khan S, Wolin K, Pakpahan R, Grubb R, Colditz G, Ragard L, et al. Body size throughout the life-course and incident benign prostatic hyperplasia-related outcomes and nocturia. BMC Urol. 2021;21:47. [PubMed] DOI: 1186/s12894-021-00816-5
- Wang YH, Wang DR, Liu JY, Pan J. Local anesthesia in oral and maxillofacial surgery: A review of current opinion. J Dent Sci. 2021;16:1055-65. [PubMed] DOI: 1016/j.jds.2020.12.003
- Khezri MB, Rezaei M, Delkhosh Reihany M, Haji Seid Javadi E. Comparison of postoperative analgesic effect of intrathecal clonidine and fentanyl added to bupivacaine in patients undergoing cesarean section: a prospective randomized double-blind study. Pain Res Treat. 2014;2014:513628. [PubMed] DOI: 1155/2014/513628
- Joshi SA, Khadke VV, Subhedar RD, Patil AW, Motghare VM. Comparative evaluation of intrathecal midazolam and low dose clonidine: efficacy, safety and duration of analgesia. A randomized, double blind, prospective clinical trial. Indian J Pharmacol. 2012;44:357. [PubMed] DOI: 4103/0253-7613.96321
- Kohaf NA, Harby SA, Abd-Ellatief AF, Elsaid MA, Abdelmottaleb NA, Abd Elsalam TF. Premedication with intranasal versus intravenous dexmedetomidine for hypotensive anesthesia during functional endoscopic sinus surgery in adults: A randomized triple-blind trial. Heliyon. 2024;10:e25175. [PubMed] DOI: 1016/j.heliyon.2024.e25175
- Manoharan MM, Paneer M, Elavarasan K, Punniyakoti KK. Dexmedetomidine versus clonidine as additives for spinal anesthesia: A comparative study. Anesth Pain Med. 2023;13. [PubMed] DOI: 5812/aapm-138274
- Kim JE, Kim NY, Lee HS, Kil HK. Effects of intrathecal dexmedetomidine on low-dose bupivacaine spinal anesthesia in elderly patients undergoing transurethral prostatectomy. Biol Pharm Bull. 2013;36:959-65. [PubMed] DOI: 1248/bpb.b12-01067
- Bhiwal AK, Sharma K, Rathore VS, Patel CMK, Chhabra A, Jaitawat SS. Comparison of two different doses of dexmedetomidine (0.25 mcg/kg and 0.5 mcg/kg) in prolonging duration of spinal anaesthesia and postoperative analgesia in patients undergoing transurethral resection of prostate: a prospective randomized double blinded study. Int J Res Med Sci. 2021;9:1569-76. DOI: 18203/2320-6012.ijrms20211930
- Mohamed T, Susheela I, Balakrishnan BP, Kaniyil S. Dexmedetomidine as adjuvant to lower doses of intrathecal bupivacaine for lower limb orthopedic surgeries. Anesth Essays Res. 2017;11:681-5. [PubMed] DOI: 4103/aer.AER_243_16
- Naaz S, Bandey J, Ozair E, Asghar A. Optimal dose of intrathecal dexmedetomidine in lower abdominal surgeries in average Indian adult. J Clin Diagn Res. 2016;10:Uc09-13. [PubMed] DOI: 7860/JCDR/2016/18008.7611
- Singh AP, Chawla S, Bajwa SJS, Khanna M. Efficacy and safety of dexmedetomidine as an intrathecal agent: a dose finding clinical study. Anaesth, Pain & Intensive Care 2017;21(1):13-18. [FreeFullText]
- Gupta M, Gupta P, Singh DK. Effect of 3 different doses of intrathecal dexmedetomidine (2.5µg, 5µg, and 10 µg) on subarachnoid block characteristics: A prospective randomized double blind dose-response trial. Pain Physician. 2016;19:E411-20. [PubMed]
- Bromage PR. A comparison of the hydrochloride and carbon dioxide salts of lidocaine and prilocaine in epidural analgesia. Acta Anaesthesiol Scand Suppl. 1965;16:55-69. [PubMed] DOI: 1111/j.1399-6576.1965.tb00523.x
- Humphries K, Page T, Donaldson T, Blaney S. Inadvertent perioperative hypothermia prevention strategies for urology surgical patients who received a blood transfusion: A retrospective analysis. J Perioper Pract. 2024;34(7-8):212-218. [PubMed] DOI: 1177/17504589231215940
