Sadia Naim Khan 1, Nazaruddin Wan Hassan 2, S. Praveena Seevaunnatum 3, Mohd Zulfakar Mazlan 4, Muhamad Hasyizan Hassan 5
Author affiliations:
Background & objective: Intubation without muscle relaxants is indicated in certain situations. The aim of this study was to compare intubating conditions without muscle relaxants between a combination of induction using target-controlled infusion (TCI) remifentanil and propofol with a topical lignocaine spray and TCI remifentanil and propofol alone in elective surgery.
Methodology: Sixty patients, aged 18 to 65 years with ASA I and II classification, were randomized into two groups: the RPL group (n = 30), which received 10 puffs of 10% lignocaine spray over the glottic area before induction followed by TCI remifentanil 4 ng/ml and TCI propofol 4 µg/ml for induction, and the control group (n = 30), which received 10 puffs of topical spray of normal saline before a similar technique of induction. The intubating conditions were assessed using a C-Mac video laryngoscope. Hemodynamic changes and the percentage of patients who required rescue muscle relaxants were recorded.
Results: The RPL group showed a significantly higher percentage of easy laryngoscopic procedures (96.7% vs. 56.7%; P < 0.01) and open vocal cord positions (86.7% vs. 56.7%; P = 0.028) than the control group. However, there were no significant differences in overall intubating conditions, requirements for rescue muscle relaxants, or hemodynamic changes between the two groups.
Conclusion: A combination of TCI remifentanil and TCI propofol with a topical lignocaine spray facilitated better intubating conditions without muscle relaxants compared to this combination without topical lignocaine.
Abbreviations: Cet: effect-site concentration, Cpt: target plasma concentration, ETT: Endotracheal tube, LOC: loss of consciousness, NS: Normal saline, TCI: Target-controlled infusion,
Keywords: Muscle Relaxants; Remifentanil; Propofol; Lignocaine; Vocal Cord
Citation: Khan SN, Wan Hassan WMN, Seevaunnatum P, Mazlan MZ, Hassan MH. Comparison of intubating conditions between induction with target-controlled infusion (TCI) of remifentanil and propofol with or without a topical lignocaine spray in elective surgery for adults. Anaesth. pain intensive care 2025;29(2):163-169. DOI: 10.35975/apic.v29i2.2699
Received: August 10, 2024; Reviewed: September 26, 2024; Accepted: September 26, 2024
Intubation of endotracheal tube (ETT) after induction of general anesthesia usually requires a good airway relaxation using muscle relaxant agents such as intravenous (IV) atracurium or rocuronium. However, there are certain conditions that muscle relaxant has to be avoided and the intubation must be done without the use of muscle relaxants such as in patients who have allergic history to muscle relaxants agent, in the airway procedure that requires the assessment of vocal cord mobility, or in patients that may have prolonged effect muscle relaxant.
There were some techniques of intubation without muscle relaxant have been discussed in the literatures using combination of various induction agents and opioid. The combination of opioids with propofol, particularly remifentanil, has shown promise in significantly improving the quality of tracheal intubation without muscle relaxant use.1–8 El-Tahan et al.9 hypothesized that the use of anesthesia without muscle-relaxants during thoracotomy can be associated with comparable surgical conditions with the standard use of muscle relaxants. Their study showed that anesthesia without muscle-relaxants offered acceptable laryngoscopy and intubating conditions when using propofol (1.5–3.0 mg/kg) and target-controlled infusion (TCI) remifentanil with a target effect-site concentration (Ce) of 4–6 ng/mL in good-to-excellent quality intubating conditions (93.9% anesthesia without muscle-relaxants vs. 100% cisatracurium; P > 0.09).9 Ide et al.,10 as proponents of the method of ETI without muscle relaxants using propofol and remifentanil, included 44 patients divided in two groups—one receiving intravenous (IV) 0.6 mg/kg rocuronium and a control group receiving IV normal saline (NS). Even though there were differences in the subcategories of intubating conditions, the overall conditions were comparable, and neither group reported postoperative hoarseness or pain.10
The mechanism of action of lignocaine in blunting the pressor response differs according to the method of administration. Lidocaine typically acts by stabilizing the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses, resulting in the rapid onset of a localized anesthetic effect. Local administration, such as gargles and sprays, may be effective for cough suppression due to the local anesthetic action at the base of the tongue and pharyngeal walls preventing receptor stimulation and improving intubating conditions.11 The inhibition of airway tactile stimulation could be mainly due to the direct blockade of the mechanoreceptors of the airways and partly due to its systemic effect.12
To the best of our knowledge, no similar study has been conducted to compare the effects of topical lignocaine in combination with TCI remifentanil and propofol on induction conditions for ETI without muscle relaxants. The aim of this study is to evaluate the effect of a combination of 10% topical lignocaine spray, TCI remifentanil, and TCI propofol on intubating conditions without the use of muscle relaxants.
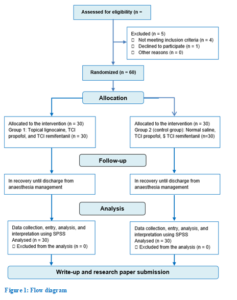
This double-blinded randomized controlled trial was conducted after receiving approval from the university’s ethics committee (study protocol code: USM/JEPeM/19120842) and written consent from the patients. Sixty patients aged between 18 and 65 years with American Society of Anesthesiologists (ASA) physical status class I or II who were scheduled for elective surgery under GA were divided into two groups through computer-generated randomization. The RPL group (n = 30) received 10% topical lignocaine spray, TCI propofol, and TCI remifentanil and the control group (n = 30) received NS spray, TCI propofol, and TCI remifentanil.
Patients with a body mass index (BMI) greater than 30 kg/m2, underlying active or recent respiratory tract infections, and/or reactive airway diseases and/or who anticipated or had a history of difficulty in intubation were excluded, as were those with a history of allergies to the study drugs; comorbidities such as poor cardiovascular reserve, neurological or psychiatric disorders, and pregnancy.
The lignocaine spray and placebo (NS) were prepared in spray bottles that were identical in appearance. These bottles were placed in sealed bags/envelopes, with their labels covered with opaque paper. They were consecutively numbered for each patient according to the randomization schedule. The allocation sequence was concealed from the patients and the anesthetist MO in-charge of the case. Corresponding envelopes were opened by the principal investigator only after the participants had completed all baseline assessments and in time for the intervention to be allocated. The interventional assignments were ascertained by the principal investigator on opening the envelopes.
All patients, anesthetist MOs in charge, and operating theatre (OT) staff were blind to the interventions that had been assigned. The outcome assessors and data analysts were blind to the allocation. To ensure the safety of the participants, the principal investigator, upon opening the envelopes was made aware of the interventions. The administration of the spray (topical 10% lignocaine or normal saline) was performed by the principal investigator. ETI and assessments of the intubating conditions were performed by the anesthetist MOs in charge of the patients.
