Heba Nasr Said Nassar 1, Madiha Metwally Zidan 2, Samia Abdelmohsen Abdellatif 3, Ahmed Mohamed El Sayed El Hennawy 4, Rania Maher Hussien 5
Authors’ affiliations:
Background & objective: The number of bariatric surgeries is rapidly increasing the world over, and the concept of fast-track surgery using laparoscopy has made bariatric surgeries a cost-effective and efficient way of treating morbidly obese patients. It has been recommended to encourage opioid free anesthesia (OFA) using non-opioid analgesics during bariatric surgery for enhanced recovery. We compared general anesthesia (GA) employing opioids with OFA in laparoscopic bariatric surgeries.
Methodology: This prospective, double-blinded, randomized controlled study compared opioid-free anesthesia with traditional opioid-based methods in 50 adult patients, ASA I and II, scheduled for non-emergency laparoscopic weight reduction procedures. Patients were randomized as 25 patients to each group: Group A patients were managed with a non-opioid regimen, while Group B was managed with opioid-based regimen. All patients received ranitidine 50 mg and metoclopramide 10 mg IV.
Results: Non-significant differences were noted between the two groups regarding heart rate, blood pressure, EtCO₂ levels, or peak inspiratory pressure (P > 0.05). Nonetheless, the group that did not receive opioids had a notably slower recovery (P < 0.05). Patients in the opioid group reported higher pain score levels, measured by the Visual Analog Scale (VAS), immediately after surgery and within the first two hours (P < 0.05). The OFA Group required extended PCA usage and received fewer supplemental narcotic doses in contrast to the opioid group (P < 0.001). A higher frequency of nausea and vomiting postoperatively was significantly increased in the opioid group (P < 0.05). No notable differences were seen regarding cases of shivering or bradycardia (P > 0.05).
Conclusion: Opioid-free anesthetic protocol in conjugation with multimodal non-opioid drugs is effective for laparoscopic bariatric surgeries and supports enhanced recovery, compared to opioid based anesthesia. Frequency of postoperative nausea and vomiting was significantly increased in the opioid group.
Abbreviations: BIS: Bispectral Index, ERAS: Enhance Recovery After Surgery, EtCO₂: End tidal carbon dioxide. GA: general anesthesia, OBA: opioid-based anesthesia, OFA: Opioid-free Anesthesia, PCA: patient controlled analgesia, VAS: Visual Analog Scale,
Keywords: bariatric procedures, enhanced recovery, laparoscopic surgery, multimodal analgesia, Opioid free anesthesia
Citation: Nassar HNS, Zidan MM, Abdellatif SA, El Hennawy AME, Hussien RM. Comparative study between opioid usage and opioid free anesthesia in laparoscopic bariatric surgeries. Anaesth. pain intensive care 2025;29(5):288-298. DOI: 10.35975/apic.v29i5.2487
Received: April 16, 2024; Revised: May 20, 2025; Accepted: May 16, 2025
Obesity is defined by an abnormal or excessive buildup of body fat, which leads to significant health risks and is recognized as one of the avoidable contributors to death globally.1
It is frequently linked to a range of other health problems, such as obstructive sleep apnea, cardiovascular diseases, type II diabetes, osteoarthritis, asthma in addition to depression.2
Obesity rates have been steadily rising worldwide, leading to an increase in bariatric surgeries. This surge has made it more crucial than ever to optimize perioperative care, particularly in terms of pain management and recovery, to address the growing number of patients requiring these procedures.3
Providing analgesia to patients complaining of obesity who are planned for laparoscopic bariatric surgery is notably complex due to their physiological changes and associated comorbidities. Therefore, the use of opioid-sparing agents during anesthesia has garnered considerable attention. Moreover, opioid use in the perioperative period for bariatric surgery is linked to increased risks, particularly respiratory depression, to which obese patients are especially susceptible. Therefore, alternatives that maintain airway tone are preferred.4
Opioid-free Anesthesia (OFA) is an anesthetic approach that completely avoids the administration of narcotics through the perioperative period to prevent opioid-induced respiratory complications while ensuring patient comfort. It is also a method used to Enhance Recovery After Surgery (ERAS).5,6
OFA relies on non-opioid medications to provide effective analgesia during surgery. Several drugs, such as dexmedetomidine, magnesium, ketamine, lidocaine, steroids, and ketorolac, have demonstrated analgesic properties. Combining these agents can enhance analgesic efficacy while minimizing adverse effects by utilizing lower effective doses of each drug.7 This multimodal approach offers a promising strategy for improving perioperative outcomes in obese patients undergoing bariatric surgery. This approach could help facilitate faster recovery, enabling bariatric surgeries to be classified as fast-track procedures, or even day-case surgeries.
Recent studies have consistently shown that Opioid-Sparing anesthesia serves as a safe and highly effective alternative approach suitable for patients undergoing bariatric surgeries. Ulbing et al. (2023) reported reduced postoperative pain, lower opioid use, and better recovery outcomes with OFA.8 Yaşar and Yıldız (2023) confirmed the safety of OFA in laparoscopic procedures, with promising pain control and higher staff satisfaction.9 Mahdy (2024) demonstrated that OFA leads to significantly lower pain scores, fewer analgesic needs, and better hemodynamic stability.10 Torre et al. (2022) found that OFA patients required fewer antiemetics and had shorter hospital stays.11 Seyam et al. (2024) highlighted that OFA also reduces inflammatory markers and prolongs analgesia duration.12 Together, these studies support integrating OFA into clinical practice for bariatric surgery due to its benefits in pain control, recovery, and safety.
However, despite the promising results, further investigation is needed to evaluate OFA in relation to Standard opioid-based anesthesia (OBA) specifically in bariatric surgeries, to confirm its benefits and establish its role as a standard practice.
The main purpose of our trial was to assess benefits of OFA in comparison to OBA for patients scheduled for bariatric surgeries, focusing on VAS pain scores and overall narcotics used in the first 24 hours after surgery. The secondary objectives aimed to analyze the differences between both groups regarding postoperative oxygen needs, the incidence of common complications such as nausea, vomiting, shivering, and delayed recovery, duration of hospital stay, and time till discharge.