- Parikh KS, Seetharamaiah S. Approach to failed spinal anaesthesia for caesarean section. Indian J Anaesth. 2018;62:691-7. [PubMed] DOI: 4103/ija.IJA_457_18
- Pirie K, Traer E, Finniss D, Myles PS, Riedel B. Current approaches to acute postoperative pain management after major abdominal surgery: a narrative review and future directions. Br J Anaesth. 2022;129:378-93. [PubMed] DOI: 1016/j.bja.2022.05.029
- Yektaş A, Belli E. The effects of 2 µg and 4 µg doses of dexmedetomidine in combination with intrathecal hyperbaric bupivacaine on spinal anesthesia and its postoperative analgesic characteristics. Pain Res Manag. 2014;19:75-81. [PubMed] DOI: 1155/2014/956825
- Halder S, Das A, Mandal D, Chandra M, Ray S, Biswas MR, et al. Effect of different doses of dexmedetomidine as adjuvant in bupivacaine-induced subarachnoid block for traumatized lower limb orthopedic surgery: a prospective, double-blinded and randomized controlled study. J Clin Diagn Res. 2014;8:Gc01-6. [PubMed] DOI: 7860/JCDR/2014/9670.5118
- Eid H, Shafie MA, Youssef H. Dose-related prolongation of hyperbaric bupivacaine spinal anesthesia by dexmedetomidine. Ain Shams J Anesthesiol. 2011;4:83-95.
- Eisenach JC, De Kock M, Klimscha W. alpha(2)-adrenergic agonists for regional anesthesia. A clinical review of clonidine (1984-1995). Anesthesiology. 1996;85:655-74. [PubMed] DOI: 1097/00000542-199609000-00026
- Abd El-Hamed Hassan A, Abd-Allah Al-Kumity A, El-Deen Mahmoud Sayed Ahmed A, Abd El-Latif Shabaiek I. Clinical comparative study between intrathecal dexmedetomidine and dexamethasone on prolonging the duration of intrathecal blockade in lower limb orthopedic surgery. Al-Azhar Med J. 2021;50:1467-78. DOI: 21608/amj.2021.158633
- Sullivan AF, Kalso EA, McQuay HJ, Dickenson AH. The antinociceptive actions of dexmedetomidine on dorsal horn neuronal responses in the anaesthetized rat. Eur J Pharmacol. 1992;215:127-33. [PubMed] DOI: 1016/0014-2999(92)90617-d
- Kimura M, Saito S, Obata H. Dexmedetomidine decreases hyperalgesia in neuropathic pain by increasing acetylcholine in the spinal cord. Neurosci Lett. 2012;529:70-4. [PubMed] DOI: 1016/j.neulet.2012.08.008
- Omara AF, Mohsen HH, Mohammed Abo Hagar A, Abdelrahman AF. Intrathecal morphine versus morphine-dexmedetomidine combination for postoperative pain control after total knee replacement: A randomized controlled trial. Local Reg Anesth. 2023;16:113-22. [PubMed] DOI: 2147/LRA.S419465
- Taha A, Shash A, Goma H, Salama A, Aly N. Intrathecal dexmedetomidine in TURP operations: A randomised controlled study. Egypt J Anaesth. 2017;33:331-7. DOI: 1016/j.egja.2017.08.006
- Demiri M, Antunes T, Fletcher D, Martinez V. Perioperative adverse events attributed to α2-adrenoceptor agonists in patients not at risk of cardiovascular events: systematic review and meta-analysis. Br J Anaesth. 2019;123:795-807. [PubMed] DOI: 1016/j.bja.2019.07.029
- Samantaray A, Hemanth N, Gunnampati K, Pasupuleti H, Mukkara M, Rao MH. Comparison of the effects of adding dexmedetomidine versus midazolam to intrathecal bupivacaine on postoperative analgesia. Pain Physician. 2015;18:71-7. [PubMed]
- Nethra SS, Sathesha M, Dixit A, Dongare PA, Harsoor SS, Devikarani D. Intrathecal dexmedetomidine as adjuvant for spinal anaesthesia for perianal ambulatory surgeries: A randomised double-blind controlled study. Indian J Anaesth. 2015;59:177-81. [PubMed] DOI: 4103/0019-5049.153040