As premedication, all patients were prescribed 7.5 mg tab. midazolam orally at night prior to their operation. On the day of the operation, questionnaires and instructions were provided to the anesthetist in charge to input the required data. Upon arrival in the operating room, each patient was monitored with an electrocardiogram (ECG), a pulse oximeter (SpO2), and a noninvasive blood pressure (NIBP) test. The baseline values of HR, systolic blood pressure (SBP), diastolic blood pressure (DBP), and MAP were taken at five intervals: at baseline before spraying with lignocaine/NS (TBL), at the time of spraying with lignocaine/NS (TL), at induction (TI), and at 0 (T0), 3 (T3), and 5 (T5) minutes post intubation. IV glycopyrrolate (0.2 mg) was administered 3 min prior to the lignocaine/NS spray.
Either lignocaine 10% oral spray or NS was administered by the principal investigator 3 min prior to induction. Each patient was asked to fully open their mouth while sitting, and a total of 10 puffs (0.1 ml per spray) of either 10% lignocaine spray or NS (2 puffs to the soft palate, 3 puffs to the posterior oropharyngeal wall, 2 puffs to the palatopharyngeal arch, and 3 puffs to the posterior third of tongue, with each puff delivering 10 mg, resulting in a total dose of 100 mg and not exceeding the toxic dose of 3–4 mg/kg) were administered using a disposable spray cannula into the mouth without the use of a laryngoscope. The patient was then pre-oxygenated until the end-tidal oxygen percentage was above 90%, after which GA was induced. The Marsh model and Minto13,14 pharmacokinetic sets were used for propofol and remifentanil, respectively. At time TI, TCI remifentanil was started at a target effect-site concentration (Cet) of 2.0 ng/ml. Once the target concentration of remifentanil had been reached, propofol TCI was started at a target plasma concentration (Cpt) of 4 μg/ml. If loss of consciousness (LOC) was not achieved within 1 min, the Cpt of propofol was stepped up by 0.5 μg/ml every 30 sec until LOC. The Ce displayed on the TCI pump at LOC was noted, and the Cpt was maintained at or above this value.
After LOC had been established by the loss of eyelash reflex and the loss of response to commands, ventilation was initiated via facemask. Muscle relaxants were not given. TCI remifentanil was then titrated upward to 4 ng/ml if BP was above 120/80 mmHg and/or not less than 15% of the baseline value. Using a C-Mac video-laryngoscope, the intubating conditions were observed and noted. Using appropriately sized endotracheal cuffed tubes, intubation was performed by the MO anesthesiologist in charge. IV rocuronium 1 mg/kg was given as a rescue strategy if poor intubating conditions were noted or if there was failure to intubate at the second attempt (under acceptable intubating conditions). These patients were not withdrawn from the study. Following successful ETI, the patients were ventilated with a mixture of oxygen and air. No topical anesthesia of the larynx was repeated. It was ensured that the TCI remifentanil was at a Ce of at least 3 ng/ml at the start of surgery if BP was above 100/60 mmHg; otherwise, it was maintained at 2 ng/ml.
During surgery, TCI propofol was titrated within the range of Cpt 3–6 μg/ml and TCI remifentanil within the range of Cet 1–8 ng/ml, according to the clinical responses of the patient and the co-administration of other agents. Since the bispectral index was not used, propofol target concentrations were gradually decreased to and then maintained at 3 μg/ml or at the level at which LOC was achieved, as per clinical judgment. Monitoring included NIBP, ECG, SpO2, and capnography. If MAP or HR fell more than 15% in comparison to the corresponding pre-induction values, IV fluid, ephedrine 3–6 mg, or atropine 0.5 mg was administered appropriately to reach at least 70% of the pre-induction value.
IV morphine and other appropriate analgesics were administered toward the end of the surgery. TCI propofol was maintained during skin closure and was stopped once the final sutures or dressings were applied. The remifentanil infusion was stopped after suturing, unless extensive dressings were involved. Extubation was performed in a standard manner when patients could open their eyes, squeeze the hand, and sustain lifting of the head on command. Reversal (0.05 mg/kg IV neostigmine and 0.02 mg/kg IV atropine) was administered if a rescue muscle relaxant had been given during intubation or maintenance. Incidence of sore throat, PONV, and pain score using a visual analogue scale (VAS; 0 for no pain, and 10 for the worst possible pain) were recorded at 15 min after arrival in the post-anesthesia care unit/recovery area. Postoperative pain was treated with analgesics appropriate to the type of surgery.
Successful intubation was defined as ETI performed at the first or second attempt with acceptable (excellent or good) intubating conditions. Intubating conditions were assessed according to a scoring system described by the guidelines for Good Clinical Research Practice.15 The variables assessed were ease of laryngoscopy (easy, fair, or difficult), vocal cords’ position (abducted, intermediate/moving, or closed), and reaction to the endotracheal tube insertion and cuff inflation (none, slight, or vigorous/sustained for longer than 5 sec). Intubating conditions were excellent if all variables were excellent, good if one or all were good with/without any being excellent, or poor if there was the presence of any quality listed as “poor” (i.e., difficult laryngoscopy, closed vocal cords, and/or vigorous/sustained reaction to tube insertion or cuff inflation). Acceptable intubation was defined as excellent or good intubating conditions. A summarized evaluation of intubating conditions is provided in Box 1.
The primary outcome was to compare (1) the intubating conditions (acceptable: excellent or good; unacceptable: poor) provided by topical lignocaine with TCI remifentanil and propofol with the control (TCI remifentanil and propofol), (2) the proportion of patients who required rescue muscle relaxants for intubation between both groups, and (3) the effects of topical lignocaine on hemodynamics (HR and MAP) during the induction of anesthesia.
We used a t-test function to calculate the sample size, with the significance level set to 0.05, a Z value of 1.96, and a power of study of 80%, using Power and Sample Size software, version 3.0.10. Based on Jin-Soo Kim et al., we concluded that the overall intubating conditions were clinically acceptable (excellent or good) in 13 out of 25 patients (52%) in the control group and in 22 out of 25 patients (88%) in the lidocaine group and that there was a significant difference between the two groups for acceptable intubating conditions (P = 0.005).16 The calculated sample size was 25 per group. After taking into consideration the 20% dropout rate and missing data, the final sample size was 30 patients per group.
Categorical data are presented as frequency and percentage (%), while numerical data are presented as mean and standard deviation (SD). The first two objectives were measured using Fisher’s exact test. A two-way repeated measure ANOVA (mixed design) was conducted to determine our third objective: whether there was a significant difference between NS and lignocaine on MAP and HR measured at six different times. The model assumptions of normality, homogeneity of covariance, and compound symmetry were checked. The significance level was set at 0.05. The statistical analysis was analyzed using SPSS software version 26.
A total of 60 patients were enrolled in this study (30 patients per group). None of the patients were excluded from the analysis. Overall, there were no significant differences in the patient’s characteristics between the two groups, as shown in Table 1.
As shown in Table 2, the RPL group showed a significantly higher percentage of easy laryngoscopy (jaw relaxed, no resistance to blade insertion) (96.7% vs. 56.7%; P < 0.01) and open vocal cord position (86.7% vs. 56.7%; P = 0.028) than the control group. There were no other significant differences in reaction to tube insertion, overall intubating conditions, or the requirement for rescue muscle relaxant.