After approval of our protocol by the Ain Shams University ethical committee and receiving signed consent forms for every participant, this prospective, double-blinded, randomized controlled study involved 50 male and female participants, aged 20 to 45, categorized as ASA I or II, with a BMI40-50 kg/m². These patients were electively scheduled for laparoscopic bariatric surgeries under general anesthesia (GA).
Participants under 20 or over 45 years old, as well as those with a previous incidence of substance abuse or opioid addiction, individuals with chronic pain, severe cardiovascular, neurological, or liver diseases, epilepsy, or allergies to any of the used medications were excluded from the trial. Additionally, Individuals whose BMI was above 50 kg/m² or who were scheduled for surgeries expected to last over three hours were not included in the study.
The sample size determination was performed with the STATA software, using a 5% alpha error and 90% statistical power. According to an earlier study (Mulier et al.,2018), 13 showed that among OA group the mean total QoR 40 score % was 74 ± 6 compared to 89 ± 3 among OFA group . Therefore, 25 participants for each group (totaling 50) was required to observe lower than the observed difference (total cases 50).
Participants were randomly assorted to one of both groups by computerized random assignment: Group A (OFA) and Group B (OBA). Patients in both groups, the anesthesiologists conducting the anesthetic management and anesthesiologists involved in data collection were blinded.
Following an 8-hour fasting period, all patients received premedication consisting of 50 mg of ranitidine (an H2 receptor blocker) and 10 mg of metoclopramide via intravenous injection one hour prior to the scheduled surgery to prevent postoperative nausea and vomiting. Additionally, ketorolac 30 mg IV, diclofenac 150 mg IM, and dexamethasone 8 mg IV were administered one hour before surgery to reduce inflammation. For sedation, 1–2 mg of midazolam was injected intravenously prior to the induction of anesthesia.
All patients underwent continuous monitoring for heart rate (HR), blood pressure, end-tidal expiratory CO₂, oxygen saturation (SpO₂), and peak inspiratory pressure using a Primus anesthesia machine (Draeger Medical, Lübeck, Germany). These parameters were recorded at the time of induction and at 30-min intervals thereafter until the conclusion of the procedure. Anesthesia depth was evaluated using the Bispectral Index (BIS) (Eagle, North American Draeger, USA), with the following scale: 0 = isoelectric line of EEG activity; <40 = deep anesthesia; 40–60 = adequate depth of anesthesia; 60–70 = deep sedation; 70–85 = light to moderate sedation; > 90 = intact reflexes; and 95–100 = fully awake.14 Infusion rates were adjusted to maintain a BIS value within the target range of 40–60.
Furthermore, the degree of neuromuscular blockade was assessed using a peripheral nerve stimulator to record the train-of-four (TOF) ratio.15
Group A (OFA Group) included 25 patients. Induction of anesthesia was achieved by administering 2 mg/kg of propofol, 0.25 mg/kg of ketamine for analgesia, 0.5–1 µg/kg ideal body weight (IBW) of dexmedetomidine, 1.5 mg/kg IBW of lidocaine, and 40 mg/kg IBW of magnesium. The calculated doses of dexmedetomidine, ketamine, lidocaine and magnesium were prepared in a 20 ml normal saline syringe given slowly over 10 min.
Group B (opioid group) included 25 patients. Anesthesia was induced with 2 mg/kg of propofol alongside an analgesic dose of 2–4 µg/kg IBW fentanyl. The calculated dose of fentanyl was diluted in a 20 ml syringe containing normal saline and administered slowly over 10 min.
In both groups, Rocuronium was provided at a dose of 0.6–1 mg/kg for endotracheal intubation. Controlled mechanical ventilation was managed with tidal volume of 7–10 mL/kg and respiratory rates set at 12–14 breaths per min, and a 5–10 cmH₂O positive end-expiratory pressure (PEEP), using a Primus anesthesia machine (Draeger Medical, Lübeck, Germany).
In Group A (OFA Group), anesthesia was maintained with isoflurane (1–1.5 vol%) and continuous infusions of ketamine (0.25–0.5 mg/kg IBW/h), dexmedetomidine (0.5–1 µg/kg/h), magnesium sulfate (5–10 mg/kg IBW/h), and lidocaine (1.5–3 mg/kg IBW/h). Ketamine infusion was titrated to keep the BIS within 40–60. Vital signs were recorded every 30 min, and dexmedetomidine infusion or isoflurane concentration was increased if signs of awareness or pain (e.g., tachycardia or hypertension) occurred.
In Group B (opioid group), anesthesia was maintained using isoflurane (1–1.5 vol%) and fentanyl infusion (1-1.5 µg/kg/hr). Vital signs were monitored every 30 min, and morphine boluses (2–3 mg) were given for signs of pain, such as tachycardia or hypertension. Muscle relaxation was sustained with rocuronium boluses (10–20 mg).
In both groups, infusions were held five min before surgery ended, and isoflurane was stopped. Using 2 mg/kg of sugammadex for reversal of the neuromuscular blocking, and extubation occurred once patient could follow commands and achieved full muscle relaxation recovery, indicated by a TOF ratio of 1.
In PACU the patient is assessed by using checklist of recovery from surgery from Johns Hopkins University includes, monitor vital signs, watch any sign of complications, careful assessment should be made for any signs of swallowing difficulties or impaired gag reflex, while continuously monitoring the patient’s level of awareness. It is important to inspect all lines, tubes, and drains. Additionally, the surgical wound should be examined for any abnormalities, and urine output must be measured. Moreover, the recovery from the anesthesia is assessed by Aldrete Scoring System.16
Total fentanyl consumption in the first 24 hours was recorded. Pain intensity was evaluated employing the Visual Analog Scale (VAS) at 30 min after recovery, then hourly for two hours, followed by measurements every four hours over a 24-hour period. Adverse effects, such as low oxygen levels (SpO₂ < 94%), obstructive breathing, delayed emergence, dizziness, somnolence, postoperative nausea and vomiting (PONV), along with shivering and sensations of cold, were closely observed in the post-anesthesia care unit (PACU) and subsequently monitored every four hours for a duration of 24 hours to ensure timely detection and management of these common postoperative complications.