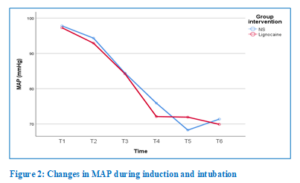
A two-way repeated measure ANOVA (mixed design) of MAP showed a significant decrease in MAP over time (Huynh–Feldt F [4.18, 242.69] = 81.0), but there was no significant interaction between time and group (Huynh–Feldt F [4.18, 242.69] = 0.85). Post hoc Bonferroni adjustment for 95% CI showed significant differences between the control group and the RPL group. Mauchly’s test of sphericity indicated that the assumption of sphericity had been violated, (χ2[14] = 41.39, df = 14, P < 0.001). Hence, the Huynh–Feldt correction was applied. There were significant changes in MAP over time (Huynh–Feldt F [4.18, 242.69] = 81.0), where the post hoc paired t-test with Bonferroni adjustment showed significant differences in the control group between baseline and induction, baseline and at 0 min, baseline and at 3 min, baseline and at 5 min, spray and induction, spray and at 0 min, spray and at 3 min, spray and at 5 min, induction and at 0 min, induction and at 3 min, and induction and at 5 min. Meanwhile, adjustment for RPL group also showed significant differences between baseline and induction, baseline and at 0 min, baseline and at 3 min, baseline and at 5 min, spray and induction, spray and at 0 min, spray and at 3 min, spray and at 5 min, induction and at 0 min, induction and at 3 min, and induction and at 5 min. The overall mean difference between the two drug interventions with regard to MAP was not significant (P > 0.05) (Table 3). Figure 2 illustrates the changes in MAP during the induction of anesthesia.
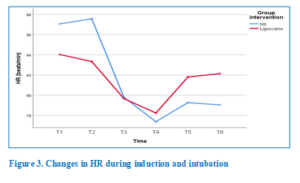
To analyze the effect of the interventions on HR, a two-way repeated measure ANOVA (mixed design) was applied. There were significant changes in HR over time (Huynh–Feldt F [4.00, 231.7] = 5.22), but there was no significant interaction between time and group (Huynh-Feldt F [4.00, 231.7] = 1.31). Post hoc Bonferroni adjustment for 95% CI showed significant differences over time in both the control group and the RPL group (Fig. 3). The overall mean difference between the two drug interventions with regard to HR was not significant (P > 0.05) (Table 4).
We postulated that topical lignocaine spray prior to induction improves intubating conditions without muscle relaxant and hemodynamic stability; hence, it may potentially decrease the requirement for remifentanil and therefore, providing insight into the future application of this technique in more sensitive patient groups, such as the elderly and children. Our results showed that the RPL group had a significantly higher percentage of easy laryngoscopic procedures (96.7% vs. 56.7%; P < 0.01) and open vocal cord positions (86.7% vs. 56.7%; P = 0.028) than the control group. Easy laryngoscopy and an open vocal cord position are considered “excellent conditions” individually.15 However, there were no significant differences in the overall intubating conditions, the requirement for rescue muscle relaxants, or the hemodynamic changes between the two groups. A previous study by Kim et al.16 showed that when topical lignocaine 120 mg was instilled into the larynx and trachea, it provided acceptable intubating conditions without hemodynamic instability in 88% of patients (compared to 52% in the control group [P = 0.005]) during TCI remifentanil and propofol without muscle relaxants. Our study showed that an oropharyngeal lignocaine spray of 100 mg provided acceptable conditions in 27 individuals (90%) compared to 25 patients (83.3%) in the control group. Excellent intubating conditions were seen in 10 patients (33.3%) in the RPL group versus four patients (13.3%) in the control group. IV rocuronium (50 mg) was administered in three cases (10%) in the RPL group and in five cases (16.7%) in the control group due to unacceptable conditions. However, the data analysis showed no significant association between the two groups in terms of intubating conditions (P = 0.706) and the need for rescue muscle relaxant in the RPL group versus the control group (P = 0.706). The major cause of unacceptable conditions was reaction to tube insertion (i.e., more than two contractions, coughing, and/or movement longer than 5 s). This was seen in four cases in the control group and in three cases in the RPL group. This is consistent with previous studies in which opioid-induced cough or cough provoked by ETT passing through the vocal cords was responsible for the majority of unacceptable conditions during ETI.17,18 Sun et al.19 conducted two studies with 1108 patients to compare incremental doses of IV lignocaine and observe the effect of this on coughing. The cough reflex was completely suppressed by plasma concentrations of lignocaine of more than 3 μg/ml.
Remifentanil has gained popularity due to the duration of apnea comparable to suxamethonium for potential or known difficult airways, especially when muscle relaxants may be undesirable. There are several case reports of induction and intubation using remifentanil and propofol for cases of malignant hyperpyrexia or lung diseases for which the avoidance of muscle relaxants was essential or desirable.20 The trials of Mencke et al.21 and Bouvet et al.22 showed no significant association between the avoidance of muscle relaxants and upper airway injury (RR 1.12, 95% CI 0.61–2.08; P = 0.14) when remifentanil was used as part of the protocol.21,22 Total intravenous anesthesia with propofol and remifentanil is considered superior to sevoflurane in terms of induction, intubation, maintenance, stress hormone response to surgical stimuli, and awakening,23 and TCI allows for the easy adjustment of anesthetic depth.
Topical lignocaine was used in the present study, and several studies have compared the two routes. However, Kumar et al.24 found no advantage of the use of nebulized lignocaine in attenuating the hemodynamic response to laryngoscopy and intubation. The use of opioids such as IV fentanyl before induction was effective in attenuating the hemodynamic response.24 Other evidence has shown contrary results, with topical lignocaine spray being effective at attenuating the pressor response when ETI was performed after 2 min of tracheal lignocaine spray. 12 Jokar et al.25 observed that MAP was not statistically lower in the topical lignocaine group than in the IV lignocaine group. However, the use of lignocaine, either topical or IV, resulted in a better hemodynamic profile compared to the control group. They observed a significant difference between the two lignocaine groups in terms of a decrease in HR; that is, a lower HR was noted during intubation in the topical group.25
There are several limitations to our study. First, the patients’ characteristics were ASA statuses of I and II and ages between 18 and 65 years old. Therefore, the findings may only be applicable to such patient populations and may have adverse effects on patients belonging to extremes of age or an ASA status higher than II, those having serious cardiovascular or respiratory diseases, and those in a state of shock. Further studies may show the beneficial role of topical lignocaine with TCI induction without muscle relaxants in such populations, since the dosage requirements might possibly be lower. Second, in the present study, the topical spray was instilled without performing laryngoscopy and was restricted to the oropharynx, in contrast to earlier studies in which topical lignocaine was instilled directly over vocal cords under direct visualization using direct laryngoscopy or nebulization and/or via the transtracheal route, possibly providing better LA spread.
Topical lignocaine spray significantly improved intubating conditions (easy laryngoscopy and open vocal cord position) during ETI without muscle relaxants using TCI remifentanil and propofol. Intubating conditions were acceptable (good to excellent) when TCI remifentanil and propofol were used for induction; therefore, their use is recommended when the avoidance of muscle relaxants is required. Topical lignocaine, such as oral spray, tracheal spray, or nebulization, is a useful adjunct for improving intubating conditions while maintaining stable hemodynamics.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Acknowledgements
We would like to thank the Malaysian Society of Anesthesiologists for supporting this study through the K Inbasegaran research grant and the Research Creativity and Management Office of University Sains Malaysia for including this grant as an external grant.
9. Conflict of interest
The authors declare no conflict of interest
10. Authors’ contribution
SNK: conception, design, execution, analysis, interpretation of the data, drafting and final approval of manuscript WMNWH: conception, design, execution, analysis, interpretation of the data, drafting, critical revision and final approval of manuscript
SPS, MZM, MHH: conception, design, critical revision and final approval of manuscript
Author affiliations:
- Sadia Naim Khan, MMed, Anesthesiologist, Department of Anesthesiology and Intensive Care, School of Medical Sciences & Hospital Universiti Sains Malaysia, Universiti Sains Malaysia, 16150 Kubang Kerian, Kota Bharu, Kelantan, Malaysia; Email: dr.sadiakhan@gmail.com,
- Wan Mohd Nazaruddin Wan Hassan, MMed, Associate Professor, Department of Anesthesiology and Intensive Care, School of Medical Sciences & Hospital Universiti Sains Malaysia, Universiti Sains Malaysia, 16150 Kubang Kerian, Kota Bharu, Kelantan, Malaysia; Email: drnaza_anaest@usm.my
- S. Praveena Seevaunnatum, Senior Lecturer, Department of Anesthesiology and Intensive Care, School of Medical Sciences & Hospital Universiti Sains Malaysia, Universiti Sains Malaysia, 16150 Kubang Kerian, Kota Bharu, Kelantan, Malaysia; Email: praveenaseeva@usm.my,
- Mohd Zulfakar Mazlan, MMed, Associate Professor, Department of Anesthesiology and Intensive Care, School of Medical Sciences & Hospital Universiti Sains Malaysia, Universiti Sains Malaysia, 16150 Kubang Kerian, Kota Bharu, Kelantan, Malaysia; Email: zulfakar@usm.my,
- Muhamad Hasyizan Hassan, MMed, Senior Lecturer, Department of Anesthesiology and Intensive Care, School of Medical Sciences & Hospital Universiti Sains Malaysia, Universiti Sains Malaysia, 16150 Kubang Kerian, Kota Bharu, Kelantan, Malaysia; Email: hasyizan@usm.my
ABSTRACT
Background & objective: Intubation without muscle relaxants is indicated in certain situations. The aim of this study was to compare intubating conditions without muscle relaxants between a combination of induction using target-controlled infusion (TCI) remifentanil and propofol with a topical lignocaine spray and TCI remifentanil and propofol alone in elective surgery.
Methodology: Sixty patients, aged 18 to 65 years with ASA I and II classification, were randomized into two groups: the RPL group (n = 30), which received 10 puffs of 10% lignocaine spray over the glottic area before induction followed by TCI remifentanil 4 ng/ml and TCI propofol 4 µg/ml for induction, and the control group (n = 30), which received 10 puffs of topical spray of normal saline before a similar technique of induction. The intubating conditions were assessed using a C-Mac video laryngoscope. Hemodynamic changes and the percentage of patients who required rescue muscle relaxants were recorded.
Results: The RPL group showed a significantly higher percentage of easy laryngoscopic procedures (96.7% vs. 56.7%; P < 0.01) and open vocal cord positions (86.7% vs. 56.7%; P = 0.028) than the control group. However, there were no significant differences in overall intubating conditions, requirements for rescue muscle relaxants, or hemodynamic changes between the two groups.
Conclusion: A combination of TCI remifentanil and TCI propofol with a topical lignocaine spray facilitated better intubating conditions without muscle relaxants compared to this combination without topical lignocaine.
Abbreviations: Cet: effect-site concentration, Cpt: target plasma concentration, ETT: Endotracheal tube, LOC: loss of consciousness, NS: Normal saline, TCI: Target-controlled infusion,
Keywords: Muscle Relaxants; Remifentanil; Propofol; Lignocaine; Vocal Cord
Citation: Khan SN, Wan Hassan WMN, Seevaunnatum P, Mazlan MZ, Hassan MH. Comparison of intubating conditions between induction with target-controlled infusion (TCI) of remifentanil and propofol with or without a topical lignocaine spray in elective surgery for adults. Anaesth. pain intensive care 2025;29(2):163-169. DOI: 10.35975/apic.v29i2.2699
Received: August 10, 2024; Reviewed: September 26, 2024; Accepted: September 26, 2024
1. INTRODUCTION
Intubation of endotracheal tube (ETT) after induction of general anesthesia usually requires a good airway relaxation using muscle relaxant agents such as intravenous (IV) atracurium or rocuronium. However, there are certain conditions that muscle relaxant has to be avoided and the intubation must be done without the use of muscle relaxants such as in patients who have allergic history to muscle relaxants agent, in the airway procedure that requires the assessment of vocal cord mobility, or in patients that may have prolonged effect muscle relaxant.
There were some techniques of intubation without muscle relaxant have been discussed in the literatures using combination of various induction agents and opioid. The combination of opioids with propofol, particularly remifentanil, has shown promise in significantly improving the quality of tracheal intubation without muscle relaxant use.1–8 El-Tahan et al.9 hypothesized that the use of anesthesia without muscle-relaxants during thoracotomy can be associated with comparable surgical conditions with the standard use of muscle relaxants. Their study showed that anesthesia without muscle-relaxants offered acceptable laryngoscopy and intubating conditions when using propofol (1.5–3.0 mg/kg) and target-controlled infusion (TCI) remifentanil with a target effect-site concentration (Ce) of 4–6 ng/mL in good-to-excellent quality intubating conditions (93.9% anesthesia without muscle-relaxants vs. 100% cisatracurium; P > 0.09).9 Ide et al.,10 as proponents of the method of ETI without muscle relaxants using propofol and remifentanil, included 44 patients divided in two groups—one receiving intravenous (IV) 0.6 mg/kg rocuronium and a control group receiving IV normal saline (NS). Even though there were differences in the subcategories of intubating conditions, the overall conditions were comparable, and neither group reported postoperative hoarseness or pain.10
The mechanism of action of lignocaine in blunting the pressor response differs according to the method of administration. Lidocaine typically acts by stabilizing the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses, resulting in the rapid onset of a localized anesthetic effect. Local administration, such as gargles and sprays, may be effective for cough suppression due to the local anesthetic action at the base of the tongue and pharyngeal walls preventing receptor stimulation and improving intubating conditions.11 The inhibition of airway tactile stimulation could be mainly due to the direct blockade of the mechanoreceptors of the airways and partly due to its systemic effect.12
To the best of our knowledge, no similar study has been conducted to compare the effects of topical lignocaine in combination with TCI remifentanil and propofol on induction conditions for ETI without muscle relaxants. The aim of this study is to evaluate the effect of a combination of 10% topical lignocaine spray, TCI remifentanil, and TCI propofol on intubating conditions without the use of muscle relaxants.
2. METHODOLOGY
This double-blinded randomized controlled trial was conducted after receiving approval from the university’s ethics committee (study protocol code: USM/JEPeM/19120842) and written consent from the patients. Sixty patients aged between 18 and 65 years with American Society of Anesthesiologists (ASA) physical status class I or II who were scheduled for elective surgery under GA were divided into two groups through computer-generated randomization. The RPL group (n = 30) received 10% topical lignocaine spray, TCI propofol, and TCI remifentanil and the control group (n = 30) received NS spray, TCI propofol, and TCI remifentanil.