Delayed recovery from GA is medically defined as the inability of the patient to regain an adequate state of awareness, remaining either unresponsive or profoundly sedated, typically assessed 30 min after the conclusion of GA.
Patients with high risk factors as > 60 BMI, severe Obstructive Sleep Apnea, preoperative abnormal respiratory functions and underlying cardiovascular disease require ICU admission for monitoring after the surgery.
Postoperative monitoring of vital signs, mainly heart rate and blood pressure readings, ICU discharge time, and hospital stay duration were also documented.
2.1. Statistical analysis:
The collected data were processed using the Statistical Package for the Social Sciences (SPSS) version 23.0 (SPSS Inc., Chicago, Illinois, USA). Mean ± standard deviation and range were used for expression of quantitative data with a normal distribution, while non-normally distributed data were presented in form of median and interquartile range (IQR).
Categorical variables were presented using frequencies and percentages. Both Shapiro-Wilk and Kolmogorov-Smirnov tests were used to evaluate the data normality.
For statistical comparisons, for parametric data the independent-samples t-test was used when comparing difference between means, whereas the Mann-Whitney U test was used for non-parametric two-group comparisons.
Qualitative data were compared between groups by the Chi-square test; however, Fisher’s exact test was employed when the expected data set count was less than five. The significance level was set at a 5% margin of error, corresponding to a 95% confidence interval. P < 0.05 indicated statistically significant, while P < 0.001 were regarded as highly significant. P > 0.05 were not significant.
Both groups had similar patient characteristics (Table 1)
and comparable intraoperative and postoperative heart rates (Figure), as well as other vital parameters (HR, MAP, PIP, EtCO₂, SpO₂) (Figures 1, 2, 3, 4 and 5).
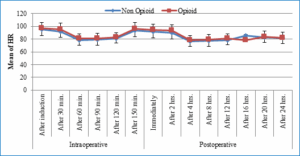
Figure 1: Comparative intraoperative and postoperative heart rates
Using: t-Independent Sample t-test for Mean ± SD
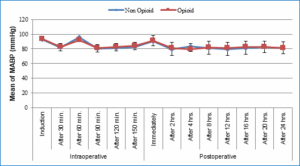
Figure 2: Comparative intraoperative and postoperative MABP (mmHg)
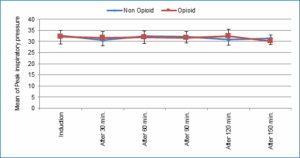
Figure 3: Comparative peak inspiratory pressures
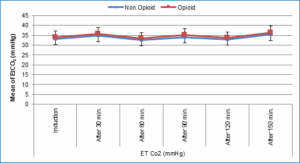
Figure 4: Comparative EtCO2 values (mmHg)
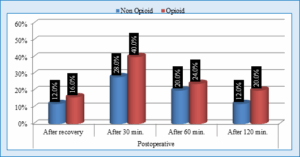
Figure 5: Comparative % of patients with SpO2 < 94%
However, the OFA Group showed a statistically significant higher delayed recovery frequency with (P < 0.05) (Figure 6) and required a longer duration of PCA for narcotic consumption (P < 0.001) Table 3), while the opioid group had higher VAS pain scores immediately postoperatively and up to 2 hours after surgery (P < 0.05) (Table 2) and required more narcotic boluses outside PCA (P < 0.001) (Table 3).
Both groups were similar in ICU recovery time and hospital stay duration (Table 4).
The opioid balanced group showed a statistically significant increased frequency of nausea and vomiting postoperatively (P < 0.05), while shivering and bradycardia differences were insignificant (P > 0.05) (Table 5).
Moreover, Bakan et al. (2015), who studied opioid-free GA using propofol, lidocaine, and dexmedetomidine for laparoscopic surgery found a statistically significant elevation of mean arterial pressure levels and heart rate values within the opioid-free group during induction, at intubation, and during early pneumoperitoneum. These findings contradict the current study, which observed no significant differences in these parameters.19 The discrepancy may be due to Bakan et al. (2015) 's reliance on dexmedetomidine alone for sedation and analgesia during induction and intubation.
The current study also emphasized that the opioid-free regimen patients experienced considerable lower postoperative pain scores and reduced opioid consumption, aligning with Soudi et al. (2022), who reported that OFA has a better outcomes than total balanced anesthesia (TBA) regarding postoperative pain intensity score and narcotics use.20 Additionally, our findings are agreeing with Berlier et al. (2022), who revealed that OFA leads to lower morphine requirements during the first 24 hours after bariatric surgical procedures in comparison to OBA.21
On the other hand, The current study's results differ from Torre et al. (2022), who reported that OFA did not reduce total postoperative opioid consumption in comparison to opioid-based anesthesia groups.11
The current study revealed non-significant difference between both regimens regarding postoperative hypoxemia. However, the opioid group experienced eight cases of nausea and vomiting, whereas the OFA Group had only two cases. This finding aligns with Elsaye et al. (2019), who found a higher postoperative complications like nausea, vomiting, hypoxia, and shivering in the opioid-based anesthesia group.22 Torre et al. (2022) also reached similar results when they revealed that OFA patients consumed less antiemetics along with faster hospital discharges.11 Similarly, Ziemann-Gimmel et al. (2014) observed a reduced incidence of nausea and vomiting postoperatively in the OFA Group, consistent with the current findings.23
Regarding recovery time, our study showed that patients receiving opioid-free anesthesia experienced significantly delayed recovery compared to those in the opioid group. This delay may be attributed to the multiple drugs used in our protocol, suggesting a need for further research to explore adjustments such as removing magnesium, reducing maintenance doses, or stopping infusions earlier. These findings align with Soudi et al.(2022), who also reported longer extubation times and a delayed achievement of an Aldrete score of 9 in patients undergoing opioid-free anesthesia.24
This study concluded that a multimodal opioid-free approach offers effective analgesia compared to traditional opioid use in bariatric surgeries, resulting in reduced postoperative pain intensity levels, less narcotics requirements, and more rapid recovery.
6. Data availability
The numerical data generated during this research is available with the authors.