Patients with a body mass index (BMI) greater than 30 kg/m2, underlying active or recent respiratory tract infections, and/or reactive airway diseases and/or who anticipated or had a history of difficulty in intubation were excluded, as were those with a history of allergies to the study drugs; comorbidities such as poor cardiovascular reserve, neurological or psychiatric disorders, and pregnancy.

The lignocaine spray and placebo (NS) were prepared in spray bottles that were identical in appearance. These bottles were placed in sealed bags/envelopes, with their labels covered with opaque paper. They were consecutively numbered for each patient according to the randomization schedule. The allocation sequence was concealed from the patients and the anesthetist MO in-charge of the case. Corresponding envelopes were opened by the principal investigator only after the participants had completed all baseline assessments and in time for the intervention to be allocated. The interventional assignments were ascertained by the principal investigator on opening the envelopes.
All patients, anesthetist MOs in charge, and operating theatre (OT) staff were blind to the interventions that had been assigned. The outcome assessors and data analysts were blind to the allocation. To ensure the safety of the participants, the principal investigator, upon opening the envelopes was made aware of the interventions. The administration of the spray (topical 10% lignocaine or normal saline) was performed by the principal investigator. ETI and assessments of the intubating conditions were performed by the anesthetist MOs in charge of the patients.
As premedication, all patients were prescribed 7.5 mg tab. midazolam orally at night prior to their operation. On the day of the operation, questionnaires and instructions were provided to the anesthetist in charge to input the required data. Upon arrival in the operating room, each patient was monitored with an electrocardiogram (ECG), a pulse oximeter (SpO2), and a noninvasive blood pressure (NIBP) test. The baseline values of HR, systolic blood pressure (SBP), diastolic blood pressure (DBP), and MAP were taken at five intervals: at baseline before spraying with lignocaine/NS (TBL), at the time of spraying with lignocaine/NS (TL), at induction (TI), and at 0 (T0), 3 (T3), and 5 (T5) minutes post intubation. IV glycopyrrolate (0.2 mg) was administered 3 min prior to the lignocaine/NS spray.
Either lignocaine 10% oral spray or NS was administered by the principal investigator 3 min prior to induction. Each patient was asked to fully open their mouth while sitting, and a total of 10 puffs (0.1 ml per spray) of either 10% lignocaine spray or NS (2 puffs to the soft palate, 3 puffs to the posterior oropharyngeal wall, 2 puffs to the palatopharyngeal arch, and 3 puffs to the posterior third of tongue, with each puff delivering 10 mg, resulting in a total dose of 100 mg and not exceeding the toxic dose of 3–4 mg/kg) were administered using a disposable spray cannula into the mouth without the use of a laryngoscope. The patient was then pre-oxygenated until the end-tidal oxygen percentage was above 90%, after which GA was induced. The Marsh model and Minto13,14 pharmacokinetic sets were used for propofol and remifentanil, respectively. At time TI, TCI remifentanil was started at a target effect-site concentration (Cet) of 2.0 ng/ml. Once the target concentration of remifentanil had been reached, propofol TCI was started at a target plasma concentration (Cpt) of 4 μg/ml. If loss of consciousness (LOC) was not achieved within 1 min, the Cpt of propofol was stepped up by 0.5 μg/ml every 30 sec until LOC. The Ce displayed on the TCI pump at LOC was noted, and the Cpt was maintained at or above this value.
| Box 1: Evaluation of intubating conditions on laryngoscopy15 | |||
| Variable assessed | Clinically acceptable | Not clinically stable | |
| Laryngoscopy# | Excellent | Good | Poor |
| Vocal cord position | Easy | Fair | Difficult |
| Reaction to insertion of the tracheal tube and cuff inflation (diaphragmatic movement/coughing) | None | Slight¥ | Vigorous/sustainedɸ |
| *Intubating conditions:
Excellent: All qualities are excellent. Good: All qualities are either excellent or good. Poor: There is the presence of a single quality listed under “poor.” #Laryngoscopy Easy: The jaw is relaxed, and there is no resistance to blade insertion. Fair: The jaw is not fully relaxed, and there is slight resistance to blade insertion. Difficult: There is poor jaw relaxation and active resistance of the patient to laryngoscopy. ¥One to two weak contractions or movement less than 2 s ɸ > 2 contractions and/or movement longer than 5 s |
|||
After LOC had been established by the loss of eyelash reflex and the loss of response to commands, ventilation was initiated via facemask. Muscle relaxants were not given. TCI remifentanil was then titrated upward to 4 ng/ml if BP was above 120/80 mmHg and/or not less than 15% of the baseline value. Using a C-Mac video-laryngoscope, the intubating conditions were observed and noted. Using appropriately sized endotracheal cuffed tubes, intubation was performed by the MO anesthesiologist in charge. IV rocuronium 1 mg/kg was given as a rescue strategy if poor intubating conditions were noted or if there was failure to intubate at the second attempt (under acceptable intubating conditions). These patients were not withdrawn from the study. Following successful ETI, the patients were ventilated with a mixture of oxygen and air. No topical anesthesia of the larynx was repeated. It was ensured that the TCI remifentanil was at a Ce of at least 3 ng/ml at the start of surgery if BP was above 100/60 mmHg; otherwise, it was maintained at 2 ng/ml.
During surgery, TCI propofol was titrated within the range of Cpt 3–6 μg/ml and TCI remifentanil within the range of Cet 1–8 ng/ml, according to the clinical responses of the patient and the co-administration of other agents. Since the bispectral index was not used, propofol target concentrations were gradually decreased to and then maintained at 3 μg/ml or at the level at which LOC was achieved, as per clinical judgment. Monitoring included NIBP, ECG, SpO2, and capnography. If MAP or HR fell more than 15% in comparison to the corresponding pre-induction values, IV fluid, ephedrine 3–6 mg, or atropine 0.5 mg was administered appropriately to reach at least 70% of the pre-induction value.
IV morphine and other appropriate analgesics were administered toward the end of the surgery. TCI propofol was maintained during skin closure and was stopped once the final sutures or dressings were applied. The remifentanil infusion was stopped after suturing, unless extensive dressings were involved. Extubation was performed in a standard manner when patients could open their eyes, squeeze the hand, and sustain lifting of the head on command. Reversal (0.05 mg/kg IV neostigmine and 0.02 mg/kg IV atropine) was administered if a rescue muscle relaxant had been given during intubation or maintenance. Incidence of sore throat, PONV, and pain score using a visual analogue scale (VAS; 0 for no pain, and 10 for the worst possible pain) were recorded at 15 min after arrival in the post-anesthesia care unit/recovery area. Postoperative pain was treated with analgesics appropriate to the type of surgery.
Successful intubation was defined as ETI performed at the first or second attempt with acceptable (excellent or good) intubating conditions. Intubating conditions were assessed according to a scoring system described by the guidelines for Good Clinical Research Practice.15 The variables assessed were ease of laryngoscopy (easy, fair, or difficult), vocal cords’ position (abducted, intermediate/moving, or closed), and reaction to the endotracheal tube insertion and cuff inflation (none, slight, or vigorous/sustained for longer than 5 sec). Intubating conditions were excellent if all variables were excellent, good if one or all were good with/without any being excellent, or poor if there was the presence of any quality listed as “poor” (i.e., difficult laryngoscopy, closed vocal cords, and/or vigorous/sustained reaction to tube insertion or cuff inflation). Acceptable intubation was defined as excellent or good intubating conditions. A summarized evaluation of intubating conditions is provided in Box 1.
The primary outcome was to compare (1) the intubating conditions (acceptable: excellent or good; unacceptable: poor) provided by topical lignocaine with TCI remifentanil and propofol with the control (TCI remifentanil and propofol), (2) the proportion of patients who required rescue muscle relaxants for intubation between both groups, and (3) the effects of topical lignocaine on hemodynamics (HR and MAP) during the induction of anesthesia.
We used a t-test function to calculate the sample size, with the significance level set to 0.05, a Z value of 1.96, and a power of study of 80%, using Power and Sample Size software, version 3.0.10. Based on Jin-Soo Kim et al., we concluded that the overall intubating conditions were clinically acceptable (excellent or good) in 13 out of 25 patients (52%) in the control group and in 22 out of 25 patients (88%) in the lidocaine group and that there was a significant difference between the two groups for acceptable intubating conditions (P = 0.005).16 The calculated sample size was 25 per group. After taking into consideration the 20% dropout rate and missing data, the final sample size was 30 patients per group.
Categorical data are presented as frequency and percentage (%), while numerical data are presented as mean and standard deviation (SD). The first two objectives were measured using Fisher’s exact test. A two-way repeated measure ANOVA (mixed design) was conducted to determine our third objective: whether there was a significant difference between NS and lignocaine on MAP and HR measured at six different times. The model assumptions of normality, homogeneity of covariance, and compound symmetry were checked. The significance level was set at 0.05. The statistical analysis was analyzed using SPSS software version 26.
3. RESULTS
A total of 60 patients were enrolled in this study (30 patients per group). None of the patients were excluded from the analysis. Overall, there were no significant differences in the patient’s characteristics between the two groups, as shown in Table 1.
As shown in Table 2, the RPL group showed a significantly higher percentage of easy laryngoscopy (jaw relaxed, no resistance to blade insertion) (96.7% vs. 56.7%; P < 0.01) and open vocal cord position (86.7% vs. 56.7%; P = 0.028) than the control group. There were no other significant differences in reaction to tube insertion, overall intubating conditions, or the requirement for rescue muscle relaxant.
A two-way repeated measure ANOVA (mixed design) of MAP showed a significant decrease in MAP over time (Huynh–Feldt F [4.18, 242.69] = 81.0), but there was no significant interaction between time and group (Huynh–Feldt F [4.18, 242.69] = 0.85). Post hoc Bonferroni adjustment for 95% CI showed significant differences between the control group and the RPL group. Mauchly’s test of sphericity indicated that the assumption of sphericity had been violated, (χ2[14] = 41.39, df = 14, P < 0.001). Hence, the Huynh–Feldt correction was applied. There were significant changes in MAP over time (Huynh–Feldt F [4.18, 242.69] = 81.0), where the post hoc paired t-test with Bonferroni adjustment showed significant differences in the control group between baseline and induction, baseline and at 0 min, baseline and at 3 min, baseline and at 5 min, spray and induction, spray and at 0 min, spray and at 3 min, spray and at 5 min, induction and at 0 min, induction and at 3 min, and induction and at 5 min. Meanwhile, adjustment for RPL group also showed significant differences between baseline and induction, baseline and at 0 min, baseline and at 3 min, baseline and at 5 min, spray and induction, spray and at 0 min, spray and at 3 min, spray and at 5 min, induction and at 0 min, induction and at 3 min, and induction and at 5 min. The overall mean difference between the two drug interventions with regard to MAP was not significant (P > 0.05) (Table 3). Figure 2 illustrates the changes in MAP during the induction of anesthesia.
| Table 1: Demographic Profile | |||
| Variable | RPL (n = 30) | Control (n = 30) | p-value |
| Age (years) | 38.83 ± 15.01 | 39.03 ± 13.17 | 0.956 |
| Weight (kg) | 62.30 ± 10.56 | 66.53 ± 11.64 | 0.146 |
| Height (cm) | 159.53 ± 8.26 | 162.35 ± 8.13 | 0.188 |
| BMI (m/kg2) | 24.19 ± 3.16 | 25.08 ± 2.99 | 0.267 |
| Gender: n (%) | |||
| · Male | 12 (40.0) | 15 (50.0) | 0.436 |
| · Female | 18 (60.0) | 15 (50.0) | |
| NYHA: n (%) | |||
| · I | 26 (86.7) | 28 (93.3) | 0.389 |
| · II | 4 (13.3) | 2 (6.7) | |
| ASA: n (%) | |||
| · I | 20 (66.7) | 21 (70.0) | 0.781 |
| · II | 10 (33.3) | 9 (30.0) | |
| Data presented as mean SD or n (%); NYHA: New York Heart Association, BMI: Basal metabolic rate | |||
| Table 2: Intubating Conditions | |||
| Parameter | RPL (n = 30) | Control (n = 30) | P-value |
| Laryngoscopy: n (%) | |||
| Easy | 29 (96.7) | 17 (56.7) | <0.01 |
| Fair | 1 (3.3) | 12 (40.0) | |
| Difficult | 0 (0.0) | 1 (3.3) | |
| Vocal cord position: n (%) | |||
| Open | 26 (86.7) | 17 (56.7) | 0.028 |
| Intermediate | 4 (13.3) | 11 (36.7) | |
| Closed | 0 (0.0) | 2 (6.7) | |
| Reaction to tube insertion: n (%) | |||
| None | 13 (43.3) | 6 (20.0) | 0.151 |
| Slight | 14 (46.7) | 20 (66.7) | |
| Vigorous | 3 (10.0) | 4 (13.3) | |
| Overall intubating conditions: n (%) | |||
| Acceptable (excellent and good) | 27 (90.0) | 25 (83.3) | 0.706 |
| Unacceptable (poor) | 3 (10.0) | 5 (16.7) | |
| Require rescue muscle relaxant: n (%) | |||
| No | 27 (90.0) | 25 (83.3) | 0.706 |
| Yes | 3 (10.0) | 5 (16.7) | |
| Data presented as n (%); P 0.05 considered as significant | |||
| Table 3: Comparative MAP between the two groups | ||
| Time | RPL (n = 30) | Control (n = 30) |
| Baseline MAP | 97.3 ± 14.3 | 97.8 ± 14.3 |
| At spray | 92.9 ± 12.1 | 94.3 ± 13.4 |
| At induction | 84.2 ± 14.8 | 84.4 ± 15.6 |
| At 0 min post intubation | 72.1 ± 9.8 | 75.9 ± 9.8 |
| At 3 min post induction | 71.9 ± 11.4 | 68.3 ± 15.3 |
| At 5 min post induction | 69.9 ± 8.9 | 71.4 ± 9.8 |
| Data given as mean ± SD | ||
To analyze the effect of the interventions on HR, a two-way repeated measure ANOVA (mixed design) was applied. There were significant changes in HR over time (Huynh–Feldt F [4.00, 231.7] = 5.22), but there was no significant interaction between time and group (Huynh-Feldt F [4.00, 231.7] = 1.31). Post hoc Bonferroni adjustment for 95% CI showed significant differences over time in both the control group and the RPL group (Fig. 3). The overall mean difference between the two drug interventions with regard to HR was not significant (P > 0.05) (Table 4).