7. Conflict of interest
All authors declare that there was no conflict of interest.
8. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
9. Ethical considerations
The research ethical committee at the medical school at Ain Shams University gave its approval to this study. (FMASU MD 358 / 2018) with Pan African Clinical Trial Registry, identifier PACTR202401561160798
10. Authors’ contribution
Each author made a significant intellectual contribution to the work, and each author read and approved the final product.
Authors’ affiliations:
- Heba Nasr Said Nassar, Assistant lecturer of Anaesthesia, Intensive Care and Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: hebanasrnassar@gmail.com; Mobile: 01015872447
- Madiha Metwally Zidan, Professor of Anaesthesia, Intensive Care and Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: madiha.zidan@hotmail.com
- Samia Abdelmohsen Abdellatif, Professor of Anaesthesia, Intensive Care and Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: samia.wahba1@hotmail.com
- Ahmed Mohamed El Sayed El Hennawy, Professor of Anaesthesia, Intensive Care and Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: anahenno@yahoo.com
- Rania Maher Hussien, Associate Professor of Anaesthesia, Intensive Care and Pain Management, Faculty of Medicine, Ain Shams University, Cairo, Egypt; Email: drrania_maamon@med.asu.edu.eg
ABSTRACT
Background & objective: The number of bariatric surgeries is rapidly increasing the world over, and the concept of fast-track surgery using laparoscopy has made bariatric surgeries a cost-effective and efficient way of treating morbidly obese patients. It has been recommended to encourage opioid free anesthesia (OFA) using non-opioid analgesics during bariatric surgery for enhanced recovery. We compared general anesthesia (GA) employing opioids with OFA in laparoscopic bariatric surgeries.
Methodology: This prospective, double-blinded, randomized controlled study compared opioid-free anesthesia with traditional opioid-based methods in 50 adult patients, ASA I and II, scheduled for non-emergency laparoscopic weight reduction procedures. Patients were randomized as 25 patients to each group: Group A patients were managed with a non-opioid regimen, while Group B was managed with opioid-based regimen. All patients received ranitidine 50 mg and metoclopramide 10 mg IV.
Results: Non-significant differences were noted between the two groups regarding heart rate, blood pressure, EtCO₂ levels, or peak inspiratory pressure (P > 0.05). Nonetheless, the group that did not receive opioids had a notably slower recovery (P < 0.05). Patients in the opioid group reported higher pain score levels, measured by the Visual Analog Scale (VAS), immediately after surgery and within the first two hours (P < 0.05). The OFA Group required extended PCA usage and received fewer supplemental narcotic doses in contrast to the opioid group (P < 0.001). A higher frequency of nausea and vomiting postoperatively was significantly increased in the opioid group (P < 0.05). No notable differences were seen regarding cases of shivering or bradycardia (P > 0.05).
Conclusion: Opioid-free anesthetic protocol in conjugation with multimodal non-opioid drugs is effective for laparoscopic bariatric surgeries and supports enhanced recovery, compared to opioid based anesthesia. Frequency of postoperative nausea and vomiting was significantly increased in the opioid group.
Abbreviations: BIS: Bispectral Index, ERAS: Enhance Recovery After Surgery, EtCO₂: End tidal carbon dioxide. GA: general anesthesia, OBA: opioid-based anesthesia, OFA: Opioid-free Anesthesia, PCA: patient controlled analgesia, VAS: Visual Analog Scale,
Keywords: bariatric procedures, enhanced recovery, laparoscopic surgery, multimodal analgesia, Opioid free anesthesia
Citation: Nassar HNS, Zidan MM, Abdellatif SA, El Hennawy AME, Hussien RM. Comparative study between opioid usage and opioid free anesthesia in laparoscopic bariatric surgeries. Anaesth. pain intensive care 2025;29(5):288-298. DOI: 10.35975/apic.v29i5.2487
Received: April 16, 2024; Revised: May 20, 2025; Accepted: May 16, 2025
1. INTRODUCTION
Obesity is defined by an abnormal or excessive buildup of body fat, which leads to significant health risks and is recognized as one of the avoidable contributors to death globally.1
It is frequently linked to a range of other health problems, such as obstructive sleep apnea, cardiovascular diseases, type II diabetes, osteoarthritis, asthma in addition to depression.2
Obesity rates have been steadily rising worldwide, leading to an increase in bariatric surgeries. This surge has made it more crucial than ever to optimize perioperative care, particularly in terms of pain management and recovery, to address the growing number of patients requiring these procedures.3
Providing analgesia to patients complaining of obesity who are planned for laparoscopic bariatric surgery is notably complex due to their physiological changes and associated comorbidities. Therefore, the use of opioid-sparing agents during anesthesia has garnered considerable attention. Moreover, opioid use in the perioperative period for bariatric surgery is linked to increased risks, particularly respiratory depression, to which obese patients are especially susceptible. Therefore, alternatives that maintain airway tone are preferred.4
Opioid-free Anesthesia (OFA) is an anesthetic approach that completely avoids the administration of narcotics through the perioperative period to prevent opioid-induced respiratory complications while ensuring patient comfort. It is also a method used to Enhance Recovery After Surgery (ERAS).5,6
OFA relies on non-opioid medications to provide effective analgesia during surgery. Several drugs, such as dexmedetomidine, magnesium, ketamine, lidocaine, steroids, and ketorolac, have demonstrated analgesic properties. Combining these agents can enhance analgesic efficacy while minimizing adverse effects by utilizing lower effective doses of each drug.7 This multimodal approach offers a promising strategy for improving perioperative outcomes in obese patients undergoing bariatric surgery. This approach could help facilitate faster recovery, enabling bariatric surgeries to be classified as fast-track procedures, or even day-case surgeries.