4. DISCUSSION
We postulated that topical lignocaine spray prior to induction improves intubating conditions without muscle relaxant and hemodynamic stability; hence, it may potentially decrease the requirement for remifentanil and therefore, providing insight into the future application of this technique in more sensitive patient groups, such as the elderly and children. Our results showed that the RPL group had a significantly higher percentage of easy laryngoscopic procedures (96.7% vs. 56.7%; P < 0.01) and open vocal cord positions (86.7% vs. 56.7%; P = 0.028) than the control group. Easy laryngoscopy and an open vocal cord position are considered “excellent conditions” individually.15 However, there were no significant differences in the overall intubating conditions, the requirement for rescue muscle relaxants, or the hemodynamic changes between the two groups. A previous study by Kim et al.16 showed that when topical lignocaine 120 mg was instilled into the larynx and trachea, it provided acceptable intubating conditions without hemodynamic instability in 88% of patients (compared to 52% in the control group [P = 0.005]) during TCI remifentanil and propofol without muscle relaxants. Our study showed that an oropharyngeal lignocaine spray of 100 mg provided acceptable conditions in 27 individuals (90%) compared to 25 patients (83.3%) in the control group. Excellent intubating conditions were seen in 10 patients (33.3%) in the RPL group versus four patients (13.3%) in the control group. IV rocuronium (50 mg) was administered in three cases (10%) in the RPL group and in five cases (16.7%) in the control group due to unacceptable conditions. However, the data analysis showed no significant association between the two groups in terms of intubating conditions (P = 0.706) and the need for rescue muscle relaxant in the RPL group versus the control group (P = 0.706). The major cause of unacceptable conditions was reaction to tube insertion (i.e., more than two contractions, coughing, and/or movement longer than 5 s). This was seen in four cases in the control group and in three cases in the RPL group. This is consistent with previous studies in which opioid-induced cough or cough provoked by ETT passing through the vocal cords was responsible for the majority of unacceptable conditions during ETI.17,18 Sun et al.19 conducted two studies with 1108 patients to compare incremental doses of IV lignocaine and observe the effect of this on coughing. The cough reflex was completely suppressed by plasma concentrations of lignocaine of more than 3 μg/ml.


| Table 4: Comparative HR Changes between the two groups | ||
| Time | RPL (n = 30) | Control (n = 30) |
| Baseline | 84.0 ± 18.9 | 87.1 ± 18.3 |
| At spray | 83.3 ± 14.6 | 87.6 ± 17.7 |
| At induction | 79.7 ± 13.5 | 79.8 ± 15.2 |
| At 0 min post intubation | 78.2 ± 16.4 | 77.4 ± 18.4 |
| At 3 min post induction | 81.8 ± 14.7 | 79.3 ± 10.8 |
| At 5 min post induction | 82.1 ± 16.7 | 79.0 ± 13.3 |
| Data given as mean ± SD | ||
Remifentanil has gained popularity due to the duration of apnea comparable to suxamethonium for potential or known difficult airways, especially when muscle relaxants may be undesirable. There are several case reports of induction and intubation using remifentanil and propofol for cases of malignant hyperpyrexia or lung diseases for which the avoidance of muscle relaxants was essential or desirable.20 The trials of Mencke et al.21 and Bouvet et al.22 showed no significant association between the avoidance of muscle relaxants and upper airway injury (RR 1.12, 95% CI 0.61–2.08; P = 0.14) when remifentanil was used as part of the protocol.21,22 Total intravenous anesthesia with propofol and remifentanil is considered superior to sevoflurane in terms of induction, intubation, maintenance, stress hormone response to surgical stimuli, and awakening,23 and TCI allows for the easy adjustment of anesthetic depth.
Topical lignocaine was used in the present study, and several studies have compared the two routes. However, Kumar et al.24 found no advantage of the use of nebulized lignocaine in attenuating the hemodynamic response to laryngoscopy and intubation. The use of opioids such as IV fentanyl before induction was effective in attenuating the hemodynamic response.24 Other evidence has shown contrary results, with topical lignocaine spray being effective at attenuating the pressor response when ETI was performed after 2 min of tracheal lignocaine spray. 12 Jokar et al.25 observed that MAP was not statistically lower in the topical lignocaine group than in the IV lignocaine group. However, the use of lignocaine, either topical or IV, resulted in a better hemodynamic profile compared to the control group. They observed a significant difference between the two lignocaine groups in terms of a decrease in HR; that is, a lower HR was noted during intubation in the topical group.25
5. LIMITATIONS
There are several limitations to our study. First, the patients’ characteristics were ASA statuses of I and II and ages between 18 and 65 years old. Therefore, the findings may only be applicable to such patient populations and may have adverse effects on patients belonging to extremes of age or an ASA status higher than II, those having serious cardiovascular or respiratory diseases, and those in a state of shock. Further studies may show the beneficial role of topical lignocaine with TCI induction without muscle relaxants in such populations, since the dosage requirements might possibly be lower. Second, in the present study, the topical spray was instilled without performing laryngoscopy and was restricted to the oropharynx, in contrast to earlier studies in which topical lignocaine was instilled directly over vocal cords under direct visualization using direct laryngoscopy or nebulization and/or via the transtracheal route, possibly providing better LA spread.
6. CONCLUSION
Topical lignocaine spray significantly improved intubating conditions (easy laryngoscopy and open vocal cord position) during ETI without muscle relaxants using TCI remifentanil and propofol. Intubating conditions were acceptable (good to excellent) when TCI remifentanil and propofol were used for induction; therefore, their use is recommended when the avoidance of muscle relaxants is required. Topical lignocaine, such as oral spray, tracheal spray, or nebulization, is a useful adjunct for improving intubating conditions while maintaining stable hemodynamics.
7. Data availability
The numerical data generated during this research is available with the authors.
8. Acknowledgements
We would like to thank the Malaysian Society of Anesthesiologists for supporting this study through the K Inbasegaran research grant and the Research Creativity and Management Office of University Sains Malaysia for including this grant as an external grant.