Recent studies have consistently shown that Opioid-Sparing anesthesia serves as a safe and highly effective alternative approach suitable for patients undergoing bariatric surgeries. Ulbing et al. (2023) reported reduced postoperative pain, lower opioid use, and better recovery outcomes with OFA.8 Yaşar and Yıldız (2023) confirmed the safety of OFA in laparoscopic procedures, with promising pain control and higher staff satisfaction.9 Mahdy (2024) demonstrated that OFA leads to significantly lower pain scores, fewer analgesic needs, and better hemodynamic stability.10 Torre et al. (2022) found that OFA patients required fewer antiemetics and had shorter hospital stays.11 Seyam et al. (2024) highlighted that OFA also reduces inflammatory markers and prolongs analgesia duration.12 Together, these studies support integrating OFA into clinical practice for bariatric surgery due to its benefits in pain control, recovery, and safety.
However, despite the promising results, further investigation is needed to evaluate OFA in relation to Standard opioid-based anesthesia (OBA) specifically in bariatric surgeries, to confirm its benefits and establish its role as a standard practice.
The main purpose of our trial was to assess benefits of OFA in comparison to OBA for patients scheduled for bariatric surgeries, focusing on VAS pain scores and overall narcotics used in the first 24 hours after surgery. The secondary objectives aimed to analyze the differences between both groups regarding postoperative oxygen needs, the incidence of common complications such as nausea, vomiting, shivering, and delayed recovery, duration of hospital stay, and time till discharge.
2. METHODOLOGY
After approval of our protocol by the Ain Shams University ethical committee and receiving signed consent forms for every participant, this prospective, double-blinded, randomized controlled study involved 50 male and female participants, aged 20 to 45, categorized as ASA I or II, with a BMI40-50 kg/m². These patients were electively scheduled for laparoscopic bariatric surgeries under general anesthesia (GA).
Participants under 20 or over 45 years old, as well as those with a previous incidence of substance abuse or opioid addiction, individuals with chronic pain, severe cardiovascular, neurological, or liver diseases, epilepsy, or allergies to any of the used medications were excluded from the trial. Additionally, Individuals whose BMI was above 50 kg/m² or who were scheduled for surgeries expected to last over three hours were not included in the study.
The sample size determination was performed with the STATA software, using a 5% alpha error and 90% statistical power. According to an earlier study (Mulier et al.,2018), 13 showed that among OA group the mean total QoR 40 score % was 74 ± 6 compared to 89 ± 3 among OFA group . Therefore, 25 participants for each group (totaling 50) was required to observe lower than the observed difference (total cases 50).
Participants were randomly assorted to one of both groups by computerized random assignment: Group A (OFA) and Group B (OBA). Patients in both groups, the anesthesiologists conducting the anesthetic management and anesthesiologists involved in data collection were blinded.
Following an 8-hour fasting period, all patients received premedication consisting of 50 mg of ranitidine (an H2 receptor blocker) and 10 mg of metoclopramide via intravenous injection one hour prior to the scheduled surgery to prevent postoperative nausea and vomiting. Additionally, ketorolac 30 mg IV, diclofenac 150 mg IM, and dexamethasone 8 mg IV were administered one hour before surgery to reduce inflammation. For sedation, 1–2 mg of midazolam was injected intravenously prior to the induction of anesthesia.
All patients underwent continuous monitoring for heart rate (HR), blood pressure, end-tidal expiratory CO₂, oxygen saturation (SpO₂), and peak inspiratory pressure using a Primus anesthesia machine (Draeger Medical, Lübeck, Germany). These parameters were recorded at the time of induction and at 30-min intervals thereafter until the conclusion of the procedure. Anesthesia depth was evaluated using the Bispectral Index (BIS) (Eagle, North American Draeger, USA), with the following scale: 0 = isoelectric line of EEG activity; <40 = deep anesthesia; 40–60 = adequate depth of anesthesia; 60–70 = deep sedation; 70–85 = light to moderate sedation; > 90 = intact reflexes; and 95–100 = fully awake.14 Infusion rates were adjusted to maintain a BIS value within the target range of 40–60.
Furthermore, the degree of neuromuscular blockade was assessed using a peripheral nerve stimulator to record the train-of-four (TOF) ratio.15
Group A (OFA Group) included 25 patients. Induction of anesthesia was achieved by administering 2 mg/kg of propofol, 0.25 mg/kg of ketamine for analgesia, 0.5–1 µg/kg ideal body weight (IBW) of dexmedetomidine, 1.5 mg/kg IBW of lidocaine, and 40 mg/kg IBW of magnesium. The calculated doses of dexmedetomidine, ketamine, lidocaine and magnesium were prepared in a 20 ml normal saline syringe given slowly over 10 min.
Group B (opioid group) included 25 patients. Anesthesia was induced with 2 mg/kg of propofol alongside an analgesic dose of 2–4 µg/kg IBW fentanyl. The calculated dose of fentanyl was diluted in a 20 ml syringe containing normal saline and administered slowly over 10 min.
In both groups, Rocuronium was provided at a dose of 0.6–1 mg/kg for endotracheal intubation. Controlled mechanical ventilation was managed with tidal volume of 7–10 mL/kg and respiratory rates set at 12–14 breaths per min, and a 5–10 cmH₂O positive end-expiratory pressure (PEEP), using a Primus anesthesia machine (Draeger Medical, Lübeck, Germany).
In Group A (OFA Group), anesthesia was maintained with isoflurane (1–1.5 vol%) and continuous infusions of ketamine (0.25–0.5 mg/kg IBW/h), dexmedetomidine (0.5–1 µg/kg/h), magnesium sulfate (5–10 mg/kg IBW/h), and lidocaine (1.5–3 mg/kg IBW/h). Ketamine infusion was titrated to keep the BIS within 40–60. Vital signs were recorded every 30 min, and dexmedetomidine infusion or isoflurane concentration was increased if signs of awareness or pain (e.g., tachycardia or hypertension) occurred.
In Group B (opioid group), anesthesia was maintained using isoflurane (1–1.5 vol%) and fentanyl infusion (1-1.5 µg/kg/hr). Vital signs were monitored every 30 min, and morphine boluses (2–3 mg) were given for signs of pain, such as tachycardia or hypertension. Muscle relaxation was sustained with rocuronium boluses (10–20 mg).
In both groups, infusions were held five min before surgery ended, and isoflurane was stopped. Using 2 mg/kg of sugammadex for reversal of the neuromuscular blocking, and extubation occurred once patient could follow commands and achieved full muscle relaxation recovery, indicated by a TOF ratio of 1.