9. Conflict of interest
The authors declare no conflict of interest
10. Authors’ contribution
SNK: conception, design, execution, analysis, interpretation of the data, drafting and final approval of manuscript WMNWH: conception, design, execution, analysis, interpretation of the data, drafting, critical revision and final approval of manuscript
SPS, MZM, MHH: conception, design, critical revision and final approval of manuscript
11. REFERENCES
- Grant S, Noble S, Woods A, Murdoch J, Davidson A. Assessment of intubating conditions in adults after induction with propofol and varying doses of remifentanil. Br J Anaesth. 1998;81(4):540-543. [PubMed] DOI: 1093/bja/81.4.540
- Stevens JB, Wheatley L. Tracheal Intubation in Ambulatory Surgery Patients. Anesth Analg. 1998;86(1):45-49. [PubMed] DOI: 1097/00000539-199801000-00009
- Erhan E, Ugur G, Alper I, Gunusen I, Ozyar B. Tracheal intubation without muscle relaxants: remifentanil or alfentanil in combination with propofol. Eur J Anaesthesiol. 2005;20(1):37-43. [PubMed] DOI: 1017/s0265021503000073
- Alexander R, Olufolabi AJ, Booth J, El-Moalem HE, Glass PS. Dosing study of remifentanil and propofol for tracheal intubation without the use of muscle relaxants. Anaesthesia. 1999;54(11):1037-1040. [PubMed] DOI: 1046/j.1365-2044.1999.00904.x
- McNeil IA, Culbert B, Russell I. Comparison of intubating conditions following propofol and succinylcholine with propofol and remifentanil 2 μg kg −1 or 4 μg kg −1. Br J Anaesth. 2000;85(4):623-625. [PubMed] DOI: 1093/bja/85.4.623
- Woods AW, Grant S, Harten J, Noble JS, Davidson JA. Tracheal intubating conditions after induction with propofol, remifentanil and lignocaine. Eur J Anaesthesiol. 1998;15(6):714-718. [PubMed] DOI: 1097/00003643-199811000-00014
- Klemola UM, Mennander S, Saarnivaara L. Tracheal intubation without the use of muscle relaxants: remifentanil or alfentanil in combination with propofol. Acta Anaesthesiol Scand. 2000;44(4):465-469. [PubMed] DOI: 1034/j.1399-6576.2000.440419.x
- Pang L, Zhuang YY, Dong S, Ma HC, Ma HS, Wang YF. Intubation without muscle relaxation for suspension laryngoscopy: A randomized, controlled study. Niger J Clin Pract. 2014;17(4):456-461. [PubMed] DOI: 4103/1119-3077.134038
- El-Tahan MR, Regal M. Target-Controlled Infusion of Remifentanil Without Muscle Relaxants Allows Acceptable Surgical Conditions During Thoracotomy Performed Under Sevoflurane Anesthesia. J Cardiothorac Vasc Anesth. 2015;29(6):1557-1566. [PubMed] DOI: 1053/j.jvca.2015.02.014
- Ide M, Sunada K, Katsuyama N. Are Muscle Relaxants Needed for Nasal Intubation in Propofol and Remifentanil Anesthesia? J Oral Maxillofac Surg. 2014;72(11):2134-2139. [PubMed] DOI: 1016/j.joms.2014.07.020
- Bolow K, Nielsen TG, Lund J. The effect of topical lignocaine on intubating conditions after propofol-alfentanil induction. Acta Anaesthesiol Scand. 1996; 40:752-756. [PubMed] DOI: 1111/j.1399-6576.1996.tb04523.x
- Takita K, Morimoto Y, Kemmotsu O. Tracheal lidocaine attenuates the cardiovascular response to endotracheal intubation. Can J Anaesth. 2001;48(8):732-736. [PubMed] DOI: 1007/BF03016686
- Minto CF, Schnider TW, Egan TD, Youngs E, Lemmens HJ, Gambus PL, et al. Influence of Age and Gender on the Pharmacokinetics and Pharmacodynamics of Remifentanil. Anesthesiology. 1997;86(1):10-23. [PubMed] DOI: 1097/00000542-199701000-00004
- Marsh B, White M, Morton N, Kenny GNC. Pharmacokinetic model driven infusion of propofol in children. Br J Anaesth. 1991;67(1):41-48. [PubMed] DOI: 1093/bja/67.1.41
- Viby-Mogensen J, Engbaek J, Eriksson LI, et al. Good Clinical Research Practice (GCRP) in pharmacodynamic studies of neuromuscular blocking agents. Acta Anaesthesiol Scand. 1996;40(1):59-74. [PubMed] DOI: 1111/j.1399-6576.1996.tb04389.x
- Kim JS, Kim DH, Joe HB, Oh CK, Kim JY. Effect of tracheal lidocaine on intubating conditions during propofol-remifentanil target-controlled infusion without neuromuscular blockade in day-case anesthesia. Korean J Anesthesiol. 2013;65(5):425-430. [PubMed] DOI: 4097/kjae.2013.65.5.425
- Ithnin F, Lim Y, Shah M, Shen L, Sia ATH. Tracheal intubating conditions Using propofol and remifentanil target-controlled infusion: A comparison of remifentanil EC50 for Glidescope and Macintosh. Eur J Anaesthesiol. 2009;26(3):223-228. [PubMed] DOI: 1097/EJA.0b013e328324b4fc
- Kim SJ, Yoo KY, Park BY, Kim WM, Jeong CW. Comparison of intubating conditions and hemodynamic responses to tracheal intubation with different effect-site concentrations of remifentanil without muscle relaxants during target-controlled infusion of propofol. Korean J Anesthesiol. 2009;57(1):13. [PubMed] DOI: 4097/kjae.2009.57.1.13
- Sun L, Guo R, Sun L. The impact of prophylactic intravenous lidocaine on opioid-induced cough: a meta-analysis of randomized controlled trials. J Anesth. 2014;28(3):325-333. [PubMed] DOI: 1007/s00540-013-1732-3
- Fotopoulou G, Theocharis S, Vasileiou I, Kouskouni E, Xanthos T. Management of the airway without the use of neuromuscular blocking agents: The use of remifentanil. Fundam Clin
- 2012;26(1):72-85. [PubMed] DOI: 10.1111/j.1472-8206.2011.00967.x
- Mencke T, Echternach M, Kleinschmidt S, Lux P, Barth V, Plinkert PK, et al. Laryngeal Morbidity and Quality of Tracheal Intubation. Anesthesiology. 2003;98(5):1049-1056. [PubMed] DOI: 1097/00000542-200305000-00005
- Bouvet L, Stoian A, Jacquot-Laperrière S, Allaouchiche B, Chassard D, Boselli E. Laryngeal injuries and intubating conditions with or without muscular relaxation: an equivalence study. Can J Anaesth. 2008;55(10):674-684. [PubMed] DOI: 1007/BF03017743
- Ihn CH, Joo JD, Choi JW, Kim DW, Jeon YS, Kim YS, et al. Comparison of Stress Hormone Response, Interleukin-6 and Anaesthetic Characteristics of Two Anaesthetic Techniques: Volatile Induction and Maintenance of Anaesthesia using Sevoflurane versus Total Intravenous Anaesthesia using Propofol and Remifentanil. J Int Med Res. 2009;37(6):1760-1771. [PubMed] DOI: 1177/147323000903700612
- Kumar A, Seth A, Prakash S, Deganwa M, Gogia A. Attenuation of the hemodynamic response to laryngoscopy and tracheal intubation with fentanyl, lignocaine nebulization, and a combination of both: A randomized controlled trial. Anesth Essays Res. 2016;10(3):661. [PubMed] DOI: 4103/0259-1162.191113
- Jokar A, Babaei M, Pourmatin S, Taheri M, Almasi-Hashiani A, Yazdanbakhsh A. Effects of intravenous and inhaled nebulized lignocaine on the hemodynamic response of endotracheal intubation patients: A randomized clinical trial. Anesth Essays Res. 2018;12(1):159. [PubMed] DOI: 4103/aer.AER_75_17