In PACU the patient is assessed by using checklist of recovery from surgery from Johns Hopkins University includes, monitor vital signs, watch any sign of complications, careful assessment should be made for any signs of swallowing difficulties or impaired gag reflex, while continuously monitoring the patient’s level of awareness. It is important to inspect all lines, tubes, and drains. Additionally, the surgical wound should be examined for any abnormalities, and urine output must be measured. Moreover, the recovery from the anesthesia is assessed by Aldrete Scoring System.16
Total fentanyl consumption in the first 24 hours was recorded. Pain intensity was evaluated employing the Visual Analog Scale (VAS) at 30 min after recovery, then hourly for two hours, followed by measurements every four hours over a 24-hour period. Adverse effects, such as low oxygen levels (SpO₂ < 94%), obstructive breathing, delayed emergence, dizziness, somnolence, postoperative nausea and vomiting (PONV), along with shivering and sensations of cold, were closely observed in the post-anesthesia care unit (PACU) and subsequently monitored every four hours for a duration of 24 hours to ensure timely detection and management of these common postoperative complications.
Delayed recovery from GA is medically defined as the inability of the patient to regain an adequate state of awareness, remaining either unresponsive or profoundly sedated, typically assessed 30 min after the conclusion of GA.
Patients with high risk factors as > 60 BMI, severe Obstructive Sleep Apnea, preoperative abnormal respiratory functions and underlying cardiovascular disease require ICU admission for monitoring after the surgery.
Postoperative monitoring of vital signs, mainly heart rate and blood pressure readings, ICU discharge time, and hospital stay duration were also documented.
2.1. Statistical analysis:
The collected data were processed using the Statistical Package for the Social Sciences (SPSS) version 23.0 (SPSS Inc., Chicago, Illinois, USA). Mean ± standard deviation and range were used for expression of quantitative data with a normal distribution, while non-normally distributed data were presented in form of median and interquartile range (IQR).
Categorical variables were presented using frequencies and percentages. Both Shapiro-Wilk and Kolmogorov-Smirnov tests were used to evaluate the data normality.
For statistical comparisons, for parametric data the independent-samples t-test was used when comparing difference between means, whereas the Mann-Whitney U test was used for non-parametric two-group comparisons.
Qualitative data were compared between groups by the Chi-square test; however, Fisher’s exact test was employed when the expected data set count was less than five. The significance level was set at a 5% margin of error, corresponding to a 95% confidence interval. P < 0.05 indicated statistically significant, while P < 0.001 were regarded as highly significant. P > 0.05 were not significant.
3. RESULTS
Both groups had similar patient characteristics (Table 1)
| Table 1: Patients’ characteristics | ||||
| Variables | OFA Group
(n = 25) |
OBA Group
(n = 25) |
Test
value |
P |
| Age | 32.15 ± 8.35 | 33.37 ± 7.13 | 0.556 | 0.581 |
| Gender | ||||
| · Female | 16 (64.0%) | 18 (72.0%) | 0.092 | 0.762 |
| · Male | 9 (36.0%) | 7 (28.0%) | ||
| BMI [kg/m2] | 48.31 ± 4.43 | 47.28 ± 4.33 | 0.831 | 0.411 |
| Duration of surgery (min) | 157.59 ± 26.27 | 160.78 ± 24.21 | 0.446 | 0.657 |
| t-Independent Sample t-test for Mean ± SD;
x2: Chi-square test for n (%) or Fisher’s exact test, where appropriate *P < 0.05 is significant |
||||
and comparable intraoperative and postoperative heart rates (Figure), as well as other vital parameters (HR, MAP, PIP, EtCO₂, SpO₂) (Figures 1, 2, 3, 4 and 5).

Figure 1: Comparative intraoperative and postoperative heart rates
Using: t-Independent Sample t-test for Mean ± SD

Figure 2: Comparative intraoperative and postoperative MABP (mmHg)

Figure 3: Comparative peak inspiratory pressures

Figure 4: Comparative EtCO2 values (mmHg)

Figure 5: Comparative % of patients with SpO2 < 94%
However, the OFA Group showed a statistically significant higher delayed recovery frequency with (P < 0.05) (Figure 6) and required a longer duration of PCA for narcotic consumption (P < 0.001) Table 3), while the opioid group had higher VAS pain scores immediately postoperatively and up to 2 hours after surgery (P < 0.05) (Table 2) and required more narcotic boluses outside PCA (P < 0.001) (Table 3).
| Table 2: Comparative VAS scores between groups at different time periods | ||||
| Time | OFA Group
(n = 25) |
OBA Group
(n = 25) |
Test value | P |
| Immediately PO | 2 (1-3) | 5 (4-6) | 3.978 | 0.004* |
| After 30 min. | 4 (3-5) | 7 (5-9) | 3.019 | 0.012* |
| After 1 hr. | 3 (2-4) | 6 (5-8) | 2.868 | 0.019* |
| After 2 hr. | 3 (2-4) | 5 (4-6) | 2.144 | 0.046* |
| After 4 hrs. | 4 (3-5) | 4 (3-5) | 0.575 | 0.180 |
| After 8 hrs. | 4 (3-5) | 4 (3-5) | 0.040 | 0.571 |
| After 12 hrs. | 4 (3-4) | 4 (3-5) | 0.719 | 0.270 |
| After 16 hrs. | 4 (3-5) | 4 (3-5) | 0.409 | 0.167 |
| After 20 hrs. | 3 (2-4) | 3 (2-4) | 0.562 | 0.179 |
| After 24 hrs. | 3 (2-4) | 3 (2-4) | 0.364 | 0.578 |
| Data are expressed as Median and Interquartile range (IQR)
Using: U = Mann-Whitney test for non-parametric data “Median (IQR)” *P < 0.05 is significant |
||||
| Table 3: Comparative total narcotic consumption postoperative with PCA | ||||
| OFA Group
(n = 25) |
OBA Group
(n = 25) |
Test value | P | |
| Duration of PCA (hrs) (total narcotic consumption) | 22.91 ± 4.56 | 18.68 ± 3.75 | 3.582 | < 0.001** |
| #Number of narcotic boluses other than PCA | 1 (0-2) | 3 (3-4) | 6.386 | < 0.001** |
| #Data are median and IQR: Interquartile range
Using: t-Independent Sample t-test for Mean ± SD; Mann-Whitney test for Non-parametric data “Median (IQR)” *P < 0.05 is significant |
||||
Both groups were similar in ICU recovery time and hospital stay duration (Table 4).
| Table 4: Comparison between groups regarding ICU and hospital stay. | |||||
| Days of ICU | OFA Group | OBA Group | Test value | P | |
| ICU stay | < 2 days | 6 (85.7%) | 7 (70.0%) | 0.565 | 0.754 |
| ≥2 days | 1 (14.3%) | 3 (30.0%) | |||
| Total | 7 100.0%) | 10 (100.0%) | |||
| Length of hospital stay (days) | Range | 2-4 | 2-4 | ||
| Mean ± SD | 3.09 ± 0.68 | 3.14 ± 0.69 | |||
| x2: Chi-square test for Number (%) or Fisher’s exact test, when appropriate; t-Independent Sample t-test for Mean ± SD
*P < 0.05 is significant; |
|||||
The opioid balanced group showed a statistically significant increased frequency of nausea and vomiting postoperatively (P < 0.05), while shivering and bradycardia differences were insignificant (P > 0.05) (Table 5).
| Table 5: Comparative side effects | ||||
| Side effects | OFA Group
(n = 25) |
OBA Group
(n = 25) |
Test value | P |
| PO nausea and vomiting | 2 (8.0%) | 8 (32.0%) | 4.410 | 0.036* |
| Bradycardia | 8 (32.0%) | 5 (20.0%) | 0.917 | 0.338 |
| Shivering | 5 (20.0%) | 7 (28.0%) | 0.430 | 0.512 |
| x2: Chi-square test for n (%) or Fisher’s exact test, when appropriate
*P < 0.05 considered as significant |
||||
4. DISCUSSION
The purpose of our study was to evaluate safety and clinical effectiveness of using non-narcotic, multimodal anesthesia technique compared to the traditional opioid-based approach in laparoscopic bariatric surgeries. Both groups had similar demographic data. Intraoperative vital data, including mean arterial blood pressure, heart rate, EtCO2, along with peak inspiratory pressure, showed no notable differences between both groups. These findings align with Mansor et al. (2013), who also reported no significant differences in vital signs between patients managed with opioid-free anesthesia and those who were given opioid-based anesthesia (17). However, Elsaye et al. (2019) observed a considerable reduction In both heart rate and mean arterial pressure levels in patients receiving opioid-free anesthesia, attributed to their use of dexmedetomidine as preoperative loading and intraoperative maintenance drug together with lidocaine and Magnesium without ketamine.18 In contrast, the current study used ketamine in the multimodal approach, which may have masked dexmedetomidine-induced bradycardia, explaining the differing results.Moreover, Bakan et al. (2015), who studied opioid-free GA using propofol, lidocaine, and dexmedetomidine for laparoscopic surgery found a statistically significant elevation of mean arterial pressure levels and heart rate values within the opioid-free group during induction, at intubation, and during early pneumoperitoneum. These findings contradict the current study, which observed no significant differences in these parameters.19 The discrepancy may be due to Bakan et al. (2015) 's reliance on dexmedetomidine alone for sedation and analgesia during induction and intubation.
The current study also emphasized that the opioid-free regimen patients experienced considerable lower postoperative pain scores and reduced opioid consumption, aligning with Soudi et al. (2022), who reported that OFA has a better outcomes than total balanced anesthesia (TBA) regarding postoperative pain intensity score and narcotics use.20 Additionally, our findings are agreeing with Berlier et al. (2022), who revealed that OFA leads to lower morphine requirements during the first 24 hours after bariatric surgical procedures in comparison to OBA.21
On the other hand, The current study's results differ from Torre et al. (2022), who reported that OFA did not reduce total postoperative opioid consumption in comparison to opioid-based anesthesia groups.11
The current study revealed non-significant difference between both regimens regarding postoperative hypoxemia. However, the opioid group experienced eight cases of nausea and vomiting, whereas the OFA Group had only two cases. This finding aligns with Elsaye et al. (2019), who found a higher postoperative complications like nausea, vomiting, hypoxia, and shivering in the opioid-based anesthesia group.22 Torre et al. (2022) also reached similar results when they revealed that OFA patients consumed less antiemetics along with faster hospital discharges.11 Similarly, Ziemann-Gimmel et al. (2014) observed a reduced incidence of nausea and vomiting postoperatively in the OFA Group, consistent with the current findings.23
Regarding recovery time, our study showed that patients receiving opioid-free anesthesia experienced significantly delayed recovery compared to those in the opioid group. This delay may be attributed to the multiple drugs used in our protocol, suggesting a need for further research to explore adjustments such as removing magnesium, reducing maintenance doses, or stopping infusions earlier. These findings align with Soudi et al.(2022), who also reported longer extubation times and a delayed achievement of an Aldrete score of 9 in patients undergoing opioid-free anesthesia.24
5. CONCLUSION
This study concluded that a multimodal opioid-free approach offers effective analgesia compared to traditional opioid use in bariatric surgeries, resulting in reduced postoperative pain intensity levels, less narcotics requirements, and more rapid recovery.
6. Data availability
The numerical data generated during this research is available with the authors.
7. Conflict of interest
All authors declare that there was no conflict of interest.
8. Funding
The study utilized the hospital resources only, and no external or industry funding was involved.
9. Ethical considerations
The research ethical committee at the medical school at Ain Shams University gave its approval to this study. (FMASU MD 358 / 2018) with Pan African Clinical Trial Registry, identifier PACTR202401561160798
10. Authors’ contribution
Each author made a significant intellectual contribution to the work, and each author read and approved the final product.
11. REFERENCES
- Luppino FS, de Wit LM, Bouvy PF, Stijnen T, Cuijpers P, Penninx BW, et al. Overweight, obesity, and depression: a systematic review and meta-analysis of longitudinal studies. Arch Gen Psychiatry. 2010;67(3):220–9. PubMedDOI: 1001/archgenpsychiatry.2010.2
- Bohringer C, Astorga C, Liu H. The benefits of opioid free anesthesia and the precautions necessary when employing it. Transl Perioper Pain Med. 2020;7(1):152–7. PubMed
- Angrisani L, Santonicola A, Iovino P, Palma R, Kow L, Prager G, et al. IFSO worldwide survey 2020–2021: current trends for bariatric and metabolic procedures. Obes Surg. 2024;34(4):1075–85. PubMedDOI: 1007/s11695-024-07118-3
- Barakat A, Hamdy MM, Elbadr MM. Uses of fluoxetine in nociceptive pain management: a literature overview. Eur J Pharmacol. 2018;829:12–25. PubMedDOI: 1016/j.ejphar.2018.03.042
- Zhou J, Du R, Wang L, Wang F, Li D, Tong G, et al. The application of enhanced recovery after surgery (ERAS) for patients undergoing bariatric surgery: a systematic review and meta-analysis. Obes Surg. 2021;31(3):1321–31. PubMedDOI: 1007/s11695-020-05209-5
- Mulier JP, editor. Opioid-free anaesthesia (OFA) to optimize enhanced recovery after laparoscopic bariatric surgery (ERAS). 3rd Int Conf Enhanced Recovery After Surg. Cannes, France; 2012.
- Liu HT, Hollmann MW, Liu WH, Hoenemann CW, Durieux ME. Modulation of NMDA receptor function by ketamine and magnesium: part I. Anesth Analg. 2001;92(5):1173–81. PubMedDOI: 1097/00000539-200105000-00019
- Ulbing S, Infanger L, Fleischmann E, Prager G, Hamp T. The performance of opioid-free anesthesia for bariatric surgery in clinical practice. Obes Surg. 2023;33(6):1687–93. PubMedDOI: 1007/s11695-023-06584-5
- Yaşar ŞÜ, Yıldız AM. Comparison of opioid and opioid-free anesthesia in bariatric surgery. Ann Clin Anal Med. 2023;14(9):825–9.
- Mahdy EW, Mohammed EA, Abdelwahab ZM. Comparative study between opioid-free anesthesia and opioid-based anesthesia in obese patients undergoing abdominal laparoscopic surgery: effective and safe analgesia challenge. Egypt J Hosp Med. 2024;97:4261–7. DOI: 21608/ejhm.2024.395543
- Torre A, Marengo M, Ledingham NS, Ajani C, Volontè F, Garofalo F, et al. Opioid-free anesthesia in bariatric surgery: a propensity score-matched analysis. Obes Surg. 2022;32(5):1673–80. PubMedDOI: 1007/s11695-022-06012-0
- Seyam SHA, Hassan AE, Elsayed MM. Assessing the impact of opioid-free anesthesia using the modified Mulimix technique on postoperative pain in bariatric surgery patients: a randomized double-blinded trial. Anaesth Pain Intensive Care. 2024;28(4). DOI: 35975/apic.v28i4.2516
- Mulier J, Wouters R, Dillemans B, Dekock M. A randomized controlled, double-blind trial evaluating the effect of opioid-free versus opioid general anaesthesia on postoperative pain and discomfort measured by the QoR-40. J Clin Anesth Pain Med. 2018;2. Full Text
- Ekman A, Lindholm ML, Lennmarken C, Sandin R. Reduction in the incidence of awareness using BIS monitoring. Acta Anaesthesiol Scand. 2004;48(1):20–6. PubMedDOI: 1111/j.1399-6576.2004.00260.x
- Foster JG, Kish SK, Keenan CH. National practice with assessment and monitoring of neuromuscular blockade. Crit Care Nurs Q. 2002;25(2):27–40. PubMedDOI: 1097/00002727-200208000-00004
- Aldrete JA, Kroulik D. A postanesthetic recovery score. Anesth Analg. 1970;49(6):924–34. PubMed
- Pavlin DJ, Rapp SE, Polissar NL, Malmgren JA, Koerschgen M, Keyes H. Factors affecting discharge time in adult outpatients. Anesth Analg. 1998;87(4):816–26. PubMedDOI: 1097/00000539-199810000-00014
- Zelcer J, Wells DG. Anaesthetic-related recovery room complications. Anaesth Intensive Care. 1987;15(2):168–74. PubMedDOI: 1177/0310057X8701500209
- Chand B, Prathanvanich P. Critical care management of bariatric surgery complications. J Intensive Care Med. 2016;31(8):511–28. PubMedDOI: 1177/0885066615593067
- Froylich D, Corcelles R, Davis M, Boules M, Daigle CR, Schauer PR, et al. Factors associated with length of stay in intensive care after bariatric surgery. Surg Obes Relat Dis. 2016;12(7):1391–6. PubMedDOI: 1016/j.soard.2015.11.031
- Mansour MA, Mahmoud AA, Geddawy M. Nonopioid versus opioid-based general anesthesia technique for bariatric surgery: a randomized double-blind study. Saudi J Anaesth. 2013;7(4):387–91. PubMedDOI: 4103/1658-354X.121045
- Elsaye RM, Gaafary AM, Elsaeid AM. Comparative study between the effect of opioid-free anesthesia versus opioid-based anesthesia in morbid obese patients. Sci J Al-Azhar Med Fac Girls. 2019;3(2):457–63. Full Text
- Bakan M, Umutoglu T, Topuz U, Uysal H, Bayram M, Kadioglu H, et al. Opioid-free total intravenous anesthesia with propofol, dexmedetomidine and lidocaine infusions for laparoscopic cholecystectomy: a prospective, randomized, double-blinded study. Braz J Anesthesiol. 2015;65(3):191–9. PubMedDOI: 1016/j.bjane.2014.05.001
- Soudi AM, Hammad RA, ElShafie MA, Ahmed IMAS, Alhadidy MA. Comparing opioid-free general anesthesia to traditional balanced general anesthesia regarding achievement of enhanced recovery in laparoscopic bariatric surgeries. Ain Shams J Anaesthesiol. 2022;14(1):24. DOI: 1186/s42077-022-00218-1